Chinese Journal of Tissue Engineering Research ›› 2022, Vol. 26 ›› Issue (13): 1985-1992.doi: 10.12307/2022.322
Previous Articles Next Articles
Protective effect of lipopolysaccharide-preconditioned astrocyte extracellular vesicles on neurons
Sun Haitao1, Huang Yonghui1, Gong Aihua2, Cao Xingbing1, Li Zhen1
- 1Affiliated Hospital of Jiangsu University, Zhenjiang 212000, Jiangsu Province, China; 2Jiangsu University, Zhenjiang 212000, Jiangsu Province, China
-
Received:2020-10-16Revised:2020-10-20Accepted:2020-11-28Online:2022-05-08Published:2021-12-18 -
Contact:Huang Yonghui, Master, Chief physician, Affiliated Hospital of Jiangsu University, Zhenjiang 212000, Jiangsu Province, China -
About author:Sun Haitao, Master candidate, Affiliated Hospital of Jiangsu University, Zhenjiang 212000, Jiangsu Province, China -
Supported by:the Scientific Research Project of Zhenjiang City, No. SH2020053 (to HYH)
CLC Number:
Cite this article
Sun Haitao, Huang Yonghui, Gong Aihua, Cao Xingbing, Li Zhen. Protective effect of lipopolysaccharide-preconditioned astrocyte extracellular vesicles on neurons[J]. Chinese Journal of Tissue Engineering Research, 2022, 26(13): 1985-1992.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
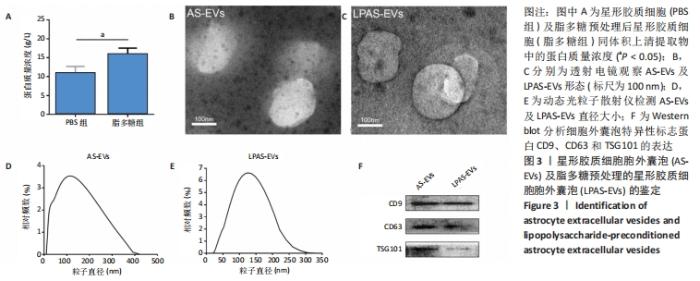
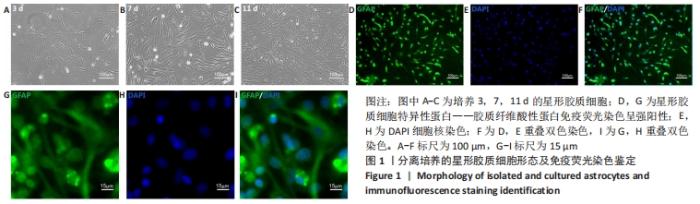
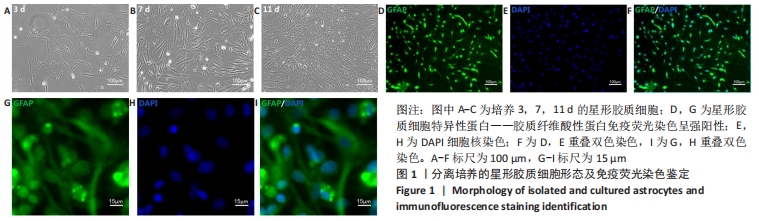
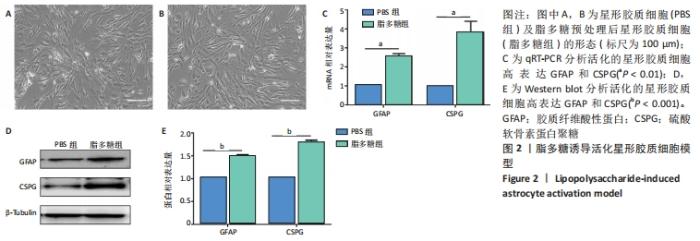
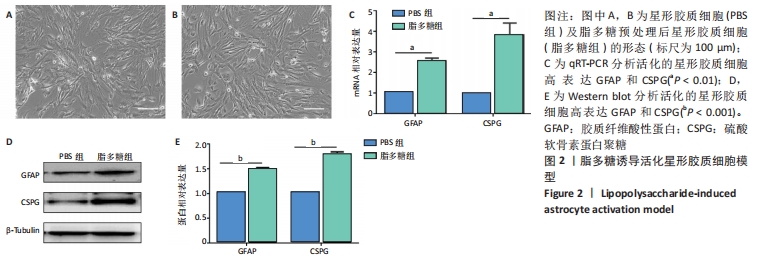
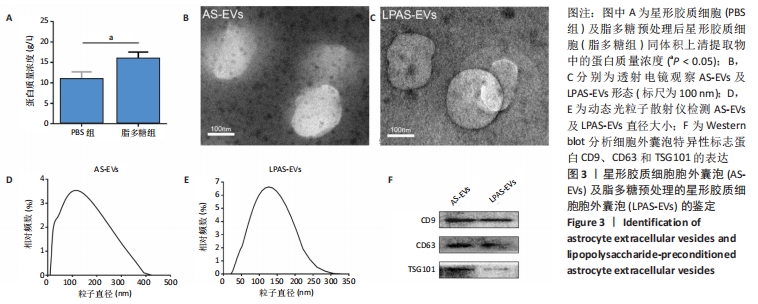
2.3 AS-EVs及LPAS-EVs的鉴定 BCA蛋白检测结果显示:PBS组和脂多糖组同体积上清提取物中,脂多糖组蛋白浓度高于PBS组,侧面反映脂多糖处理活化后的星形胶质细胞所释放的细胞外囊泡较未被活化的增多(P < 0.05,n=3),见图3A。透射电镜显示AS-EVs和LPAS-EVs形态近似圆形或椭圆形杯状,直径20-200 nm,见图3B,C。动态光粒子散射仪显示AS-EVs直径10-400 nm,平均(115±4.55) nm(n=3),LPAS-EVs直径20-300 nm,平均 (127.67±3.09) nm(n=3),见图3D,E,LPAS-EVs直径略大于AS-EVs。透射电镜及动态光粒子散射仪结果显示所提取的2种星形胶质细胞胞外囊泡均符合细胞外囊泡形态学标准,且二者形态大小存在差异,提示二者在功能上存在差异。Western blot检测结果显示,细胞外囊泡标记蛋白CD9、CD63和Tsg101阳性,见图3F。"
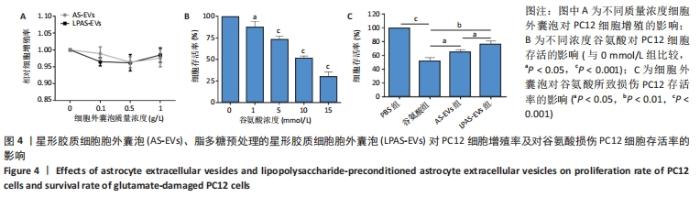
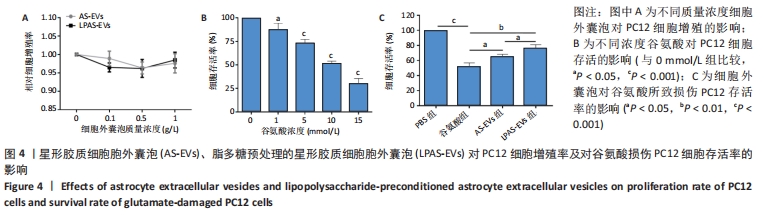
2.4 AS-EVs及LPAS-EVs对PC12细胞增殖和对谷氨酸损伤PC12细胞存活率的影响 CCK-8分析不同浓度AS-EVs和LPAS-EVs对PC12细胞增殖的影响,结果显示,PC12细胞并不随着AS-EVs和LPAS-EVs浓度增加而出现明显的细胞增殖抑制或促进现象,即AS-EVs和LPAS-EVs对PC12细胞增殖没有明显的影响,见图4A。在谷氨酸损伤PC12细胞模型中,CCK-8结果显示,与0 mmol/L相比,随着谷氨酸浓度的增加,PC12细胞存活率降低,呈一定浓度依赖性。在谷氨酸浓度达到10 mmol/L时,PC12细胞存活率(51.28±2.21)%,显著低于0 mmol/L组。因此,选用10 mmol/L谷氨酸进行后续实验,见图4B,后续实验结果显示,与PBS组相比,谷氨酸组PC12细胞存活率明显降低(P < 0.001,n=3),而AS-EVs组和LPAS-EVs组细胞存活率高于谷氨酸组(P均< 0.05,n=3),且LPAS-EVs组高于AS-EVs组(P < 0.05,n=3),见图4C。这些结果表明AS-EVs和LPAS-EVs对PC12细胞存活率的影响是通过改善细胞活性而非促进增殖来实现,且LPAS-EVs的作用强于AS-EVs。"
