Chinese Journal of Tissue Engineering Research ›› 2020, Vol. 24 ›› Issue (19): 3063-3070.doi: 10.3969/j.issn.2095-4344.2023
Previous Articles Next Articles
Applications, roles and problems of exosomes derived from different stem cells in the treatment of cardiovascular diseases
Liu Zu, Han Shen, Li Yaxiong, Li Kunlin, Zhang Yayong, Jiang Lihong
- Department of Cardiac Vascular Surgery, Yan’an Hospital, Kunming Medical University, Kunming Yan’an Hospital, Department of Cardiovascular Surgery, Yunnan Institute of Cardiovascular Surgery, Yunnan Key Laboratory of Cardiovascular Diseases, the First People’s Hospital of Yunnan Province, Kunming 650051, Yunnan Province, China
-
Received:2019-07-04Revised:2019-07-06Accepted:2019-08-15Online:2020-07-08Published:2020-04-09 -
Contact:Jiang Lihong, MD, Professor, Doctoral supervisor, Master’s supervisor, Department of Cardiac Vascular Surgery, Yan’an Hospital, Kunming Medical University, Kunming Yan'an Hospital, Department of Cardiovascular Surgery, Yunnan Institute of Cardiovascular Surgery, Yunnan Key Laboratory of Cardiovascular Diseases, the First People’s Hospital of Yunnan Province, Kunming 650051, Yunnan Province, China Zhang Yayong, Master, Attending physician, Department of Cardiac Vascular Surgery, Yan’an Hospital, Kunming Medical University, Kunming Yan’an Hospital, Department of Cardiovascular Surgery, Yunnan Institute of Cardiovascular Surgery, Yunnan Key Laboratory of Cardiovascular Diseases, the First People’s Hospital of Yunnan Province, Kunming 650051, Yunnan Province, China -
About author:Liu Zu, Master candidate, Department of Cardiac Vascular Surgery, Yan’an Hospital, Kunming Medical University, Kunming Yan’an Hospital, Department of Cardiovascular Surgery, Yunnan Institute of Cardiovascular Surgery, Yunnan Key Laboratory of Cardiovascular Diseases, the First People’s Hospital of Yunnan Province, Kunming 650051, Yunnan Province, China -
Supported by:the Science and Technology Talents and Platform Program of Yunnan Province, Yunnan Provincial Key Laboratory of Cardiovascular Diseases, No. 2018DG008; the Yunnan Provincial Science and Technology Innovation Talents Program, Provincial Innovation Team of Key Diagnosis and Treatment Technology Research in Cardiovascular Surgery, Kunming Yan’an Hospital, No. 2018HC027; the Health Science and Technology Program of Yunnan Province, No. 2017NS328
CLC Number:
Cite this article
Liu Zu, Han Shen, Li Yaxiong, Li Kunlin, Zhang Yayong, Jiang Lihong. Applications, roles and problems of exosomes derived from different stem cells in the treatment of cardiovascular diseases[J]. Chinese Journal of Tissue Engineering Research, 2020, 24(19): 3063-3070.
share this article
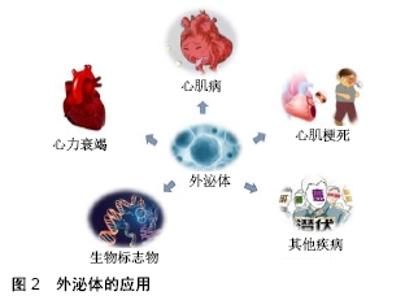
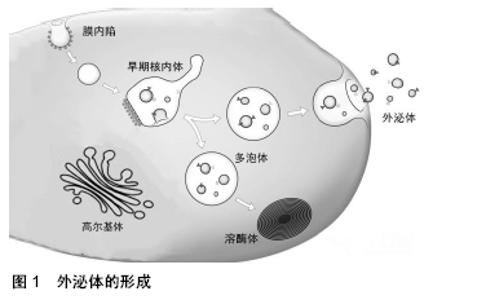
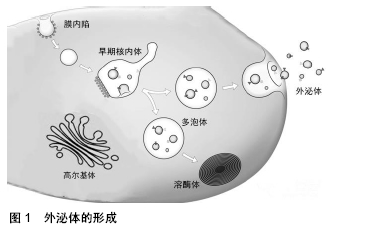
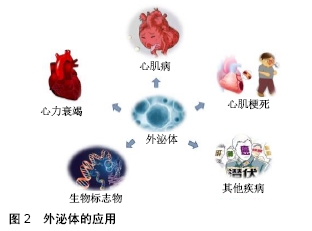
外泌体表面携带特定标记物,如CD9、CD63、CD81、Alix、Foltillin和TSG101;囊泡内包含多种生物活性物质,如蛋白质、核酸(DNA、mRNA、非编码RNA)和脂质等[11]。外泌体所携带的内容物赋予其丰富的生物学信息,通过外泌体传输到其他细胞发挥特定的生物学效应[12]。 外泌体可以通过多种机制与靶细胞相互作用:①外泌体通过靶细胞的内吞作用被摄取[13],COSTA VERDERA等[14]研究发现外泌体的摄取取决于酪氨酸激酶和胆固醇活性,这与不依赖网格蛋白的内吞作用有关,而磷酸肌醇3-激酶活性和Na+/H+交换对大胞饮有重要作用。此外,通过siRNA介导的caveolin-1、flotillin-1、RhoA、Rac1和Pak1敲低能够抑制外泌体被细胞摄取,但网格蛋白重链不起作用。这些结果表明,外泌体主要通过不依赖网格蛋白的内吞作用和大胞饮作用进入细胞。②外泌体通过与靶细胞的质膜融合,将内容物释放到靶细胞中[15],VALADI等[12]观察到小鼠肥大细胞外泌体中的RNA可转移至其他小鼠和人肥大细胞,在将小鼠外泌体RNA转移至人肥大细胞后,在受体细胞中发现新的小鼠蛋白质,表明转移的外泌体mRNA可在进入另一细胞后翻译成蛋白质。这种信息转运方式能在机体中广泛存在,如间充质干细胞来源外泌体产生的心肌保护作用[16]。外泌体表面存在多种细胞因子及生长因子,当外泌体进入靶细胞时,这些分泌出来的因子能够抑制细胞凋亡并刺激血管生成[17]。此外,外泌体能够将miRNA转移到受体细胞,在此过程中可传递遗传信息,调节细胞功能。③外泌体通过识别靶细胞表面的特定受体[18-19],例如外泌体可通过与内皮细胞表面的受体相结合起到促血管生成的作用[20]。干细胞来源外泌体促进细胞存活、抑制细胞凋亡及促血管新生等作用,可能与多种机制共同作用有关,具体作用机制仍待进一步探究。 2.2 干细胞来源外泌体及其在心血管疾病治疗中的应用 自1968年第一次骨髓细胞移植治疗成功以来[21],以细胞疗法为代表的再生医学被应用于许多疾病的治疗。在心血管疾病领域,尤其是心肌梗死的治疗中,干细胞疗法表现出了巨大的潜力。研究表明,干细胞疗法可以促进心肌细胞的存活,从而改善心肌收缩力以治疗心血管疾病,尤其是心肌梗死[22]。然而,细胞疗法具有一定的局限性,其移植成功率低、具有免疫排斥性和致瘤性等风险都限制了其临床应用。近年来的研究表明,干细胞除了可诱导分化为心肌细胞以修复梗死组织外,还可以通过外泌体的旁分泌机制参与修复受损心肌组织[23]。因此,外泌体有望取代干细胞疗法成为治疗心血管疾病尤其是心肌梗死的新方法[24-26],为其在生物学、再生医学、疾病诊断及预防等领域带来了新的机遇,见图2。 "
