Chinese Journal of Tissue Engineering Research ›› 2023, Vol. 27 ›› Issue (6): 935-940.doi: 10.12307/2023.266
Previous Articles Next Articles
Schwann cell-derived exosomes promote the repair and regeneration of injured peripheral nerves
Yuan Bo1, 2, Xie Lide1, Fu Xiumei1, 3
- 1Chengde Medical University, Chengde 067000, Hebei Province, China; 2Chengde Nursing Vocational College, Chengde 067000, Hebei Province, China; 3Hebei Key Laboratory of Nerve Injury and Repair, Chengde 067000, Hebei Province, China
-
Received:2022-03-24Accepted:2022-05-13Online:2023-02-28Published:2022-08-12 -
Contact:Fu Xiumei, MD, Associate professor, Chengde Medical University, Chengde 067000, Hebei Province, China; Hebei Key Laboratory of Nerve Injury and Repair, Chengde 067000, Hebei Province, China Xie Lide, MD, Professor, Chengde Medical University, Chengde 067000, Hebei Province, China -
About author:Yuan Bo, Master candidate, Lecturer, Chengde Medical University, Chengde 067000, Hebei Province, China; Chengde Nursing Vocational College, Chengde 067000, Hebei Province, China -
Supported by:Natural Science Foundation of Hebei Province, No. H20214066056 (to FXM); Key Project of Education Department of Hebei Province, No. ZD2020178 (to FXM); Hebei Provincial Department of Science and Technology "Technology Innovation Guidance Special Project-Science and Technology Work Conference" Project
CLC Number:
Cite this article
Yuan Bo, Xie Lide, Fu Xiumei. Schwann cell-derived exosomes promote the repair and regeneration of injured peripheral nerves[J]. Chinese Journal of Tissue Engineering Research, 2023, 27(6): 935-940.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
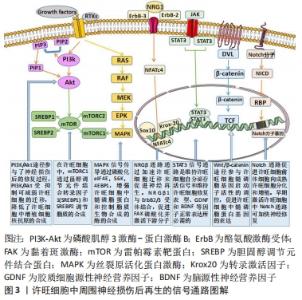
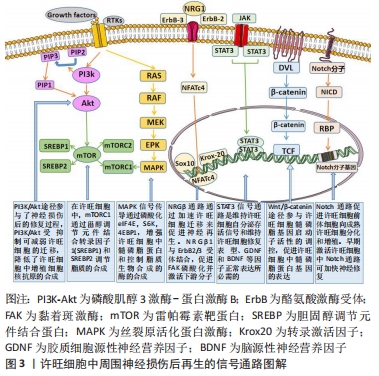
2.1 许旺细胞在促进受损周围神经再生修复中的作用 2.1.1 许旺细胞的可塑性及其作用 许旺细胞起源于神经嵴干细胞,它通过髓鞘形成独立的轴突,并允许神经冲动在较长距离上完成快速传播。许旺细胞可以分化成不同的表型,包括髓鞘化许旺细胞、非髓鞘化许旺细胞和修复型许旺细胞[19-20]。不同的分化亚型有着不同的生理和病理作用。随着环境信号刺激和控制基因的改变,非髓鞘表型许旺细胞可以转变为髓鞘表型许旺细胞,这体现出了许旺细胞的可塑性[21]。 周围神经损伤后因缺乏营养支持,神经远端会出现变形、解体、髓鞘及轴突破坏等一系列连锁反应,即Wallerian变性[22]。在Wallerian变性过程中,成熟的髓鞘许旺细胞和非髓鞘许旺细胞会在细胞层面和分子层面发生复杂的变化,形成修复型许旺细胞,从而启动整体修复过程。首先髓鞘分化发生逆转,促髓鞘转录因子表达水平下调,随后未成熟的许旺细胞的生物标志物、神经营养因子和相应的细胞因子上调,最后结构发生重组形成修复许旺细胞表型。修复的许旺细胞与未成熟的许旺细胞有着显著的区别,前者可利用受体酪氨酸激酶亚家族(TAM家族)对其他表型许旺细胞进行调控[23],利用神经营养因子促使损伤的轴突出芽和延长,并通过分泌相应的细胞因子来影响神经变性的速率,然后通过募集临近区域内的巨噬细胞,吞噬并清除损伤区域内的髓鞘碎片,从而启动局部免疫反应,为损伤神经的再生修复开辟通道。通过在两断端神经之间形成半透明的带状外观的“神经桥”——Büngner带,以此来维系轴突再生与修复的微环境[22]。许旺细胞还可以促进血管再生,从而为轴突的再生恢复提供营养支持。发生去分化的许旺细胞可为神经元的轴突再生提供生长通道,使再生轴突穿过瘢痕组织并被重新引导至相应的靶组织[20]。在轴突完成再生后,修复型的许旺细胞再次分化成髓鞘表型或非髓鞘化表型以支持神经功能恢复[24]。许旺细胞的可塑性使得它在细胞发育、轴突的生长和维持以及周围神经损伤后的再生修复过程中具有重要意义。 2.1.2 许旺细胞调控损伤神经再生修复的机制 周围神经损伤后,许旺细胞先后发生去分化、突触传递、轴突再生等一系列复杂的级联反应,这一过程是神经轴突、许旺细胞和微环境3者之间通过多种信号通路相互作用的结果[25]。许旺细胞调控损伤神经再生修复的信号通路,见图3。 "
