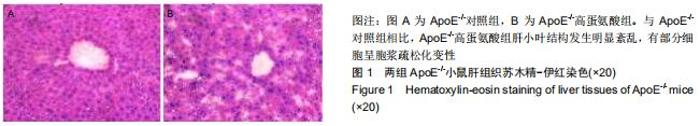
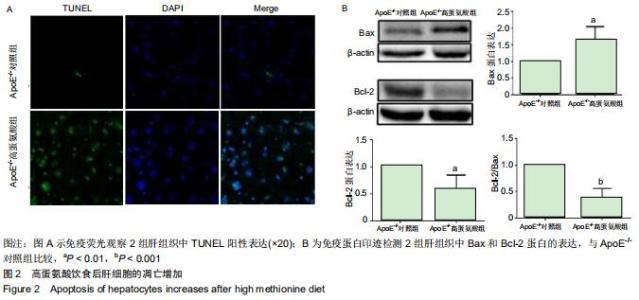
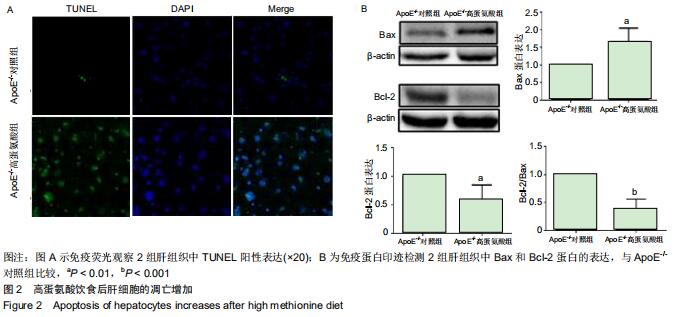
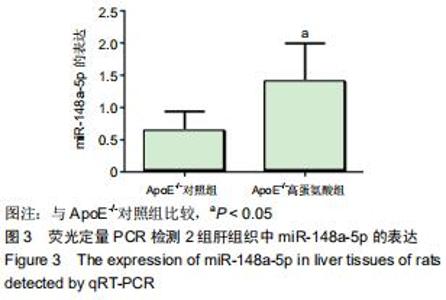
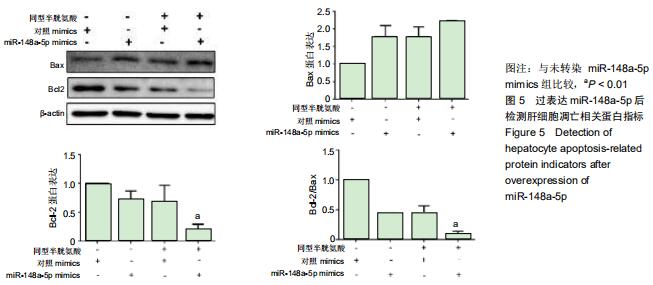
|
[1] ZHANG D, LOU J, ZHANG X, et al. Hyperhomocysteinemia results from and promotes hepatocellular carcinoma via CYP450 metabolism by CYP2J2 DNA methylation. Oncotarget. 2017;8(9):15377-15392.
[2] AI Y, SUN Z, PENG C, et al. Homocysteine Induces Hepatic Steatosis Involving ER Stress Response in High Methionine Diet-Fed Mice. Nutrients. 2017;9(4):346.
[3] KUMAR A, PALFREY HA, PATHAK R, et al. The metabolism and significance of homocysteine in nutrition and health. Nutr Metab (Lond) 2017;14 78.
[4] YANG A, JIAO Y, YANG S, et al. Homocysteine activates autophagy by inhibition of CFTR expression via interaction between DNA methylation and H3K27me3 in mouse liver. Cell Death Dis. 2018;9(2):169.
[5] 朱姗姗,何太平,邹堂斌.miRNAs与非酒精性脂肪肝相关信号通路[J].实用医学杂志,2016,32(23):3820-3822.
[6] GEBERT LFR, MACRAE IJ. Regulation of microRNA function in animals. Nat Rev Mol Cell Biol. 2019;20(1):21-37.
[7] CHEN Z, MA T, HUANG C, et al. MicroRNA-148a: a potential therapeutic target for cancer. Gene. 2014;533:456-457.
[8] KALANI A, CHATURVEDI P, KALANI K, et al. A high methionine, low folate and vitamin B6/B12 containing diet can be associated with memory loss by epigenetic silencing of netrin-1. Neural Regen Res. 2019;14(7):1247-1254.
[9] PERŁA-KAJÁN J, JAKUBOWSKI H. Dysregulation of epigenetic mechanisms of gene expression in the pathologies of hyperhomocysteinemia. Int J Mol Sci. 2019;20(13). pii: E3140. doi: 10.3390/ijms20133140.