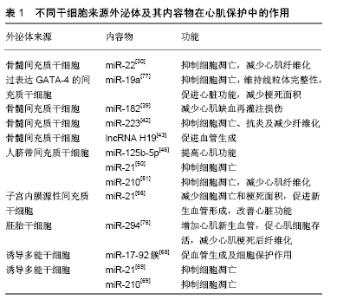
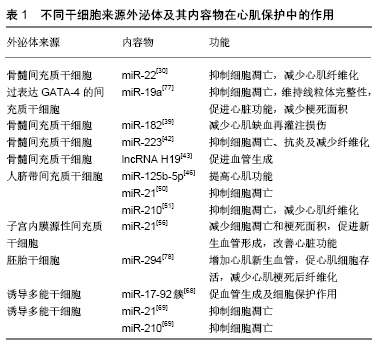
2.2.1 间充质干细胞来源的外泌体 间充质干细胞因其具有自我更新、向不同组织分化及免疫调节功能,而被作为治疗急性心肌梗死、心力衰竭等心血管疾病的有效方法之一。针对细胞移植,FREYMAN等[27]研究发现急性心肌梗死后14 d,注射的间充质干细胞只有0.06%保留在猪心肌梗死区。此外,ANGOULVANT等[28]通过体外实验表明,间充质干细胞或间充质干细胞来源的心肌细胞都能显著降低心肌梗死或室性心动过速损伤,这种保护作用可通过大鼠新生心肌细胞中PI3K途径的旁分泌激活进行介导。因此,作为间充质干细胞旁分泌系统的一个重要组成部分,外泌体已经在心肌再生医学领域进行广泛的研究,证明它们在治疗心血管疾病方面的潜力。有研究证实间充质干细胞和间充质干细胞来源外泌体之间有类似的miRNA表达模式,在急性心肌梗死大鼠模型中,通过比较二者在促进心肌细胞增殖、抗细胞凋亡及抑制心肌纤维化的能力,证实两种治疗方法对于改善急性心肌梗死大鼠心脏功能的效果相当[29]。 (1)骨髓间充质干细胞:在各种间充质干细胞中,骨髓间充质干细胞在心血管疾病中的研究最为广泛。LAI等[16]研究发现骨髓间充质干细胞培养基上清液中能发挥组织损伤修复作用的物质可能是外泌体,后续研究也发现外泌体可参与促进细胞存活、抑制细胞凋亡、促进血管生成、抗炎、免疫调节和抑制纤维化等环节,从而发挥组织修复作用。 促进细胞存活和抑制细胞凋亡:在正常条件下,功能性心肌细胞主要通过脂肪酸β氧化磷酸化合成三磷酸腺苷,但在缺血缺氧环境下,心肌细胞氧化磷酸化途径受到抑制,减少三磷酸腺苷的生成,严重制约了三磷酸腺苷依赖性的基因转录、蛋白合成、离子泵等多种生物途径进程,最终导致心肌细胞功能异常甚至细胞凋亡。FENG等[30]研究发现在缺血预处理后,小鼠骨髓间充质干细胞分泌富含miR-22的外泌体,将其作用于缺血心肌中可减少心肌细胞凋亡,并且miR-22的抗凋亡作用是通过直接靶向甲基CpG结合蛋白2(methyl CpG binding protein 2,Mecp2)介导的,在小鼠心肌梗死模型中,通过其外泌体给药可显著减少梗死面积和改善心肌纤维化。YU等[31]研究表明,过表达GATA-4的骨髓间充质干细胞来源外泌体可显著提高在缺氧环境下体外培养的心肌细胞存活率和线粒体膜电位,并在小鼠心肌梗死模型中恢复心肌收缩功能和减小梗死面积,随后他们发现miR-19a在GATA-4过表达的骨髓间充质干细胞来源外泌体中更高,通过抑制磷酸酶和数十种同源物参与心肌保护作用。 促进新生血管的形成:心肌缺血性损伤后,心肌细胞开始出现凋亡/死亡,心脏处于代偿/修复状态,最终导致心室重塑、心肌梗死区瘢痕形成,那么促进心肌梗死区域新生血管的形成显得尤为重要。BIAN等[32]收集了骨髓间充质干细胞在缺氧刺激下分泌直径约为100 nm的细胞外囊泡,心肌内注射这些囊泡可促进新生血管生成,可通过增强心肌梗死模型血管生成来保护心肌组织免受缺血性损伤。此外,TENG等[33]研究表明,在大鼠心肌梗死后,骨髓间充质干细胞来源外泌体显著增加血管生成,减小梗死面积,并保持心脏收缩或舒张功能。 抗炎、免疫调节和抑制纤维化:炎症是心肌梗死后细胞因子和趋化因子从受损心肌中释放,进而吸引和激活多种免疫系统细胞而引发的一个重要过程[34]。缺血性心肌发生氧化应激、线粒体功能障碍、心肌细胞坏死和细胞凋亡,从而触发损伤相关分子模式的表达和释放,并激活局部巨噬细胞释放促炎细胞因子,如白细胞介素1β、白细胞介素6、白细胞介素8和肿瘤坏死因子α。除了损伤相关分子模式,缺血心肌还激活Toll样受体信号和补体系统,从而在心肌梗死早期持续使中性粒细胞向受损组织的募集和迁移[35]。单核细胞依次被招募并渗透到梗死心肌中,变成M1或M2巨噬细胞[36]。成纤维细胞在炎症过程中起着重要作用,在炎症早期,成纤维细胞通过产生细胞因子(如白细胞介素1β、白细胞介素6、白细胞介素8、肿瘤坏死因子α)和基质金属蛋白酶(如基质金属蛋白酶2和基质金属蛋白酶9)发挥促炎和基质降解作用[37]。心肌成纤维细胞的增殖和大量细胞外基质蛋白的产生对于维持缺血心脏的结构完整性至关重要,最后形成成熟的交联瘢痕,伴随着修复细胞的凋亡和清除[38]。因此,炎症反应与心肌损伤、修复和重塑密切相关。 ZHAO等[39]研究发现骨髓间充质干细胞来源外泌体对心肌缺血再灌注损伤模型中的巨噬细胞具有免疫调节作用,主要通过传递外泌体中的miR-182来抑制Toll样受体4的激活,从而减轻心肌缺血再灌注损伤。FRAN?OIS等[40]研究发现骨髓间充质干细胞来源外泌体能够通过白细胞介素6的调节,进一步延长中性粒细胞寿命,促进巨噬细胞向M2抗炎表型转化,进而影响T细胞免疫应答,在心肌梗死中发挥保护效应[18]。LIU等[41]在大鼠心肌梗死/再灌注模型中,注射骨髓间充质干细胞来源外泌体可通过AMPK/mTOR和Akt/mTOR途径诱导心肌细胞自噬,从而减少细胞凋亡和梗死面积,改善心功能。WANG等[42]在利用盲肠结扎穿孔术建立的脓毒症模型中,通过靶向SEMA3A和STAT-3,骨髓间充质干细胞分泌的富含miR-223的外泌体可保护盲肠结扎穿孔术诱发的心功能障碍、凋亡和炎症反应抑制纤维化。 除此之外,外泌体中含有的lncRNA对心肌保护起着重要的调节作用,如lncRNA H19。HUANG等[43]研究发现,在体外细胞培养实验中,经阿托伐他汀预处理的骨髓间充质干细胞来源外泌体可加速内皮细胞的迁移、促进管状结构的形成、增加内皮细胞的存活率。在大鼠急性心肌梗死模型中,与骨髓间充质干细胞来源外泌体相比,经阿托伐他汀预处理的骨髓间充质干细胞来源外泌体可促进心功能恢复,进一步缩小梗死面积,减少心肌细胞凋亡。在机制上,他们鉴定出lncRNA H19是骨髓间充质干细胞来源外泌体调控miR-675表达、激活促血管生成因子和细胞间黏附分子1的中介物,lncRNA H19至少介导了其部分促进血管生成的心脏保护作用。近来也有研究发现lncRNA H19是内皮细胞衰老的重要调节因子,可促进内皮细胞增殖[44]。 (2)其他类型的间充质干细胞:人脐带间充质干细胞来源外泌体对心血管疾病,尤其是对急性心肌梗死的潜在保护作用已被探索。例如,ZHAO等[45]研究表明,在急性心肌梗死动物模型中,人脐带间充质干细胞来源外泌体可以通过保护心肌细胞凋亡和促进血管生成来改善心脏收缩功能,这与Bcl-2家族的表达有关。在另一项动物研究中,人脐带间充质干细胞来源外泌体可通过抑制miR-125b-5p促进Smad7表达,从而改善急性心肌梗死后心肌修复[46]。MA等[47]研究表明Akt修饰的人脐带间充质干细胞来源外泌体在大鼠急性心肌梗死模型中具有良好的促血管生成效果。他们发现Akt修饰的外泌体可向内皮细胞传递高水平的血小板衍生生长因子D,并显著增强内皮细胞的增殖、迁移和管状结构及血管生成。此外,SHI等[48]研究表明,人脐带间充质干细胞来源外泌体可促进炎症环境中成纤维细胞向肌成纤维细胞的分化,并有利于心脏保护作用。LIU等[49]研究表明,人脐带间充质干细胞来源外泌体可能通过激活PI3K/Akt/mTOR通路调节自噬,从而有助于防止H9C2细胞凋亡。此外,他们发现缺氧预处理能促进人脐带间充质干细胞中外泌体的分泌而提高抗凋亡作用。缺氧预处理产生的外泌体的抗凋亡效应也可能与其特异性miRNA (miRNA-21[50]、miRNA-210[51]、miRNA-125b- 5p[52])以及血管内皮生长因子、表皮生长因子、成纤维细胞生长因子等细胞因子有关[53]。 除了骨髓间充质干细胞及人脐带间充质干细胞来源外泌体外,其他类型的间充质干细胞来源外泌体也被证明对心肌保护有益。CUI等[54]研究发现,脂肪源间充质干细胞来源外泌体在体外导致H9C2细胞在缺氧/复氧条件下的细胞活力显著增加,而脂肪间充质干细胞来源外泌体通过激活体内Wnt/β-连环蛋白信号来保护缺血心肌免受梗死/再灌注损伤。