Chinese Journal of Tissue Engineering Research ›› 2021, Vol. 25 ›› Issue (13): 2127-2132.doi: 10.3969/j.issn.2095-4344.2189
Application and prospect of induced pluripotent stem cells in tumor diseases
Ni Jinghua1, 2, 3, 4, Luo Jia1, 2, 3, 4, Jiang Sen1, 2, 3, 4, Li Heng5, Zhu Jianzhong1, 2, 3, 4
- 1College Veterinary Medicine, 2Comparative Medicine Research Institute, Yangzhou University, Yangzhou 225009, Jiangsu Province, China; 3Jiangsu Co-innovation Center for Prevention and Control of Important Animal Infectious Diseases and Zoonoses, Yangzhou 225009, Jiangsu Province, China; 4Joint International Research Laboratory of Agriculture and Agri-Product Safety, Yangzhou 225009, Jiangsu Province, China; 5Suzhou Truway Biotech Inc., Suzhou 215411, Jiangsu Province, China
-
Received:2020-04-11Revised:2020-04-23Accepted:2020-05-27Online:2021-05-08Published:2020-12-29 -
Contact:Zhu Jianzhong, MD, Professor, College Veterinary Medicine, Yangzhou University, Yangzhou 225009, Jiangsu Province, China; Comparative Medicine Research Institute, Yangzhou University, Yangzhou 225009, Jiangsu Province, China; Jiangsu Co-innovation Center for Prevention and Control of Important Animal Infectious Diseases and Zoonoses, Yangzhou 225009, Jiangsu Province, China; Joint International Research Laboratory of Agriculture and Agri-Product Safety, Yangzhou 225009, Jiangsu Province, China -
About author:Ni Jinghua, Master candidate, College Veterinary Medicine, Yangzhou University, Yangzhou 225009, Jiangsu Province, China; Comparative Medicine Research Institute, Yangzhou University, Yangzhou 225009, Jiangsu Province, China; Jiangsu Co-innovation Center for Prevention and Control of Important Animal Infectious Diseases and Zoonoses, Yangzhou 225009, Jiangsu Province, China; Joint International Research Laboratory of Agriculture and Agri-Product Safety, Yangzhou 225009, Jiangsu Province, China
CLC Number:
Cite this article
Ni Jinghua, Luo Jia, Jiang Sen, Li Heng, Zhu Jianzhong. Application and prospect of induced pluripotent stem cells in tumor diseases[J]. Chinese Journal of Tissue Engineering Research, 2021, 25(13): 2127-2132.
share this article
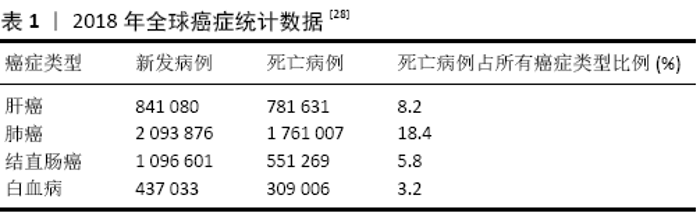
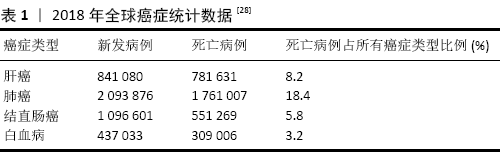
2.1 诱导多能干细胞技术 诱导多能干细胞最初是日本科学家SHINYA YAMANAKA于2006年利用病毒载体将4个转录因子OSKM(Oct4、Sox2、Klf4和c-Myc)组合转入分化的体细胞中,使其重编程而得到的类似胚胎干细胞的一种细胞类 型[12]。随后世界各地不同科学家陆续发现其他方法同样也可以制备这种细胞。迄今为止,诱导多能干细胞可以从各种物种中产生,包括人类、小鼠、大鼠和恒河猴等[13]。不同的转录因子组合可用于诱导不同细胞类型的间充质干细胞,例如,小鼠成纤维细胞需要OSKM来重新编程,神经干细胞只需要Oct4诱导重编程。造成这种现象的原因可能是不同细胞类型的基因表达水平不同[14]。由于c-Myc和Klf4在细胞中过表达时具有直接的致瘤潜能,为提高重编程效率,降低潜在成瘤性,重新编程也可以采用其他转录因子,例如Lin28与Oct4和Sox2结合,成功生成了诱导多能干细胞[15]。通过这种方法可以比OKSM更高效地获得高质量的诱导多能干细胞。同样,GLIS1(Jdp2、Id1、Jhdm1b、Lrh1、Sall4和Glis1)也可以代替OKSM[16]。目前,有许多产生诱导多能干细胞的方法和途径,包括反转录病毒转导[17]、DNA构建[18]、非整合游离体[19]、非整合仙台病毒[20]、非整合修饰的mRNA[21]、转座子[22]、一些小分子化合物等[23]。诱导多能干细胞技术在干细胞、表观遗传学以及生物医学等研究领域都引起了强烈的反响,使人们对多能性调控机制有了突破性的新认识,进一步拉近了干细胞和临床疾病治疗的距离。 2.2 诱导多能干细胞在肿瘤疾病中的应用 癌症是人和动物主要疾病之一,在世界范围内发病率不断上升,见表1。与此同时,癌症预防和治疗方面取得了进展,延长了患者生存期,甚至得到治愈。癌症治疗创新有赖于对肿瘤生物学基础的了解,针对癌症信号通路关键蛋白的治疗性小分子和抗体提高了特定癌症患者的存活率,但对许多其他种类癌症来说,治疗选择仍然有限,对耐药机制的了解也很有限[24]。因此,科学家努力确定癌症的遗传特征,从而加深对其在癌症发生和治疗作用的认识[25]。肿瘤干细胞的存在被认为是肿瘤的起源,其在肿瘤组织中存在的数量较少。与正常干细胞相似,肿瘤干细胞具有自我更新和分化的能力,可以增加肿瘤细胞的数量[26]。目前,诱导多能干细胞或重编程技术在生物医学研究中得到了广泛的应用,利用这些技术可以将癌细胞重新编程为多能细胞,并使其分化为目标细胞系,从而可以观察到癌症的不同进展阶段,研究不同亚群的肿瘤发生过程[27]。诱导多能干细胞的出现为肿瘤疾病的研究和治疗带来了新的思路,下面以肝癌、肺癌、结直肠癌、骨肉瘤和白血病等为代表,简述诱导多能干细胞在肿瘤疾病中的应用。 "
