Chinese Journal of Tissue Engineering Research ›› 2021, Vol. 25 ›› Issue (19): 3083-3089.doi: 10.3969/j.issn.2095-4344.3528
Previous Articles Next Articles
Effect of stem cell derived exosomes on repairing peripheral nerve injury
Wang Kang1, Zhi Xiaodong1, 2, Wang Wei1, 2, 3
- 1Department of Orthopedics, First Affiliated Hospital of Jinzhou Medical University, Jinzhou 121000, Liaoning Province, China; 2Institute of Orthopedics, Jinzhou Medical University, Jinzhou 121000, Liaoning Province, China; 3Liaoning Key Laboratory of Medical Tissue Engineering, Jinzhou 121000, Liaoning Province, China
-
Received:2020-08-07Revised:2020-08-11Accepted:2020-09-15Online:2021-07-09Published:2021-01-14 -
Contact:Wang Wei, MD, Chief physician, Professor, Department of Orthopedics, First Affiliated Hospital of Jinzhou Medical University, Jinzhou 121000, Liaoning Province, China; Institute of Orthopedics, Jinzhou Medical University, Jinzhou 121000, Liaoning Province, China; Liaoning Key Laboratory of Medical Tissue Engineering, Jinzhou 121000, Liaoning Province, China -
About author:Wang Kang, Master candidate, Department of Orthopedics, First Affiliated Hospital of Jinzhou Medical University, Jinzhou 121000, Liaoning Province, China Zhi Xiaodong, MD, Chief physician, Department of Orthopedics, First Affiliated Hospital of Jinzhou Medical University, Jinzhou 121000, Liaoning Province, China; Institute of Orthopedics, Jinzhou Medical University, Jinzhou 121000, Liaoning Province, China -
Supported by:the Science and Technology Research Local Service Project of Department of Education of Liaoning Province, No. JYTFUDF201757 (to WW); the School-Enterprise Cooperation Fund Project of Jinzhou Medical University, No. 2020002 (to WW); the Science and Technology Fund Project of Liaoning Province, No. 20180551216 (to ZXD)
CLC Number:
Cite this article
Wang Kang, Zhi Xiaodong, Wang Wei. Effect of stem cell derived exosomes on repairing peripheral nerve injury[J]. Chinese Journal of Tissue Engineering Research, 2021, 25(19): 3083-3089.
share this article
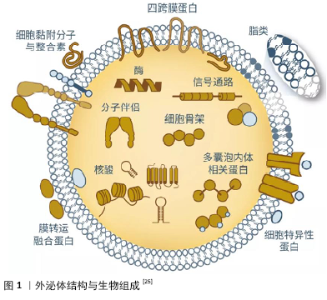
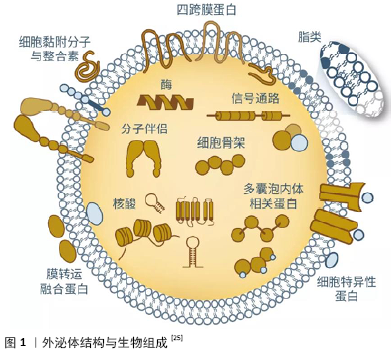
2.1 文献筛选 初检文献8 157篇,去除重复文献5 509篇,通过阅读文题、摘要排除无关文献2 325篇,阅读全文,排除不符合纳入标准的文献259篇,最终纳入64篇文章。 2.2 简要回顾:干细胞及干细胞源性外泌体在创伤修复中的研究进展 干细胞具有强大的自我更新能力,能够良好地维持机体组织器官内的稳定性,从而使得组织器官保持生长和衰亡的动态平衡。即使经连续传代培养冷冻保存后,干细胞仍可在体内或体外特定的诱导条件下用于衰老和病变引起的组织器官损伤修复。目前研究较多的干细胞包括骨髓间充质干细胞、胚胎干细胞、造血干细胞、脂肪间充质干细胞、牙髓干细胞、人脐带间充质干细胞等[11-15]。研究发现,在组织修复过程中,干细胞可通过肝细胞生长因子、转化生长因子β、白细胞介素10等抑制促炎细胞因子的产生和刺激抗炎细胞因子、抗原特异性T细胞的产生,还可通过促进细胞增殖、迁移或抑制细胞凋亡,介导免疫反应等途径修复创伤组织[16-18]。 随着干细胞治疗研究工作的深入,人们逐渐发现干细胞治疗具有诸多局限性,如细胞表型不稳定、成本高、移植前细胞来源和运输、干细胞移植时免疫排斥反应、致瘤性和安全性问题,限制了干细胞的临床应用。另外,许多先前被认为是干细胞的再生特性,被证明是通过分泌的外泌体来介导的,受到了广泛的关注[19-20]。 外泌体是存在于血液、尿液、乳汁等各种体液中的一种囊泡样小体,直径40-200 nm,呈杯状结构。外泌体由细胞质膜初次内陷形成内吞小体后,相互融合形成早期核内体,再次内陷包裹细胞内物质,形成多个腔内小囊泡,形成晚期核内体,即多泡体,多泡体与细胞质膜融合,释放腔内小囊泡到胞外[21-22]。