DENG等[55]研究发现来自脂肪间充质干细胞的外泌体通过激活S1P/SK1/S1PR1信号传导和促进巨噬细胞M2极化来改善心肌梗死后的心脏损伤。此外,WANG等[56]发现与骨髓间充质干细胞和脂肪间充质干细胞相比,在大鼠心肌梗死模型中,子宫内膜源性间充质干细胞具有更好的心肌保护作用。这些差异可能是由于某些miRNA,特别是子宫内膜间充质干细胞来源外泌体中的miR-21富集,通过靶向PTEN对细胞存活和血管生成产生影响。 2.2.2 胚胎干细胞来源外泌体 胚胎干细胞来源于胚胎未分化的内部团块细胞,其具有全能性,可以通过自我更新和分化为包括心肌细胞在内的各种类型的细胞。通过将胚胎干细胞诱导分化的心肌细胞植入到心肌梗死动物模型中,可以促进受损心肌组织心肌细胞再生并增强心肌收缩功能[57-58],但伦理问题和致畸性限制了其临床应用。近来的研究表明其旁分泌作用可介导心血管系统内细胞间的通讯,胚胎干细胞分泌的外泌体被认为将是治疗心血管疾病的替代治疗策略。 胚胎干细胞来源外泌体同样可通过抑制心肌细胞凋亡、促进心肌细胞存活、增强新生血管形成和减少梗死后纤维化,来发挥心肌保护作用。TAVAKOLI等[59]研究发现胚胎干细胞来源外泌体能抑制多柔比星诱导的H9C2细胞凋亡。KHAN等[60]发现在小鼠急性心肌梗死模型中,从胚胎干细胞中提取的外泌体通过心肌内注射可改善心肌收缩功能。他们还发现胚胎干细胞外泌体中富含miR-290-295簇(尤其是miR-294),能够增强新生血管形成、促进心肌细胞存活和减少梗死后心肌纤维化。还有研究表明,胚胎干细胞中的miR-294富集对细胞增殖有促进作用,对其分化有抑制作用[61-62]。除此之外,胚胎干细胞衍生细胞分泌的外泌体也可发挥心肌保护作用。LAI等[63]研究发现人胚胎干细胞来源的间充质干细胞分泌的外泌体是旁分泌作用中的心肌保护成分,可介导组织损伤和心肌修复。在小鼠梗死后心力衰竭模型中,KERVADEC等[64]研究人胚胎干细胞来源的心血管祖细胞释放的外泌体在心肌梗死后给药是否能提供与单独使用人胚胎干细胞来源的心血管祖细胞给药同等的益处,他们通过超声心动图引导下将其外泌体经皮注射进入梗死周围心肌,结果表明外泌体在改善心脏功能方面与干细胞有同等效果,可促进心肌细胞存活和增殖。 2.2.3 诱导多能干细胞来源外泌体 诱导多能干细胞是日本科学家山中申弥教授在2006年将Oct-4、Sox-2、Klf4、c-Myc 4种干细胞特异性转录因子转入人成纤维细胞中重新编程的一种类似胚胎干细胞的细胞类型[65-66]。在心血管疾病的研究中,诱导多能干细胞有诱导分化为心肌细胞的潜力,能促进小鼠梗死心肌的修复和再生,改善心脏功能[67]。虽然在诱导多能干细胞的治疗中可以排除免疫排斥及伦理等问题,然而诱导多能干细胞同样可能受到致瘤性的影响。因此,诱导多能干细胞来源外泌体同样成为了研究的重点。研究表明,诱导多能干细胞来源外泌体也可通过抑制心肌细胞凋亡、促进心肌细胞存活、增强新生血管形成和减少心肌纤维化,来发挥心肌保护作用。JUNG等[10]研究发现包括诱导多能干细胞来源外泌体在内的分泌小泡对心血管疾病动物模型的新生血管形成和心肌细胞存活均有积极影响。ADAMIAK等[68]研究发现诱导多能干细胞来源外泌体中高浓度miR-17-92簇(例如miR-19a、miR-19b、miR-20a)可能在心肌缺血再灌注损伤后具有促血管生成和心肌细胞保护作用,他们通过蛋白组学数据发现诱导多能干细胞来源外泌体有几个促血管新生分子的富集,包括骨形成蛋白4、血小板衍生生长因子α、胚胎瘤衍生因子1、血小板反应蛋白1和血管内皮生长因子C。WANG等[69]从小鼠心脏成纤维细胞来源的诱导多能干细胞获得的外泌体中发现含有心脏保护分子miRNA,其中包括Nanog调节的miR-21和缺氧诱导因子1α调节的miR-210,它们能保护心肌细胞免受H2O2诱导的体外氧化应激和体内心肌缺血再灌注损伤。此外,其他研究也有类似的观察结果,心肌细胞含丰富的miR-21和miR-210能减轻氧化应激诱导的心肌细胞凋亡[70-72]。 除了诱导多能干细胞来源外泌体外,诱导多能干细胞衍生细胞来源的外泌体对受损心肌也有保护作用,如诱导多能干细胞来源的间充质干细胞和诱导多能干细胞来源的心肌细胞在心肌梗死中发挥重要作用[10]。YANG[73]研究表明,诱导多能干细胞来源心肌细胞分泌的外泌体及其内容物是挽救心脏梗死区受损心肌细胞凋亡、坏死、炎症反应、重塑和纤维化作用机制的基础。EL HARANE等[74]发现人类诱导多能干细胞来源的心血管祖细胞分泌的细胞外囊泡在治疗充血性心力衰竭方面是有效的,可能通过其特异性的16个高度富集、进化保守的miRNA来实现,大部分与组织修复途径相关。此外,HU等[75]发现诱导多能干细胞来源的间充质干细胞分泌的外泌体激活了血管生成相关的基因表达,并促进人脐静脉内皮细胞迁移、增殖和导管形成。ZHANG等[76]研究表明将人诱导多能干细胞来源的间充质干细胞分泌的外泌体移植到伤口部位,可加速再上皮化、减少瘢痕宽度和促进胶原蛋白成熟。 以上研究总结了不同干细胞来源外泌体及其内容物通过促细胞存活、抑制细胞凋亡、促进血管生成、抗炎、免疫调节和减少纤维化等作用来改善心肌功能,在心肌再生及心肌保护中发挥重要作用,这可能是今后治疗心血管疾病的潜在治疗手段,见表1[30,39,42-43,46,50-51,56,68-69,77-78]。 "

2.3 外泌体的优势、局限及安全性 干细胞来源外泌体提供有益的miRNA和生长因子等,促进血管生成、分化和细胞存活,此外外泌体可以通过抑制炎症反应改善微环境,从而改善心肌功能。与干细胞疗法相比,外泌体具有储存方便、使用方便、不会引发免疫排斥反应等优势。例如,心肌梗死患者往往需要立即干预,而干细胞疗法需要时间来分离细胞并且生长到所需的数量。虽然可以通过使用冷冻细胞来解决,但许多细胞不能在冷冻/解冻过程中存活;其次,干细胞在梗死后的促炎症微环境中移植和存活能力较差,降低了其治疗效果,相比之下,外泌体是无细胞的颗粒,可以用标准技术纯化并储存在低温环境中直到使用。外泌体由脂质双层膜包裹,不需要使用有毒的冷冻保存剂,可以承受冻融循环。虽然动物实验表明干细胞治疗效果较好,但通常需要直接注射到心肌中,不适合应用于人心肌梗死。理想的干预措施是静脉注射,但当通过静脉注射时,大多数干细胞会被停留在肺内,无法到达靶器官,而外泌体足够小,可以通过肺进入循环,更加精确地到达目标细胞,从而发挥治疗作用;另外,外泌体在疾病治疗中的另一个主要优势是外泌体的内容物可以通过预处理或基因工程进行修饰,从而更加准确的发挥靶向治疗作用。尽管外泌体具有显著的优势,但其应用也有一些局限性:①外泌体含量较低,提取过程繁琐,采用经典的高速离心法提取外泌体耗时较长;②外泌体的促血管生成和促生存作用可能会促进癌细胞的生长。例如,间充质干细胞来源外泌体通过激活ERK1/2通路增强了血管内皮生长因子在肿瘤细胞中的表达[79]。 外泌体是否能真正应用于临床,其自身安全性是最为重要的考量因素,通过在不同动物模型中进行的大量体内实验证实了外泌体的自身安全性。SUN等[80]将人脐带间充质干细胞来源外泌体静脉输注到急性心肌梗死大鼠中,通过测量大鼠体质量并收集尾静脉血以评估肝肾功能,结果表明人脐带间充质干细胞来源外泌体对肝肾功能无不良影响。其他检测如溶血、血管、全身性过敏反应、热原和血液学指标,也表明人脐带间充质干细胞来源外泌体是适用的。ADAMIAK等[81]比较了诱导多能干细胞和诱导多能干细胞来源的外泌体在体内给药对于心肌修复的安全性和有效性,两种治疗方法均保留了梗死区存活心肌,而且外泌体可显著减少凋亡。6周后随访观察发现,注射诱导多能干细胞的小鼠在心脏不同位置均有畸胎瘤发生,而注射诱导多能干细胞来源外泌体的小鼠未发现这种肿瘤转化。因此,注射具有生物活性的、无细胞的由干细胞分泌的外泌体是缺血性损伤后心脏修复的一种有效和安全的方法。 "

