Chinese Journal of Tissue Engineering Research ›› 2021, Vol. 25 ›› Issue (19): 3090-3096.doi: 10.3969/j.issn.2095-4344.3504
Previous Articles Next Articles
Bladder cancer stem cell markers and related signaling pathways: antibody targeted therapy
Chen Jie1, Liao Chengcheng2, Chen Zhiwei1, Wang Yan3
- 1The Third Affiliated Hospital of Zunyi Medical University, Zunyi 563000, Guizhou Province, China; 2Key Laboratory of Oral Disease Research in Guizhou Province, Zunyi 563000, Guizhou Province, China; 3Department of Urology, The Third Affiliated Hospital of Zunyi Medical University (The First People’s Hospital of Zunyi City), Zunyi 563000, Guizhou Province, China
-
Received:2020-05-14Revised:2020-05-16Accepted:2020-06-19Online:2021-07-09Published:2021-01-14 -
Contact:Wang Yan, MD, Professor, Department of Urology, The Third Affiliated Hospital of Zunyi Medical University (The First People’s Hospital of Zunyi City), Zunyi 563000, Guizhou Province, China -
About author:Chen Jie, Master candidate, The Third Affiliated Hospital of Zunyi Medical University, Zunyi 563000, Guizhou Province, China -
Supported by:the National Natural Science Foundation of China, No. 81660121 (to WY)
CLC Number:
Cite this article
Chen Jie, Liao Chengcheng, Chen Zhiwei, Wang Yan. Bladder cancer stem cell markers and related signaling pathways: antibody targeted therapy[J]. Chinese Journal of Tissue Engineering Research, 2021, 25(19): 3090-3096.
share this article

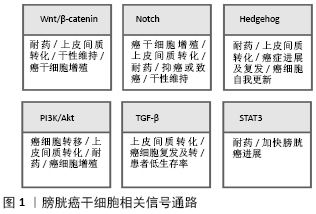
2.1 膀胱癌组织中的干细胞 膀胱癌干细胞是一类膀胱组织中具有多向分化潜能的特殊细胞,被认为是膀胱癌发生的启动细胞,可以诱发肿瘤的形成[11],主要表达包括CD44、CK5、CK17和层粘连蛋白受体等标志物[12]。为了识别和确定肿瘤来源干细胞,对来自同一个体的正常细胞和肿瘤干细胞进行了分析,发现在肿瘤干细胞中特异性表达一些干细胞标志物[11],其中OCT4是胚胎干细胞自我更新的关键调控因子,在人膀胱癌中过表达,与膀胱癌快速进展和侵袭性有关[13],另一种标记物是CD44,它是位于肿瘤尿路上皮基底细胞层的一种常见干细胞标记物[14]。基底层尿路上皮细胞的增殖依赖于基质成纤维细胞产生的Wnt信号[12]。对于依赖Gli-1的肿瘤,最初通过基质成纤维细胞SHH信号通路的旁分泌作用将切换到自分泌模式。一系列临床膀胱癌的发生与Hedgehog信号的激活有关[15]。目前大量研究证明以 Hedgehog、Wnt和Notch为主的信号通路参与了膀胱癌干细胞的生物学行为。 肿瘤干细胞是能够启动肿瘤的克隆性细胞,具有维持细胞异质性、自我更新和分化的能力[14],它们驱动肿瘤的生长、转移和对常规抗癌药物的耐药[16]。人们普遍认为肿瘤干细胞可能来自于通过复杂机制发生了基因突变的正常干细胞[17]。 此外,正常的尿路上皮干细胞和分化的基底细胞、中间细胞和移行上皮细胞也可能获得致癌潜能并转化为肿瘤干细 胞[8,11]。 2.2 膀胱癌干细胞标志物的研究进展 近年来,膀胱癌干细胞的分离和分子特征研究备受关注,建立一组肿瘤干细胞特异性的生物标志物显得非常重要,CD44、ALDH1A1、OCT4、YAP1、CD47、67LR和CEACAM6等蛋白质被认为是膀胱癌干细胞的标记物[18-19]。一部分肿瘤干细胞表面标志物已被确定与膀胱癌的发生、进展、侵袭性、干性的维持、转移和复发有关[20],见表1。识别具有重要影响的预后性标志物有助于为针对膀胱癌干细胞的治疗提供靶点从而更有效地治疗膀胱癌。 "

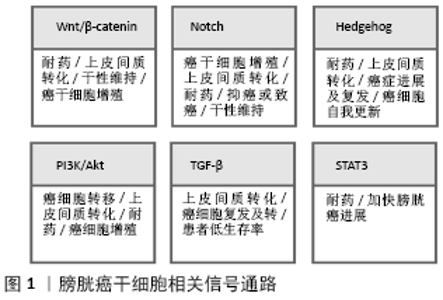
2.2.1 CD44 CD44是一个跨膜糖蛋白,在多种肿瘤中过表 达[18]。CD44+细胞位于正常尿路上皮的基底层[21]。CD44可与细胞外基质蛋白透明质酸相结合,透明质酸是细胞外基质的主要成分,由基质细胞和肿瘤细胞表达[22]。透明质酸与CD44配体结合域结合,引起构象改变,进而激活几条信号通路,导致细胞增殖、黏附、迁移和侵袭[23]。例如,CD44可以作为透明质酸的受体将信号传导入胞内激活Wnt/β-catenin 信号通路,故抑制CD44与透明质酸的结合会导致肿瘤细胞运动性丧失[24]。CD44还是一种独特的黏附分子,介导肿瘤细胞的迁移并黏附于细胞微环境中的基质上,从而增强肿瘤细胞的侵袭能力,促进肿瘤的增殖,抑制CD44的表达可显著降低肿瘤细胞形成克隆以及形成肿瘤的能力。此外,抑制透明质酸合成酶1、CD44v3、CD44v6和CD44s会使膀胱癌的生长、侵袭和血管生成行为消失[25]。 CHAN等[26]研究发现CD44+肿瘤细胞在免疫缺陷小鼠体内的癌变能力是CD44-肿瘤细胞的10-200倍,并且在300例膀胱癌患者组织中的表达分析表明,CD44+细胞亚群约占所有肿瘤细胞的40%。另一项研究也显示了类似的肿瘤干细胞亚群,并探讨了EMA-CD44v6+是膀胱癌启动细胞的分子标志物可能性:EMA-CD44v6+细胞约占肿瘤细胞总数的30%,体外单细胞克隆实验显示CD44+细胞具有更高的自我更新能力和与亲代肿瘤类似的克隆能力[27]。此外,特定的CD44 RNA剪接变异体,即CD44v8-10在膀胱癌中的过度表达已被证明与肿瘤的进展、侵袭性和转移密切相关[28]。有相关研究证明,CD44可作为膀胱癌的预后标志物[29]。 2.2.2 ALDH1A1 ALDH1属于乙醛脱氢酶家族,能够将细胞内乙醛氧化为乙酸,从而调控多种组织细胞分化和基因表达[30]。 ALDH1A1阳性细胞群在膀胱癌中富集,其上调与膀胱癌的进展、侵袭性、复发、生存期缩短和预后不良有关[31]。