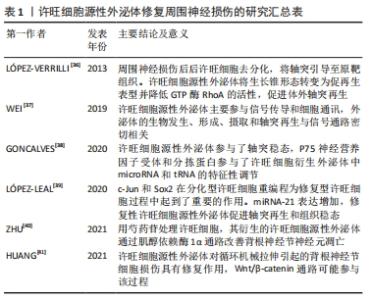
2.2 外泌体在周围神经损伤修复中的应用研究 外泌体的形成经历以下阶段:首先细胞质膜发生内陷形成早期的分泌内体;随后向内发芽形成腔内囊泡;分泌内体继续酸化进一步发育成熟;最终通过与质膜融合向胞外释放囊泡,即外泌体[26-27]。这些释放到胞外的外泌体将携带的多种生物活性物质释放到细胞质中,通过与受体结合、吞噬作用、巨胞饮作用,以及质膜融合等方式在细胞脂质双层结构内完成远距离运输,在翻译和转录过程中调节特定的受体细胞,从而实现与受体细胞之间的相互作用。 研究表明,几乎所有体液中都可以检测到外泌体,包括血液、尿液、脑脊液、唾液、母乳等[28]。外泌体具有可调节的免疫原性、能够穿过血脑屏障、输送细胞因子、分泌特异性的脂质和蛋白质等广泛的功能特性[17,29]。根据外泌体数据库的最新显示,外泌体含有9 769种蛋白质,3 408种mRNA,2 838种miRNA和1 116种脂质[30],并成为肿瘤、心血管疾病和免疫反应等多种生理病理过程中的重要参与者[31]。除此以外,巨噬细胞、多种干细胞、许旺细胞分泌的外泌体可以有效刺激受损神经的轴突再生,刺激血管生成以及细胞增殖,为神经修复营造理想的微环境[32]。因此,外泌体在周围神经修复再生领域具有广阔的前景。 随着对外泌体研究的不断进展,如何高效快速地分离纯化外泌体是实验研究和蛋白组学分析的关键。外泌体体积微小,且不同外泌体的成分含量和形态也存在差异,因此外泌体在分离提取阶段面临着巨大的挑战。目前常用的分离提纯方法有差速离心法、密度梯度离心法、超滤法、免疫亲和法、聚合物沉淀法、凝胶过滤色谱法等[33-35]。对于外泌体的提取目前还没有标准化的流程,为了高效率得到高纯度的外泌体,可以在实验过程中结合具体情况将不同方法相互联合以达到最好的提取效果。 2.3 许旺细胞源性外泌体修复周围神经损伤的应用进展 2.3.1 许旺细胞源性外泌体修复周围神经损伤的研究成果汇总 见表1[36-41]。 "


2.3.2 内源性许旺细胞衍生的外泌体对周围神经损伤的修复作用 许旺细胞源性外泌体对周围神经系统损伤后轴突再生的促进作用已被广泛证实。WEI等[37]对许旺细胞源性外泌体进行蛋白质组学分析,发现内源性许旺细胞衍生的外泌体在修复损伤神经过程中主要促进信号传导和细胞通讯,通过PI3K/Akt和CAMP 信号通路、神经营养因子在修复受损神经过程中发挥着重要的作用。另一项研究对内源性许旺细胞衍生的外泌体进行RNA测序,发现许旺细胞源性外泌体参与了轴突的稳态,通过敲除许旺细胞中p75神经营养因子受体或sortilin基因来改变tRNAs和miRNAs的表达水平,可以影响自噬和磷脂酰肌醇等相关信号通路[38]。许旺细胞分泌的外泌体携带的表达最高的20种miRNA主要与轴突发生、轴突导向和轴突延伸的调节有关[42],其中内源性许旺细胞分泌的外泌体携带的miRNA-93-5p、miRNA-16-5p、miRNA21-5p、miRNA-99b-5p和miRNA-9a-5p是调节轴突生长过程的最重要的miRNA[38],这些生物活性物质的作用和机制将是接下来重点研究的问题。 LóPEZ-LEAL等[39]研究认为周围神经的再生修复需要在机体的微环境下将分化型许旺细胞重编程为修复型许旺细胞,而重编程这一过程需要c-Jun和Sox2的参与,c-Jun和Sox2的上调抑制了分化表型许旺细胞的髓鞘形成,促进了修复型许旺细胞分泌的外泌体被外周神经元内化,从而介导了轴突的再生。虽然分化表型许旺细胞分泌的外泌体没有表现出增强轴突再生的能力,但用环腺苷酸诱导分化型许旺细胞并对其分泌的外泌体进行测序,结果发现分化表型许旺细胞分泌的外泌体携带的miRNA通过自分泌或旁分泌途径传递相关信息来抑制内源性许旺细胞的迁移,促进髓鞘形成,从而发挥许旺细胞修复周围神经损伤的功能[35]。分化表型许旺细胞分泌的外泌体通过改变miRNA的表达来调节许旺细胞的迁移,这取决于磷酸酶张力蛋白同源物基因(phosphatase and tensin homology deleted on chromosome ten,PTEN)表达水平的下调和磷酸肌醇(phosphatidylinositol-3-kinase,PI3K)的活化[39]。这表明不同分化表型的内源性许旺细胞衍生的外泌体功能是不同的,因为不同外泌体携带的物质成分、含量是有差异的,修复表型许旺细胞分泌的外泌体可以特异性修复周围神经损伤。但受时间影响较大,修复型许旺细胞在长时间缺失轴突接触后,许旺细胞凋亡数量增加,再生功能也会相应减低[40]。因此争取周围神经损伤后的时间非常重要。 2.3.3 外源性许旺细胞分泌的外泌体在周围神经损伤修复过程中促进作用 HRANG等[41]发现输入外源性许旺细胞衍生的外泌体可以通过促进背根神经节细胞增殖,促进细胞周期向G2期转变、抑制细胞凋亡、促进轴突再生等途径对机械损伤后的背根神经节细胞发挥治疗作用,Wnt/β-catenin信号通路参与其中。同时外源性许旺细胞分泌的外泌体可以通过增加自噬和减少细胞凋亡来诱导脊髓损伤后的轴突再生,从而促进运动功能的恢复,EGFR/Akt/mTOR信号通路参与了这一过程,研究还显示增加的许旺细胞源性外泌体可导致EGFR表达水平降低,对Akt/mTOR信号通路产生抑制作用[43]。PAN等[44]实验证明,外源性许旺细胞分泌的外泌体通过NF-κB/PI3K信号通路增加星形胶质细胞上Toll样受体2的表达、减少硫酸软骨素蛋白聚糖的沉积,来促进轴突再生、神经功能恢复,从而促进创伤性脊髓损伤后小鼠的功能恢复。