| [1] STAMM C, NASSERI B, HETZER R. Cardiac stem cells in patients with ischaemic cardiomyopathy. Lancet. 2012;379(9819):891. [2] MAKKAR RR, SMITH RR, CHENG K, et al. Intracoronary cardiosphere-derived cells for heart regeneration after myocardial infarction (CADUCEUS): a prospective, randomised phase 1 trial. Lancet. 2012;379(9819):895-904. [3] URBANELLI L, BURATTA S, SAGINI K, et al. Exosome-based strategies for Diagnosis and Therapy. Recent Pat CNS Drug Discov. 2015;10(1):10-27. [4] MÜLLER-EHMSEN J, WHITTAKER P, KLONER RA, et al. Survival and development of neonatal rat cardiomyocytes transplanted into adult myocardium. J Mol Cell Cardiol. 2002;34(2):107-116. [5] PAGANI FD, DERSIMONIAN H, ZAWADZKA A, et al. Autologous skeletal myoblasts transplanted to ischemia-damaged myocardium in humans. Histological analysis of cell survival and differentiation. J Am Coll Cardiol. 2003;41(5):879-888. [6] CAI H, LIN L, CAI H, et al. Prognostic evaluation of microRNA-210 expression in pediatric osteosarcoma. Med Oncol. 2013;30(2):499. [7] KISHORE R, KHAN M. More Than Tiny Sacks: Stem Cell Exosomes as Cell-Free Modality for Cardiac Repair. Circ Res. 2016;118(2):330-343. [8] KHAN M, KISHORE R. Stem Cell Exosomes: Cell-FreeTherapy for Organ Repair. Methods Mol Biol. 2017;1553:315-321. [9] ZHANG Z, YANG J, YAN W, et al. Pretreatment of Cardiac Stem Cells With Exosomes Derived From Mesenchymal Stem Cells Enhances Myocardial Repair. J Am Heart Assoc. 2016;5(1):e002856. [10] JUNG JH, FU X, YANG PC. Exosomes Generated From iPSC-Derivatives: New Direction for Stem Cell Therapy in Human Heart Diseases. Circ Res. 2017;120(2):407-417. [11] BANG C, THUM T. Exosomes: new players in cell-cell communication. Int J Biochem Cell Biol. 2012;44(11):2060-2064. [12] VALADI H, EKSTRÖM K, BOSSIOS A, et al. Exosome-mediated transfer of mRNAs and microRNAs is a novel mechanism of genetic exchange between cells. Nat Cell Biol. 2007;9(6):654-659. [13] FITZNER D, SCHNAARS M, VAN ROSSUM D, et al. Selective transfer of exosomes from oligodendrocytes to microglia by macropinocytosis. J Cell Sci. 2011;124(Pt 3):447-458. [14] COSTA VERDERA H, GITZ-FRANCOIS JJ, SCHIFFELERS RM, et al. Cellular uptake of extracellular vesicles is mediated by clathrin-independent endocytosis and macropinocytosis. J Control Release. 2017;266:100-108. [15] PAROLINI I, FEDERICI C, RAGGI C, et al. Microenvironmental pH is a key factor for exosome traffic in tumor cells. J Biol Chem. 2009; 284(49):34211-34222. [16] LAI RC, ARSLAN F, LEE MM, et al. Exosome secreted by MSC reduces myocardial ischemia/reperfusion injury. Stem Cell Res. 2010;4(3):214-222. [17] PIRONTI G, STRACHAN RT, ABRAHAM D, et al. Circulating Exosomes Induced by Cardiac Pressure Overload Contain Functional Angiotensin II Type 1 Receptors. Circulation. 2015;131(24): 2120-2130. [18] SEGURA E, GUÉRIN C, HOGG N, et al. CD8+ dendritic cells use LFA-1 to capture MHC-peptide complexes from exosomes in vivo. J Immunol. 2007;179(3):1489-1496. [19] O'LOUGHLIN AJ, WOFFINDALE CA, WOOD MJ. Exosomes and the emerging field of exosome-based gene therapy. Curr Gene Ther. 2012;12(4):262-274. [20] SKOG J, WÜRDINGER T, VAN RIJN S, et al. Glioblastoma microvesicles transport RNA and proteins that promote tumour growth and provide diagnostic biomarkers. Nat Cell Biol.2008;10(12):1470-1476. [21] BACH FH, ALBERTINI RJ, JOO P, et al. Bone-marrow transplantation in a patient with the Wiskott-Aldrich syndrome. Lancet. 1968;2(7583): 1364-1366. [22] SANGANALMATH SK, BOLLI R. Cell therapy for heart failure: a comprehensive overview of experimental and clinical studies, current challenges, and future directions. Circ Res. 2013;113(6):810-834. [23] GNECCHI M, ZHANG Z, NI A, et al. Paracrine mechanisms in adult stem cell signaling and therapy. Circ Res. 2008;103(11):1204-1219. [24] SINGLA DK. Stem cells and exosomes in cardiac repair. Curr Opin Pharmacol. 2016;27:19-23. [25] SUZUKI E, FUJITA D, TAKAHASHI M, et al. Stem cell-derived exosomes as a therapeutic tool for cardiovascular disease. World J Stem Cells. 