Chinese Journal of Tissue Engineering Research ›› 2023, Vol. 27 ›› Issue (2): 177-183.doi: 10.12307/2022.1007
Previous Articles Next Articles
Ginsenoside Rg3 protects PC12 cells against oxygen-glucose deprivation/reoxygenation-induced damage
Zhong Jinglin, Cao Huimin, Pan Yaru, Jian Wenxuan, Wang Qi
- Science and Technology Innovation Center, Guangzhou University of Chinese Medicine, Guangzhou 510000, Guangdong Province, China
-
Received:2022-02-07Accepted:2022-03-02Online:2023-01-18Published:2022-06-20 -
Contact:Wang Qi, Master, Professor, Doctoral supervisor, Science and Technology Innovation Center, Guangzhou University of Chinese Medicine, Guangzhou 510000, Guangdong Province, China -
About author:Zhong Jinglin, Master candidate, Science and Technology Innovation Center, Guangzhou University of Chinese Medicine, Guangzhou 510000, Guangdong Province, China -
Supported by:the Key Laboratory Project of Colleges and Universities in Guangdong Province, No. 2019KSYS005 (to WQ); Guangdong Province Science and Technology Plan International Cooperation Project, No. 2020A0505100052 (to WQ)
CLC Number:
Cite this article
Zhong Jinglin, Cao Huimin, Pan Yaru, Jian Wenxuan, Wang Qi. Ginsenoside Rg3 protects PC12 cells against oxygen-glucose deprivation/reoxygenation-induced damage[J]. Chinese Journal of Tissue Engineering Research, 2023, 27(2): 177-183.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
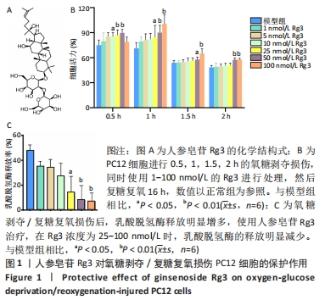
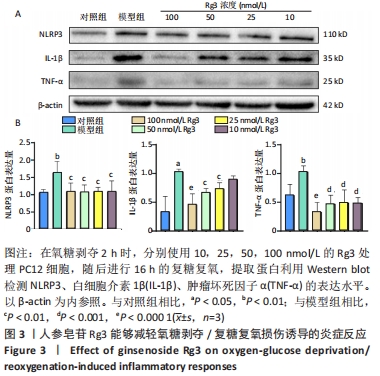
2.1 人参皂苷Rg3对氧糖剥夺/复糖复氧损伤PC12细胞的保护作用 图1A是人参皂苷Rg3的结构式,使用1-100 nmol/L的Rg3处理受到不同时间氧糖剥夺损伤的PC12细胞,如图1B所示,在不同损伤时间内,Rg3在10-100 nmol/L浓度范围时,都可以不同程度地升高细胞活力。通过测定乳酸脱氢酶的释放来评估PC12细胞受损的程度,如图1C所示,在氧糖剥夺2 h时,Rg3在25-100 nmol/L时均可以明显减少乳酸脱氢酶的释放(P < 0.05)。因为在0.5,1,1.5,2 h时,细胞活力都出现明显下降,分别为81%,75%,55%,51%,为保证模型的稳定性与损伤的效果,后续实验使用氧糖剥夺2 h,复糖复氧16 h进行造模。"
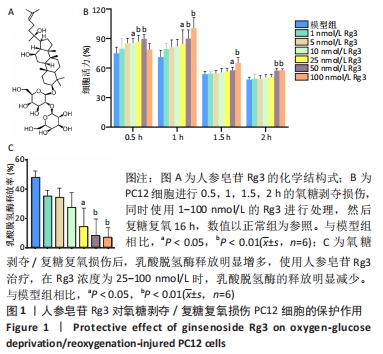
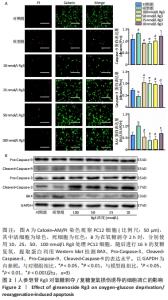
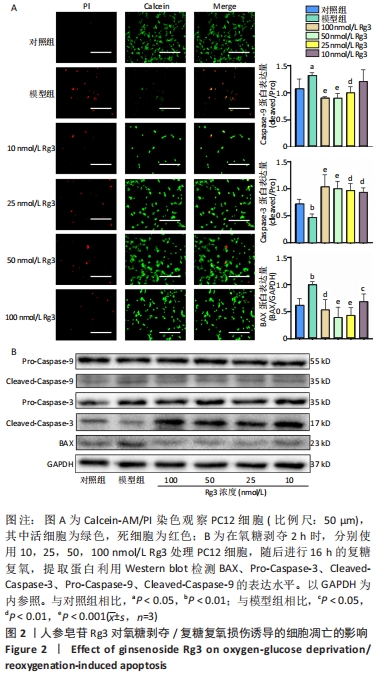
2.2 人参皂苷Rg3对氧糖剥夺/复糖复氧损伤诱导的细胞凋亡的影响 Calcein-AM/PI双染色结果显示Rg3可以减少氧糖剥夺/复糖复氧损伤诱导的细胞凋亡,见图2A。利用Western blot检测凋亡相关蛋白BAX、ProCaspase-3、Pro-Caspase-9、CleavedCaspase-3、CleavedCaspase-9的表达水平,与模型组相比,Rg3在100 nmol/L时可以明显降低BAX蛋白的表达(P < 0.01)。意外的是,与对照组相比,模型组Cleaved-Caspase-9活性升高,Cleaved-Caspase-3活性降低,而Pro-Caspasse-9和Pro-Caspase-3蛋白的活性无明显变化;在使用不同浓度的Rg3处理后,Cleaved-Caspase-9表达降低,Cleaved-Caspase-3表达明显升高,对Pro-Caspasse-9和Pro-Caspase-3的表达无明显影响,见图2B。"
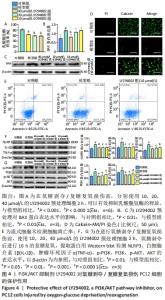
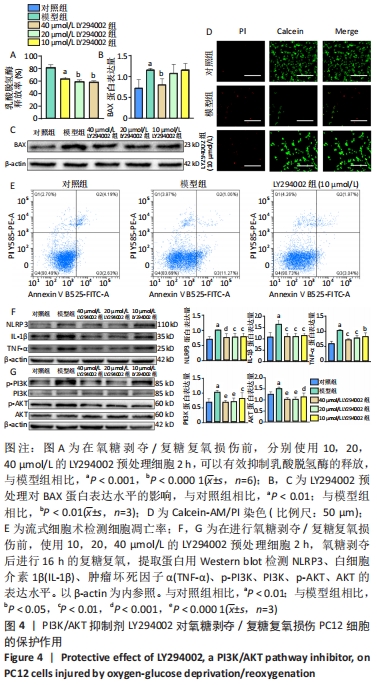
