Chinese Journal of Tissue Engineering Research ›› 2021, Vol. 25 ›› Issue (13): 2097-2102.doi: 10.3969/j.issn.2095-4344.2190
Previous Articles Next Articles
Therapeutic application of adipose stem cell-free liquid extracts: skin aging, wound healing, scar recovery and nerve regeneration
Cai Yuan1, Deng Chengliang2
- 1Department of Dermatology, 2Plastic Surgery, Affiliated Hospital of Zunyi Medical University, Zunyi 563003, Guizhou Province, China
-
Received:2020-03-09Revised:2020-03-13Accepted:2020-05-09Online:2021-05-08Published:2020-12-29 -
Contact:Deng Chengliang, MD, Master’s supervisor, Associate chief physician, Plastic Surgery, Affiliated Hospital of Zunyi Medical University, Zunyi 563003, Guizhou Province, China -
About author:Cai Yuan, Master candidate, Department of Dermatology, Affiliated Hospital of Zunyi Medical University, Zunyi 563003, Guizhou Province, China -
Supported by:the National Natural Science Foundation of China, No. 81801921 (to DCL)
CLC Number:
Cite this article
Cai Yuan, Deng Chengliang. Therapeutic application of adipose stem cell-free liquid extracts: skin aging, wound healing, scar recovery and nerve regeneration[J]. Chinese Journal of Tissue Engineering Research, 2021, 25(13): 2097-2102.
share this article
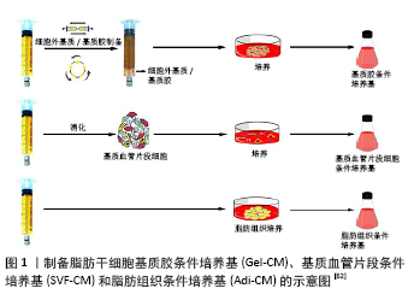
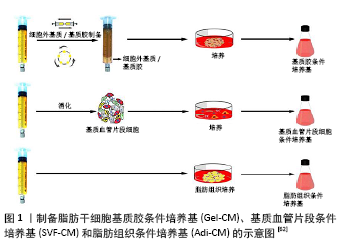
2.1 脂肪干细胞条件培养基 2.1.1 脂肪干细胞条件培养基的制备 将脂肪干细胞添加到含体积分数为10%胎牛血清的基础培养基中培养一段时间,待细胞生长状态良好后将基础培养基替换成无血清培养基,培养两三天后收集上清液,即为脂肪干细胞条件培养基。体外培养的条件不同,脂肪干细胞条件培养基中含有的细胞因子的量也不同。多项研究在正常氧含量(体积分数为20%-21%)的体外培养条件下获取脂肪干细胞条件培养基,也有研究报道在低氧条件(体积分数为2%O2)下可获取含有更多细胞因子的脂肪干细胞条件培养基,可能原因是脂肪干细胞在低氧条件下激活低氧诱导因子1α,介导其下游的各类信号通路[5],继而刺激脂肪干细胞增殖,促使脂肪干细胞分泌更多的血管内皮生长因子、肝细胞生长因子和碱性成纤维细胞生长因子等[6-7]。