2016;8(9):297-305. [26] PRATHIPATI P, NANDI SS, MISHRA PK. Stem Cell-Derived Exosomes, Autophagy, Extracellular Matrix Turnover, and miRNAs in Cardiac Regeneration during Stem Cell Therapy. Stem Cell Rev Rep. 2017; 13(1):79-91. [27] FREYMAN T, POLIN G, OSMAN H, et al. A quantitative, randomized study evaluating three methods of mesenchymal stem cell delivery following myocardial infarction. Eur Heart J. 2006;27(9):1114-1122. [28] ANGOULVANT D, IVANES F, FERRERA R, et al. Mesenchymal stem cell conditioned media attenuates in vitro and ex vivo myocardial reperfusion injury. J Heart Lung Transplant. 2011;30(1):95-102. [29] SHAO L, ZHANG Y, LAN B, et al. MiRNA-Sequence Indicates That Mesenchymal Stem Cells and Exosomes Have Similar Mechanism to Enhance Cardiac Repair. Biomed Res Int. 2017;2017:4150705. [30] FENG Y, HUANG W, WANI M, et al. Ischemic preconditioning potentiates the protective effect of stem cells through secretion of exosomes by targeting Mecp2 via miR-22. PLoS One. 2014;9(2): e88685. [31] YU B, KIM HW, GONG M, et al. Exosomes secreted from GATA-4 overexpressing mesenchymal stem cells serve as a reservoir of anti-apoptotic microRNAs for cardioprotection. Int J Cardiol. 2015;182: 349-360. [32] BIAN S, ZHANG L, DUAN L, et al. Extracellular vesicles derived from human bone marrow mesenchymal stem cells promote angiogenesis in a rat myocardial infarction model. J Mol Med (Berl). 2014;92(4): 387-397. [33] TENG X, CHEN L, CHEN W, et al. Mesenchymal Stem Cell-Derived Exosomes Improve the Microenvironment of Infarcted Myocardium Contributing to Angiogenesis and Anti-Inflammation. Cell Physiol Biochem. 2015;37(6):2415-2424. [34] FRANGOGIANNIS NG. The inflammatory response in myocardial injury, repair, and remodelling. Nat Rev Cardiol. 2014;11(5):255-265. [35] CHANDRASEKAR B, SMITH JB, FREEMAN GL. Ischemia- reperfusion of rat myocardium activates nuclear factor-KappaB and induces neutrophil infiltration via lipopolysaccharide-induced CXC chemokine. Circulation. 2001;103(18):2296-2302. [36] HORCKMANS M, RING L, DUCHENE J, et al. Neutrophils orchestrate post-myocardial infarction healing by polarizing macrophages towards a reparative phenotype. Eur Heart J. 2017;38(3):187-197. [37] SHINDE AV, FRANGOGIANNIS NG. Fibroblasts in myocardial infarction: a role in inflammation and repair. J Mol Cell Cardiol. 2014; 70:74-82. [38] KAIN V, PRABHU SD, HALADE GV. Inflammation revisited: inflammation versus resolution of inflammation following myocardial infarction. Basic Res Cardiol. 2014;109(6):444. [39] ZHAO J, LI X, HU J, et al. Mesenchymal stromal cell-derived exosomes attenuate myocardial ischaemia-reperfusion injury through miR-182-regulated macrophage polarization. Cardiovasc Res. 2019; 115(7):1205-1216. [40] FRANÇOIS M, ROMIEU-MOUREZ R, LI M, et al. Human MSC suppression correlates with cytokine induction of indoleamine 2,3-dioxygenase and bystander M2 macrophage differentiation. Mol Ther. 2012;20(1):187-195. [41] LIU L, JIN X, HU CF, et al. Exosomes Derived from Mesenchymal Stem Cells Rescue Myocardial Ischaemia/Reperfusion Injury by Inducing Cardiomyocyte Autophagy Via AMPK and Akt Pathways. Cell Physiol Biochem. 2017;43(1):52-68. [42] WANG X, GU H, QIN D, et al. Exosomal miR-223 Contributes to Mesenchymal Stem Cell-Elicited Cardioprotection in Polymicrobial Sepsis. Sci Rep. 2015;5:13721. [43] HUANG P, WANG L, LI Q, et al. Atorvastatin Enhances the Therapeutic Efficacy of Mesenchymal Stem Cells Derived Exosomes in Acute Myocardial Infarction via Up-regulating Long Non-coding RNA H19. Cardiovasc Res. 2019 May 22. doi: 10.1093/cvr/cvz139. [Epub ahead of print] [44] HOFMANN P, SOMMER J, THEODOROU K, et al. Long non-coding RNA H19 regulates endothelial cell aging via inhibition