与正常膀胱组织相比,它是膀胱癌类器官中上调最多的基因之一[21]。SU等[30]采用Aldefluor法从膀胱癌细胞中分离ALDH1A1+细胞,然后通过体外和体内方法研究ALDH1A1+细胞的干细胞特性。研究表明ALDH1A1在216例膀胱癌组织中的表达率为26%(56/216),与膀胱移行上皮细胞癌的分期、组织学分级、复发进展及转移密切相关(P < 0.05)。此外,ALDH1A1的表达与患者的癌症特异性和总体生存率呈负相关(分别为P=0.027和0.030)。这些研究结果表明ALDH1A1+细胞可能特异性富集于膀胱癌的肿瘤干细胞。临床研究中发现,无论是低度恶性还是高度恶性的膀胱癌患者血清中ALDH总活性均显著升高,且具有较高的敏感性和特异性[32-33]。也有相关文献报道ALDH1的表达与非浸润性膀胱癌的临床病理分期、分级呈正相关关系,ALDH1蛋白高表达可能促进了非浸润性膀胱癌的发生、发展[34]。 因此,ALDH1A1可能是一个潜在的预后标志物,可以提高复发和进展患者临床诊断的准确性和选择最佳的治疗方案。目前,ALDH1作为一种新的肿瘤干细胞标志物应用于实体肿瘤干细胞的诊疗方案已获专利授权[35]。 2.2.3 OCT4 OCT4(又称POU5F1)是胚胎干细胞自我更新的关键调控因子之一,是在膀胱癌样本中表达的胚胎干细胞标记物[36]。ATLASI等[13]采用半定量RT-PCR方法检测32例膀胱肿瘤组织、13例非膀胱肿瘤组织和9例正常尿路上皮组织中Oct-4的mRNA表达。采用Western blot和免疫组织化学分析进一步检测Oct-4在蛋白水平的表达。实验结果表明OCT-4在几乎所有受检肿瘤中均有表达(31/32),但在部分非肿瘤性肿瘤(6/22)中表达水平较低(P < 0.001)。赵朋朋等[37]实验发现OCT4在膀胱癌组织中的表达率(76.1%)明显高于正常膀胱组织,其表达量与肿瘤病理分级、复发有密切相关性。同时也有文献报道OCT4在膀胱癌中的高表达被证明与人类膀胱癌的高级别及其手术后的复发有关[38]。这些结果表明OCT4可能是判断膀胱癌进展的一个有价值的临床诊断和预后标志物,并可能成为尿路上皮癌分子靶向治疗的一个引人注目的治疗靶点。 2.2.4 CD47 CD47是一种细胞表面蛋白,根据CHAN等[26]的研究,膀胱肿瘤的CD44+肿瘤干细胞表达免疫球蛋白样跨膜整合蛋白相关蛋白(IAP/CD47),被认为是巨噬细胞的抑制信号。CHAN等[10]在分选膀胱癌干细胞时,发现CD47不仅在膀胱癌细胞中广泛表达,并且在膀胱癌干细胞中具有显著的特异性表达。在体外使用单克隆抗体阻断CD47信号通路可以在诱导巨噬细胞对膀胱癌细胞的吞噬作用,在体内CD47mAb(抗CD47单克隆抗体)可使膀胱癌异种移植物的肿瘤体积减小和远处转移能力降低;此外,CD47在膀胱癌干细胞中高水平表达[8]。已有研究表明阻断CD47后会激活并增加免疫系统对肿瘤的清除能力[39]。CD47可能成为膀胱癌干细胞的一个潜在的治疗靶点,在体外单克隆抗体阻断CD47作为膀胱癌的一种潜在免疫治疗方式可诱导膀胱癌细胞中巨噬细胞吞噬,这提示CD47免疫治疗可有效运用肌层浸润性膀胱癌上。 2.2.5 YAP1 Yes相关蛋白(Yes-associated protein,YAP1)是Hippo肿瘤抑制途径的核心效应因子[40]。越来越多的证据表明,YAP1在肿瘤生长、免疫抑制和化疗耐药中具有重要的作用[41]。虽然其潜在机制还不清楚,但在不同的细胞成分中激活YAP1通路会诱导产生一个免疫抑制的肿瘤微环境。YAP1被证明是通过Hippo信号通路诱导膀胱癌细胞生长和侵袭[42]。相关研究使用Western blot和qRT-PCR方法检测YAP蛋白及其mRNA在正常膀胱上皮细胞及膀胱癌细胞中的表达,实验结果表明,膀胱癌细胞相对正常膀胱上皮细胞YAP高表达 (P < 0.05),且该实验进一步表明了YAP高表达能够增强膀胱癌干细胞特性[43]。 另一项研究表明,YAP1的辅因子MASK2对于YAP1促进膀胱癌的生长和迁移是必需的[44]。OOKI等[45]观察到YAP1和COX2/PGE2通路共同作用于膀胱癌干细胞的增殖,并且它们的抑制剂可成功阻止膀胱癌的进展。此外,YAP1具有将非肿瘤干细胞重新编程为具有肿瘤干细胞样细胞的能 力[46],并通过诱导自噬来维持肿瘤细胞的干性[47]。这些报道提示YAP1在膀胱癌干细胞和膀胱癌的生长和发展中起重要作用。 2.2.6 67LR和CEACAM6 67L-kD层粘连蛋白受体(67 kD laminin receptor,67LR)和癌胚抗原相关的细胞黏附分子6 (human carcinoembryonic antigen-related cell adhesion molecule 6,CEACAM6)是肌层浸润性膀胱癌的生物标志物,它们也可以用于膀胱癌干细胞的分离[48]。67LR在高侵袭性尿路上皮癌的肿瘤基质界面表达,约在80%的膀胱癌中表达上调。有趣的是,虽然67LR和CEACAM6通常都在基底细胞中表达,但通过选择67LR+和CEACAM6-表型也可以分离出肿瘤干细胞[49]。67LR和CEACAM6可能在膀胱癌治疗中作为有效的标志物而发挥重要作用。 2.3 膀胱癌干细胞相关信号通路的研究进展 了解肿瘤干细胞在膀胱癌中的作用及其调控机制可能有助于治疗和提供更好的预后。维持正常干细胞自我更新和分化的信号通路中某些关键基因发生突变,将会引起整个信号通路的紊乱或是过度激活,最终导致肿瘤的发生。SonicHedgeho信号通路[50-51]、与EMT相关的通路(如Wnt/β-catenin、Notch和TGF-β[52]、JAK-STAT通路[53]、MAPK[54]、PI3K/AKT通路[50]),都与膀胱癌干细胞的发生和进展有关,这些信号通路在膀胱癌细胞的增殖、分化、凋亡、衰老、自我更新等方面发挥着重要作用。一部分信号通路(如Notch、TGF-β、PI3K/Akt、SHH、Wnt/β-catenin)是通过参与激活上皮-间质转化而调控膀胱癌干细胞。PI3K/Akt可能通过与TGF-β和Wnt/β-catenin等信号通路相互作用而激活上皮-间质转化参与促进膀胱癌的进展,见图1。TGF-β诱导的SHH可能参与调节膀胱癌细胞的上皮-间质转化和致瘤性。因此,靶向这些信号通路在逆转膀胱癌细胞上皮-间质转化表型和潜在抑制膀胱癌进展和转移方面可能在临床上是有益的。 2.3.1 Wnt/β-catenin通路 Wnt信号在尿路上皮发育以及成人尿路上皮组织内稳态的维持中起重要作用[55]。分泌的Wnt配体与富含半胱氨酸结构域的7个跨膜G蛋白偶联受体家族成员Frizzleds(FZD)及其相关辅助受体结合,可以刺激复杂的酯化反应[56]。Wnt信号被分为2个路径:经典的Wnt (β-catenin依赖的)通路和非经典的Wnt(β-catenin不依赖的)通路。Wnt/β-catenin通路被证明是小鼠正常尿路上皮损伤后修复所必不可少的[57-58],并且在Uroplakin II作用下过度表达的β-catenin引发了转基因小鼠尿路上皮的良性增生[59]。在膀胱癌组织中,Wnt/β-catenin信号调节通路的突变或基因改变导致上皮-间质转化、尿路上皮出现干细胞表型,促进膀胱癌耐药、生存能力增强及膀胱癌成瘤[60]。膀胱癌高分化细胞中含有Wnt信号抑制分子,抑制Wnt信号通路分子的活性,导致下游分子β-catenin不能激活,而Wnt信号的过度激活可以引起肿瘤干细胞过度增殖[61]。在尿路上皮干细胞中,Wnt/β-catenin信号通路中40个基因的单核苷酸多态性基因分型揭示了Wnt/β-catenin通路的变异,这些变异被证明在膀胱癌的发病机制中起作用[62]。 