2.4 LY294002对氧糖剥夺/复糖复氧损伤PC12细胞的保护作用 为明确PI3K/AKT信号通路在氧糖剥夺/复糖复氧损伤模型中发挥的作用,使用特异性PI3K/AKT通路抑制剂LY294002进行阳性对照实验。在氧糖剥夺2 h的条件下,单独使用LY294002预处理细胞,结果表明不同浓度的LY294002均能够减少乳酸脱氢酶的释放,见图4A。 Calcein-AM/PI染色和流式结果均表明,LY294002能够抑制氧糖剥夺/复糖复氧损伤介导的细胞凋亡,Western blot结果发现LY294002能够明显降低炎症相关因子NLRP3、白细胞介素1β、肿瘤坏死因子α和凋亡蛋白BAX的表达水平,并且随着LY294002浓度的升高,其对PI3K和AKT的磷酸化抑制程度逐渐增强,见图4B-F。 "
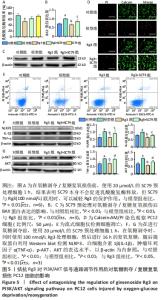
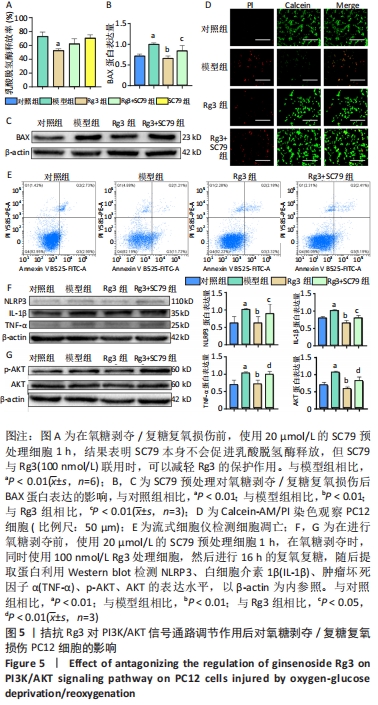
2.5 拮抗Rg3对PI3K/AKT信号通路调节作用后对氧糖剥夺/复糖复氧损伤PC12细胞的影响 为进一步探索Rg3对PI3K/AKT信号通路的调节作用,将Rg3与特异性AKT激活剂SC79联合使用。乳酸脱氢酶释放实验结果显示,与模型组相比,单独使用SC79的乳酸脱氢酶释放率无明显差异,而Rg3和SC79联合使用与单独使用Rg3相比,乳酸脱氢酶释放会增多,但无统计学差异,见图5A。Calcein-AM/PI染色和流式结果均表明,SC79能够促进细胞凋亡,升高细胞凋亡率,Western blot结果表明SC79能恢复炎症相关因子NLRP3、白细胞介素1β、肿瘤坏死因子α和凋亡蛋白BAX的水平,并且能够明显促进AKT的磷酸化,图5B-F。"
| [1] DONNAN GA, FISHER M, MACLEOD M, et al. Stroke. Lancet. 2008; 371(9624):1612-1623. [2] MENON BK, CAMPBELL BC, LEVI C, et al. Role of imaging in current acute ischemic stroke workflow for endovascular therapy. Stroke. 2015; 46(6):1453-1461. [3] ZHANG H, LIU X, YANG F, et al. Overexpression of HIF-1α protects PC12 cells against OGD/R-evoked injury by reducing miR-134 expression. Cell Cycle. 2020;19(9):990-999. [4] LI S, SUN X, XU L, et al. Baicalin attenuates in vivo and in vitro hyperglycemia-exacerbated ischemia/reperfusion injury by regulating mitochondrial function in a manner dependent on AMPK. Eur J Pharmacol. 2017;815:118-126. [5] REVATHIDEVI S, MUNIRAJAN AK. Akt in cancer: Mediator and more. Semin Cancer Biol. 2019;59:80-91. [6] GABBOUJ S, RYHÄNEN S, MARTTINEN M, et al. Altered Insulin Signaling in Alzheimer’s Disease Brain - Special Emphasis on PI3K-Akt Pathway. Front Neurosci. 2019;13:629. [7] KWON O, KIM KW, KIM MS. Leptin signalling pathways in hypothalamic neurons. Cell Mol Life Sci. 2016;73(7):1457-1477. [8] ZHANG Z, YAO L, YANG J, et al. PI3K/Akt and HIF‑1 signaling pathway in hypoxia‑ischemia (Review). Mol Med Rep. 2018;18(4):3547-3554. [9] 蔡苏娜,李强,周慧,等.白杨素通过抑制PI3K/AKT/mTOR信号通路发挥抗炎和抗氧化作用:基于蛋白质芯片方法[J].南方医科大学学报,2021,41(10):1554-1561. [10] LI C, ZHAO B, LIN C, et al. TREM2 inhibits inflammatory responses in mouse microglia by suppressing the PI3K/NF-κB signaling. Cell Biol Int. 2019;43(4):360-372. [11] WONG AS, CHE CM, LEUNG KW. Recent advances in ginseng as cancer therapeutics: a functional and mechanistic overview. Nat Prod Rep. 2015;32(2):256-272. [12] WU W, SUN L, ZHANG Z, et al. Profiling and multivariate statistical analysis of Panax ginseng based on ultra-high-performance liquid chromatography coupled with quadrupole-time-of-flight mass spectrometry. J Pharm Biomed Anal. 2015;107:141-150. [13] ATTELE AS, WU JA, YUAN CS. Ginseng pharmacology: multiple constituents and multiple actions. Biochem Pharmacol. 