外泌体包含着膜联蛋白、肌动蛋白和β-微管蛋白等与膜运输和融合相关的大量蛋白质,外泌体还富含特定的蛋白质标记,例如热休克蛋白和四跨膜蛋白(CD9、CD63、CD81、CD82)等,其中膜结合蛋白(例如主要组织相容性复合物分子)数量较少,因此其免疫原性低于其亲代细胞,外泌体还富含磷脂和胆固醇等脂类,其含量、组成与起源细胞类型密切相关,见图1。另外,外泌体携带有mRNA、核糖体RNA、长非编码RNA和一些细胞因子。外泌体通过水平转移这些物质来参与细胞间各种信息交流及物质交换,进行生物合成和吸收、免疫监视、微环境修饰、炎症调节等[23-25]。 干细胞来源外泌体的再生修复能力与其来源细胞功能密切相关,来源细胞的基本特性和分离获取难易程度决定了干细胞来源外泌体的应用前景。目前研究较多的外泌体来源于骨髓间充质干细胞、脂肪间充质干细胞、人脐带间充质干细胞等[26]。骨髓间充质干细胞取材较为方便,含量也相对较为丰富,可向外胚层和内胚层来源的神经元样细胞、胶质样细胞分化,且具有强大的成骨、软骨和脂肪等分化潜能。骨髓间充质干细胞来源外泌体具有与骨髓间充质干细胞相似的促进受损组织修复的作用,但骨髓间充质干细胞来源外泌体可能随着传代次数的增加对组织修复能力逐渐减弱[27];脂肪间充质干细胞是最易取材的一类干细胞,且脂肪间充质干细胞提取分离方法的不断改进,使其在组织修复中的研究工作逐渐迈向新阶段,但是由于脂肪间充质干细胞在增殖、分化、旁分泌、抗凋亡能力上存在性别和年龄差异,故脂肪间充质干细胞来源外泌体在临床研究和应用时可能面临解决个体化治疗等相关问题[28-29];人脐带间充质干细胞较其他干细胞分化能力更强、免疫原性低,对供者无影响,避免伦理争议等优势,且人脐带间充质干细胞相比于其他类型干细胞具有最强的神经分化能力,因此人脐带间充质干细胞来源外泌体可能较其他干细胞来源外泌体更能促进神经组织修复[30]。然而,目前各种干细胞来源外泌体的优缺点研究并未得到深入开展,相信这将是干细胞被进一步广泛应用于临床治疗的重要研究内容。 干细胞来源外泌体在各种病理诊断和治疗中的研究已广泛开展,如神经退行性疾病、创伤修复、抗衰老及肿瘤抑制等,有望成为预后分析和监测的新思路及方法[31-32]。与干细胞相比,较长时间反复局部给予相对低剂量干细胞来源外泌体,观察不到严重的免疫反应,其机制与免疫调节分子如程序性细胞死亡配体1和Fas/FasL的表达有关,因此干细胞来源外泌体具有较低的免疫原性,也避免了致瘤性问题,为进一步实现“无细胞治疗”策略提供了新的思路[33-34]。干细胞来源外泌体作为生物活性分子天然载体,被用于递送不同治疗物的有效携带物,包括短干扰RNA、反义寡核苷酸、化疗药物和免疫调节剂,并具有将其递送到预期目的地的能 力[35]。另外,在制备、储存、保存及其作为现成生物治疗剂方面,干细胞来源外泌体同样比干细胞具有更广泛的应用前景,如干细胞来源外泌体已应用于促进皮肤伤口愈合、促进肝再生、减少心肌梗死面积等领域,获得了良好的修复效果[36-38]。干细胞来源外泌体包含的多种miRNA和蛋白质在组织再生过程中发挥重要作用,如miRNA-191、miRNA-222和miRNA-21等参与调节细胞周期,miRNA-222、miRNA-21、let-7f参与调控血管再生[39];血小板源性生长因子受体B、表皮生长因子受体和尿激酶型纤溶酶原激活物受体通过参与调控信号通路调节干细胞分化方向;神经营养因子、表皮生长因子和转化生长因子β等具有营养神经元、促进轴突再生及轴突髓鞘化等作用[40-41]。MCBRIDE等[42]研究发现骨髓间充质干细胞来源外泌体能通过将Wnt3a蛋白分泌到无血清条件培养基,激活Wnt 信号通路,促进皮肤成纤维细胞的增殖、迁移和体外血管生成。HAN等[43]应用功能性肽水凝胶封装的人脐带间充质干细胞来源外泌体促进心脏修复,发现其可通过减少炎症、纤维化和细胞凋亡促进血管生成,改善心肌梗死模型大鼠的心脏功能。综上,干细胞来源外泌体已成功应用于转化医学、组织工程、临床治疗等方面,在组织损伤修复和再造方面具有重要的应用潜力。 2.3 干细胞来源外泌体对周围神经损伤修复的作用 2.3.1 干细胞来源外泌体调控miRNA细胞间的运输和表达促进轴突生长 外泌体介导的miRNA信息交换是一种重要的细胞间通讯方式,在神经系统损伤修复过程中起着关键作用。Wallerian变性过程中,在轴突或神经末梢发现大量影响细胞骨架组织的miRNA,其能抑制轴突生长,影响许旺细胞向非髓鞘表型去分化,是许旺细胞对神经元损伤反应的调节剂。YU等[44]对受损的大鼠坐骨神经组织进行了高通量测序,共识别出225个在坐骨神经受损后有显著表达差异的miRNA。通过生物信息学分析表明,这些特异性表达的miRNA,如miRNA-23a,miRNA-200,miRNA-133b,miRNA-17-92过表达 参与了许旺细胞的表型转换、轴突生长和巨噬细胞迁移、囊泡介导的转运、细胞黏附和细胞凋亡负调控。LOPEZ-LEAL等[10]研究结果显示干细胞来源外泌体携带的miRNA-9、 miRNA-19、miRNA-219、miRNA-222在损伤轴突中过表达,参与到许旺细胞增殖分化和神经发生的修复过程;CHING 等[45] 发现干细胞来源外泌体携带的miRNA-222通过靶向LASS2促进许旺细胞增殖和迁移,并促进PTEN表达使神经突向外生长,进一步揭示了干细胞来源外泌体通过miRNA促进神经再生修复的机制。EIRIN等[46]研究也证实了骨髓间充质干细胞来源外泌体可将miRNAs传递到轴突,上调许旺细胞的特征分子如p75神经营养因子受体、胶质纤维酸性蛋白和Sox10蛋白的表达,且骨髓间充质干细胞来源外泌体中的miRNA148a、miRNA532-5p、miRNA378和let-7f含量比骨髓间充质干细胞中更高。许多学者将干细胞来源外泌体介导miRNA参与微环境物质交流这一特性与基因工程相结合,取得了良好的效果,如DIDIOT等[47]将外泌体中的miRNA进行基因编辑后应用于Huntington疾病研究中,发现携带修饰化小干扰RNAs的外泌体能够被宿主神经元内化,并且能够引起miRNA和蛋白的差异性表达,这项研究提示可通过承载干细胞来源外泌体携带的miRNA来修饰原代神经细胞或许旺细胞,调节其增殖分化活性以及特定的生物活性分子分泌,从而转变其细胞生物特性和基因表达水平,靶向促进神经可塑性和轴突生长。 2.3.2 干细胞来源外泌体介导生长因子促进轴突再生 周围神经损伤后,神经元胞体出现变性损害,靶源性生长因子的供应链中断,此时神经细胞和许旺细胞中的生长因子信号通路不能被快速激活,阻碍神经再生[48]。研究表明在受损的神经组织中进行外源性神经生长因子补充,可提高残端神经生长因子的含量和促进循环稳态的恢复,有助于神经组织的再生与功能恢复[49]。BUCAN等[50]评估了脂肪间充质干细胞来源外泌体对坐骨神经再生和神经突生长的影响,发现脂肪间充质干细胞来源外泌体含有的神经营养因子、成纤维细胞生长因子1、脑源性神经营养因子、胰岛素样生长因子1和神经生长因子等可转移至许旺细胞,促进轴突再生和髓鞘形成。