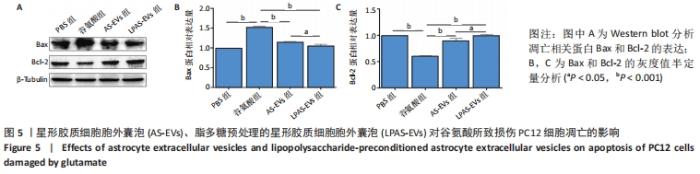
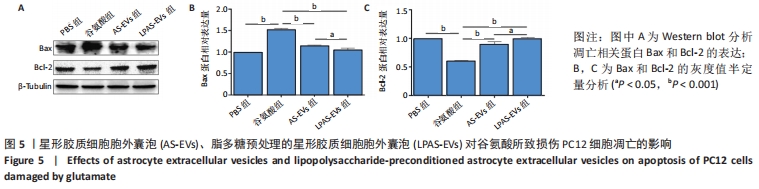
2.5 AS-EVs及LPAS-EVs对谷氨酸损伤PC12细胞凋亡的影响 Western blot检测结果显示,与PBS组相比,谷氨酸组Bax蛋白表达水平明显升高,而Bcl-2表达水平降低(P均 < 0.001,n=3);与谷氨酸组相比,AS-EVs组和LPAS-EVs组Bax 蛋白表达水平降低(P均 < 0.001,n=3),LPAS-EVs组略低于AS-EVs组(P < 0.05,n=3),而Bcl-2表达水平升高(P均 < 0.001,n=3),LPAS-EVs组略高于AS-EVs组(P < 0.05,n=3),见图5。结果提示AS-EVs和LPAS-EVs能够增强PC12细胞的抗凋亡作用,且LPAS-EVs的作用强于AS-EVs。"

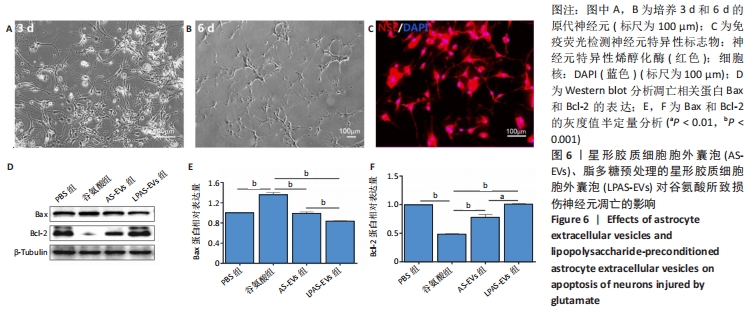
2.6 AS-EVs及LPAS-EVs对谷氨酸所致损伤神经元凋亡的影响 成功提取神经元并进行纯化和鉴定,见图6A-C,利用10 mmol/L谷氨酸制作神经元损伤模型并分组。Western blot检测凋亡相关蛋白Bax和Bcl-2的表达,得到了与“2.5”相似的结果,见图6D-F,与PBS组相比,谷氨酸组Bax蛋白表达水平明显升高,而Bcl-2表达水平降低(P均 < 0.001,n=3);与谷氨酸组相比,AS-EVs组和LPAS-EVs组Bax蛋白表达水平降低,LPAS-EVs组略低于AS-EVs组(P均< 0.001,n=3),而Bcl-2表达水平升高,LPAS-EVs组略高于AS-EVs组(P均 < 0.001,n=3)。结果提示AS-EVs和LPAS-EVs能够增强神经元的抗凋亡能力,且LPAS-EVs的作用强于AS-EVs。"
| [1] GBD 2016 HEADACHE COLLABORATORS. Global, regional, and national burden of migraine and tension-type headache, 1990-2016: a systematic analysis for the Global Burden of Disease Study 2016. Lancet Neurol. 2018;17(11):954-976. [2] MARKIEWICZ I, LUKOMSKA B. The role of astrocytes in the physiology and pathology of the central nervous system. Acta Neurobiol Exp (Wars). 2006;66(4):343-358. [3] ORR MB, GENSEL JC. Spinal Cord Injury Scarring and Inflammation: Therapies Targeting Glial and Inflammatory Responses. Neurotherapeutics. 2018;15(3):541-553. [4] LEE YJ, HAN SB, NAM SY, et al. Inflammation and Alzheimer’s disease. Arch Pharm Res. 2010;33(10):1539-1556. [5] FRICKER M, TOLKOVSKY AM, BORUTAITE V, et al. Neuronal Cell Death. Physiol Rev. 2018;98(2):813-880. [6] SOFRONIEW MV, VINTERS HV. Astrocytes: biology and pathology. Acta Neuropathol. 2010;119(1):7-35. [7] CLARKE LE, BARRES BA. Emerging roles of astrocytes in neural circuit development. Nat Rev Neurosci. 2013;14(5):311-321. [8] BERGERSEN LH. Lactate in the brain--without turning sour. Tidsskr Nor Laegeforen. 2006;126(16):2094-2097. [9] ZIL’BERTER IUI, ZIL’BERTER TM. Power metabolism from neurons and a glia to the whole brain: norm, pathology and correction. Usp Fiziol Nauk. 2012;43(2):37-54. [10] BAK LK, SCHOUSBOE A, WAAGEPETERSEN HS. The glutamate/GABA-glutamine cycle: aspects of transport, neurotransmitter homeostasis and ammonia transfer. J Neurochem. 