1999;58(11): 1685-1693. [14] HE B, CHEN P, YANG J, et al. Neuroprotective effect of 20(R)-ginsenoside Rg(3) against transient focal cerebral ischemia in rats. Neurosci Lett. 2012;526(2):106-111. [15] LIU H, LU X, HU Y, et al. Chemical constituents of Panax ginseng and Panax notoginseng explain why they differ in therapeutic efficacy. Pharmacol Res. 2020;161:105263. [16] LEE H, KONG G, TRAN Q, et al. Relationship Between Ginsenoside Rg3 and Metabolic Syndrome. Front Pharmacol. 2020;11:130. [17] CHENG Z, ZHANG M, LING C, et al. Neuroprotective Effects of Ginsenosides against Cerebral Ischemia. Molecules. 2019;24(6): 1102. [18] BROUGHTON BR, REUTENS DC, SOBEY CG. Apoptotic mechanisms after cerebral ischemia. Stroke. 2009;40(5):e331-e339. [19] CAMPANELLA M, SCIORATI C, TAROZZO G, et al. Flow cytometric analysis of inflammatory cells in ischemic rat brain. Stroke. 2002;33(2): 586-592. [20] JIN R, LIU L, ZHANG S, et al. Role of inflammation and its mediators in acute ischemic stroke. J Cardiovasc Transl Res. 2013;6(5):834-851. [21] GONG Z, PAN J, SHEN Q, et al. Mitochondrial dysfunction induces NLRP3 inflammasome activation during cerebral ischemia/reperfusion injury. J Neuroinflammation. 2018;15(1):242. [22] WANG G, WANG T, ZHANG Y, et al. Schizandrin Protects against OGD/R-Induced Neuronal Injury by Suppressing Autophagy: Involvement of the AMPK/mTOR Pathway. Molecules. 2019;24(19):3624. [23] JIA Y, CUI R, WANG C, et al. Metformin protects against intestinal ischemia-reperfusion injury and cell pyroptosis via TXNIP-NLRP3-GSDMD pathway. Redox Biol. 2020;32:101534. [24] KIM S, KIM T, AHN K, et al. Ginsenoside Rg3 antagonizes NMDA receptors through a glycine modulatory site in rat cultured hippocampal neurons. Biochem Biophys Res Commun. 2004;323(2):416-424. [25] SUN ZG, CHEN LP, WANG FW, et al. Protective effects of ginsenoside Rg1 against hydrogen peroxide-induced injury in human neuroblastoma cells. Neural Regen Res. 2016;11(7):1159-1164. [26] FAN C, SONG Q, WANG P, et al. Neuroprotective Effects of Ginsenoside-Rg1 Against Depression-Like Behaviors via Suppressing Glial Activation, Synaptic Deficits, and Neuronal Apoptosis in Rats. Front Immunol. 