Chinese Journal of Tissue Engineering Research ›› 2020, Vol. 24 ›› Issue (35): 5625-5631.doi: 10.3969/j.issn.2095-4344.2922
Previous Articles Next Articles
Pathological and behavioral changes after ischemic stroke in adult mice
Liang Yanfeng1, Hao Peng1, Duan Hongmei1, Zhao Wen1, Gao Yudan1, Li Xiaoguang1, 2, Yang Chaoyang1
1Department of Neurobiology, Capital Medical University, Beijing 100069, China; 2School of Biological and Medical Engineering, Beijing Laboratory of Biomaterials and Nerve Regeneration, Big Data Precision Medical Center for Advanced Innovation, Beihang University, Beijing 100191, China
-
Received:2020-02-25Revised:2020-03-03Accepted:2020-04-23Online:2020-12-18Published:2020-10-17 -
Contact:Yang Chaoyang, MD, Professor, Department of Neurobiology, Capital Medical University, Beijing 100069, China -
About author:Liang Yanfeng, Master, Department of Neurobiology, Capital Medical University, Beijing 100069, China -
Supported by:the National Key Research and Development Projects of China, No. 2017YFC1104002 and 2017YFC1104001; the National Natural Science Foundation of China, No. 81941011, 31730030, 31650001, 31320103903, 31670988, 31971279 , 31771053; Beijing Science and Technology Plan, No. Z181100001818007; 2018 Key Science and Technology Plan of Beijing Municipal Education Commission, No. KZ201810025030
CLC Number:
Cite this article
Liang Yanfeng, Hao Peng, Duan Hongmei, Zhao Wen, Gao Yudan, Li Xiaoguang, Yang Chaoyang. Pathological and behavioral changes after ischemic stroke in adult mice[J]. Chinese Journal of Tissue Engineering Research, 2020, 24(35): 5625-5631.
share this article
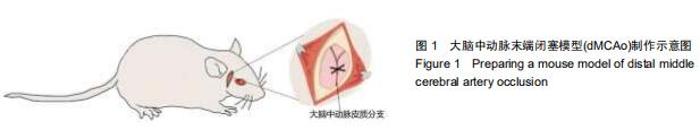
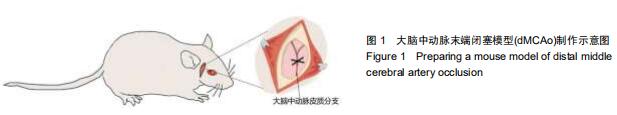
1.4.1 脑卒中模型制作 实验小鼠腹腔注射0.3%戊巴比妥钠(20 mL/kg)麻醉,左侧卧位,无菌环境下,沿眼外眦与外耳道连线中点的垂线划开约0.5 cm的切口,划开附着在颞骨上的颞肌筋膜(注意不要损伤颞肌,避免影响小鼠咀嚼功能),将颞肌向下推至颧骨后,暴露右侧颞骨。由于小鼠颅骨较薄,可透过颅骨找到大脑中动脉皮质分支,在其上方用牙科钻钻开直径1 mm骨窗,骨窗位置尽量靠近颧骨侧,经针尖挑开硬脑膜后,用电凝笔电凝大脑中动脉皮质分支(图1),此时电凝可造成约0.1 mL的出血量,这可作为判断手术成功的标志。之后用无菌棉球压迫止血,无出血后缝合皮肤,手术总时长在30 min以内,手术完成后对动物进行复温归笼。后期连续注射3 d青霉素抗感染处理。dMCAo诱导的缺血性损伤局限于部分运动皮质和感觉皮质(图2B),术后第2天对该模型进行行为学测试,患侧肢体出现明显感觉运动缺失则进一步确定模型制备成功。假手术组仅进行开直径1 mm骨窗后缝合皮肤,后期处理与卒中组相同。 "
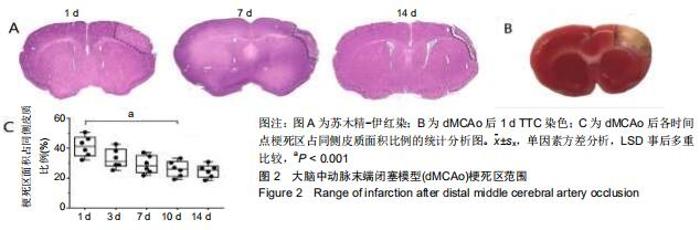
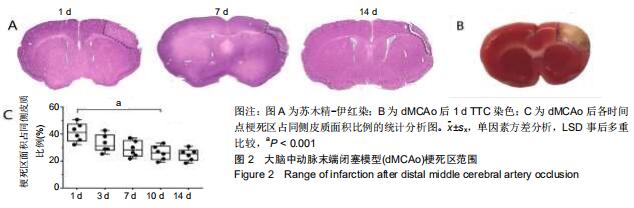
2.2 梗死范围及区域 首先在dMCAo后1 d进行苏木精-伊红染(图2A)和TTC染色(图2B),dMCAo后3,7,10和14 d进行苏木精-伊红染色,以观察此模型的梗死范围及影响的脑功能区域。dMCAO后1 d,由于细胞水肿,梗死区域面积较大;随着水肿消退,细胞死亡,梗死区逐渐缩小,术后 10 d时候梗死区范围趋于稳定(图2A,C)。之后,对dMCAo后梗死面积进行统计分析,梗死后第1天,梗死面积占同侧皮质面积百分比为(41.2±6.9)%,第10天缩小至(26.1± 5.2)%(图2C;梗死面积占比:第10天比第1天,P < 0.001),之后稳定在(24.7±4.4)%(图2C;梗死面积占比:第14天比第10天,P=0.67)。通过对比小鼠脑图谱,梗死区域主要位于皮质初级运动皮质(M1区)、初级感觉皮质(S1区)和次级感觉皮质(S2区)。 "