尽管这项研究并未深入揭示周围神经再生的具体机制,但它是第一个证明外泌体富含多种营养神经的生长因子。LIAO等[51-52]对干细胞来源外泌体传递神经营养因子机制进行了探究,发现生长因子可能协同联合干扰素γ和白细胞介素4,作用于肌苷激酶受体或低亲和力神经生长因子受体,在早期引起内源性炎症信号的激活,加速残端神经组织的再生,晚期抑制炎症反应,减少神经元变性,加速周围神经再生,且神经生长因子和胶质细胞源性神经营养因子促进许旺细胞迁移和细胞因子产生呈剂量依赖性,表现出协同作用和时间效应。除了本身携带有生长因子,干细胞来源外泌体还可通过激活损伤的神经细胞分泌各种活性生长因子,如Akt、ERK和STAT3等信号通路分子,诱导肝细胞生长因子、胰岛素样生长因子1、神经生长因子和基质衍生生长因子1分泌,这一研究进一步揭示了干细胞来源外泌体介导生长因子促进神经组织再生的作用机制[53]。干细胞来源外泌体中生长因子的存在以及诱导分泌信号通路的激活提供了调节萎缩神经存活和轴突生长的可能性,增加干细胞来源外泌体生物活性分子水平可能是加速周围神经损伤恢复的重要方法,如与基因工程技术相结合,人为合理调控细胞的神经生长因子目标基因表达,分泌理想的细胞生长因子,从而获得更好的周围神经损伤修复效果。 2.3.3 干细胞来源外泌体的炎症调节特性 周围神经损伤后,远端轴突开始退化并迅速产生促炎反应,许旺细胞首先在神经远端被激活,趋化因子和促炎细胞因子通过许旺细胞的促神经炎性反应得到释放。循环巨噬细胞、常驻巨噬细胞和其他外周免疫细胞也聚集到损伤部位促进髓鞘和轴突碎片的清除[54]。干细胞来源外泌体包含的内容物如转化生长因子β、干扰素γ、前列腺素E2、环氧合酶2等炎症调节因子的表达参与介导免疫炎症反应过程,在调控免疫炎症应答方面与干细胞相当,通过参与B细胞增殖活化,诱导调节性T细胞迁移,激活许旺细胞固有的免疫反应,从而使许旺细胞与炎症细胞相互协调以促进神经再生[55]。另外,干细胞来源外泌体的炎症调节特性还可能与其含有的炎症调节因子刺激树突状细胞、淋巴细胞、中性粒细胞等不同炎症细胞来源外泌体分泌,刺激免疫细胞来源外泌体分泌大量与炎症调节相关的miRNA、lncRNA、趋化因子、运载组胺、中性蛋白酶等炎症递质和细胞活性分子[56-57]。研究表明,绒毛膜间充质干细胞、骨髓间充质干细胞和人脐带间充质干细胞均能够调控炎症反应,下调受损背根神经节中炎性细胞因子水平,并有效触发骨髓衍生巨噬细胞从M1到M2表型的极化。有学者证明干细胞来源外泌体可以通过传递miRNA-30d-5p抑制小胶质细胞的自噬,从而最终将小胶质细胞极化为抗炎表型,从而改善神经元损伤,这项研究不仅解释了干细胞来源外泌体介导神经炎症促进神经发生的机制,而且还表明干细胞来源外泌体可能有希望作为减轻神经炎症基因表达的介导载体[58]。干细胞来源外泌体治疗有望成为周围神经损伤后局部免疫炎症调节的重要方法之一。 2.3.4 干细胞来源外泌体参与调控细胞凋亡、增殖过程 在周围神经损伤后所经历一系列复杂变化中,细胞组织凋亡的加速尤为显著,其中许旺细胞凋亡是重要的病理生理变化之一。有学者在研究中已注意到,干细胞来源外泌体可释放多种具有抗凋亡作用的miRNA,通过激活信号通路发挥抗许旺细胞凋亡作用,如AKT和 ERK信号通路[59]。干细胞来源外泌体还可促进神经调节蛋白的合成,而神经调节蛋白与许旺细胞增殖有关,促进许旺细胞表达成髓相关蛋白,干细胞来源外泌体还可影响氧化应激所引起的线粒体膜电位增加,从而抑制细胞凋亡,其机制与其携带的某些细胞因子如白细胞介素8、肿瘤坏死因子α等激活ERK1/2通路有关[60]。LIU等[61]研究也证实了脂肪间充质干细胞来源外泌体能够上调抗凋亡的Bcl-2 mRNA表达和下调促凋亡的Bax mRNA表达,Bcl-2/Bax的比率增加,降低周围神经损伤后许旺细胞的凋亡。因此,脂肪间充质干细胞来源外泌体通过减少许旺细胞凋亡、减轻其自噬作用并促进Büngner带形成,有利于神经元轴突再生,从而加速周围神经损伤的修复[62]。 2.3.5 干细胞来源外泌体参与受损周围神经的血管再生 周围神经损伤后微循环障碍是导致周围神经继发性损伤的重要原因,血管网络的重建为轴突生长提供了可再生的微环境,维持血管完整性是治疗周围神经再生的重要目标。研究表明干细胞来源外泌体可以诱导中枢神经系统的血管生成,充当血管内皮细胞的通讯介质,可提高神经损伤后血管的可塑性,促进内源性血管生成和神经生成,并减少大鼠的血管炎症反应[63]。VRIJSEN等[64]研究表明干细胞来源外泌体可介导内皮细胞的迁移和血管网形成,其机制与外泌体中高表达的细胞外基质金属蛋白酶诱导因子激活ERK/AKT信号通路有关。然而,干细胞来源外泌体在周围神经损伤中的血管修复作用尚未报道,有待进一步研究。 2.4 干细胞来源外泌体在周围神经损伤修复中的优势和挑战 干细胞来源外泌体所含的miRNA、circRNA、lncRNA和蛋白质等细胞因子参与细胞内的信号调控,通过经典的配体/受体结合作用和膜融合作用在细胞间传递生物活性物质,介导细胞间的信息交流,激活受体细胞内的信号通路,抑制免疫炎症反应、营养神经、调控细胞凋亡、促进血管生成等生理过程,发挥与干细胞类似的强大的组织修复再生和保护能力,并且还可避免干细胞移植时出现的免疫排斥、致瘤风险等问题。由于干细胞来源外泌体是脂溶性的,能自由穿过血管壁,通过血脑屏障,且具有良好的稳定性,有利于储存及运输,可作为良好的非免疫原性药物载体,增加装载药物的生物利用度,干细胞来源外泌体比干细胞具有更好的细胞外基质结合能力,这让干细胞来源外泌体成为了神经组织损伤修复“无细胞”治疗的新型策略。 目前,许多研究已经观察到干细胞来源外泌体可以在损伤后加速周围神经再生,但是尚未阐明神经再生的分子机制,就干细胞来源外泌体基础研究仍然有待深入。例如:①人羊膜间充质干细胞、肌源干细胞、牙髓干细胞等均是具有强大分化潜力的原始细胞,然而这些干细胞的外泌体在周围神经损伤修复中的应用研究并未得到深入开展;②干细胞来源外泌体研究仍受限于粒子检测和分离实验技术,以及无法在体内以可靠的分辨率成像和跟踪外泌体;③移植干细胞来源外泌体之后,是否能够与宿主神经细胞形成新的神经环路,起到恢复神经功能的作用;④干细胞来源外泌体储存方式对于修复效果的影响,以及干细胞来源外泌体制备的标准化程序及质控标准的构建等;⑤外源的干细胞来源外泌体与宿主细胞(许旺细胞、血管内皮细胞、炎症细胞等)自身产生的外泌体的相互作用。另外,干细胞来源外泌体与神经组织工程导管、基因治疗等方法高效结合的新型疗法也需进一步探索。因此,需要大量工作来克服上述困难与挑战,以实现干细胞来源外泌体早日广泛应用于临床研究中。 "