of STAT3 signalling. Cardiovasc Res. 2019;115(1):230-242. [45] ZHAO Y, SUN X, CAO W, et al. Exosomes Derived from Human Umbilical Cord Mesenchymal Stem Cells Relieve Acute Myocardial Ischemic Injury. Stem Cells Int. 2015;2015:761643. [46] WANG XL, ZHAO YY, SUN L, et al. Exosomes derived from human umbilical cord mesenchymal stem cells improve myocardial repair via upregulation of Smad7. Int J Mol Med. 2018;41(5):3063-3072. [47] MA J, ZHAO Y, SUN L, et al. Exosomes Derived from Akt-Modified Human Umbilical Cord Mesenchymal Stem Cells Improve Cardiac Regeneration and Promote Angiogenesis via Activating Platelet- Derived Growth Factor D. Stem Cells Transl Med. 2017;6(1):51-59. [48] SHI Y, YANG Y, GUO Q, et al. Exosomes Derived from Human Umbilical Cord Mesenchymal Stem Cells Promote Fibroblast-to- Myofibroblast Differentiation in Inflammatory Environments and Benefit Cardioprotective Effects. Stem Cells Dev. 2019;28(12):799-811. [49] LIU H, SUN X, GONG X, et al. Human umbilical cord mesenchymal stem cells derived exosomes exert antiapoptosis effect via activating PI3K/Akt/mTOR pathway on H9C2 cells. J Cell Biochem. 2019; 120(9): 14455-14464. [50] CUI GH, WU J, MOU FF, et al. Exosomes derived from hypoxia-preconditioned mesenchymal stromal cells ameliorate cognitive decline by rescuing synaptic dysfunction and regulating inflammatory responses in APP/PS1 mice. FASEB J. 2018;32(2): 654-668. [51] ZHU J, LU K, ZHANG N, et al. Myocardial reparative functions of exosomes from mesenchymal stem cells are enhanced by hypoxia treatment of the cells via transferring microRNA-210 in an nSMase2-dependent way. Artif Cells Nanomed Biotechnol. 2018; 46(8):1659-1670. [52] ZHU LP, TIAN T, WANG JY, et al. Hypoxia-elicited mesenchymal stem cell-derived exosomes facilitates cardiac repair through miR-125b-mediated prevention of cell death in myocardial infarction. Theranostics. 2018;8(22):6163-6177. [53] HAN Y, REN J, BAI Y, et al. Exosomes from hypoxia-treated human adipose-derived mesenchymal stem cells enhance angiogenesis through VEGF/VEGF-R. Int J Biochem Cell Biol. 2019;109:59-68. [54] CUI X, HE Z, LIANG Z, et al. Exosomes From Adipose-derived Mesenchymal Stem Cells Protect the Myocardium Against Ischemia/Reperfusion Injury Through Wnt/β-Catenin Signaling Pathway. J Cardiovasc Pharmacol. 2017;70(4):225-231. [55] DENG S, ZHOU X, GE Z, et al. Exosomes from adipose-derived mesenchymal stem cells ameliorate cardiac damage after myocardial infarction by activating S1P/SK1/S1PR1 signaling and promoting macrophage M2 polarization. Int J Biochem Cell Biol. 2019;114: 105564. [56] WANG K, JIANG Z, WEBSTER KA, et al. Enhanced Cardioprotection by Human Endometrium Mesenchymal Stem Cells Driven by Exosomal MicroRNA-21. Stem Cells Transl Med. 2017;6(1):209-222. [57] LAFLAMME MA, CHEN KY, NAUMOVA AV, et al. Cardiomyocytes derived from human embryonic stem cells in pro-survival factors enhance function of infarcted rat hearts. Nat Biotechnol. 2007;25(9): 1015-1024. [58] CHONG JJ, YANG X, DON CW, et al. Human embryonic-stem- cell-derived cardiomyocytes regenerate non-human primate hearts. Nature. 2014;510(7504):273-277. [59] TAVAKOLI DARGANI Z, SINGLA DK. Embryonic stem cell-derived exosomes inhibit doxorubicin-induced TLR4-NLRP3-mediated cell death-pyroptosis. Am J Physiol Heart Circ Physiol. 2019;317(2): H460-H471. [60] KHAN M, NICKOLOFF E, ABRAMOVA T, et al. Embryonic stem cell-derived exosomes promote endogenous repair mechanisms and enhance cardiac function following myocardial infarction. Circ Res. 2015;117(1):52-64. [61] WANG Y, MELTON C, LI YP, et al. miR-294/miR-302 promotes proliferation, suppresses G1-S restriction point, and inhibits ESC differentiation through separable mechanisms. Cell Rep. 2013;4(1): 99-109. [62] GUO WT, WANG XW, YAN YL, et al. Suppression of epithelial-mesenchymal transition and apoptotic pathways by miR-294/302 family synergistically blocks let-7-induced silencing of self-renewal in embryonic stem cells. Cell Death Differ. 