另一份报道还显示,β-catenin的积聚和Wnt/β-catenin通路的失调诱导了膀胱癌的肿瘤细胞增殖并增加了侵袭的可能性[63]。此外,通过Lnc-RNA UCA1(尿路上皮癌相关1)增强Wnt6的表达,增加了膀胱癌细胞对顺铂和吉西他滨的耐药性[64]。这些研究结果表明,不管是Wnt/β-catenin信号通路本身的基因还是由于其他基因改变导致Wnt/β-catenin通路基因发生异常变化,对肿瘤干细胞的自我更新和肿瘤发生转移均有重要影响。 2.3.2 Notch信号通路 Notch信号通路主要调控正常干细胞的增殖、分化、凋亡和细胞间通讯;一部分Notch信号通路分子在正常干细胞中高表达,说明Notch信号通路与干细胞的自我更新密切相关[65]。此外,Notch通路在肿瘤发生过程中也很关键,Notch信号通路既有致癌作用又有肿瘤抑制作用,这取决于器官和细胞环境,它涉及4个受体(Notch1-4)和5个配体,并调节多个靶基因的转录:Notch1受体在膀胱癌中的表达减少,它的激活减少了细胞的增殖,这表明它具有肿瘤抑制作用[66]。另一方面,Notch2的过度表达促进了细胞的增殖、上皮-间质转化、侵袭和干性的维持,抑制Notch2的表达可以降低恶性表型[67]。此外,Notch3过表达促进了膀胱癌细胞的生长和耐药[68]。而有研究表明Notch通路对上皮-间质转化具有负调控作用,膀胱癌中的DNA甲基化可调控miR-193a-3p的高表达,从而抑制Notch通路,促进上皮-间质转化诱导的多药耐药[69]。由于Notch的这些相反作用和避免对其他器官产生不良反应,将需要更多的证据来支持癌症中Notch靶向治疗的发展。 2.3.3 Hedgehog信号通路 SonicHedgehog(SHH)是Hedgehog信号通路中的一种,是胚胎发育过程中器官发育和分化的重要组成部分。无论是Hedgehog信号通路中相关蛋白未被准确表达,还是干扰SHH信号通路的其他细胞因子表达,都可能引起不同类型肿瘤的发生,而这正是由于正常干细胞或祖细胞增殖分化的平衡被破坏而导致细胞失控增长引起的[70]。一旦SHH信号通路被异常激活,它就会触发癌症的发生和发展,并对化疗产生耐药[71]。SHH的激活促进了膀胱癌的上皮-间质转化,从而增加了膀胱癌的侵袭性、克隆性、致癌性和干性[52]。通过糖基转移酶GALNT1介导的糖基化激活SHH,并被证明能维持膀胱癌干细胞的自我更新和致癌潜能[72]。因此,将SHH作为靶点的治疗在逆转上皮-间质转化表型方面可能在临床上是有益的,并可能潜在地抑制膀胱癌的进展和侵袭。 2.3.4 PI3K/Akt通路 PI3K/Akt信号通路在维持细胞增殖和肿瘤细胞的生物学特性,包括细胞生长、分化、细胞周期、代谢、存活、凋亡、血管生成和迁移等方面起着至关重要的作用[73]。PI3K/Akt可能通过与NF-κB、RAS、TGF-β和Wnt/β-catenin等信号通路相互作用而激活上皮-间质转化参与调控肿瘤干细胞,影响肿瘤的侵袭性,包括整合素连接激酶活性和一系列相关转录因子(Twist、Snail和Slug)的激活,以及刺激基质降解蛋白酶(如基质金属蛋白酶)[74]。另外该途径与顺铂化疗耐药有关[75]。目前已有研究支持靶向PI3K/AKT通路能够起到抑制膀胱癌干细胞增殖和转移的治疗作用[76]。这些研究说明PI3K/AKT通路在膀胱癌的进展行为中有重要意义,可能成为膀胱癌干细胞治疗的一个潜在靶点。 2.3.5 TGF-β通路 TGF-β已被证明在细胞增殖、转移、生存和分化中发挥重要作用[77]。TGF-β调节的上皮-间质转化能使肿瘤上皮细胞转变为具有间质特性的细胞,肿瘤细胞由此获得侵袭性和转移性,进而从原发灶中逸出,再发生转移[78]。 根据是否被 Smads蛋白家族的调控,TGF-β调节上皮-间质转化的信号通路主要分为 Smads 依赖性和 Smads 非依赖性的2种类型。Islam等[51]实验结果证明了由TGF-β1诱导的细胞能够通过SHH途径显示干性样特征。LIANG等[77]通过建立膀胱癌模型,发现TGF-β信号传导被阻断后导致了膀胱癌干细胞数量减少。在膀胱癌中,TGF-β信号的激活增加了肿瘤的发生,并通过上调MALAT1的表达促进了上皮-间质转化,这与膀胱癌患者的低生存率有关。此外,MALAT1基因的敲除抑制了TGF-β诱导的上皮内皮细胞转化,其靶向抑制了TGF-β引发的转移和侵袭特性。这些研究提示MALAT1介导的TGF-β通路可能是阻止膀胱癌进展的一种有前途的治疗选择。 2.3.6 STAT3通路 STAT3在膀胱癌干细胞中被激活能加快侵袭性膀胱癌临床进程[79]。磷酸化的STAT3蛋白水平在膀胱癌肿瘤组织中上调,并与膀胱癌细胞的生长和存活有关[80]。Ho等[79]发现STAT3基因小鼠可以直接从原位癌发展为侵袭性癌症,绕过了非侵袭性乳头状肿瘤阶段,而由激活的STAT3驱动的侵袭性膀胱癌主要由膀胱癌干细胞组成。此外,KMT1A被证明可以通过KMT1A/GATA3/STAT3通路积极调节人类膀胱癌干细胞的自我更新和致癌性[81]。这些临床前数据表明,阻断癌细胞中STAT3激活的药物和方法在提高抗癌药物的敏感性或防止抗癌耐药性方面可能具有更大价值。"
| [1] FERLAY J, SHIN HR, BRAY F, et al. Estimates of worldwide burden of cancer in 2008: GLOBOCAN 2008. Int J Cancer. 2010;127(12): 2893-2917. [2] KAMAT AM, HAHN NM, EFSTATHIOU JA, et al. Bladder cancer. Lancet. 2016;388(10061):2796-2810. [3] LERNER SP, BAJORIN DF, DINNEY CP, et al. Summary and Recommendations from the National Cancer Institute’s Clinical Trials Planning Meeting on Novel Therapeutics for Non-Muscle Invasive Bladder Cancer. Bladder Cancer. 2016;2(2):165-202. [4] MALKOWICZ SB, VAN POPPEL H, MICKISCH G, et al. Muscle-invasive urothelial carcinoma of the bladder. Urology. 2007;69(1 Suppl):3-16. [5] ŚLUSARCZYK A, ZAPAŁA P, ZAPAŁA Ł, et al. Prediction of BCG responses in non-muscle-invasive bladder cancer in the era of novel immunotherapeutics. Int Urol Nephrol. 2019;51(7):1089-1099. [6] MITRA AP, COTE RJ. Searching for novel therapeutics and targets: insights from clinical trials. Urol Oncol. 