许旺细胞源性外泌体携带的诸如siRNA,miRNA和蛋白质等生物活性分子可以在外泌体形成期间或者外泌体分离后加载到外泌体中,通过外泌体递送到相应的靶细胞[45]。CONG等[46]研究表明许旺细胞源性外泌体携带miRNA-21-5p负调节PTEN,同时调节PI3K途径的下游信号分子,从而促进氧-葡萄糖剥夺损伤的神经元恢复。这些生物活性物质在周围神经损伤后的再生修复过程中起到不同程度的修饰作用。 2.3.4 外源性物质对许旺细胞源性外泌体功能的影响 研究发现皮肤前体细胞经N2、神经调节蛋白1β和毛喉素体外诱导后可衍生为许旺细胞[47],并具有分泌外泌体的功能,利用蛋白印迹实验证实,外泌体通过PI3K/Akt信号通路改善氧-葡萄糖剥夺后神经元的活力,通过调节Bax/Bcl-2增强氧-葡萄糖剥夺损伤后感觉神经元的抗凋亡作用,从而促进受损神经的轴突再生。另一项研究发现高葡萄糖环境会对原代许旺细胞造成一定程度的伤害,并通过激活细胞外信号调节激酶这一信号通路抑制许旺细胞活性和分化[48]。通过体外研究表明,高葡萄糖刺激可使许旺细胞源性外泌体携带的miRNA-28、miRNA-31a和miRNA-130a的表达水平增加,其相关靶蛋白DNA甲基转移酶3a、NUMB内吞衔接蛋白、轴突中的突触体相关蛋白25等表达减少,从而使远端轴突生成减少[49]。利用芍药苷来干预许旺细胞源性外泌体,可显著降低葡萄糖调节蛋白78、肌醇依赖酶1α的表达以及肌醇依赖酶1α的磷酸化水平,调节肌醇依赖酶1α 通路下游蛋白的表达,以此来抑制背根神经节神经元的凋亡[40]。这进一步证实了外泌体携带的生物活性物质参与了轴突-神经胶质细胞间的通讯,并在促进轴突再生和增强神经元存活方面起到了一定的修饰作用[39]。 2.3.5 许旺细胞源性外泌体的优势与挑战 周围神经损伤后的再生修复涉及许旺细胞表型的变化、巨噬细胞的激活和血管网络的重建[49]。许旺细胞是周围神经损伤后神经修复的重要细胞。首先从许旺细胞和成纤维细胞中提取外泌体,然后将提取的外泌体分别与骨髓间充质干细胞共培养,结果发现许旺细胞源性外泌体显著增加了骨髓间充质干细胞中许旺细胞的特异标志物的表达;而成纤维细胞衍生的外泌体并未表现出相同的效果[50],揭示了许旺细胞源性外泌体在修复受损神经方面的优越性。LóPEZ-VERRILLI等[36]用PKH67标记许旺细胞源性外泌体和原代成纤维细胞分泌的外泌体,将两种不同来源的外泌体在体外作用于背根神经节神经元,结果显示轴突会选择性地与许旺细胞源性外泌体结合,也证实了许旺细胞源性外泌体在神经修复方面的作用。COURT等[51]研究进一步表明轴突的延长和再生依赖于许旺细胞的调节,许旺细胞通过其分泌的外泌体提供mRNAs,来协助轴突合成相应蛋白质,许旺细胞源性外泌体为受损轴突提供高效、特异的支持。同时许旺细胞源性外泌体的不同表型中只有修复表型许旺细胞分泌的外泌体能够促进周围神经损伤后的轴突再生,分化表型许旺细胞衍生的外泌体未表现出此功能[39]。许旺细胞源性外泌体可以通过增强表皮神经纤维、促进髓鞘和轴突再生,来提高运动神经元的存活和抑制细胞凋亡来保护周围神经[52],而许旺细胞源性外泌体介导的神经细胞活力增强的潜在机制与caspase3依赖性细胞凋亡有关,这也部分揭示了许旺细胞源性外泌体发挥作用的可能机制。 许旺细胞源性外泌体在修复周围神经损伤过程中也存在一些挑战,一方面,许旺细胞源性外泌体来源有限,许旺细胞体外增殖相对困难,易被成纤维细胞污染,无法大规模提取外泌体,因此在投入临床使用过程中存在一定的局限性。另一方面,对于外泌体的分离纯化目前尚没有标准流程,现有的外泌体分离提取方法有着不同的局限性,例如回收率低、纯度低、耗时、费力等。因此如何高效率得到纯度高的外泌体是利用许旺细胞源性外泌体修复周围神经损伤面临的巨大挑战。第三方面,许旺细胞源性外泌体的给药途径、使用安全性、能否在临床上广泛应用等问题也有待更加深入的探究,利用许旺细胞源性外泌体作为治疗载体将生物活性分子递送至受损神经元以改善周围神经损伤后的神经功能、促进受损神经的轴突再生有着良好的前景。 "

| [1] CASTILLO-GALVÁN ML, MARTÍNEZ-RUIZ FM, DE LA GARZA-CASTERO O, et al. Study of peripheral nerve injury in trauma patients. Gac Med Mex. 2014;150(6):527-532. [2] NOCERA G, JACOB C. Mechanisms of Schwann cell plasticity involved in peripheral nerve repair after injury. Cell Mol Life Sci. 2020;77(20): 3977-3989. [3] BHANDARI PS. Management of peripheral nerve injury. J Clin Orthop Trauma. 2019;10(5):862-866. [4] KLIMOVICH P, RUBINA K, SYSOEVA V, et al. New frontiers in peripheral nerve regeneration: concerns and remedies. Int J Mol Sci. 2021; 22(24):13380. [5] 宋凯凯, 张锴, 贾龙. 周围神经系统损伤的微环境与修复方式[J].中国组织工程研究,2021,25(4):651-656. [6] LKUMI A,GINGERY A,TOYOSHIMA Y, et al. Administration of purified exosome product in a rat sciatic serve reverse autograft model. Plast Reconstr Surg. 2021;148(2):200-211. [7] KUBIAK CA,KUNG TA,BOWN DL, et al. State-of-the-art techniques in treating peripheral nerve injury. Plast Reconstr Surg. 2018;141(3):702-710. [8] RAZA C, RIAZ HA, ANJUM R, et al. Repair strategies for injured peripheral nerve: review. Life Sci. 2020;243:117308. [9] 朱芳慧.