2015;22(7): 1158-1169. [63] LAI RC, ARSLAN F, LEE MM, et al. Exosome secreted by MSC reduces myocardial ischemia/reperfusion injury. Stem Cell Res. 2010;4(3):214-222. [64] KERVADEC A, BELLAMY V, EL HARANE N, et al. Cardiovascular progenitor-derived extracellular vesicles recapitulate the beneficial effects of their parent cells in the treatment of chronic heart failure. J Heart Lung Transplant. 2016;35(6):795-807. [65] TAKAHASHI K, YAMANAKA S. Induction of pluripotent stem cells from mouse embryonic and adult fibroblast cultures by defined factors. Cell. 2006;126(4):663-676. [66] TAKAHASHI K, TANABE K, OHNUKI M, et al. Induction of pluripotent stem cells from adult human fibroblasts by defined factors. Cell. 2007; 131(5):861-872. [67] SINGLA DK, LONG X, GLASS C, et al. Induced pluripotent stem (iPS) cells repair and regenerate infarcted myocardium. Mol Pharm. 2011; 8(5):1573-1581. [68] ADAMIAK M, CHENG G, BOBIS-WOZOWICZ S, et al. Induced Pluripotent Stem Cell (iPSC)-Derived Extracellular Vesicles Are Safer and More Effective for Cardiac Repair Than iPSCs. Circ Res. 2018; 122(2):296-309. [69] WANG Y, ZHANG L, LI Y, et al. Exosomes/microvesicles from induced pluripotent stem cells deliver cardioprotective miRNAs and prevent cardiomyocyte apoptosis in the ischemic myocardium. Int J Cardiol. 2015;192:61-69. [70] ZHU H, FAN GC. Role of microRNAs in the reperfused myocardium towards post-infarct remodelling. Cardiovasc Res. 2012;94(2): 284-292. [71] XIAO J, PAN Y, LI XH, et al. Cardiac progenitor cell-derived exosomes prevent cardiomyocytes apoptosis through exosomal miR-21 by targeting PDCD4. Cell Death Dis. 2016;7(6):e2277. [72] DIAO H, LIU B, SHI Y, et al. MicroRNA-210 alleviates oxidative stress-associated cardiomyocyte apoptosis by regulating BNIP3. Biosci Biotechnol Biochem. 2017;81(9):1712-1720. [73] YANG PC. Induced Pluripotent Stem Cell (iPSC)-Derived Exosomes for Precision Medicine in Heart Failure. Circ Res. 2018;122(5): 661-663. [74] EL HARANE N, KERVADEC A, BELLAMY V, et al. Acellular therapeutic approach for heart failure: in vitro production of extracellular vesicles from human cardiovascular progenitors. Eur Heart J. 2018;39(20):1835-1847. [75] HU GW, LI Q, NIU X, et al. Exosomes secreted by human-induced pluripotent stem cell-derived mesenchymal stem cells attenuate limb ischemia by promoting angiogenesis in mice. Stem Cell Res Ther. 2015;6:10. [76] ZHANG J, GUAN J, NIU X, et al. Exosomes released from human induced pluripotent stem cells-derived MSCs facilitate cutaneous wound healing by promoting collagen synthesis and angiogenesis. J Transl Med. 2015;13:49. [77] YU B, KIM HW, GONG M, et al. Exosomes secreted from GATA-4 overexpressing mesenchymal stem cells serve as a reservoir of anti-apoptotic microRNAs for cardioprotection. Int J Cardiol. 2015; 182:349-360. [78] KHAN M, NICKOLOFF E, ABRAMOVA T, et al. Embryonic stem cell-derived exosomes promote endogenous repair mechanisms and enhance cardiac function following myocardial infarction. Circ Res. 2015;117(1):52-64. [79] ZHU W, HUANG L, LI Y, et al. Exosomes derived from human bone marrow mesenchymal stem cells promote tumor growth in vivo. Cancer Lett. 2012;315(1):28-37. [80] SUN L, XU R, SUN X, et al. Safety evaluation of exosomes derived from human umbilical cord mesenchymal stromal cell. Cytotherapy. 2016;18(3):413-422. [81] ADAMIAK M, CHENG G, BOBIS-WOZOWICZ S, et al. Induced Pluripotent Stem Cell (iPSC)-Derived Extracellular Vesicles Are Safer and More Effective for Cardiac Repair Than iPSCs. Circ Res. 2018; 122(2):296-309. |