2007;25(4):341-343. [7] VAN DER HORST G, BOS L, VAN DER PLUIJM G. Epithelial plasticity, cancer stem cells, and the tumor-supportive stroma in bladder carcinoma. Mol Cancer Res. 2012;10(8):995-1009. [8] OHISHI T, KOGA F, MIGITA T. Bladder Cancer Stem-Like Cells: Their Origin and Therapeutic Perspectives. Int J Mol Sci. 2015;17(1):43. [9] 王海明,石爱平,许宁.膀胱癌干细胞标记物的研究进展[J].中国老年学杂志,2014,34(21):6224-6226. [10] CHAN KS, VOLKMER JP, WEISSMAN I. Cancer stem cells in bladder cancer: a revisited and evolving concept. Curr Opin Urol. 2010;20(5): 393-397. [11] GARG M. Urothelial cancer stem cells and epithelial plasticity: current concepts and therapeutic implications in bladder cancer. Cancer Metastasis Rev. 2015;34(4):691-701. [12] HATINA J, SCHULZ WA. Stem cells in the biology of normal urothelium and urothelial carcinoma. Neoplasma. 2012;59(6):728-736. [13] ATLASI Y, MOWLA SJ, ZIAEE SA, et al. OCT-4, an embryonic stem cell marker, is highly expressed in bladder cancer. Int J Cancer. 2007;120(7): 1598-1602. [14] PASCAL LE, DEUTSCH EW, CAMPBELL DS, et al. The urologic epithelial stem cell database (UESC) - a web tool for cell type-specific gene expression and immunohistochemistry images of the prostate and bladder. BMC Urol. 2007;7:19. [15] FEI DL, LI H, KOZUL CD, et al. Activation of Hedgehog signaling by the environmental toxicant arsenic may contribute to the etiology of arsenic-induced tumors. Cancer Res. 2010 ;70(5):1981-1988. [16] ZHANG Y, WANG Z, YU J, et al. Cancer stem-like cells contribute to cisplatin resistance and progression in bladder cancer. Cancer Lett. 2012;322(1):70-77. [17] JORDAN CT. Cancer stem cells: controversial or just misunderstood? Cell Stem Cell. 2009;4(3):203-205. [18] KLONISCH T, WIECHEC E, HOMBACH-KLONISCH S, et al. Cancer stem cell markers in common cancers - therapeutic implications. Trends Mol Med. 2008;14(10):450-460. [19] WOODWARD WA, SULMAN EP. Cancer stem cells: markers or biomarkers? Cancer Metastasis Rev. 2008;27(3):459-470. [20] LI Y, LIN K, YANG Z, et al. Bladder cancer stem cells: clonal origin and therapeutic perspectives. Oncotarget. 2017;8(39):66668-66679. [21] ELBADAWY M, USUI T, MORI T, et al. Establishment of a novel experimental model for muscle-invasive bladder cancer using a dog bladder cancer organoid culture. Cancer Sci. 2019;110(9):2806-2821. [22] BANERJEE S, MODI S, MCGINN O, et al. Impaired Synthesis of Stromal Components in Response to Minnelide Improves Vascular Function, Drug Delivery, and Survival in Pancreatic Cancer. Clin Cancer Res. 2016; 22(2):415-425. [23] WANG L, ZUO X, XIE K, et al. The Role of CD44 and Cancer Stem Cells. Methods Mol Biol. 2018;1692:31-42. [24] SUGAHARA KN, HIRATA T, HAYASAKA H, et al. Tumor cells enhance their own CD44 cleavage and motility by generating hyaluronan fragments. J Biol Chem. 2006;281(9):5861-5868. [25] GOLSHANI R, LOPEZ L, ESTRELLA V, et al. Hyaluronic acid synthase-1 expression regulates bladder cancer growth, invasion, and angiogenesis through CD44. Cancer Res. 2008;68(2):483-491. [26] CHAN KS, ESPINOSA I, CHAO M, et al. Identification, molecular characterization, clinical prognosis, and therapeutic targeting of human bladder tumor-initiating cells. Version 2. Proc Natl Acad Sci U S A. 2009; 106(33):14016-14021. [27] YANG YM, CHANG JW. Bladder cancer initiating cells (BCICs) are among EMA-CD44v6+ subset: novel methods for isolating undetermined cancer stem (initiating) cells. Cancer Invest. 