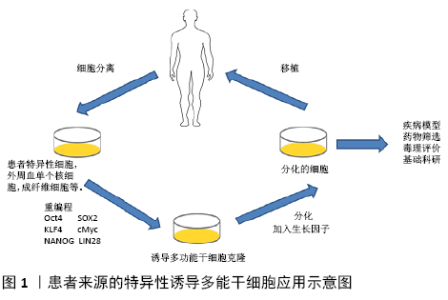
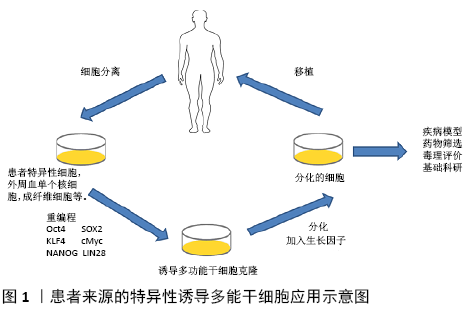
2.2.1 肝癌 肝癌是异质性最强、死亡率最高的难治性恶性肿瘤。肝癌的病死率位居肿瘤第二,仅次于肺癌,发病率逐年上升[29]。肝细胞是肝实质的主要细胞类型,表达多种肝脏功能所必需的代谢酶。在肝发育过程中,胎肝的肝祖细胞同时分化为成熟的肝细胞和胆管细胞。肝祖细胞具有双潜能和高增殖能力,可作为肝移植细胞的潜在来源或肝脏研究模型。通过诱导多能干细胞可以获得具有个体特异性的肝细胞,能有效避免排斥反应,是极具潜力的供移植应用的细胞类别[30]。肝脏肿瘤干细胞标志物包括CD13、CD24、CD44、CD90、CD133和EpCAM,这些标记可引起肿瘤的高度侵袭性特点及耐药性。近年来,利用激光诱导单细胞融合技术将肝癌HepG2细胞系与人胚胎干细胞融合,这种融合细胞表现出肝脏肿瘤干细胞的特征,如CD133和EPCAM标记的存在,以及阿霉素耐药性。融合细胞在小鼠异种移植模型中能够促进肿瘤的发生[31]。CD44是一种肿瘤干细胞标记,存在肝癌细胞、急性白血病中。HAN等[32]利用来源于HepG2的人肝细胞癌细胞系C3A,将OSKM转染到C3A细胞中,得到了C3A衍生的肝肿瘤干细胞模型(C3A-iCSCs),然后使用CRISPER/Cas9 系统敲除C3A-iCSCs中的CD44,与CD44+C3A-iCSCs相比,CD44-C3A-iCSCs在小鼠中表现出较高的多能性,在异种移植后分化增加。这项研究表明,CD44通过保持低分化的肿瘤细胞特性,在肝细胞中发挥重要功能。乙型肝炎病毒 (hepatitis B virus,HBV)和丙型肝炎病毒(hepatitis C virus,HCV)感染常导致肝癌,目前许多研究利用正常的诱导多能干细胞分化成肝细胞作为体外平台来研究HBV和HCV感染[33]。 KANEKO等[34]开发了用于HBV感染的诱导多能干细胞来源的肝祖细胞系(IPS-HPCs)培养模型。受感染的细胞可以产生HBV颗粒并启动先天免疫反应。此外,牛磺胆酸钠共转运多肽(NTCP)在IPS-HPCs中的过表达可为研究HBV感染提供平台。这些模型有助于了解宿主病毒的相互作用,并为HBV的治疗确定新的靶点。C8orf4又叫做甲状腺癌1(TC1),广泛存在于多种脊椎动物中并具有进化保守性,在多种肿瘤中具有高表达并对肿瘤发生具有重要作用。ZHU等[35]通过分析C8orf4在胚胎干细胞和诱导多能干细胞中的表达谱,发现C8orf4的基因在肝细胞癌和肝癌干细胞中表达非常微弱。C8orf4能够减弱肝癌干细胞的自我更新能力和肿瘤扩散能力,同时在肝癌干细胞中NOTCH2信号途径被激活。C8orf4定位于肝癌肿瘤细胞的细胞质,能够与NOTCH2胞内结构域发生相互作用,进而减弱N2ICD的核转定位。删除C8orf4会引起N2ICD的核转定位,触发NOTCH2信号途径,从而维持肝癌干细胞的活性,同时NOTCH2激活水平与临床肝癌患者的病情严重性和预后具有一致性。这项研究表明C8orf4能够通过抑制NOTCH2信号途径负向调控肝癌干细胞的自我更新,对于解决肝癌治疗过程中出现的化疗抵抗和复发难题具有重要生物学意义。 2.2.2 肺癌 肺癌是起源于肺部支气管黏膜或腺体的恶性肿瘤[36]。全球范围内,肺癌的发病率、死亡率都极高且呈上升趋势。2018年全球统计数据显示,男性肺癌的发病率和死亡率占恶性肿瘤的第一位[36]。目前的治疗方法只能在有限的时间范围内减轻终末期肺部疾病患者的症状或延缓疾病的进展。从自我更新的细胞群中复制出人类肺部疾病模型,将为更广泛地研究人类肺部疾病创造机会。利用新的基因编辑技术,从患有肺部疾病的患者身上获得经过基因修正的呼吸道上皮祖细胞可以提供一种潜在的治疗方法。诱导多能干细胞提供了来自患者和疾病的无限来源的特异型细胞,这些细胞可以分化成各种不同的细胞类型,这使它成为一个理想的模型系统,以研究肺部疾病病理并开发新的临床前治疗方法。FIRTH等[37]在研究中描述了人类诱导多能干细胞向功能性呼吸道上皮细胞的分化,展示了极化上皮层中多纤毛细胞和基底细胞的产生。诱导多能干细胞通过定形内胚层分化为前部前肠内胚层、肺内胚层,然后在气液界面培养中成熟,获得功能性假复层极化上皮。在气液界面培养中,将肺上皮基面暴露于液体介质和空气中,产生极化的上皮,显示有CC10阳性囊泡的Club细胞、MUC5A/C阳性杯状细胞以及CK5、P63和PDPN阳性基底细胞。基底层表达的间充质干细胞标记物是极化上皮层分化和维持所必需的,因此,诱导多能干细胞定向分化为包括多纤毛细胞在内的人类气道上皮细胞提供了一种可再生来源。最近,GHAEDI等[38]描述了一种产生效率更高的肺泡上皮细胞的方案。研究表明,在相对同质的Ⅱ型肺泡上皮细胞(AEC-II)中,SP-C的表达高达97%,粘蛋白的表达为95%,SP-B的表达为93%,CD54的表达为89%。将诱导多能干细胞衍生的AEC-Ⅱ细胞暴露于Wnt/β-catenin抑制剂IWR-1中,使表型从AEC-Ⅱ细胞转化为AEC-Ⅰ细胞,超过90%表达AEC-Ⅰ细胞标记T-1和Calveolin-1。在适当的培养条件下,诱导多能干细胞衍生的肺祖细胞黏附并重新整合了脱细胞化的肺细胞外基质。该研究组还描述了气液界面培养在旋转生物反应器中的应用,用于组织工程和药物研发的肺泡上皮细胞的大规模生产。肺移植是许多终末期患者的唯一确定治疗方法,但由于缺乏合适的供肺和移植排斥反应,其临床影响有限。每年有超过10%的等待移植患者在移植前死亡[39]。干细胞研究的最新发现引起了人们对开发可再生受伤/病变肺的极大兴趣。对动物肺损伤模型的研究表明,在不同的解剖区域有不同的祖细胞群负责维持肺上皮细胞的稳态。近年来,从胚胎干细胞和诱导多能干细胞中提取的肺祖细胞的纯化和鉴定取得了重大进展。这些来自胚胎干细胞/诱导多能干细胞的肺祖细胞对理解肺干细胞生物学和疾病过程、开发基于细胞治疗不可治愈的肺部疾病以及产生生物工程肺有很大的希望[40]。组织工程的研究已经强有力地证明工程功能器官如心脏、肝脏和肾脏的潜力[41]。工程肺是治疗终末期肺部疾病的一个有希望的方向,一些研究表明已经能够利用合成支架在三维培养系统中培养胎儿和成人肺细胞,从而在体外构建工程肺组织[42]。 