| [1] BHANDARI PS. Management of peripheral nerve injury. J Clin Orthop Trauma. 2019;10(5):862-866. [2] EHMEDAH A, NEDELJKOVIC P, DACIC S, et al. Vitamin B Complex Treatment Attenuates Local Inflammation after Peripheral Nerve Injury. Molecules. 2019;24(24):4615. [3] HUANG L, XIA B, SHI X, et al. Time-restricted release of multiple neurotrophic factors promotes axonal regeneration and functional recovery after peripheral nerve injury. FASEB J. 2019;33(7):8600-8613. [4] KORNFELD T, VOGT PM, RADTKE C. Nerve grafting for peripheral nerve injuries with extended defect sizes. Wien Med Wochenschr. 2019;169(9-10):240-251. [5] GU X, DING F, WILLIAMS DF. Neural tissue engineering options for peripheral nerve regeneration. Biomaterials. 2014;35(24):6143-6156. [6] GEUNA S, GNAVI S, PERROTEAU I, et al. Tissue engineering and peripheral nerve reconstruction: an overview. Int Rev Neurobiol. 2013;108:35-57. [7] BUSUTTIL F, RAHIM AA, PHILLIPS JB. Combining Gene and Stem Cell Therapy for Peripheral Nerve Tissue Engineering. Stem Cells Dev. 2017;26(4):231-238. [8] HAASTERT K, GROTHE C. Gene therapy in peripheral nerve reconstruction approaches. Curr Gene Ther. 2007;7(3):221-228. [9] SHI Y, ZHOU L, TIAN J, et al. Transplantation of neural stem cells overexpressing glia-derived neurotrophic factor promotes facial nerve regeneration. Acta Otolaryngol. 2009;129(8):906-914. [10] LOPEZ-LEAL R, COURT FA. Schwann Cell Exosomes Mediate Neuron-Glia Communication and Enhance Axonal Regeneration. Cell Mol Neurobiol. 2016;36(3):429-436. [11] CLEVERS H. STEM CELLS. What is an adult stem cell? Science. 2015; 350(6266):1319-1320. [12] DULAK J, SZADE K, SZADE A, et al. Adult stem cells: hopes and hypes of regenerative medicine. Acta Biochim Pol. 2015;62(3):329-337. [13] KOLIOS G, MOODLEY Y. Introduction to stem cells and regenerative medicine. Respiration. 2013;85(1):3-10. [14] SOBHANI A, KHANLARKHANI N, BAAZM M, et al. Multipotent Stem Cell and Current Application. Acta Med Iran. 2017;55(1):6-23. [15] LIU L, MICHOWSKI W, KOLODZIEJCZYK A, et al. The cell cycle in stem cell proliferation, pluripotency and differentiation. Nat Cell Biol. 2019; 21(9):1060-1067. [16] LIN Y, ZHANG F, LIAN XF, et al. Mesenchymal stem cell-derived exosomes improve diabetes mellitus-induced myocardial injury and fibrosis via inhibition of TGF-β1/Smad2 signaling pathway. Cell Mol Biol (Noisy-le-grand). 2019;65(7):123-126. [17] JI L, BAO L, GU Z, et al. Comparison of immunomodulatory properties of exosomes derived from bone marrow mesenchymal stem cells and dental pulp stem cells. Immunol Res. 2019;67(4-5):432-442. [18] CUI K, CHEN Y, ZHONG H, et al. Transplantation of IL-10-Overexpressing Bone Marrow-Derived Mesenchymal Stem Cells Ameliorates Diabetic-Induced Impaired Fracture Healing in Mice. Cell Mol Bioeng. 2019;13(2):155-163. [19] PARK S, CHOI Y, JUNG N, et al. Myogenic differentiation potential of human tonsil-derived mesenchymal stem cells and their potential for use to promote skeletal muscle regeneration. Int J Mol Med. 2016; 37(5):1209-1220. [20] MERINO A, RIPOLL E, DE RAMON L, et al. The Timing of Immunomodulation Induced by Mesenchymal Stromal Cells Determines the Outcome of the Graft in Experimental Renal Allotransplantation. Cell Transplant. 