2008;26(7):725-733. [28] MURAMAKI M, MIYAKE H, KAMIDONO S, et al. Over expression of CD44V8-10 in human bladder cancer cells decreases their interaction with hyaluronic acid and potentiates their malignant progression. J Urol. 2004;171(1):426-430. [29] JUNG M, KIM B, MOON KC. Immunohistochemistry of cytokeratin (CK) 5/6, CD44 and CK20 as prognostic biomarkers of non-muscle-invasive papillary upper tract urothelial carcinoma. Histopathology. 2019;74(3):483-493. [30] SU Y, QIU Q, ZHANG X, et al. Aldehyde dehydrogenase 1 A1-positive cell population is enriched in tumor-initiating cells and associated with progression of bladder cancer. Cancer Epidemiol Biomarkers Prev. 2010;19(2):327-337. [31] NAMEKAWA T, IKEDA K, HORIE-INOUE K, et al. ALDH1A1 in patient-derived bladder cancer spheroids activates retinoic acid signaling leading to TUBB3 overexpression and tumor progression. Int J Cancer. 2020;146(4):1099-1113. [32] ORYWAL K, JELSKI W, WEREL T, et al. The Diagnostic Significance of Serum Alcohol Dehydrogenase Isoenzymes and Aldehyde Dehydrogenase Activity in Urinary Bladder Cancer Patients. Anticancer Res. 2017;37(7):3537-3541. [33] ORYWAL K, JELSKI W, WEREL T, et al. The activity of class I, II, III and IV alcohol dehydrogenase isoenzymes and aldehyde dehydrogenase in the sera of bladder cancer patients. Acta Biochim Pol. 2017;64(1):81-84. [34] 张海涛,金美善,石爱平,等.肿瘤干细胞标记物ALDH1在非浸润性膀胱癌组织中的表达及其临床意义[J].吉林大学学报(医学版), 2013,39(2):326-329. [35] TOMITA H, TANAKA K, TANAKA T, et al. Aldehyde dehydrogenase 1A1 in stem cells and cancer. Oncotarget. 2016;7(10):11018-11032. [36] EZEH UI, TUREK PJ, REIJO RA, et al. Human embryonic stem cell genes OCT4, NANOG, STELLAR, and GDF3 are expressed in both seminoma and breast carcinoma. Cancer. 2005;104(10):2255-2265. [37] 赵朋朋,刘春晓,许凯,等. OCT4在膀胱癌中的表达及其临床意义[J].南方医科大学学报,2012,32(5):643-646. [38] HUANG P, CHEN J, WANG L, et al. Implications of transcriptional factor, OCT-4, in human bladder malignancy and tumor recurrence. Med Oncol. 2012;29(2):829-834. [39] MCCRACKEN MN, CHA AC, WEISSMAN IL. Molecular Pathways: Activating T Cells after Cancer Cell Phagocytosis from Blockade of CD47 “Don’t Eat Me” Signals. Clin Cancer Res. 2015;21(16):3597-3601. [40] BEVERDAM A, CLAXTON C, ZHANG X, et al. Yap controls stem/progenitor cell proliferation in the mouse postnatal epidermis. J Invest Dermatol. 2013;133(6):1497-1505. [41] SHIBATA M, HAM K, HOQUE MO. A time for YAP1: Tumorigenesis, immunosuppression and targeted therapy. Int J Cancer. 2018;143(9): 2133-2144. [42] DONG L, LIN F, WU W, et al. Verteporfin inhibits YAP-induced bladder cancer cell growth and invasion via Hippo signaling pathway. Int J Med Sci. 2018;15(6):645-652. [43] 杨勇,姜睿.膀胱癌YAP表达及YAP对膀胱癌干细胞特性的影响[J].实用医学杂志,2018,34(19):3181-3184. [44] DONG L, LIN F, WU W, et al. Transcriptional cofactor Mask2 is required for YAP-induced cell growth and migration in bladder cancer cell. J Cancer. 2016;7(14):2132-2138. [45] OOKI A, DEL CARMEN RODRIGUEZ PENA M, MARCHIONNI L, et al. YAP1 and COX2 Coordinately Regulate Urothelial Cancer Stem-like Cells. Cancer Res. 2018;78(1):168-181. [46] ZANCONATO F, CORDENONSI M, PICCOLO S. YAP/TAZ at the Roots of Cancer. Cancer Cell. 2016;29(6):783-803. [47] GARCÍA-PRAT L, MARTÍNEZ-VICENTE M, PERDIGUERO E, et al. Autophagy maintains stemness by preventing senescence. Nature. 