2.2.3 结直肠癌 结直肠癌是临床上最常见的消化系统恶性肿瘤,近年来其发病率和病死率在世界范围内均呈上升趋势[43]。诱导多能干细胞分化为结肠干细胞的能力可以为模拟患者特异性胃肠疾病提供一个平台,如2009年大肠类器官已经通过Lgr5肠干细胞生成[44]。此外表达OKSM的人原发性结肠癌细胞转化为类肿瘤干细胞并呈现转移状态,在小鼠体内连续移植后仍得以被保留。与肿瘤干细胞相关的信号分子GLI2在转移细胞中高度表达,导致患者预后不良。因此,OSKM诱导的结肠癌细胞也可以作为研究肿瘤干细胞的模型,或作为癌症治疗和癌症治疗药物筛选的模型[45]。近年来,通过使用CRISPR/Cas9来靶向结肠癌的腺瘤-癌序列,将健康的人类结肠器官转化为相应的癌组织得到应用。APC、TP53、SMAD等抑癌基因的破坏以及KRAS、PI3K等致癌基因的遗传编辑使生成体外结肠癌的类器官模型成为可能[46-47]。 FAP是一种遗传性结直肠癌,由腺瘤性息肉病基因(APC)突变引起[48]。2017年,CRESPO等[49]从胚胎干细胞/诱导多能干细胞中分化出结肠类器官细胞。利用家族性腺瘤性息肉病患者的成纤维细胞生成诱导多能干细胞(FAP-IPSCs),然后生成结肠类器官细胞。在FAP-IPSCs中可以观察到Wnt活性与促进细胞增殖和疾病表型相关。该平台可用于药物筛选实验,是研究肿瘤进展的良好工具。DROST等[50]也通过删除结肠器官中的DNA修复基因,模拟了不匹配修复缺陷的结肠直肠癌。除了贴壁细胞系外,ROPER等[51]最近的一项研究证明了CRISPR编辑的结肠类器官原位移植的可能:患者在原位移植时,肿瘤来源器官样体再现了组织浸润和转移。因此,将基于CRISPR的器官筛选与原位移植相结合可能是研究疾病进展不同阶段中肿瘤驱动因素的合适方法。JI等[52]利用正常结肠干细胞的类组织发现了腺苷酸激酶HCINAP可以通过增强LDHA的磷酸化和活性来促进结直肠癌干细胞的有氧糖酵解,进而促进上皮间充质转化、迁移、侵袭、重新成瘤、自我更新。HCINAP的敲低能抑制细胞有氧糖酵解,促进线粒体呼吸和活性氧的产生,进而促进结直肠癌干细胞的凋亡。研究表明,葡萄糖水平低等代谢压力引起的细胞ATP水平降低会抑制LDHA的磷酸化并促进HCINAP和LDHA的相互作用,而在结直肠癌中HCINAP的异常高表达能通过促进LDHA磷酸化来促进糖酵解产生能量,从而提高结直肠癌干细胞对营养匮乏的抵抗性,证明HCINAP是一个具有促癌特性的腺苷酸激酶,结直肠癌干细胞的代谢过程被HCINAP严格调控。因此,利用HCINAP的小分子抑制剂或单克隆抗体杀灭结直肠癌细胞具有潜在的临床价值。 2.2.4 骨肉瘤 骨肉瘤是最常见的恶性骨肿瘤,其几乎占儿童骨肉瘤组织亚类的60%,它最常见于骨骼的骨骺[53]。目前,骨肉瘤的主要治疗方法是手术和化疗。关于骨肉瘤的细胞来源有两种主要的假说:间充质干细胞和成骨细胞。间充质干细胞Tp53、Rb1、Myc、Ras等特异性突变可能导致间充质干细胞形成骨肉瘤。另一方面,成骨细胞分化缺陷被认为是骨肉瘤发生的原因[54]。然而,骨肉瘤形成的分子机制仍需进一步研究。结合诱导多能干细胞模式研究与骨肉瘤相关的基因功能,无疑将为更好地理解骨肿瘤的发生、发展、进展和治疗提供可评估的平台。LFS是p53抑癌基因突变引起的常染色体显性综合征。LFS患者通常出现多种肿瘤的早期病症,包括骨肉瘤、乳腺癌、肉瘤和白血病[55]。由于骨肉瘤是影响LFS家族的主要癌症之一,2015年,LEE等[56]研究了p53突变对成骨细胞分化的影响,发现来自LFS-iPSC的成骨细胞具备骨肉瘤特征,包括缺陷分化和致瘤潜能。此外,他们发现印迹H19基因在LFS衍生的成骨细胞中表达受损,H19表达的恢复改善了成骨细胞的分化特性。LFS小鼠模型还不能完全概括该疾病,但使用LFS患者来源的诱导多能干细胞,它们可以具备骨肉瘤特征和基因表达特征,并识别具有潜在临床意义的显著失调基因。利用该模型,P53-H19被确定为骨肉瘤发生的关键调控通路。这些发现证明了用诱导多能干细胞研究人类遗传性肿瘤综合征的可行性。2019年HAN等[57]鉴定了一类新的骨膜中胚层间质前体细胞,在此细胞群中敲除肿瘤抑制基因Lkb1可导致骨肉瘤的发生。通过在组织蛋白酶K(CathepsinK,Ctsk)阳性细胞中特异性敲除Lkb1,发现敲除小鼠出现类似人类骨肉瘤的表现,并且通过谱系追踪的方法证明骨膜来源的Ctsk阳性细胞是成骨前体细胞,具有形成骨肉瘤的潜能。这项工作借助骨肉瘤模型,论证了Ctsk阳性细胞的成骨干性,并进一步通过基因敲除小鼠和药物处理模型论证了LKB1-mTORC1通路在骨肉瘤中的作用,为骨肉瘤细胞分型和靶向治疗提供了新的思路和研究基础。 2.2.5 白血病 白血病是一类造血干细胞恶性克隆性疾病,克隆性白血病细胞因为增殖失控、分化障碍、凋亡受阻等机制在骨髓和其他造血组织中大量增殖累积,并浸润其他非造血组织和器官,同时抑制正常造血功能,进而导致造血组织功能障碍的血液系统疾病。临床上患者常出现不同程度的贫血、出血、感染发热以及肝、脾、淋巴结肿大。白血病的发病率为3/10万-4/10 万,白血病为中、青年发病率和病死率最高的恶性肿瘤[58]。白血病可分为4种类型:急性髓系白血病、急性淋巴细胞白血病、慢性髓系白血病和慢性淋巴细胞白血病[59]。诱导多能干细胞为治疗包括白血病在内的多种疾病打开了一扇有希望的窗户。造血干细胞或造血母细胞是一种多能干细胞,可通过造血作用向所有血细胞分化。造血干细胞移植是目前治疗白血病最先进、最有效的方法。造血干细胞移植可用于治疗急性淋巴细胞白血病、急性髓系白血病、慢性髓系白血病、霍奇金淋巴瘤(复发/难治性)、非霍奇金淋巴瘤、骨髓增生异常综合征、多发性骨髓瘤等恶性疾病;还有非恶性疾病,如地中海贫血、镰状细胞贫血、凡可尼贫血、再生障碍性贫血、免疫缺陷综合征和先天性代谢紊乱[60]。造血干细胞移植最重要的临床挑战是在移植的黄金时期缺乏相容的供体和免疫排斥风险。诱导多能干细胞技术的进步使得从血液恶性肿瘤患者中创造出健康的多能干细胞成为可能。2015年,SALCI等[61]利用诱导多能干技术从急性髓系白血病患者的成纤维细胞中产生造血干细胞,他们发现这些造血干细胞具有正常的祖细胞状态,没有白血病突变。LIU等[62]成功制作了小鼠急性髓系白血病模型。他们通过在含有OKSM因子的造血细胞中过度表达人MLL-AF9融合基因,创造了嵌合诱导多能干细胞的小鼠。这些小鼠由于MLL-AF9的激活而自发发展为急性髓系白血病。研究表明,操纵MLL-AF9基因表达模式的表观遗传调控因子是MLL-AF9白血病细胞与衍生的诱导多能干细胞之间的桥梁。KUMANO等[63]从伊马替尼敏感的慢性髓系白血病患者中提取了诱导多能干细胞。这些诱导多能干细胞继续表达BCR-ABL蛋白,但对伊马替尼耐药。他们被证明可以维持诱导多能干细胞信号来补偿BCR-ABL的抑制。这些细胞在转化为造血系后恢复了对伊马替尼的易感性。2017年,KOTINI等[64]收集了包括白血病前期、低风险骨髓增生异常综合征、高风险骨髓增生异常综合征和继发性急性髓系白血病等一系列疾病阶段的诱导多能干细胞系。在分化过程中发现了严重程度分级的造血表型和阶段特异性,描绘了疾病进展的表型路线图。这表明疾病阶段的转变,包括逆转和进展,可以在这个系统中使用基因编辑引入突变进行建模,并且这种基于诱导多能干细胞的方法可以用于揭示药物对疾病阶段的特异性反应。此研究为区分骨髓转化起始和进展提供了方向,并为测试遗传和药理干预提供了一个新的平台。2019年,XU等[65]首次利用第3代基因编辑技术CRISPR-Cas9在造血干细胞和祖细胞中编辑CCR5基因并成功移植到1名同时患有HIV和急性淋巴细胞白血病的27岁男性患者。研究表明,基因编辑后的造血干细胞移植治疗使患者的急性淋巴白血病得到完全缓解,携带CCR5突变的供者细胞能够在受者体内长期存活可达19个月,初步探索了该方法的可行性和安全性。在成体造血干细胞上的基因编辑并不会对其他组织器官及生殖系统产生影响。该研究初步证明了基因编辑的成体造血干细胞移植的可行性和在人体内的安全性,促进和推动基因编辑技术在临床应用领域的发展。 "