2006;98(3):641-653. [11] SONNEWALD U, SCHOUSBOE A. Introduction to the Glutamate-Glutamine Cycle. Adv Neurobiol. 2016;13:1-7. [12] MICHINAGA S, KOYAMA Y. Dual Roles of Astrocyte-Derived Factors in Regulation of Blood-Brain Barrier Function after Brain Damage. Int J Mol Sci. 2019;20(3):571. [13] LI-NA Z, DENG C, DA X, et al. Mesencephalic astrocyte-derived neurotrophic factor and its role in nervous system disease. Neurol Sci. 2017;38(10):1741-1746. [14] DURKEE CA, ARAQUE A. Diversity and Specificity of Astrocyte-neuron Communication. Neuroscience. 2019;396:73-78. [15] SOFRONIEW MV. Molecular dissection of reactive astrogliosis and glial scar formation. Trends Neurosci. 2009;32(12):638-647. [16] CHUN H, LEE CJ. Reactive astrocytes in Alzheimer’s disease: A double-edged sword. Neurosci Res. 2018;126:44-52. [17] HAMBY ME, SOFRONIEW MV. Reactive astrocytes as therapeutic targets for CNS disorders. Neurotherapeutics. 2010;7(4):494-506. [18] PEKNY M, PEKNA M, MESSING A, et al. Astrocytes: a central element in neurological diseases. Acta Neuropathol. 2016;131(3):323-345. [19] WANG K, YE L, LU H, et al. TNF-α promotes extracellular vesicle release in mouse astrocytes through glutaminase. J Neuroinflammation. 2017; 14(1):87. [20] VAN NIEL G, D’ANGELO G, RAPOSO G. Shedding light on the cell biology of extracellular vesicles. Nat Rev Mol Cell Biol. 2018;19(4):213-228. [21] 高振橙,刘欣.间充质干细胞外泌体在神经系统疾病修复过程中的作用与应用[J].中国组织工程研究,2020,24(19):3048-3054. [22] SUN G, LI G, LI D, et al. hucMSC derived exosomes promote functional recovery in spinal cord injury mice via attenuating inflammation. Mater Sci Eng C Mater Biol Appl. 2018;89:194-204. [23] MATHIEU M, MARTIN-JAULAR L, LAVIEU G, et al. Specificities of secretion and uptake of exosomes and other extracellular vesicles for cell-to-cell communication. Nat Cell Biol. 2019;21(1):9-17. [24] LIU W, RONG Y, WANG J, et al. Exosome-shuttled miR-216a-5p from hypoxic preconditioned mesenchymal stem cells repair traumatic spinal cord injury by shifting microglial M1/M2 polarization. J Neuroinflammation. 2020;17(1):47. [25] LUO Z, WANG H, FANG S, et al. Annexin-1 Mimetic Peptide Ac2-26 Suppresses Inflammatory Mediators in LPS-Induced Astrocytes and Ameliorates Pain Hypersensitivity in a Rat Model of Inflammatory Pain. Cell Mol Neurobiol. 