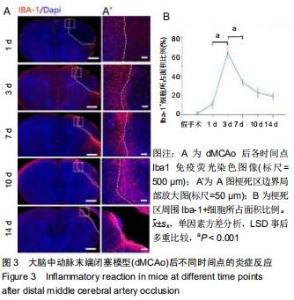
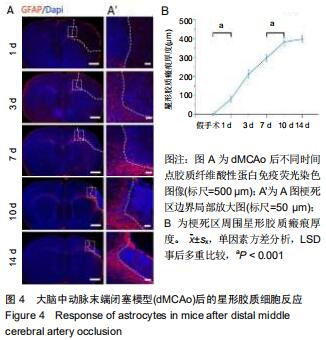
2.3 脑卒中后炎症反应及星型胶质细胞反应 dMCAo后最为突出的病理变化,即炎症反应和星形胶质反应,用Iba-1标记小胶质细胞和/或巨噬细胞,胶质纤维酸性蛋白标记星形胶质细胞,对dMCAo后不同时间点的脑组织切片进行免疫荧光染色。损伤后第1天,炎症反应并不明显;第3天小胶质细胞开始出现明显的活化状态(细胞胞体肿胀,突起变短增厚),大量Iba-1阳性细胞从损伤周围皮质、胼胝体及纹状体向梗死区迁移,梗死周围区域Iba-1阳性细胞面积所占总面积(64.0±5.1)%(将梗死区周围400 μm内面积定为总面积,将总面积内包含Iba-1阳性细胞区域面积定为Iba-1阳性细胞面积),此时炎症反应达到峰值(图3A,B;Iba-1阳性细胞面积占比,第3天比第1天,P < 0.001;第3天比第7天,P < 0.001);卒中后7 d时,同侧胼胝体、纹状体炎症反应明显减轻,激活状态的小胶质细胞集中于梗死区边界;卒中后10 d时大量Iba-1阳性细胞进入梗死区内(图3A,B)。炎症反应发生的同时,星形胶质瘢痕壁的形成可能是防止因炎症扩散以及保护梗死周围正常组织不受损害的关键措施[11],因此检测了胶质瘢痕形成的时间点。卒中后3 d,梗死区周围出现反应性星形胶质细胞,这些聚集于梗死区周围的反应性星形胶质细胞突起广泛重叠和交叉,在第10-14天形成稳定的胶质瘢痕(图4A,B;瘢痕厚度:第10天(382±20) μm;第14天(397±16) μm;P=0.205;瘢痕厚度为梗死区周围发生重叠的胶质纤维酸性蛋白阳性细胞区域面积除以此区域长度)。绘制出小胶质细胞和星形胶质细胞的动态变化,这对于后期在此模型上进行相关实验有重要指导意义。 "
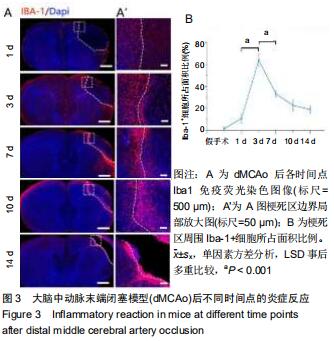
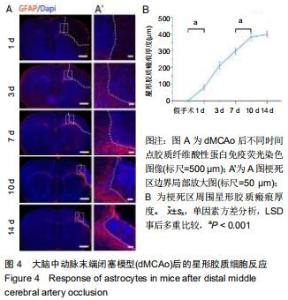
2.3 脑卒中后炎症反应及星型胶质细胞反应 dMCAo后最为突出的病理变化,即炎症反应和星形胶质反应,用Iba-1标记小胶质细胞和/或巨噬细胞,胶质纤维酸性蛋白标记星形胶质细胞,对dMCAo后不同时间点的脑组织切片进行免疫荧光染色。损伤后第1天,炎症反应并不明显;第3天小胶质细胞开始出现明显的活化状态(细胞胞体肿胀,突起变短增厚),大量Iba-1阳性细胞从损伤周围皮质、胼胝体及纹状体向梗死区迁移,梗死周围区域Iba-1阳性细胞面积所占总面积(64.0±5.1)%(将梗死区周围400 μm内面积定为总面积,将总面积内包含Iba-1阳性细胞区域面积定为Iba-1阳性细胞面积),此时炎症反应达到峰值(图3A,B;Iba-1阳性细胞面积占比,第3天比第1天,P < 0.001;第3天比第7天,P < 0.001);卒中后7 d时,同侧胼胝体、纹状体炎症反应明显减轻,激活状态的小胶质细胞集中于梗死区边界;卒中后10 d时大量Iba-1阳性细胞进入梗死区内(图3A,B)。炎症反应发生的同时,星形胶质瘢痕壁的形成可能是防止因炎症扩散以及保护梗死周围正常组织不受损害的关键措施[11],因此检测了胶质瘢痕形成的时间点。卒中后3 d,梗死区周围出现反应性星形胶质细胞,这些聚集于梗死区周围的反应性星形胶质细胞突起广泛重叠和交叉,在第10-14天形成稳定的胶质瘢痕(图4A,B;瘢痕厚度:第10天(382±20) μm;第14天(397±16) μm;P=0.205;瘢痕厚度为梗死区周围发生重叠的胶质纤维酸性蛋白阳性细胞区域面积除以此区域长度)。绘制出小胶质细胞和星形胶质细胞的动态变化,这对于后期在此模型上进行相关实验有重要指导意义。 "
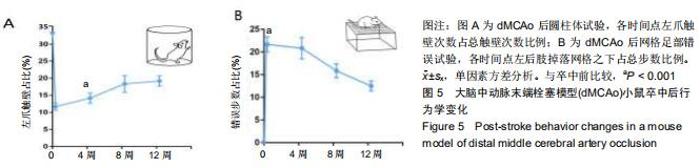
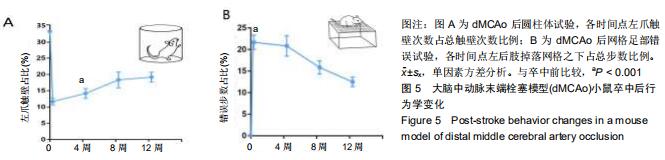
2.4 脑卒中对行为学的影响 在圆柱体试验中,dMCAo后小鼠患侧前肢触壁次数占总触壁次数明显减少[图5A,卒中前33.3%,卒中后(11.3±3.1)%,P < 0.001]。在网格试验中,正常小鼠后肢基本不会掉落在网格之下,而卒中后由于患侧后肢感觉的缺失,患肢会掉落到网格之下,小鼠错误步数占总步数比率明显增加[图5B,卒中前0;卒中后(21.7±4.1)%;P < 0.001]。在dMCAo后4-12周小鼠患侧前肢触壁次数逐渐回升,患侧后肢错误步数也逐渐减少,这是长时间的脑自发恢复过程,实验中模型小鼠的自发功能恢复在dMCAo后4-8周时速度最快,一直持续到12周,但仍有明显的感觉运动功能缺失(图5A,B)。 "
|