2016;529(7584):37-42. [48] EDRIS B, WEISKOPF K, VOLKMER AK, et al. Antibody therapy targeting the CD47 protein is effective in a model of aggressive metastatic leiomyosarcoma. Proc Natl Acad Sci U S A. 2012;109(17):6656-6661. [49] MOREL AP, LIÈVRE M, THOMAS C, et al. Generation of breast cancer stem cells through epithelial-mesenchymal transition. PLoS One. 2008; 3(8):e2888. [50] SHIN K, LIM A, ODEGAARD JI, et al. Cellular origin of bladder neoplasia and tissue dynamics of its progression to invasive carcinoma. Nat Cell Biol. 2014;16(5):469-478. [51] ISLAM SS, MOKHTARI RB, NOMAN AS, et al. Sonic hedgehog (Shh) signaling promotes tumorigenicity and stemness via activation of epithelial-to-mesenchymal transition (EMT) in bladder cancer. Mol Carcinog. 2016;55(5):537-551. [52] FANG D, KITAMURA H. Cancer stem cells and epithelial-mesenchymal transition in urothelial carcinoma: Possible pathways and potential therapeutic approaches. Int J Urol. 2018;25(1):7-17. [53] LU R, ZHANG YG, SUN J. STAT3 activation in infection and infection-associated cancer. Mol Cell Endocrinol. 2017;451:80-87. [54] ELBADAWY M, USUI T, YAMAWAKI H, et al. Novel Functions of Death-Associated Protein Kinases through Mitogen-Activated Protein Kinase-Related Signals. Int J Mol Sci. 2018;19(10):3031. [55] DIMOV I, VISNJIC M, STEFANOVIC V. Urothelial cancer stem cells. ScientificWorldJournal. 2010;10:1400-1415. [56] GARG M, MAURYA N. WNT/β-catenin signaling in urothelial carcinoma of bladder. World J Nephrol. 2019;8(5):83-94. [57] SHIN K, LEE J, GUO N, et al. Hedgehog/Wnt feedback supports regenerative proliferation of epithelial stem cells in bladder. Nature. 2011;472(7341):110-114. [58] PAPAFOTIOU G, PARASKEVOPOULOU V, VASILAKI E, et al. KRT14 marks a subpopulation of bladder basal cells with pivotal role in regeneration and tumorigenesis. Nat Commun. 2016;7:11914. [59] AHMAD I, MORTON JP, SINGH LB, et al. β-Catenin activation synergizes with PTEN loss to cause bladder cancer formation. Oncogene. 2011; 30(2):178-189. [60] FODDE R, BRABLETZ T. Wnt/beta-catenin signaling in cancer stemness and malignant behavior. Curr Opin Cell Biol. 2007;19(2):150-158. [61] YANG Y, CHENG Z, TANG H, et al. Neonatal Maternal Separation Impairs Prefrontal Cortical Myelination and Cognitive Functions in Rats Through Activation of Wnt Signaling. Cereb Cortex. 2017;27(5):2871-2884. [62] PIERZYNSKI JA, HILDEBRANDT MA, KAMAT AM, et al. Genetic Variants in the Wnt/β-Catenin Signaling Pathway as Indicators of Bladder Cancer Risk. J Urol. 2015;194(6):1771-1776. [63] KASTRITIS E, MURRAY S, KYRIAKOU F, et al. Somatic mutations of adenomatous polyposis coli gene and nuclear b-catenin accumulation have prognostic significance in invasive urothelial carcinomas: evidence for Wnt pathway implication. Int J Cancer. 2009;124(1):103-108. [64] PAN J, LI X, WU W, et al. Long non-coding RNA UCA1 promotes cisplatin/gemcitabine resistance through CREB modulating miR-196a-5p in bladder cancer cells. Cancer Lett. 2016;382(1):64-76. [65] CHEN J, CRABBE A, VAN DUPPEN V, et al. The notch signaling system is present in the postnatal pituitary: marked expression and regulatory activity in the newly discovered side population. Mol Endocrinol. 