| [1] HEMBERGER M, HANNA CW, DEAN W. Mechanisms of early placental development in mouse and humans. Nat Rev Genet. 2020;21(1):27-43. [2] TU X, ZHANG QR, ZHANG W, et al. Single-cell data-driven mathematical model reveals possible molecular mechanisms of embryonic stem-cell differentiation. Math Biosci Eng. 2019;16(5):5877-5896. [3] D’SOUZA GX, ROSE SE, KNUPP A, et al. The application of in vitro-derived human neurons in neurodegenerative disease modeling. J Neurosci Res. 2020 Mar 13. doi: 10.1002/jnr.24615. Online ahead of print. [4] SAMAK M, HINKEL R. Stem Cells in Cardiovascular Medicine: Historical Overview and Future Prospects. Cells. 2019;8(12):1530. [5] DANIEL MG, RAPP K, SCHANIEL C, et al. Induction of developmental hematopoiesis mediated by transcription factors and the hematopoietic microenvironment. Ann N Y Acad Sci. 2020;1466(1):59-72. [6] DEGUCHI S, TAKAYAMA K, MIZUGUCHI H. Generation of Human Induced Pluripotent Stem Cell-Derived Hepatocyte-Like Cells for Cellular Medicine. Biol Pharm Bull. 2020;43(4):608-615. [7] ALAVI M, BARANOV P. The iPSc-Derived Retinal Tissue as a Tool to Study Growth Factor Production in the Eye. Adv Exp Med Biol. 2018;1074:619-624. [8] MIRANDA CC, FERNANDES TG, DIOGO MM, et al. Human Pluripotent Stem Cells: Applications and Challenges for Regenerative Medicine and Disease Modeling. Adv Biochem Eng Biotechnol. 2020;171:189-224. [9] TAKAHASHI K, YAMANAKA S. Induction of pluripotent stem cells from mouse embryonic and adult fibroblast cultures by defined factors. Cell. 2006;126(4): 663-676. [10] ISHINO Y, SHINAGAWA H, MAKINO K, et al. Nucleotide sequence of the iap gene, responsible for alkaline phosphatase isozyme conversion in Escherichia coli, and identification of the gene product. J Bacteriol. 1987;169(12):5429-5433. [11] HSU PD, LANDER ES, ZHANG F. Development and applications of CRISPR-Cas9 for genome engineering. Cell. 2014;157(6):1262-1278. [12] OKITA K, ICHISAKA T, YAMANAKA S. Generation of germline-competent induced pluripotent stem cells. Nature. 2007;448(7151):313-317. [13] YOSHIDA Y, YAMANAKA S. Induced Pluripotent Stem Cells 10 Years Later: For Cardiac Applications. Circ Res. 2017;120(12):1958-1968. [14] WEN W, ZHANG JP, CHEN W, et al. Generation of Integration-free Induced Pluripotent Stem Cells from Human Peripheral Blood Mononuclear Cells Using Episomal Vectors. J Vis Exp. 2017;(119):55091. [15] WANG L, SU Y, HUANG C, et al. NANOG and LIN28 dramatically improve human cell reprogramming by modulating LIN41 and canonical WNT activities. Biol Open. 2019;8(12):bio047225. [16] LIU J, HAN Q, PENG T, et al. The oncogene c-Jun