| [1] | Pu Rui, Chen Ziyang, Yuan Lingyan. Characteristics and effects of exosomes from different cell sources in cardioprotection [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(在线): 1-. |
| [2] | Zhao Min, Feng Liuxiang, Chen Yao, Gu Xia, Wang Pingyi, Li Yimei, Li Wenhua. Exosomes as a disease marker under hypoxic conditions [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1104-1108. |
| [3] | Chen Ziyang, Pu Rui, Deng Shuang, Yuan Lingyan. Regulatory effect of exosomes on exercise-mediated insulin resistance diseases [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(25): 4089-4094. |
| [4] | Gao Kun, Chen Dayu, Zhang Yong, Liu Weidong, Sun Shufen, Lai Wenqiang, Ma Dujun, Wu Yihong, Lin Zhanpeng, Jiang Yinglu, Yu Weiji. Achyranthes bidentata alcohol extract inhibits extracellular matrix degradation of the cartilage by regulating synovial fibroblast exosomes [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(23): 3636-3640. |
| [5] | Xia Guoming, Xu Qiang, Liu Xuqiang, Yu Xiaolong, Dai Min. Application and effect of induced pluripotent stem cells in bone surgery tissue engineering [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(19): 3077-3082. |
| [6] | Wang Kang, Zhi Xiaodong, Wang Wei. Effect of stem cell derived exosomes on repairing peripheral nerve injury [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(19): 3083-3089. |
| [7] | Zhao Shuangdan, Zheng Jiahua, Qi Wenbo, Huang Xianghua. Role and mechanism of exosomes derived from mesenchymal stem cells in reproductive system diseases [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(19): 3097-3102. |
| [8] | Liu Feng, Zhang Yu, Wang Yanli, Luo Wei, Han Chaoshan, Li Yangxin. Application of temperature-sensitive chitosan hydrogel encapsulated exosomes in ischemic diseases [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(16): 2479-2487. |
| [9] | Cai Yuan, Deng Chengliang. Therapeutic application of adipose stem cell-free liquid extracts: skin aging, wound healing, scar recovery and nerve regeneration [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(13): 2097-2102. |
| [10] | Liu Tao, Zhang Nini, Huang Guilin . Relationship between extracellular vesicles and radiation-induced tissue injury [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(13): 2121-2126. |
| [11] | Ni Jinghua, Luo Jia, Jiang Sen, Li Heng, Zhu Jianzhong. Application and prospect of induced pluripotent stem cells in tumor diseases [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(13): 2127-2132. |
| [12] | Zhang Xiaobo, Zhang Jie, Sun Yu. Future hot spots for cartilage repair: laminin promotes stem cell proliferation [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(1): 141-145. |
| [13] | Chen Ganghong, Zeng Chaoming, Chen Ziming, Liao Junxing, Ma Yuanchen, Zheng Qiujian . Intra-articular injection of optimal concentration of bone marrow mesenchymal stem cells for treating rabbit cartilage defects [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(7): 996-1001. |
| [14] |
Xie Fei, Li Yanle, Lin Xinxiao, Hu Haiwei, Sang Zhicheng, Sun Yongsheng, Jiang Kewei, Cheng Ying, Wen Guannan, Wen Jianmin, Sun Weidong.
Mechanism underlying mechanical stress regulating fibroblasts-derived
exosomes at the osteotomized end following hallux valgus correction |
| [15] | Hu Chaoran, Qiu Bing, Zhou Zhuxing, Yang Yang, Li Jia. In vitro biocompatibility of 3D printed polycaprolactone/nano-hydroxyapatite composite scaffold with bone marrow mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(4): 589-595. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||