[1] WRITING GROUP MEMBERS, MOZAFFARIAN D, BENJAMIN EJ, et al. Heart Disease and Stroke Statistics-2016 Update: A Report From the American Heart Association. Circulation.2016;33(4):e38. [2] VALERY L FEIGIN, THEO VOS, EMMA NICHOLS, et al.Global, regional, and national burden of neurological disorders, 1990-2016: a systematic analysis for the Global Burden of Disease Study 2016.Lancet Neurol. 2019;18(5):459-480. [3] DONNAN GA, FISHER M, MACLEOD M, et al.Stroke.Lancet. 2008;371(9624):1612-1623. [4] JAYARAJ RL, AZIMULLAH S, BEIRAM R, et al. Neuroinflammation: friend and foe for ischemic stroke. J Neuroinflammation 2019;16(1):142. [5] JOSHUA E BURDA, MICHAEL V SOFRONIEW. Reactive Gliosis and the Multicellular Response to CNS Damage and Disease. Neuron.2014;81(2):229-248. [6] BERNHEISEL CR, SCHLAUDECKER JD, LEOPOLD K.Subacute Management of Ischemic Stroke. Am Fam Physician. 2011;84(12):1383-1388. [7] MEMEZAWA H, MINAMISAWA H, SMITH ML, et al. Ischemic penumbra in a model of reversible middle cerebral artery occlusion in the rat.Exp Brain Res.1992; 89(1):67-78. [8] DAVID W HOWELLS, MICHELLE J PORRITT, SARAH SJ REWELL, et al. Different strokes for different folks: the rich diversity of animal models of focal cerebral ischemia.J Cereb Blood Flow Metab. 2010;30(8):1412-1431. [9] TAMURA A, GRAHAM DI, MCCULLOCH J, et al. Focal Cerebral Ischaemia in the Rat: 1. Description of Technique and Early Neuropathological Consequences Following Middle Cerebral Artery Occlusion.J Cereb Blood Flow Metab.1981; 1(1):53-60. [10] MCAULEY MA. Rodent models of focal ischemia. Cerebrovasc Brain Metab Rev.1995;7(2):153-180. [11] RANSOM BR, RANSOM CB. Astrocytes: multitalented stars of the central nervous system. Methods Mol Biol.2012;814: 3-7. [12] SHIMIN LIU, GEHUA ZHEN, BRUNO P MELONI, et al. Rodent Stroke Model Guidelines for Preclinical Stroke Trials (1st Edition).J Exp Stroke Transl Med.2009;2(2):2-27. [13] KONSTANTIN-ALEXANDER HOSSMANN. The two pathophysiologies of focal brain ischemia: implications for translational stroke research. J Cereb Blood Flow Metab 2012;32(7):1310-1316. [14] SARAH LUNARDI BACCETTO, CHRISTIAN LEHMANN. Microcirculatory Changes in Experimental Models of Stroke and CNS-Injury Induced Immunodepression.Int J Mol Sci. 2019;20(20):5184. [15] DOYLE KP, BUCKWALTER MS.A Mouse Model of Permanent Focal Ischemia: Distal Middle Cerebral Artery Occlusion.Methods Mol Biol. 2014;1135:103-110. [16] GELDERBLOM M, LEYPOLDT F, STEINBACH K, et al. Temporal and Spatial Dynamics of Cerebral Immune Cell Accumulation in Stroke. Stroke.2009;40(5):1849-1857. [17] XIAOMING HU, REHANA K LEAK, YEJIE SHI, et al.Microglial and macrophage polarization-new prospects for brain repair. Nat Rev Neurol. 