2017;26(6):1017-1030. [21] KALLURI R, LEBLEU VS. The biology, function, and biomedical applications of exosomes. Science. 2020;367(6478):eaau6977. [22] BANG C, THUM T. Exosomes: new players in cell-cell communication. Int J Biochem Cell Biol. 2012;44(11):2060-2064. [23] SKOTLAND T, SANDVIG K, LLORENTE A. Lipids in exosomes: Current knowledge and the way forward. Prog Lipid Res. 2017;66:30-41. [24] FAMILTSEVA A, JEREMIC N, TYAGI SC. Exosomes: cell-created drug delivery systems. Mol Cell Biochem. 2019;459(1-2):1-6. [25] COLOMBO M, RAPOSO G, THÉRY C. Biogenesis, secretion, and intercellular interactions of exosomes and other extracellular vesicles. Annu Rev Cell Dev Biol. 2014;30:255-289. [26] ABDIK H, AVSAR ABDIK E, HIZLI DENIZ AA, et al. A Novel Virtue in Stem Cell Research: Exosomes and Their Role in Differentiation. Adv Exp Med Biol. 2019;1144:133-146. [27] ZHOU W, LIN J, ZHAO K, et al. Single-Cell Profiles and Clinically Useful Properties of Human Mesenchymal Stem Cells of Adipose and Bone Marrow Origin. Am J Sports Med. 2019;47(7):1722-1733. [28] YANG D, LI N, ZHANG G. Spontaneous adipogenic differentiation potential of adipose‑derived stem cells decreased with increasing cell passages. Mol Med Rep. 2018;17(4):6109-6115. [29] SOWA Y, IMURA T, NUMAJIRI T, et al. Adipose-derived stem cells produce factors enhancing peripheral nerve regeneration: influence of age and anatomic site of origin. Stem Cells Dev. 2012;21(11): 1852-1862. [30] HENDRIJANTINI N, HARTONO P. Phenotype Characteristics and Osteogenic Differentiation Potential of Human Mesenchymal Stem Cells Derived from Amnion Membrane (HAMSCs) and Umbilical Cord (HUC-MSCs). Acta Inform Med. 2019;27(2):72-77. [31] LUDWIG N, WHITESIDE TL, REICHERT TE. Challenges in Exosome Isolation and Analysis in Health and Disease. Int J Mol Sci. 2019;20(19): 4684. [32] YANG B, CHEN Y, SHI J. Exosome Biochemistry and Advanced Nanotechnology for Next-Generation Theranostic Platforms. Adv Mater. 2019;31(2):e1802896. [33] CHEN G, HUANG AC, ZHANG W, et al. Exosomal PD-L1 contributes to immunosuppression and is associated with anti-PD-1 response. Nature. 2018;560(7718):382-386. [34] NING Y, SHEN K, WU Q, et al. Tumor exosomes block dendritic cells maturation to decrease the T cell immune response. Immunol Lett. 2018;199:36-43. [35] LI SP, LIN ZX, JIANG XY, et al. Exosomal cargo-loading and synthetic exosome-mimics as potential therapeutic tools. Acta Pharmacol Sin. 2018;39(4):542-551. [36] FERREIRA ADF, GOMES DA. Stem Cell Extracellular Vesicles in Skin Repair. Bioengineering (Basel). 