2018;9:2889. [27] CHO M, CHOI G, SHIM I, et al. Enhanced Rg3 negatively regulates Th1 cell responses. J Ginseng Res. 2019;43(1):49-57. [28] SUN M, YE Y, XIAO L, et al. Anticancer effects of ginsenoside Rg3 (Review). Int J Mol Med. 2017;39(3):507-518. [29] QIN XY, WANG YN, LIU HF, et al. Anti-cancer activities of metal-based complexes by regulating the VEGF/VEGFR2 signaling pathway and apoptosis-related factors Bcl-2, Bax, and caspase-9 to inhibit angiogenesis and induce apoptosis. Metallomics. 2020;12(1):92-103. [30] BUDIHARDJO I, OLIVER H, LUTTER M, et al. Biochemical pathways of caspase activation during apoptosis. Annu Rev Cell Dev Biol. 1999; 15:269-290. [31] BRENTNALL M, RODRIGUEZ-MENOCAL L, DE GUEVARA RL, et al. Caspase-9, caspase-3 and caspase-7 have distinct roles during intrinsic apoptosis. BMC Cell Biol. 2013;14:32. [32] YUAN J, SHAHAM S, LEDOUX S, et al. The C. elegans cell death gene ced-3 encodes a protein similar to mammalian interleukin-1 beta-converting enzyme. Cell. 1993;75(4):641-652. [33] PORTER AG, JÄNICKE RU. Emerging roles of caspase-3 in apoptosis. Cell Death Differ. 1999;6(2):99-104. [34] LOSSI L, CASTAGNA C, MERIGHI A. Caspase-3 Mediated Cell Death in the Normal Development of the Mammalian Cerebellum. Int J Mol Sci. 2018;19(12):3999. [35] POMPURA SL, DOMINGUEZ-VILLAR M. The PI3K/AKT signaling pathway in regulatory T-cell development, stability, and function. J Leukoc Biol. 2018;103:1065-1076. [36] XU F, NA L, LI Y, et al. Roles of the PI3K/AKT/mTOR signalling pathways in neurodegenerative diseases and tumours. Cell Biosci. 2020;10(1):54. [37] ZHANG J, ZHANG JX, ZHANG QL. PI3K/AKT/mTOR-mediated autophagy in the development of autism spectrum disorder. Brain Res Bull. 2016; 125:152-158. [38] MANTHARI RK, TIKKA C, OMMATI MM, et al. Arsenic induces autophagy in developmental mouse cerebral cortex and hippocampus by inhibiting PI3K/Akt/mTOR signaling pathway: involvement of blood-brain barrier’s tight junction proteins. Arch Toxicol. 2018;92(11):3255-3275. [39] GARABADU D, VERMA J. Exendin-4 attenuates brain mitochondrial toxicity through PI3K/Akt-dependent pathway in amyloid beta (1-42)-induced cognitive deficit rats. Neurochem Int. 2019;128:39-49. [40] KITAGAWA H, WARITA H, SASAKI C, et al. Immunoreactive Akt, PI3-K and ERK protein kinase expression in ischemic rat brain. Neurosci Lett. 1999;274(1):45-48. [41] ZHAO M, HOU S, FENG L, et al. Vinpocetine Protects Against Cerebral Ischemia-Reperfusion Injury by Targeting Astrocytic Connexin43 via the PI3K/AKT Signaling Pathway. Front Neurosci. 