2020;40(4):569-585. [26] KARABABA A, GROOS-SAHR K, ALBRECHT U, et al. Ammonia Attenuates LPS-Induced Upregulation of Pro-Inflammatory Cytokine mRNA in Co-Cultured Astrocytes and Microglia. Neurochem Res. 2017;42(3):737-749. [27] CHEN A, WANG H, ZHANG Y, et al. Paeoniflorin exerts neuroprotective effects against glutamate‑induced PC12 cellular cytotoxicity by inhibiting apoptosis. Int J Mol Med. 2017;40(3):825-833. [28] CHANG CH, CHEN HX, YÜ G, et al. Curcumin-Protected PC12 Cells Against Glutamate-Induced Oxidative Toxicity. Food Technol Biotechnol. 2014;52(4):468-478. [29] RYU KY, LEE HJ, WOO H, et al. Dasatinib regulates LPS-induced microglial and astrocytic neuroinflammatory responses by inhibiting AKT/STAT3 signaling. J Neuroinflammation. 2019;16(1):190. [30] 张冬梅, 陶涛, 夏小鹏,等.Cyclin D3,CDK11p58在脂多糖诱导大鼠星形胶质细胞活化过程中的表达及相互作用[J]. 南通大学学报(医学版), 2013, 33(4):237-241. [31] 郭生龙, 朱洁, 谢瑱,等.Che-1通过mTOR通路调控自噬在谷氨酸所致神经元损伤中的作用研究[J].现代生物医学进展,2019,19(18): 66-70,145. [32] ZHU S, CHEN M, CHEN M, et al. Fibroblast Growth Factor 22 Inhibits ER Stress-Induced Apoptosis and Improves Recovery of Spinal Cord Injury. Front Pharmacol. 2020;11:18. [33] CHEN Y, QIN C, HUANG J, et al. The role of astrocytes in oxidative stress of central nervous system: A mixed blessing. Cell Prolif. 2020;53(3): e12781. [34] SONG HH, SONG TC, YANG T, et al. High mobility group box 1 mediates inflammatory response of astrocytes via cyclooxygenase 2/prostaglandin E2 signaling following spinal cord injury. Neural Regen Res. 2021;16(9):1848-1855. [35] VERKHRATSKY A, HO MS, VARDJAN N, et al. General Pathophysiology of Astroglia. Adv Exp Med Biol. 2019;1175:149-179. [36] 李剑锋, 闫金玉, 夏润福,等.脊髓损伤胶质瘢痕形成及星形胶质细胞作用的研究与转化意义[J].中国组织工程研究,2016,20(37): 5609-5616. [37] IBÁÑEZ F, MONTESINOS J, UREÑA-PERALTA JR, et al. TLR4 participates in the transmission of ethanol-induced neuroinflammation via astrocyte-derived extracellular vesicles. J Neuroinflammation. 2019;16(1):136. [38] TARASSISHIN L, SUH HS, LEE SC. LPS and IL-1 differentially activate mouse and human astrocytes: role of CD14. Glia. 2014;62(6):999-1013. [39] OLTVAI ZN, MILLIMAN CL, KORSMEYER SJ. Bcl-2 heterodimerizes in vivo with a conserved homolog, Bax, that accelerates programmed cell death. Cell. 1993;74(4):609-619. [40] HUANG JL, QU Y, TANG J, et al. Protective effect of astrocyte exosomes on hypoxic-ischemic neurons. Zhongguo Dang Dai Er