此外,研究报道在三维培养条件下获取的脂肪干细胞条件培养基中含有更多的转化生长因子β和血管内皮生长因子[8],这可能与三维培养更能模拟脂肪干细胞的体内环境有关[9]。低氧联合三维培养可能是获取脂肪干细胞条件培养基较好的体外培养条件。 2.1.2 脂肪干细胞条件培养基与皮肤老化 皮肤老化可分为内源性老化和外源性老化。内源性老化是人体的一个自然老化过程,主要由于成纤维细胞受损、减少,逐渐丧失其增殖及合成胶原的能力[10],从而出现皱纹、细纹、色素沉着、皮肤弹性减退等表现。体外研究发现,脂肪干细胞条件培养基中的旁分泌因子如表皮生长因子、碱性成纤维细胞生长因子、转化生长因子β、血管内皮生长因子等通过促进皮肤成纤维细胞增殖、迁移,增加细胞外基质和胶原合成,从而改善皱纹及面部凹陷[11]。研究还发现脂肪干细胞条件培养基可恢复体外连续传代后衰老的成纤维细胞的生物学活性,包括促进增殖和分泌细胞外基质蛋白(Ⅰ型胶原、Ⅲ型胶原以及弹性蛋白[12-13])。此外,张坤等[14]通过D-半乳糖制备小鼠皮肤成纤维细胞体外衰老模型,添加脂肪干细胞条件培养基后可降低小鼠皮肤成纤维细胞中caspase3、p53的表达,从而降低衰老相关标志物β-半乳糖苷酶的活性。对体内研究而言,脂肪干细胞条件培养基治疗需克服机体的皮肤屏障。众所周知,位于表皮最外层的角质层是抵御外界环境损伤、防止水分丢失的主要结构,也是各种化妆品和大分子药物不能渗透进入真皮的原因,因此,临床上常使用激光和微针制造微型孔道以增加药物吸收。研究发现,脂肪干细胞条件培养基联合点阵CO2激光治疗不仅可以提高皮肤弹性,改善皮肤粗糙度以及降低经皮水分丢失等皮肤老化现象,还可减轻激光后色素沉着[15]。还有研究报道微针联合脂肪干细胞条件培养基可显著改善皮肤粗糙度,降低黑色素含量,提高皮肤亮度、光泽、弹性和抗皱等多种功效[16]。研究表明,脂肪干细胞条件培养基中的各种细胞因子通过产生的微孔进到真皮,从而激活成纤维细胞,刺激胶原的生成和重塑,促进皮肤再生[17]。 紫外线是引起皮肤外源性老化的重要因素,也称为光老化[18]。紫外线(290-320 nm)通过产生活性氧上调基质金属蛋白酶的表达和促炎症细胞因子的分泌,诱导真皮中的成纤维细胞损伤,进而使细胞外基质生成减少[19]。SON等[20]研究发现脂肪干细胞条件培养基与紫外线照射的人真皮成纤维细胞共培养可显著上调Ⅰ型前胶原的表达,下调基质金属蛋白酶1的表达。此外,GUO等[21]探讨脂肪干细胞条件培养基对紫外线照射后不同衰老程度成纤维细胞的保护作用,结果表明脂肪干细胞条件培养基预处理显著降低了成纤维细胞的衰老率。LI等[22]研究表明脂肪干细胞条件培养基通过下调紫外线B诱导的MAPK和TGF-β/Smad信号通路减少基质金属蛋白酶1的产生和白细胞介素6的分泌,从而保护人角质形成细胞及人皮肤成纤维细胞免受紫外线B诱导的光老化。 上述研究表明,无论是内源性还是外源性老化,脂肪干细胞条件培养基均能改善成纤维细胞的功能,促使其合成更多的细胞外基质,其机制可能与脂肪干细胞条件培养基中含有的生长因子有关。 2.1.3 脂肪干细胞条件培养基与创面愈合 创面愈合是一个非常复杂的过程,涉及浸润细胞、驻扎细胞和细胞因子的相互作用,角质形成细胞、内皮细胞、成纤维细胞的迁移、增殖及细胞外基质的沉积是创面愈合的主要生物学过程。近年来,已有证据表明脂肪干细胞条件培养基对创面愈合起重要作用。COOPER等[23]研究发现脂肪干细胞条件培养基能促进成纤维细胞的增殖及迁移,在随后的大鼠缺血创面模型中发现脂肪干细胞条件培养基可加速成年大鼠缺血性创面的愈合。