2020;14:223. [42] JIN W, XU W, ZHANG X, et al. Ischemic Preconditioning Upregulates Decoy Receptors to Protect SH-SY5Y Cells from OGD Induced Cellular Damage by Inhibiting TRAIL Pathway and Agitating PI3K/Akt Pathway. Mol Neurobiol. 2020;57(9):3658-3670. [43] WANG HL, LIU FL, LI RQ, et al. Electroacupuncture improves learning and memory functions in a rat cerebral ischemia/reperfusion injury model through PI3K/Akt signaling pathway activation. Neural Regen Res. 2021;16(6):1011-1016. [44] CARPENTER CL, CANTLEY LC. Phosphoinositide kinases. Curr Opin Cell Biol. 1996;8(2):153-158. [45] MOREIRA JB, WOHLWEND M, ALVES MN, et al. A small molecule activator of AKT does not reduce ischemic injury of the rat heart. J Transl Med. 2015;13:76. |
| [1] | Wu Yongli, Liu Di, Wang Duo, Liu Junwei, Ma Yuyuan. Effect of warm acupuncture on PI3K/Akt signaling pathway in articular cartilage of a rabbit knee osteoarthritis model [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(35): 5596-5601. |
| [2] | Sun Haitao, Huang Yonghui, Gong Aihua, Cao Xingbing, Li Zhen. Protective effect of lipopolysaccharide-preconditioned astrocyte extracellular vesicles on neurons [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(13): 1985-1992. |
| [3] | Cheng Ning, Zhang Zhizhong. Regulatory effects of LncRNA MEG3 on the proliferation and apoptosis of nucleus pulposus cells in rat intervertebral disc [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(26): 4106-4111. |
| [4] | Yu Hui, Zhao Yang, Fei Jiayue, Zhang Wenli, Zhao Luosha. Isoliquiritigenin inhibits SETD7 expression against oxidative damage in cardiomyocytes induced by hypoxia/reoxygenation [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(35): 5613-5618. |
| [5] | Shi Dongmei, Dong Ming, Lu Ying, Niu Weidong. PI3K/Akt signaling pathway and bone destruction: problems and mechanisms [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(23): 3716-3722. |
| [6] | Lü Cong, Sun Lin, Feng Haoyu, Ma Xun, He Yajun, Li Jisheng. Inhibition of high mobility group box 1/nuclear factor-kappa B pathway reduces apoptosis in spinal cord astrocytes after oxygen-glucose deprivation/reoxygenation [J]. Chinese Journal of Tissue Engineering Research, 2019, 23(33): 5353-5359. |
| [7] | Dang Hui1, Zhang Shuxiang1, Guan Xutao1, Wan Jiangwei1, Shi Lin2. Effects of Chinese medicine for detoxifying and removing blood stasis in model mice with acute lymphocytic leukemia [J]. Chinese Journal of Tissue Engineering Research, 2019, 23(27): 4392-4396. |