2015;11(1):56-64. [18] AMANTEA D, TASSORELLI C, PETRELLI F, et al. Understanding the Multifaceted Role of Inflammatory Mediators in Ischemic Stroke.Curr Med Chem.2014;21(18): 2098-2117. [19] YOUL KJ, JOOHYUN P, YOUNG CJ, et al. Inflammation after Ischemic Stroke: The Role of Leukocytes and Glial Cells.Exp Neurobiol.2016;25(5):241-251. [20] XU S, LU J, SHAO A, et al. Glial Cells: Role of the Immune Response in Ischemic Stroke.Front Immunol. 2020;11:294. [21] CHECHNEVA OV, DENG W. Mitochondrial translocator protein (TSPO), astrocytes and neuroinflammation. Neural Regen Res. 2016;11(7):1056-1057. [22] ASYA ROLLS, RAVID SHECHTER, MICHAL SCHWARTZ. The bright side of the glial scar in CNS repair. Nat Rev Neurosci. 2009;10(3):235-241. [23] MOFTAH M, JADAVJI NM. Role of behavioral training in reducing functional impairments after stroke. Neural Regen Res. 2019;14(9):1507-1508. [24] CLAUDIA ALIA, CRISTINA SPALLETTI, STEFANO LAI, et al.Neuroplastic Changes Following Brain Ischemia and their Contribution to Stroke Recovery: Novel Approaches in Neurorehabilitation.Front Cell Neurosci.2017;11:76. [25] PATRICE VOSS, MARYSE E THOMAS, J MIGUEL CISNEROS-FRANCO, et al. Dynamic Brains and the Changing Rules of Neuroplasticity: Implications for Learning and Recovery.Front Psychol.2017;8:1657. [26] YANG JP, LIU HJ, LIU XF. VEGF Promotes Angiogenesis and Functional Recovery in Stroke Rats.J Invest Surg. 2010;23(3): 149-155. [27] ANTINA DE BOER, ANNET STORM, MARICEL GOMEZ-SOLER, et al.Environmental enrichment during the chronic phase after experimental stroke promotes functional recovery without synergistic effects of EphA4 targeted therapy.Hum Mol Genet. 2020;29(4):605-617. [28] CARACCIOLO L, MAROSI M, MAZZITELLI J, et al. CREB controls cortical circuit plasticity and functional recovery after stroke.Nat Commun. 2018;9(1):2250. [29] LI F, ZHANG T, LI BJ, et al. Motor imagery training induces changes in brain neural networks in stroke patients. Neural Regen Res. 2018;13(10):1771-1781. [30] GERLINDE A METZ, IWA ANTONOW-SCHLORKE, OTTO W WITTEMETZ GA. Motor improvements after focal cortical ischemia in adult rats are mediated by compensatory mechanisms.Behav Brain Res. 2005;162(1):71-82. [31] THERESA A. JONES, STEPHANIE C, JEFFERSON. Reflections of experience-expectant development in repair of the adult damaged brain.Dev Psychobiol.2011;53(5): 466-475. [32] C LOIS, A ALVAREZ-BUYLLA. Proliferating Subventricular Zone Cells in the Adult Mammalian Forebrain can Differentiate into Neurons and Glia.Proc Natl Acad Sci U S A. 1993;90(5):2074-2077. [33] DUAN HM, LI XG, WANG C, et al. Functional hyaluronate collagen scaffolds induce NSCs differentiation into functional neurons in repairing the traumatic brain injury.Acta Biomater. 