甲钴胺联合硫辛酸治疗糖尿病周围神经病变的临床疗效[J].实用心脑肺血管病杂志,2017,25(S2):32-34. [10] 赵华飞,王维新,崔硕,等.几丁糖对鼠坐骨神经损伤后再生的影响[J].中国临床药理学杂志,2018,34(14):1659-1661. [11] MODRAK M, TALUKDER MAH, GURGENASHVILI K, et al. Peripheral nerve injury and myelination: Potential therapeutic strategies. J Neurosci Res. 2020;98(5):780-795. [12] LI Y, MA Z, REN Y, et al. Tissue engineering strategies for peripheral nerve regeneration. Front Neurol. 2021;12:768267. [13] BUSUTTIL F, RAHIM AA, PHILLIPS JB. Cand stem cell therapy for peripheral nerve tissue engineering. Stem Cells Dev. 2017;26(4):231-238. [14] 陈渝,邓忠良,翁政,等.NGF/MAG双基因共表达腺病毒修复大鼠周围神经损伤的试验研究[J].中国修复重建外科杂志,2016,30(8):1026-1033. [15] LI H, CHANG LJ, NEUBAUER DR, et al. Immortalization of human normal and NF1 neurofibroma Schwann cells. Laboratory Investigation. 2016; 96(10):1105-1115. [16] CHEN J, REN S, DUSCHER D, et al. Exosomes from human adipose-derived stem cells promote sciatic nerve regeneration via optimizing Schwann cell function. J Cell Physiol. 2019;234(12):23097-23110. [17] DAI J, SU Y, ZHONG S, et al. Exosomes: key players in cancer and potential therapeutic strategy. Signal Transduct Target Ther. 2020; 5(1):145. [18] 袁一鸣,王艳,陈程程,等.外泌体在周围神经损伤进程中的效应[J].中国组织工程研究,2020,24(31):5079-5084. [19] MONK KR, FELTRI ML, TAVEGGIA C. New insights on Schwann cell development. Glia. 2015;63(8):1376-1393. [20] JESSEN KR, MIRSKY R. The success and failure of the Schwann cell response to nerve injury. Front Cell Neurosci. 2019;13:33. [21] HARTY BL, MONK KR. Unwrapping the unappreciated: recent progress in remak Schwann cell biology. Curr Opin Neurobiol. 2017;47:131-137. [22] 蒋玲丽,魏在荣.周围神经损伤修复过程以及miRNA在周围神经修复中的研究进展[J].遵义医科大学学报,2020,43(6):801-806. [23] MADISON RD, MCGEE C, RAWSON R, et al. Extracellular vesicles from a muscle cell line (C2C12) enhance cell survival and neurite outgrowth of a motor neuron cell line (NSC-34). J Extracell Vesicles. 2014;3(1): 22865. [24] BOERBOOM A, DION V, CHARIOT A, et al. Molecular mechanisms involved in Schwann cell plasticity. Front Mol Neurosci. 2017;10:38. [25] WONG FC, YE L, DEMIR IE, et al. Schwann cell-derived exosomes: Janus-faced mediators of regeneration and disease. Glia. 2022;70(1):20-34. [26] IBRAHIM A, MARBÁN E. Exosomes: Fundamental biology and roles in cardiovascular physiology. Annu Rev Physiol. 