2018;6(1):4. [37] LOU G, CHEN Z, ZHENG M, et al. Mesenchymal stem cell-derived exosomes as a new therapeutic strategy for liver diseases. Exp Mol Med. 2017;49(6):e346. [38] KHAN M, NICKOLOFF E, ABRAMOVA T, et al. Embryonic stem cell-derived exosomes promote endogenous repair mechanisms and enhance cardiac function following myocardial infarction. Circ Res. 2015;117(1):52-64. [39] DENG H, SUN C, SUN Y, et al. Lipid, Protein, and MicroRNA Composition Within Mesenchymal Stem Cell-Derived Exosomes. Cell Reprogram. 2018;20(3):178-186. [40] LI R, ZHAO K, RUAN Q, et al. Bone marrow mesenchymal stem cell-derived exosomal microRNA-124-3p attenuates neurological damage in spinal cord ischemia-reperfusion injury by downregulating Ern1 and promoting M2 macrophage polarization. Arthritis Res Ther. 2020;22(1):75. [41] MEAD B, CHAMLING X, ZACK DJ, et al. TNFα-Mediated Priming of Mesenchymal Stem Cells Enhances Their Neuroprotective Effect on Retinal Ganglion Cells. Invest Ophthalmol Vis Sci. 2020;61(2):6. [42] MCBRIDE JD, RODRIGUEZ-MENOCAL L, GUZMAN W, et al. Bone Marrow Mesenchymal Stem Cell-Derived CD63+ Exosomes Transport Wnt3a Exteriorly and Enhance Dermal Fibroblast Proliferation, Migration, and Angiogenesis In Vitro. Stem Cells Dev. 2017;26(19):1384-1398. [43] HAN C, ZHOU J, LIANG C, et al. Human umbilical cord mesenchymal stem cell derived exosomes encapsulated in functional peptide hydrogels promote cardiac repair. Biomater Sci. 2019;7(7):2920-2933. [44] YU B, ZHOU S, WANG Y, et al. Profile of microRNAs following rat sciatic nerve injury by deep sequencing: implication for mechanisms of nerve regeneration. PLoS One. 2011;6(9):e24612. [45] CHING RC, WIBERG M, KINGHAM PJ. Schwann cell-like differentiated adipose stem cells promote neurite outgrowth via secreted exosomes and RNA transfer. Stem Cell Res Ther. 2018;9(1):266. [46] EIRIN A, RIESTER SM, ZHU XY, et al. MicroRNA and mRNA cargo of extracellular vesicles from porcine adipose tissue-derived mesenchymal stem cells. Gene. 2014;551(1):55-64. [47] DIDIOT MC, HALL LM, COLES AH, et al. Exosome-mediated Delivery of Hydrophobically Modified siRNA for Huntingtin mRNA Silencing. Mol Ther. 2016;24(10):1836-1847. [48] RICHNER M, ULRICHSEN M, ELMEGAARD SL, et al. Peripheral nerve injury modulates neurotrophin signaling in the peripheral and central nervous system. Mol Neurobiol. 2014;50(3):945-970. [49] MOATTARI M, KOUCHESFEHANI HM, KAKA G, et al. Evaluation of nerve growth factor (NGF) treated mesenchymal stem cells for recovery in neurotmesis model of peripheral nerve injury. J Craniomaxillofac Surg. 2018;46(6):898-904. [50] BUCAN V, VASLAITIS D, PECK CT, et al. Effect of Exosomes from Rat Adipose-Derived Mesenchymal Stem Cells on Neurite Outgrowth and Sciatic Nerve Regeneration After Crush Injury. Mol Neurobiol. 2019;56(3):1812-1824. [51] LIAO CF, CHEN CC, LU YW, et al. Effects of endogenous inflammation signals elicited by nerve growth factor, interferon-γ, and interleukin-4 on peripheral nerve regeneration. J Biol Eng. 2019;13:86. [52] LACKINGTON WA, KOČÍ Z, ALEKSEEVA T, et al. Controlling the dose-dependent, synergistic and temporal effects of NGF and GDNF by encapsulation in PLGA microparticles for use in nerve guidance conduits for the repair of large peripheral nerve defects. J Control Release. 