impedes somatic cell reprogramming. Nat Cell Biol. 2015;17(7):856-867. [17] AL ABBAR A, NORDIN N, GHAZALLI N, et al. Generation of induced pluripotent stem cells by a polycistronic lentiviral vector in feeder- and serum- free defined culture. Tissue Cell. 2018 ;55:13-24. [18] LEE CH, INGROLE RSJ, GILL HS. Generation of induced pluripotent stem cells using elastin like polypeptides as a non-viral gene delivery system. Biochim Biophys Acta Mol Basis Dis. 2020;1866(4):165405. [19] SLAMECKA J, SALIMOVA L, MCCLELLAN S, et al. Non-integrating episomal plasmid-based reprogramming of human amniotic fluid stem cells into induced pluripotent stem cells in chemically defined conditions. Cell Cycle. 2016;15(2):234-249. [20] TAI L, TEOH HK, CHEONG SK. Reprogramming human dermal fibroblast into induced pluripotent stem cells using nonintegrative Sendai virus for transduction. Malays J Pathol. 2018;40(3):325-329. [21] MCGRATH PS, DIETTE N, KOGUT I, et al. RNA-based Reprogramming of Human Primary Fibroblasts into Induced Pluripotent Stem Cells. J Vis Exp. 2018; (141): 10.3791/58687. [22] HEW BE, SATO R, MAURO D, et al. RNA-guided piggyBac transposition in human cells. Synth Biol (Oxf). 2019;4(1):ysz018. [23] CHEN G, GUO Y, LI C, et al. Small Molecules that Promote Self-Renewal of Stem Cells and Somatic Cell Reprogramming. Stem Cell Rev Rep. 2020;16(3):511-523. [24] KIM C. Disease modeling and cell based therapy with iPSC: future therapeutic option with fast and safe application. Blood Res. 2014;49(1):7-14. [25] WEN WS, YUAN ZM, MA SJ, et al. CRISPR-Cas9 systems: versatile cancer modelling platforms and promising therapeutic strategies. Int J Cancer. 2016;138(6): 1328-1336. [26] BATLLE E, CLEVERS H. Cancer stem cells revisited. Nat Med. 2017;23(10): 1124-1134. [27] CHAO HM, CHERN E. Patient-derived induced pluripotent stem cells for models of cancer and cancer stem cell research. J Formos Med Assoc. 2018;117(12): 1046-1057. [28] BRAY F, FERLAY J, SOERJOMATARAM I, et al. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2018;68(6):394-424. [29] DASGUPTA P, HENSHAW C, YOULDEN DR, et al. Global trends in incidence rates of childhood liver cancers: A systematic review and meta-analysis. Paediatr Perinat Epidemiol. 2020 Apr 26. doi: 10.1111/ppe.12671. Online ahead of print. [30] ASHMORE-HARRIS C, BLACKFORD SJ, GRIMSDELL B, et al. Reporter gene-engineering of human induced pluripotent stem cells during differentiation renders in vivo traceable hepatocyte-like cells accessible. Stem Cell Res. 2019; 41:101599. [31] WANG R, CHEN S, LI C, et al. Fusion with stem cell makes the hepatocellular carcinoma cells similar to liver tumor-initiating cells. BMC Cancer. 2016;16:56. [32] HAN S, GUO J, LIU Y, et al. Knock out CD44 in reprogrammed liver cancer cell C3A increases CSCs stemness and promotes differentiation. Oncotarget. 2015; 6(42):44452-44465. [33] KIDO T, KOUI Y, SUZUKI K, et al. CPM Is a Useful Cell Surface Marker to Isolate Expandable Bi-Potential Liver Progenitor Cells Derived from Human iPS Cells. Stem Cell Reports. 