2016;45:182-195. [34] PENG HAO, HONGMEI DUAN, FEI HAO, et al. Neural repair by NT3-chitosan via enhancement of endogenous neurogenesis after adult focal aspiration brain injury. Biomaterials. 2017;140:88-102. [35] LINHONG MO, ZHAOYANG YANG, AIFENG ZHANG, et al. The repair of the injured adult rat hippocampus with NT-3-chitosan carriers.Biomaterials.2010;31(8):2184-2192. [36] HIROFUMI NAKATOMI, TOSHIHIKO KURIU, SHIGEO OKABE, et al. Regeneration of Hippocampal Pyramidal Neurons after Ischemic Brain Injury by Recruitment of Endogenous Neural Progenitors.Cell. 2002;110(4):429-441. [37] JACK M PARENT, ZINAIDA S VEXLER, CHAO GONG, et al. Rat forebrain neurogenesis and striatal neuron replacement after focal stroke.Ann Neurol. 2002;52(6):802-813. |
| [1] | Zhang Yu, Tian Shaoqi, Zeng Guobo, Hu Chuan. Risk factors for myocardial infarction following primary total joint arthroplasty [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1340-1345. |
| [2] | Yuan Mei, Zhang Xinxin, Guo Yisha, Bi Xia. Diagnostic potential of circulating microRNA in vascular cognitive impairment [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1299-1304. |
| [3] | Li Jing, Xie Jianshan, Cui Huilin, Cao Ximei, Yang Yanping, Li Hairong. Expression and localization of diacylglycerol kinase zeta and protein kinase C beta II in mouse back skin with different coat colors [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1196-1200. |
| [4] | Tang Hui, Yao Zhihao, Luo Daowen, Peng Shuanglin, Yang Shuanglin, Wang Lang, Xiao Jingang. High fat and high sugar diet combined with streptozotocin to establish a rat model of type 2 diabetic osteoporosis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1207-1211. |
| [5] | Chai Le, Lü Jianlan, Hu Jintao, Hu Huahui, Xu Qingjun, Yu Jinwei, Quan Renfu. Signal pathway variation after induction of inflammatory response in rats with acute spinal cord injury [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1218-1223. |