SEO等[24]研究发现脂肪干细胞条件培养基联合Exendin-4 (Ex-4)可促进糖尿病鼠创面愈合,两者具有协同作用,可能机制是Ex-4作为一种胰高血糖素样肽受体激动剂,促进胰岛素分泌,降低血糖,联合脂肪干细胞条件培养基后可显著促进人脐静脉内皮细胞的增殖及迁移进而促进血管生成。此外,脂肪干细胞条件培养基可联合富血小板纤维蛋白加速创面愈合。富血小板纤维蛋白中含有多种生长因子和基质蛋白,同样具有促进组织再生和修复的功能,两者联合后更能刺激成纤维细胞分泌细胞外基质,肉芽组织形成更多,从而更好地促进创面愈合[25]。ZHAO等[26]进一步探讨了富血小板纤维蛋白中哪些细胞因子对皮肤成纤维细胞增殖和迁移发挥影响,结果表明:表皮生长因子、血小板源性生长因子AA、血管内皮生长因子、碱性成纤维细胞生长因子这4种细胞因子浓度较高,其中碱性成纤维细胞生长因子是促进成纤维细胞增殖和迁移的主要细胞因子,血管内皮生长因子和血小板源性生长因子AA主要对成纤维细胞的迁移起促进作用。此外,与单一细胞因子相比,脂肪干细胞条件培养基对成纤维细胞增殖和迁移的促进作用更为明显。这些结果表明,脂肪干细胞条件培养基中的众多因子具有协同作用,共同促进创面愈合。 2.1.4 脂肪干细胞条件培养基与瘢痕 瘢痕分为病理性瘢痕和生理性瘢痕,病理性瘢痕主要指瘢痕疙瘩和增生性瘢痕。文献报道脂肪干细胞条件培养基可抑制瘢痕疙瘩形成。WANG等[27]将瘢痕疙瘩组织与脂肪干细胞条件培养基体外共培养,能显著降低瘢痕疙瘩组织中金属蛋白酶组织抑制因子1水平,减少Ⅰ型胶原的沉积,表明脂肪干细胞条件培养基通过调节胞外基质的重塑与胶原合成,从而改善瘢痕疙瘩的形成。 增生性瘢痕通常以细胞外基质异常沉积为特征,而转化生长因子β1因其强烈的致纤维化作用被认为是瘢痕纤维化修复过程中的关键因子。宋菲等[28]在转化生长因子β1的刺激下研究了不同浓度脂肪干细胞条件培养基(0,10%,50%,100%)对成纤维细胞增殖及凋亡的影响,结果发现脂肪干细胞条件培养基浓度越高,抑制增殖及促进凋亡作用越明显。MA等[29]研究发现脂肪干细胞条件培养基中的肝细胞生长因子可有效抑制纤维化并调节细胞外基质重塑。此外,P38/MAPK信号通路在增生性瘢痕纤维化中也起重要作用,LI等[30]研究了脂肪干细胞条件培养基通过抑制p38/MAPK信号通路治疗增生性瘢痕的机制,发现经脂肪干细胞条件培养基处理的增生性瘢痕成纤维细胞p-p38蛋白水平呈浓度依赖性下调,其抑制剂SB203580的应用进一步降低了p-p38的表达,而p38的激活剂茴香霉素通过上调p-p38的表达,促进胶原的生成和沉积。因此,该研究显示脂肪干细胞条件培养基可通过抑制p38/MAPK信号通路达到抗瘢痕作用。另外,ZHANG等[31-32]在兔耳增生性瘢痕模型注射脂肪干细胞条件培养基后,发现兔耳增生性瘢痕变得不明显,组织学分析显示经脂肪干细胞条件培养基处理的一侧瘢痕更扁平、更薄,其胶原纤维排列规则,胶原沉积减少。综上所述,脂肪干细胞条件培养基通过减少胶原沉积及抗纤维化在减轻瘢痕形成中具有重要作用,推测可能与培养基中含有的细胞因子有关,但具体机制尚不明确,仍需进行更深入的研究。 2.1.5 脂肪干细胞条件培养基与神经保护 脂肪干细胞条件培养基局部移植可营养神经。新生儿缺氧缺血性脑病是引起新生儿急性死亡和慢性神经系统损伤的主要原因之一,具有相当高的发病率和死亡率。WEI等[33]在大鼠脑缺氧缺血性损伤模型中测试了脂肪干细胞条件培养基保护神经元的能力。结果表明脂肪干细胞条件培养基中含有的神经营养因子如胰岛素样生长因子1和脑源性神经营养因子,对神经病变损伤的恢复起着重要作用,改善了缺氧缺血性脑损伤大鼠模型的认知功能和运动功能。