2019;304:51-64. [53] SHARMA P, MESCI P, CARROMEU C, et al. Exosomes regulate neurogenesis and circuit assembly. Proc Natl Acad Sci U S A. 2019; 116(32):16086-16094. [54] DUBOVÝ P, JANČÁLEK R, KUBEK T. Role of inflammation and cytokines in peripheral nerve regeneration. Int Rev Neurobiol. 2013;108:173-206. [55] CHAN BD, WONG WY, LEE MM, et al. Exosomes in Inflammation and Inflammatory Disease. Proteomics. 2019;19(8):e1800149. [56] TAN DBA, ARMITAGE J, TEO TH, et al. Elevated levels of circulating exosome in COPD patients are associated with systemic inflammation. Respir Med. 2017;132:261-264. [57] WANG T, NASSER MI, SHEN J, et al. Functions of Exosomes in the Triangular Relationship between the Tumor, Inflammation, and Immunity in the Tumor Microenvironment. J Immunol Res. 2019; 2019:4197829. [58] JIANG M, WANG H, JIN M, et al. Exosomes from MiR-30d-5p-ADSCs Reverse Acute Ischemic Stroke-Induced, Autophagy-Mediated Brain Injury by Promoting M2 Microglial/Macrophage Polarization. Cell Physiol Biochem. 2018;47(2):864-878. [59] SUN Z, SHI K, YANG S, et al. Effect of exosomal miRNA on cancer biology and clinical applications. Mol Cancer. 2018;17(1):147. [60] FIGLIOLINI F, RANGHINO A, GRANGE C, et al. Extracellular Vesicles From Adipose Stem Cells Prevent Muscle Damage and Inflammation in a Mouse Model of Hind Limb Ischemia: Role of Neuregulin-1. Arterioscler Thromb Vasc Biol. 2020;40(1):239-254. [61] LIU CY, YIN G, SUN YD, et al. Effect of exosomes from adipose-derived stem cells on the apoptosis of Schwann cells in peripheral nerve injury. CNS Neurosci Ther. 2020;26(2):189-196. [62] YIN G, LIU C, LIN Y, et al. Effect of exosomes from adipose-derived stem cells on peripheral nerve regeneration. Zhongguo Xiu Fu Chong Jian Wai Ke Za Zhi. 2018;32(12):1592-1596. [63] ZHANG Y, CHOPP M, MENG Y, et al. Effect of exosomes derived from multipluripotent mesenchymal stromal cells on functional recovery and neurovascular plasticity in rats after traumatic brain injury. J Neurosurg. 2015;122(4):856-867. [64] VRIJSEN KR, MARING JA, CHAMULEAU SA, et al. Exosomes from Cardiomyocyte Progenitor Cells and Mesenchymal Stem Cells Stimulate Angiogenesis Via EMMPRIN. Adv Healthc Mater. 2016;5(19): 2555-2565. |
| [1] | Pu Rui, Chen Ziyang, Yuan Lingyan. Characteristics and effects of exosomes from different cell sources in cardioprotection [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(在线): 1-. |
| [2] | Lin Qingfan, Xie Yixin, Chen Wanqing, Ye Zhenzhong, Chen Youfang. Human placenta-derived mesenchymal stem cell conditioned medium can upregulate BeWo cell viability and zonula occludens expression under hypoxia [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(在线): 4970-4975. |