2015;5(4):508-515. [34] KANEKO S, KAKINUMA S, ASAHINA Y, et al. Human induced pluripotent stem cell-derived hepatic cell lines as a new model for host interaction with hepatitis B virus. Sci Rep. 2016;6:29358. [35] ZHU P, WANG Y, DU Y, et al. C8orf4 negatively regulates self-renewal of liver cancer stem cells via suppression of NOTCH2 signalling. Nat Commun. 2015;6: 7122. [36] HOFMAN P. Advances in theranostic biomarkers for lung cancer from clinical to molecular pathology. J Thorac Dis. 2019;11(Suppl 1):S1-S2. [37] FIRTH AL, DARGITZ CT, QUALLS SJ, et al. Generation of multiciliated cells in functional airway epithelia from human induced pluripotent stem cells. Proc Natl Acad Sci U S A. 2014;111(17):E1723-1730. [38] GHAEDI M, MENDEZ JJ, BOVE PF, et al. Alveolar epithelial differentiation of human induced pluripotent stem cells in a rotating bioreactor. Biomaterials. 2014;35(2):699-710. [39] KIRK R, DIPCHAND AI, ROSENTHAL DN, et al. The International Society for Heart and Lung Transplantation Guidelines for the management of pediatric heart failure: Executive summary. [Corrected]. J Heart Lung Transplant. 2014;33(9): 888-909. [40] QUAN Y, WANG D. Clinical potentials of human pluripotent stem cells in lung diseases. Clin Transl Med. 2014;3:15. [41] ASNAGHI A, MACCHIARINI P, MANTERO S. Tissue engineering toward organ replacement: a promising approach in airway transplant. Int J Artif Organs. 2009; 32(11):763-768. [42] GUENTHART BA, O’NEILL JD, KIM J, et al. Cell replacement in human lung bioengineering. J Heart Lung Transplant. 2019;38(2):215-224. [43] FERLAY J, COLOMBET M, SOERJOMATARAM I, et al. Estimating the global cancer incidence and mortality in 2018: GLOBOCAN sources and methods. Int J Cancer. 2019;144(8):1941-1953. [44] SATO T, VRIES RG, SNIPPERT HJ, et al. Single Lgr5 stem cells build crypt-villus structures in vitro without a mesenchymal niche. Nature. 2009;459(7244): 262-265. [45] SINGOVSKI G, BERNAL C, KUCIAK M, et al. In vivo epigenetic reprogramming of primary human colon cancer cells enhances metastases. J Mol Cell Biol. 2016; 8(2):157-173. [46] CONG L, RAN FA, COX D, et al. Multiplex genome engineering using CRISPR/Cas systems. Science. 2013;339(6121):819-823. [47] MATANO M, DATE S, SHIMOKAWA M, et al. Modeling colorectal cancer using CRISPR-Cas9-mediated engineering of human intestinal organoids. Nat Med. 2015;21(3):256-262. [48] SASIKUMAR R, REJITHA JR, BINUMON PK, et al. Role of heterozygous APC mutation in niche succession and initiation of colorectal cancer--a computational study. PLoS One. 2011;6(8):e22720. [49] CRESPO M, VILAR E, TSAI SY, et al. Colonic organoids derived from human induced pluripotent stem cells for modeling colorectal cancer and drug testing. Nat Med. 2017;23(7):878-884. [50] DROST J, VAN BOXTEL R, BLOKZIJL F, et al. Use of CRISPR-modified human stem cell organoids to study the origin of mutational signatures in cancer. Science. 2017;358(6360):234-238. [51] ROPER J, TAMMELA T, CETINBAS NM, et al. In vivo genome editing and organoid transplantation models of colorectal cancer and metastasis. Nat Biotechnol. 