2006;20(12):3293-3307. [66] GORIKI A, SEILER R, WYATT AW, et al. Unravelling disparate roles of NOTCH in bladder cancer. Nat Rev Urol. 2018;15(6):345-357. [67] HAYASHI T, GUST KM, WYATT AW, et al. Not all NOTCH Is Created Equal: The Oncogenic Role of NOTCH2 in Bladder Cancer and Its Implications for Targeted Therapy. Clin Cancer Res. 2016;22(12):2981-2992. [68] ZHANG H, LIU L, LIU C, et al. Notch3 overexpression enhances progression and chemoresistance of urothelial carcinoma. Oncotarget. 2017;8(21):34362-34373. [69] CAI Z, ZHANG F, CHEN W, et al. miRNAs: A Promising Target in the Chemoresistance of Bladder Cancer. Onco Targets Ther. 2019;12: 11805-11816. [70] MEDINA V, CALVO MB, DÍAZ-PRADO S, et al. Hedgehog signalling as a target in cancer stem cells. Clin Transl Oncol. 2009;11(4):199-207. [71] USUI T, SAKURAI M, UMATA K, et al. Hedgehog Signals Mediate Anti-Cancer Drug Resistance in Three-Dimensional Primary Colorectal Cancer Organoid Culture. Int J Mol Sci. 2018;19(4):1098. [72] LI C, DU Y, YANG Z, et al. GALNT1-Mediated Glycosylation and Activation of Sonic Hedgehog Signaling Maintains the Self-Renewal and Tumor-Initiating Capacity of Bladder Cancer Stem Cells. Cancer Res. 2016;76(5):1273-1283. [73] LUNARDI A, WEBSTER KA, PAPA A, et al. Role of aberrant PI3K pathway activation in gallbladder tumorigenesis. Oncotarget. 2014;5(4): 894-900. [74] XU W, YANG Z, LU N. A new role for the PI3K/Akt signaling pathway in the epithelial-mesenchymal transition. Cell Adh Migr. 2015;9(4): 317-324. [75] HO JN, BYUN SS, LEE SE, et al. Multikinase inhibitor motesanib enhances the antitumor effect of cisplatin in cisplatin‑resistant human bladder cancer cells via apoptosis and the PI3K/Akt pathway. Oncol Rep. 2019;41(4):2482-2490. [76] ISKENDER B, IZGI K, CANATAN H. Novel anti-cancer agent myrtucommulone-A and thymoquinone abrogate epithelial-mesenchymal transition in cancer cells mainly through the inhibition of PI3K/AKT signalling axis. Mol Cell Biochem. 2016;416(1-2):71-84. [77] LIANG Y, ZHU F, ZHANG H, et al. Conditional ablation of TGF-β signaling inhibits tumor progression and invasion in an induced mouse bladder cancer model. Sci Rep. 2016;6:29479. [78] TAN EJ, OLSSON AK, MOUSTAKAS A. Reprogramming during epithelial to mesenchymal transition under the control of TGFβ. Cell Adh Migr. 2015;9(3):233-246. [79] HO PL, LAY EJ, JIAN W, et al. Stat3 activation in urothelial stem cells leads to direct progression to invasive bladder cancer. Cancer Res. 2012;72(13):3135-3142. [80] SUN Y, CHENG MK, GRIFFITHS TR, et al. Inhibition of STAT signalling in bladder cancer by diindolylmethane: relevance to cell adhesion, migration and proliferation. Curr Cancer Drug Targets. 2013;13(1): 57-68. [81] YANG Z, HE L, LIN K, et al. The KMT1A-GATA3-STAT3 Circuit Is a Novel Self-Renewal Signaling of Human Bladder Cancer Stem Cells. Clin Cancer Res. 2017;23(21):6673-6685. |
| [1] | Pu Rui, Chen Ziyang, Yuan Lingyan. Characteristics and effects of exosomes from different cell sources in cardioprotection [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(在线): 1-. |