2016;78:67-83. [27] DE GREGORIO C, DíAZ P, LÓPEZ-LEAL R, et al. Purification of exosomes from primary Schwann cells, RNA extraction, and next-generation sequencing of exosomal RNAs. Methods Mol Biol. 2018;1739:299-315. [28] 谢欢. HUVECs分泌的外泌体通过抑制miR-21-3p减弱缺氧/复氧诱导的神经细胞凋亡[D].南昌:南昌大学,2019. [29] KALLURI R, LEBLEU VS. The biology, function, and biomedical applications of exosomes. Science. 2020;367(6478):6977. [30] XIE F, ZHOU X, FANG M, et al. Extracellular vesicles in cancer immune microenvironment and cancer immunotherapy. Adv Sci (Weinh). 2019; 6(24):1901779. [31] ZHOU B, XU K, ZHENG X, et al. Application of exosomes as liquid biopsy in clinical diagnosis. Signal Transduct Target Ther. 2020;5(1):144. [32] 周敏,洪莉,胡鸣,等.外泌体在周围神经损伤中的研究进展[J].医学综述,2017,23(13):2497-2500. [33] 高方园, 焦丰龙, 张养军,等.外泌体分离技术及其临床应用研究进展[J].色谱,2019,37(10):1071-1083. [34] HYUNG S, KIM JY, YU CJ, et al. Neuroprotective effect of glial cell-derived exosomes on neurons. Immunotherapy: Open Access. 2019; 5(1):156. [35] SOHN E J, PARK H T, SHIN YK. Exosomes derived from differentiated Schwann cells inhibit Schwann cell migration via microRNAs. Neuroreport. 2020;31(7):515-522. [36] LÓPEZ-VERRILLI MA, PICOU F, COURT FA. Schwann cell-derived exosomes enhance axonal regeneration in the peripheral nervous system. Glia. 2013;61(11):1795-1806. [37] WEI Z, FAN B, DING H, et al. Proteomics analysis of Schwann cell-derived exosomes: a novel therapeutic strategy for central nervous system injury. Mol Cell Biochem. 2019;457(1-2):51-59. [38] GONCALVES NP, YAN Y, ULRICHSEN M, et al. Modulation of small RNA signatures in Schwann-cell-derived extracellular vesicles by the p75 neurotrophin receptor and Sortilin. Biomedicines. 2020;8(11):450. [39] LÓPEZ-LEAL R, DÍAZ-VIRAQUÉ F, CATALÁN RJ, et al. Schwann cell reprogramming into repair cells increases miRNA-21 expression in exosomes promoting axonal growth. J Cell Sci. 2020;133(12):239004. [40] ZHU Y, HAN S, LI X, et al. Paeoniflorin effect of Schwann cell-derived exosomes ameliorates dorsal root ganglion neurons apoptosis through IRE1α pathway. Evid Based Complement Alternat Med. 2021; 2021:6079305. [41] HRANG G, HU M, LU D, et al. Protective effect and potential mechanism of Schwann cell-derived exosomes on mechanical damage of rat dorsal root ganglion cells. J Obstet Gynaecol Res. 2021;47(10):3691-3701. [42] GONÇALVES NP, VÆGTER CB, ANDERSEN H, et al. Schwann cell interactions with axons and microvessels in diabetic neuropathy. Nat Rev Neurol. 