2017;35(6):569-576. [52] JI Y, YANG C, TANG Z, et al. Adenylate kinase hCINAP determines self-renewal of colorectal cancer stem cells by facilitating LDHA phosphorylation. Nat Commun. 2017;8:15308. [53] SADYKOVA LR, NTEKIM AI, MUYANGWA-SEMENOVA M, et al. Epidemiology and Risk Factors of Osteosarcoma. Cancer Invest. 2020. doi: 10.1080/07357907.2020.1768401. Online ahead of print. [54] LIN YH, JEWELL BE, GINGOLD J, et al. Osteosarcoma: Molecular Pathogenesis and iPSC Modeling. Trends Mol Med. 2017;23(8):737-755. [55] MASCIARI S, DEWANWALA A, STOFFEL EM, et al. Gastric cancer in individuals with Li-Fraumeni syndrome. Genet Med. 2011;13(7):651-657. [56] LEE DF, SU J, KIM HS, et al. Modeling familial cancer with induced pluripotent stem cells. Cell. 2015;161(2):240-254. [57] HAN Y, FENG H, SUN J, et al. Lkb1 deletion in periosteal mesenchymal progenitors induces osteogenic tumors through mTORC1 activation. J Clin Invest. 2019;129(5):1895-1909. [58] 杨鹏,冯娜,李莎莎,等.白血病患者并发肺部感染的临床分析[J].中华肺部疾病杂志(电子版),2018,11(6): 726-728. [59] MAALI A, ATASHI A, GHAFFARI S, et al. A Review on Leukemia and iPSC Technology: Application in Novel Treatment and Future. Curr Stem Cell Res Ther. 2018;13(8):665-675. [60] SUNDARAMURTHI T, WEHRLEN L, FRIEDMAN E, et al. Hematopoietic Stem Cell Transplantation Recipient and Caregiver Factors Affecting Length of Stay and Readmission. Oncol Nurs Forum. 2017;44(5):571-579. [61] SALCI KR, LEE JH, LARONDE S, et al. Cellular Reprogramming Allows Generation of Autologous Hematopoietic Progenitors From AML Patients That Are Devoid of Patient-Specific Genomic Aberrations. Stem Cells. 2015;33(6):1839-1849. [62] LIU Y, CHENG H, GAO S, et al. Reprogramming of MLL-AF9 leukemia cells into pluripotent stem cells. Leukemia. 2014;28(5):1071-1080. [63] KUMANO K, ARAI S, HOSOI M, et al. Generation of induced pluripotent stem cells from primary chronic myelogenous leukemia patient samples. Blood. 2012; 119(26):6234-6242. [64] KOTINI AG, CHANG CJ, CHOW A, et al. Stage-Specific Human Induced Pluripotent Stem Cells Map the Progression of Myeloid Transformation to Transplantable Leukemia. Cell Stem Cell. 2017;20(3):315-328.e7. [65] XU L, WANG J, LIU Y, et al. CRISPR-Edited Stem Cells in a Patient with HIV and Acute Lymphocytic Leukemia. N Engl J Med. 2019;381(13):1240-1247. |
| [1] | Pu Rui, Chen Ziyang, Yuan Lingyan. Characteristics and effects of exosomes from different cell sources in cardioprotection [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(在线): 1-. |
| [2] | Lin Qingfan, Xie Yixin, Chen Wanqing, Ye Zhenzhong, Chen Youfang. Human placenta-derived mesenchymal stem cell conditioned medium can upregulate BeWo cell viability and zonula occludens expression under hypoxia [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(在线): 4970-4975. |
| [3] | Jiang Yong, Luo Yi, Ding Yongli, Zhou Yong, Min Li, Tang Fan, Zhang Wenli, Duan Hong, Tu Chongqi. Von Mises stress on the influence of pelvic stability by precise sacral resection and clinical validation [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1318-1323. |