[10] FU Y, WANG X, KONG W. Hyperhomocysteinaemia and vascular injury: advances in mechanisms and drug targets. Br J Pharmacol. 2018;175:1173-1189.
[11] WU X, ZHANG L, MIAO Y, et al. Homocysteine causes vascular endothelial dysfunction by disrupting endoplasmic reticulum redox homeostasis. Redox Biol. 2019;20:46-59.
[12] KAR S, SHAHSHAHAN HR, KAMBIS TN, et al. Hydrogen sulfide ameliorates homocysteine-induced cardiac remodeling and dysfunction. Front Physiol. 2019;10:598.
[13] WANG D, ZHAO R, QU YY, et al. Colonic lysine homocysteinylation induced by high-fat diet suppresses DNA damage repair. Cell Rep. 2018;25:398-412.
[14] JAKUBOWSKI H. Protein n-homocysteinylation and colorectal cancer. Trends Cancer. 2019;5:7-10.
[15] GUIRAUD SP, MONTOLIU I, DA SILVA L, et al. High-throughput and simultaneous quantitative analysis of homocysteine–methionine cycle metabolites and co-factors in blood plasma and cerebrospinal fluid by isotope dilution LC–MS/MS. Anal Bioanal Chem. 2017;409(1):295-305.
[16] 谢琳,丁宁,徐灵博,等.FoxO1 DNA甲基化在同型半胱氨酸致肝细胞凋亡中的作用[J].实用医学杂志, 2018,34(16):2659-2662+ 2669.
[17] PENG YT, CHEN P, OUYANG RY, et al. Multifaceted role of prohibitin in cell survival and apoptosis. Apoptosis. 2015;20: 1135-1149.
[18] CUI J, PLACZEK WJ. Post-Transcriptional Regulation of Anti-Apoptotic BCL2 Family Members. Int J Mol Sci. 2018; 19(1). pii: E308.
[19] CAVALCANTE GC, SCHAAN AP, CABRAL GF, et al. A Cell's Fate: An Overview of the Molecular Biology and Genetics of Apoptosis. Int J Mol Sci. 2019;20(17). pii: E4133.
[20] BRAMBILLA E, NEGOESCU A, GAZZERI S, et al. Apoptosis-related factors p53, Bcl2, and Bax in neuroendocrine lung tumors. Am J Pathol. 1996;149:1941-1952.
[21] WEISS CN, ITO K. A Macro View of MicroRNAs: the discovery of micrornas and their role in hematopoiesis and hematologic disease. Int Rev Cell Mol Biol. 2017;334: 99-175.
[22] BERTOLI G, CAVA C, CASTIGLIONI I. MicroRNAs: New biomarkers for diagnosis, prognosis, therapy prediction and therapeutic tools for breast cancer. Theranostics. 2015; 5: 1122-1143.
[23] MCALINDEN A, IM GI. MicroRNAs in Orthopaedic Research: Disease Associations, Potential Therapeutic Applications, and Perspectives. J Orthop Res. 2018;36(1):33-51.
[24] ZHOU L, LIM MYT, KAUR P, et al. Importance of miRNA stability and alternative primary miRNA isoforms in gene regulation during Drosophila development. Elife. 2018;7:e38389.
[25] MORA AL, ROJAS M, PARDO A, et al. Emerging therapies for idiopathic pulmonary fibrosis, a progressive age-related disease. Nat Rev Drug Discov. 2017;16(11):810.
[26] 高红强,李志强,王海富,等.减体积肝移植模型大鼠肝脏组织miRNAs表达谱变化[J].中国组织工程研究,2016,20(5): 712-717.
[27] SONG L, ZHANG Z, ZHANG J, et al. Ratio of microRNA-122/155 in isoniazid-induced acute liver injury in mice. Exp Ther Med. 2016;12(2):889-894.
[28] YANG Y, JIN Z, DONG R, et al. MicroRNA-29b/142-5p contribute to the pathogenesis of biliary atresia by regulating the IFN-γ gene. Cell Death Dis. 2018;9(5):545.
[29] YOGI K, SRIDHAR E, GOEL N, et al. MiR-148a, a microRNA upregulated in the WNT subgroup tumors, inhibits invasion and tumorigenic potential of medulloblastoma cells by targeting Neuropilin 1.Oncoscience. 2015;2(4):334-348.
[30] JUNG KH, ZHANG J, ZHOU C, et al. Differentiation Therapy for Hepatocellular Carcinoma: multifaceted effects of miR-148a on tumor growth and phenotype and liver fibrosis. Hepatology. 2016;63(3):864-879.
[31] BABU KR, MUCKENTHALER MU. miR-148a regulates expression of the transferrin receptor 1 in hepatocellular carcinoma. Sci Rep. 2019;9(1):1518.
|