2017;13(3):135-147. [43] PAN D, ZHU S, ZHANG W, et al. Autophagy induced by Schwann cell-derived exosomes promotes recovery after spinal cord injury in rats. Biotechnol Lett. 2022;44(1):129-142. [44] PAN D, LI Y, YANG F, et al. Increasing toll-like receptor 2 on astrocytes induced by Schwann cell-derived exosomes promotes recovery by inhibiting CSPGs deposition after spinal cord injury. J Neuroinflammation. 2021;18(1):172. [45] HU M, HONG L, LIU C, et al. Electrical stimulation enhances neuronal cell activity mediated by Schwann cell derived exosomes. Sci Rep. 2019; 9(1):4206. [46] CONG M, SHEN M, WU X, et al. Improvement of sensory neuron growth and survival via negatively regulating PTEN by miR-21-5p-contained small extracellular vesicles from skin precursor-derived Schwann cells. Stem Cell Res Ther. 2021;12(1):80. [47] Liu D, Liang X, Zhang H. Effects of High Glucose on Cell Viability and Differentiation in Primary Cultured Schwann Cells: Potential Role of ERK Signaling Pathway. Neurochem Res. 2016;41(6):1281-1290. [48] JIA L, CHOPP M, WANG L, et al. Exosomes derived from high-glucose-stimulated Schwann cells promote development of diabetic peripheral neuropathy. FASEB J. 2018;32(12):fj201800597R. [49] Dong R, Liu Y, Yang Y, et al. MSC-Derived Exosomes-Based Therapy for Peripheral Nerve Injury: A Novel Therapeutic Strategy. Biomed Res Int. 2019;2019:6458237. [50] WANG H, JIA Y, LI J, et al. Schwann cell derived exosomes induce bone marrow derived mesenchymal stem cells to express Schwann cell markers in vitro. Mol Med Rep. 2020;21(3):1640-1646. [51] COURT FA, ALVAREZ J. Schwann cell and axon: an interlaced unit-from action potential to phenotype expression. Adv Exp Med Biol. 2016; 949:183-201. [52] WANG L, CHOPP M, SZALAD A, et al. Exosomes derived from schwann cells ameliorate peripheral neuropathy in type 2 diabetic mice. Diabetes. 2020;69(4):749-759. |
| [1] | Sun Kexin, Zeng Jinshi, Li Jia, Jiang Haiyue, Liu Xia. Mechanical stimulation enhances matrix formation of three-dimensional bioprinted cartilage constructs [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(在线): 1-7. |
| [2] | Pan Zhongjie, Qin Zhihong, Zheng Tiejun, Ding Xiaofei, Liao Shijie. Targeting of non-coding RNAs in the pathogenesis of the osteonecrosis of the femoral head [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(9): 1441-1447. |
| [3] | Zhao Lu, Zhao Yifei, Gao Da, Liu Yanfang, Fu Tingting, Xu Jiangyan. Expression of suppressor of Zeste 12 in kidney tissues of rats with diabetic nephropathy [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(8): 1179-1186. |