| [4] | Zhang Tongtong, Wang Zhonghua, Wen Jie, Song Yuxin, Liu Lin. Application of three-dimensional printing model in surgical resection and reconstruction of cervical tumor [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1335-1339. |
| [5] | Zhang Chao, Lü Xin. Heterotopic ossification after acetabular fracture fixation: risk factors, prevention and treatment progress [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1434-1439. |
| [6] | Zhou Jihui, Li Xinzhi, Zhou You, Huang Wei, Chen Wenyao. Multiple problems in the selection of implants for patellar fracture [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1440-1445. |
| [7] | Wang Debin, Bi Zhenggang. Related problems in anatomy mechanics, injury characteristics, fixed repair and three-dimensional technology application for olecranon fracture-dislocations [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1446-1451. |
| [8] | Gu Xia, Zhao Min, Wang Pingyi, Li Yimei, Li Wenhua. Relationship between hypoxia inducible factor 1 alpha and hypoxia signaling pathway [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1284-1289. |
| [9] | Ji Zhixiang, Lan Changgong. Polymorphism of urate transporter in gout and its correlation with gout treatment [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1290-1298. |
| [10] | Yuan Mei, Zhang Xinxin, Guo Yisha, Bi Xia. Diagnostic potential of circulating microRNA in vascular cognitive impairment [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1299-1304. |
| [11] | Zhang Xiumei, Zhai Yunkai, Zhao Jie, Zhao Meng. Research hotspots of organoid models in recent 10 years: a search in domestic and foreign databases [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1249-1255. |
| [12] | Hou Jingying, Yu Menglei, Guo Tianzhu, Long Huibao, Wu Hao. Hypoxia preconditioning promotes bone marrow mesenchymal stem cells survival and vascularization through the activation of HIF-1α/MALAT1/VEGFA pathway [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 985-990. |
| [13] | Shi Yangyang, Qin Yingfei, Wu Fuling, He Xiao, Zhang Xuejing. Pretreatment of placental mesenchymal stem cells to prevent bronchiolitis in mice [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 991-995. |
| [14] | Liang Xueqi, Guo Lijiao, Chen Hejie, Wu Jie, Sun Yaqi, Xing Zhikun, Zou Hailiang, Chen Xueling, Wu Xiangwei. Alveolar echinococcosis protoscolices inhibits the differentiation of bone marrow mesenchymal stem cells into fibroblasts [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 996-1001. |
| [15] | Fan Quanbao, Luo Huina, Wang Bingyun, Chen Shengfeng, Cui Lianxu, Jiang Wenkang, Zhao Mingming, Wang Jingjing, Luo Dongzhang, Chen Zhisheng, Bai Yinshan, Liu Canying, Zhang Hui. Biological characteristics of canine adipose-derived mesenchymal stem cells cultured in hypoxia [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1002-1007. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||