| [8] | Zhang Yong, Ma Xun, Sun Lin, Zhang Li, Guan Xiaoming, Lü Cong, Chen Xu. Exosomes derived from human umbilical cord mesenchymal stem cells attenuate edema of spinal astrocytes after oxygen-glucose deprivation/reoxygenation injury in rats [J]. Chinese Journal of Tissue Engineering Research, 2019, 23(25): 4011-4017. |
| [9] | Gao Junyan, Cao Yanfei, Zhang Yun, Liu Xuemin, Hou Yanhong, Li Jianbin, Wu Zhibing. Timosaponin B-II reduces necroptosis of VSC4.1 cells induced by oxygen-glucose deprivation [J]. Chinese Journal of Tissue Engineering Research, 2019, 23(25): 4062-4067. |
| [10] | Li Chenchen1, 2, Liu Baimei1, 2, Liu Yang1, 2, Lü Ying1, 2, Cui Pudong1, 2, An Meiwen1, 2 . Trend similarity of PI3K/AKT and MEK/ERK signaling pathways related protein expression in the hypertrophic scar of Bama pigs and human [J]. Chinese Journal of Tissue Engineering Research, 2019, 23(23): 3673-3679. |
| [11] | Shi Jiang, Gao Shilun, Liu Jinduo, Gu Tianxiang, Shi Enyi. Bone marrow mesenchymal stem cell exosomes alleviate oxygen-glucose deprivation/reperfusion injury in hippocampal neurons [J]. Chinese Journal of Tissue Engineering Research, 2019, 23(21): 3316-3322. |
| [12] | Zhang Xu1, Chen Wangsheng2, Feng Jian3, Zhang Mengjiao1, Jiang Wenjie1, Liang Xuemei1. Protective effect of Ginkgolide B against hypoxia/reoxygenation-induced cardiomyocyte injury and its underlying mechanisms [J]. Chinese Journal of Tissue Engineering Research, 2019, 23(19): 3097-3101. |
| [13] | Qiu Shi1, Li Jun-guo1, Qiu Qian2, Chen Hui1, Xiang Zi-min3. Caffeic acid phenethyl ester against cellular injuries in the rotenone-induced Parkinson’s disease model [J]. Chinese Journal of Tissue Engineering Research, 2016, 20(40): 5979-5985. |
| [14] | Guan Li-na, Ji Yong-qiang, Ba Mao-wen, Lian Pei-wen, Li Ning, Mu Xian-yu, Yu Guo-ping. Mechanism of pituitary adenylate cyclase-activating polypeptide against the cytotoxicity of lactacystin [J]. Chinese Journal of Tissue Engineering Research, 2015, 19(46): 7461-7465. |
| [15] | Li Xiao, Zhang Xin-sheng, Luo Hai-jie, Luan Shuo, Wan Qing, Wu Shao-ling, Ma Chao . Growth and secretion features of microencapsulated human adrenal pheochromocytoma cells in artificial cerebrospinal fluid [J]. Chinese Journal of Tissue Engineering Research, 2015, 19(38): 6114-6120. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||