| [3] | Zhang Tongtong, Wang Zhonghua, Wen Jie, Song Yuxin, Liu Lin. Application of three-dimensional printing model in surgical resection and reconstruction of cervical tumor [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1335-1339. |
| [4] | Zhang Chao, Lü Xin. Heterotopic ossification after acetabular fracture fixation: risk factors, prevention and treatment progress [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1434-1439. |
| [5] | Zhou Jihui, Li Xinzhi, Zhou You, Huang Wei, Chen Wenyao. Multiple problems in the selection of implants for patellar fracture [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1440-1445. |
| [6] | Wang Debin, Bi Zhenggang. Related problems in anatomy mechanics, injury characteristics, fixed repair and three-dimensional technology application for olecranon fracture-dislocations [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1446-1451. |
| [7] | Zhang Xiumei, Zhai Yunkai, Zhao Jie, Zhao Meng. Research hotspots of organoid models in recent 10 years: a search in domestic and foreign databases [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1249-1255. |
| [8] | Ji Zhixiang, Lan Changgong. Polymorphism of urate transporter in gout and its correlation with gout treatment [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1290-1298. |
| [9] | Yuan Mei, Zhang Xinxin, Guo Yisha, Bi Xia. Diagnostic potential of circulating microRNA in vascular cognitive impairment [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1299-1304. |
| [10] | Wang Zhengdong, Huang Na, Chen Jingxian, Zheng Zuobing, Hu Xinyu, Li Mei, Su Xiao, Su Xuesen, Yan Nan. Inhibitory effects of sodium butyrate on microglial activation and expression of inflammatory factors induced by fluorosis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1075-1080. |
| [11] | Wang Xianyao, Guan Yalin, Liu Zhongshan. Strategies for improving the therapeutic efficacy of mesenchymal stem cells in the treatment of nonhealing wounds [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1081-1087. |
| [12] | Wan Ran, Shi Xu, Liu Jingsong, Wang Yansong. Research progress in the treatment of spinal cord injury with mesenchymal stem cell secretome [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1088-1095. |
| [13] | Liao Chengcheng, An Jiaxing, Tan Zhangxue, Wang Qian, Liu Jianguo. Therapeutic target and application prospects of oral squamous cell carcinoma stem cells [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1096-1103. |
| [14] | Zhao Min, Feng Liuxiang, Chen Yao, Gu Xia, Wang Pingyi, Li Yimei, Li Wenhua. Exosomes as a disease marker under hypoxic conditions [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1104-1108. |
| [15] | Xie Wenjia, Xia Tianjiao, Zhou Qingyun, Liu Yujia, Gu Xiaoping. Role of microglia-mediated neuronal injury in neurodegenerative diseases [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1109-1115. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||