| [6] | Chen Jiming, Wu Xiaojing, Liu Tianfeng, Chen Haicong, Huang Chengshuo. Effects of silymarin on liver injury and bone metabolism induced by carbon tetrachloride in mice [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1224-1228. |
| [7] | Zhang Xiumei, Zhai Yunkai, Zhao Jie, Zhao Meng. Research hotspots of organoid models in recent 10 years: a search in domestic and foreign databases [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1249-1255. |
| [8] | Jiao Hui, Zhang Yining, Song Yuqing, Lin Yu, Wang Xiuli. Advances in research and application of breast cancer organoids [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1122-1128. |
| [9] | Wang Feng, Zhou Liyu, Saijilafu, Qi Shibin, Ma Yanxia, Wei Shanwen. CaMKII-Smad1 promotes axonal regeneration of peripheral nerves [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1064-1068. |
| [10] | Yuan Xinping, Shao Yanbo, Wu Chao, Wang Jianling, Tong Liangcheng, Li Ying. Accuracy of target bone segments in personalized differential modeling and simulation of CT scanning parameters at fracture end [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(6): 912-916. |
| [11] | Zhang Mi, Wu Saixuan, Dong Ming, Lu Ying, Niu Weidong. Expression of interleukin-24 in a mouse model of periapical periodontitis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 679-684. |
| [12] | Xie Yang, Zhang Shujiang, Liu Menglan, Luo Ying, Yang Yang, Li Zuoxiao. Mechanism by which rapamycin protects spinal cord neurons in experimental autoimmune encephalomyelitis mice [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 695-700. |
| [13] | Ruan Guangping, Yao Xiang, Liu-Gao Miyang, Cai Xuemin, Li Zian, Pang Rongqing, Wang Jinxiang, Pan Xinghua. Umbilical cord mesenchymal stem cell transplantation for traumatic systemic inflammatory response syndrome in tree shrews [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(25): 3994-4000. |
| [14] | Wang Qiufei, Gu Ye, Peng Yuqin, Xue Feng, Ju Rong, Zhu Feng, Wang Yijun, Geng Dechun, Xu Yaozeng. Effect of Wnt/beta-catenin signaling pathway on osteoblasts under the action of wear particles [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(24): 3894-3901. |
| [15] | Su Baotong, Wang Hanyu, Xu Yilang, Xie Yajuan, Cheng Zhian. Construction of a Nomogram prediction model for postoperative delirium after hip fracture in the elderly based on medical records from a hospital [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(24): 3844-3849. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||