Ke Za Zhi. 2018; 20(5):397-402. [41] PEI X, LI Y, ZHU L, et al. Astrocyte-derived exosomes suppress autophagy and ameliorate neuronal damage in experimental ischemic stroke. Exp Cell Res. 2019;382(2):111474. [42] KARTHIKEYAN A, PATNALA R, JADHAV SP, et al. MicroRNAs: Key Players in Microglia and Astrocyte Mediated Inflammation in CNS Pathologies. Curr Med Chem. 2016;23(30):3528-3546. [43] FANG Z, ZHAO J, XIE W, et al. LncRNA UCA1 promotes proliferation and cisplatin resistance of oral squamous cell carcinoma by sunppressing miR-184 expression. Cancer Med. 2017;6(12):2897-2908. [44] DU J, YANG ST, LIU J, et al. Silence of LncRNA GAS5 Protects Cardiomyocytes H9c2 against Hypoxic Injury via Sponging miR-142-5p. Mol Cells. 2019;42(5):397-405. |
| [1] | Wen Dandan, Li Qiang, Shen Caiqi, Ji Zhe, Jin Peisheng. Nocardia rubra cell wall skeleton for extemal use improves the viability of adipogenic mesenchymal stem cells and promotes diabetes wound repair [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1038-1044. |
| [2] | Zhang Yujie, Yang Jiandong, Cai Jun, Zhu Shoulei, Tian Yuan. Mechanism by which allicin inhibits proliferation and promotes apoptosis of rat vascular endothelial cells [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1080-1084. |
| [3] | Deng Shuang, Pu Rui, Chen Ziyang, Zhang Jianchao, Yuan Lingyan . Effects of exercise preconditioning on myocardial protection and apoptosis in a mouse model of myocardial remodeling due to early stress overload [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(5): 717-723. |
| [4] | Li Anan, Jiang Tao, Zhan Min, Cai Yuning, Song Min, Li Congcong, Lin Wenzheng, Zhang Jiayuan, Liu Wengang. Pharmacological mechanism of Shenling Baizhu San in the treatment of knee osteoarthritis based on network pharmacology and molecular docking [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(2): 197-204. |
| [5] | Wei Wenyue, Wang Yuyin, Guo Minfang, Zhang Jing, Gu Qingfang, Song Lijuan, Chai Zhi, Yu Jiezhong, Ma Cungen. Fasudil inhibits neuronal apoptosis via regulating mitochondrial dynamics in APP/PS1 mice [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(2): 232-238. |
| [6] | Huang Mei, Wang Feiqing, Liang Huiling, Yang Xu, Zhao Jianing, Wang Kun, Liu Yanqing, Zhou Yuan, Wang Jishi, Li Yanju, Liu Yang. Effect of bone marrow mesenchymal stem cell conditioned medium on the proliferation of multiple myeloma cells [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(19): 3024-3029. |