| [4] | Xu Xingxing, Wen Chaoju, Meng Maohua, Wang Qinying, Chen Jingqiao, Dong Qiang. Carbon nanomaterials in oral implant [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1062-1070. |
| [5] | Yang Yitian, Wang Lu, Yao Wei, Zhao Bin. Application of the interaction between biological scaffolds and macrophages in bone regeneration [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1071-1079. |
| [6] | Li Cheng, Zheng Guoshuang, Kuai Xiandong, Yu Weiting. Alginate scaffold in articular cartilage repair [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1080-1088. |
| [7] | Shi Yehong, Wang Cheng, Chen Shijiu. Early thrombosis and prevention of small-diameter blood vessel prosthesis [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1110-1116. |
| [8] | Tang Haotian, Liao Rongdong, Tian Jing. Application and design of piezoelectric materials for bone defect repair [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1117-1125. |
| [9] | Xu Yan, Li Ping, Lai Chunhua, Zhu Peijun, Yang Shuo, Xu Shulan. Piezoelectric materials for vascularized bone regeneration [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1126-1132. |
| [10] | Liu Wentao, Feng Xingchao, Yang Yi, Bai Shengbin. Effect of M2 macrophage-derived exosomes on osteogenic differentiation of bone marrow mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(6): 840-845. |
| [11] | Li Xinyue, Li Xiheng, Mao Tianjiao, Tang Liang, Li Jiang. Three-dimensional culture affects morphology, activity and osteogenic differentiation of human periodontal ligament stem cells [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(6): 846-852. |
| [12] | Li Qicheng, Deng Jin, Fu Xiaoyang, Han Na. Effects of bone marrow mesenchymal stem cells-derived exosomes on hypoxia-treated myoblasts [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(6): 853-859. |
| [13] | Wang Min, Yin Xiushan, Wang Yingxi, Zhang Yan, Zhao Long, Xia Shuyue. Inhalation of bone marrow mesenchymal stem cells-derived exosomes alleviates inflammatory injury in chronic obstructive pulmonary disease [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(6): 827-834. |
| [14] | Zhang Houjun, Deng Bowen, Jiang Shengyuan, Zhao Yi, Ren Jingpei, Xu Lin, Mu Xiaohong. Proteomic analysis of cerebrospinal fluid exosomes derived from cerebral palsy children [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(6): 903-908. |
| [15] | Gao Ting, Ma Xiaohong, Li Xiaorong. Extraction and identification of exosomes from three different sources of ovarian granulosa cells [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(6): 860-865. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||