| [2] | Lin Qingfan, Xie Yixin, Chen Wanqing, Ye Zhenzhong, Chen Youfang. Human placenta-derived mesenchymal stem cell conditioned medium can upregulate BeWo cell viability and zonula occludens expression under hypoxia [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(在线): 4970-4975. |
| [3] | Yuan Jiawei, Zhang Haitao, Jie Ke, Cao Houran, Zeng Yirong. Underlying targets and mechanism of Taohong Siwu Decoction in prosthetic joint infection on network pharmacology [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1428-1433. |
| [4] | Zhang Chao, Lü Xin. Heterotopic ossification after acetabular fracture fixation: risk factors, prevention and treatment progress [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1434-1439. |
| [5] | Zhou Jihui, Li Xinzhi, Zhou You, Huang Wei, Chen Wenyao. Multiple problems in the selection of implants for patellar fracture [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1440-1445. |
| [6] | Wang Debin, Bi Zhenggang. Related problems in anatomy mechanics, injury characteristics, fixed repair and three-dimensional technology application for olecranon fracture-dislocations [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1446-1451. |
| [7] | Gu Xia, Zhao Min, Wang Pingyi, Li Yimei, Li Wenhua. Relationship between hypoxia inducible factor 1 alpha and hypoxia signaling pathway [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1284-1289. |
| [8] | Ji Zhixiang, Lan Changgong. Polymorphism of urate transporter in gout and its correlation with gout treatment [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1290-1298. |
| [9] | Yuan Mei, Zhang Xinxin, Guo Yisha, Bi Xia. Diagnostic potential of circulating microRNA in vascular cognitive impairment [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1299-1304. |
| [10] | Zhang Xiumei, Zhai Yunkai, Zhao Jie, Zhao Meng. Research hotspots of organoid models in recent 10 years: a search in domestic and foreign databases [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1249-1255. |
| [11] | Wang Zhengdong, Huang Na, Chen Jingxian, Zheng Zuobing, Hu Xinyu, Li Mei, Su Xiao, Su Xuesen, Yan Nan. Inhibitory effects of sodium butyrate on microglial activation and expression of inflammatory factors induced by fluorosis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1075-1080. |
| [12] | Wang Xianyao, Guan Yalin, Liu Zhongshan. Strategies for improving the therapeutic efficacy of mesenchymal stem cells in the treatment of nonhealing wounds [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1081-1087. |
| [13] | Wan Ran, Shi Xu, Liu Jingsong, Wang Yansong. Research progress in the treatment of spinal cord injury with mesenchymal stem cell secretome [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1088-1095. |
| [14] | Liao Chengcheng, An Jiaxing, Tan Zhangxue, Wang Qian, Liu Jianguo. Therapeutic target and application prospects of oral squamous cell carcinoma stem cells [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1096-1103. |
| [15] | Zhao Min, Feng Liuxiang, Chen Yao, Gu Xia, Wang Pingyi, Li Yimei, Li Wenhua. Exosomes as a disease marker under hypoxic conditions [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1104-1108. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||