| [7] | Qiu Hanke, Zhang Fei, Peng Wuxun. Effect and mechanism of mesenchymal stem cell-related long non-coding RNA on cell proliferation and apoptosis [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(19): 3078-3083. |
| [8] | Xu Xuezhen, Song Lixian, Li Aiqun, Yang Jian, Li Xiaokun, Wang Zhanqing, Shi Qingpeng. Bone morphogenetic protein 7 inhibits apoptosis of nucleus pulposus cells [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(17): 2726-2731. |
| [9] | Li Min, Yu Yang, Cheng Jiyan. Mechanism of paeoniflorin on bone marrow mesenchymal stem cells intervened by lipopolysaccharide [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(13): 2000-2005. |
| [10] | Shan Wen, Shi Wei. Expression and function of colony-stimulating factor 1 in differentiation of neural stem cells induced by activated astrocyte conditioned medium [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(13): 2069-2074. |
| [11] | Jiang Shengyuan, Deng Bowen, Xu Lin, Liu Gang, He Feng, Zhao Yi, Ren Jingpei, Mu Xiaohong. Role and mechanism of tetramethylpyrazine in spinal cord injury repair [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(11): 1799-1804. |
| [12] | Yu Dong, Liu Kan, Shi Zongting, Yang Xiaoxia, Liu Hengping, Zhang Qingfeng. Pathological changes of the cervical intervertebral discs and rules of migration and apoptosis in endplate chondrocytes in a rabbit model of dynamic disequilibrium [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(11): 1675-1679. |
| [13] | Xing Hongchang, Cao Jianping, Zhu Jing, Yao Kun. Mechanism by which enalapril alleviates myocardial injury in a rat model of limb ischemia-reperfusion [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(11): 1747-1751. |
| [14] | Cai Shengsheng, Mei Heng, Zhang Xuequan, Deng Jin, Cao Jun, He Bin. Prepared HPe6DF composite nanoparticles enhance the effect of photodynamic therapy [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(10): 1566-1573. |
| [15] | Meng Defeng, Li Changzai, Wu Chuntao. Effects of LINC02532 targeting miR-145 on proliferation, migration, invasion and apoptosis of pancreatic cancer stem cells [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(1): 52-58. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||