SALGADO等[34]分析了在各种微环境下脂肪干细胞分泌的可溶性因子,发现脂肪干细胞可分泌神经生长因子、胶质源性神经营养因子及脑源性神经营养因子等与神经再生密切相关的生长因子,这些神经再生相关的生长因子可能是脂肪干细胞条件培养基促进神经损伤修复的根本原因。 2.1.6 脂肪干细胞条件培养基与呼吸系统 呼吸系统是脂肪干细胞条件培养基应用的另一领域。急性呼吸窘迫综合征是临床上急危重症患者死亡的重要原因。研究表明,脂肪干细胞条件培养基中的旁分泌因子能有效减轻急性呼吸窘迫综合征肺损伤的发展,在脂多糖诱导的小鼠急性呼吸窘迫综合征模型中发现使用脂肪干细胞条件培养基可改善肺部炎症组织学变化、抑制支气管肺泡灌洗液蛋白外渗、减少炎症递质肿瘤坏死因子和白细胞介素6的分泌[35]。此外,肺动脉高压和肺纤维化也是呼吸系统的难治愈性疾病,现有的药物干预措施未能改善其临床结果或降低疾病相关死亡率,故而RATHINASABAPATHY等[36]研究了脂肪干细胞条件培养基对肺动脉高压和肺纤维的影响,结果表明:在用野百合碱和博莱霉素诱导的大鼠肺动脉高压和肺纤维化模型中,脂肪干细胞条件培养基通过改善肺血流量和抑制心脏重塑减轻野百合碱诱导的肺动脉高压,以及通过减轻胶原沉积阻止博莱霉素模型肺纤维化的进展,从而改善肺动脉高压和肺纤维化的临床症状,可能为肺动脉高压和肺纤维化的临床治疗提供一种新的方法,但其具体机制仍需进一步的研究。 2.2 脂肪干细胞外泌体 2.2.1 脂肪干细胞外泌体的制备 外泌体是由多泡体分泌的直径为30-150 nm不等的膜囊泡,属于胞外囊泡的一种[37]。当前,制备外泌体的方法较多,包括基于外泌体大小和密度的蔗糖梯度离心法[38],基于不同膜孔径分离的超滤法[39],以及基于表面标志物的表达吸附、分离的免疫亲和磁珠分离法[40],还有根据聚合物共沉淀原理的ExoQuick提取法[41],其中最常用的是基于外泌体大小的超速离心法[42],即先进行脂肪干细胞体外培养,将达到70%-80%融合的脂肪干细胞用PBS冲洗,接种到无血清培养基中培养48 h后在4 ℃下收集上清液,分别进行300×g离心10 min,2 000×g离心 10 min,10 000×g离心30 min以去除细胞和细胞碎片;继续将培养基100 000×g超速离心70 min,将离心管底部的沉淀物重新悬浮于PBS中并冲洗,最后进行100 000×g离心 70 min去除外泌体中污染的蛋白质,最终获得外泌体颗粒。 2.2.2 脂肪干细胞外泌体与创面愈合 研究表明脂肪干细胞外泌体通过促进成纤维细胞迁移、增殖而促进创面愈合。HU等[43]研究报道外泌体能被成纤维细胞内化,增加N-cadherin、cyclin-1、PCNA和Ⅰ型胶原的基因表达,同时促进成纤维细胞的增殖和迁移;有趣的是,与局部注射相比,静脉注射脂肪干细胞外泌体促进创面愈合效果更好。ZHANG等[44]研究发现脂肪干细胞外泌体主要通过激活PI3K/Akt信号途径,刺激真皮成纤维细胞增殖及迁移,从而促进创面愈合。脂肪干细胞外泌体不仅影响成纤维细胞,还可影响角质形成细胞。受损人角质形成细胞与脂肪干细胞外泌体共培养后,可显著恢复受损人角质形成细胞的迁移能力[45]。田新立等[46]研究发现脂肪干细胞外泌体与角质形成细胞共培养后ERK1/2、AKT和STAT3的磷酸化水平显著增高,说明外泌体可能通过激活ERK1/2、AKT和STAT3信号通路进而影响其增殖及迁移。此外,MA等[47]将人角质形成细胞置于过氧化氢建立皮肤损伤模型,发现脂肪干细胞外泌体也可通过激活Wnt/β‐catenin信号通路从而促进其增殖和迁移并抑制凋亡。还有研究报道高表达micoRNA-21的脂肪干细胞外泌体通过增强基质金属蛋白酶9和抑制基质金属蛋白酶抑制剂2的表达从而促进角质形成细胞的增殖和迁移[48]。此外,脂肪干细胞外泌体还可影响内皮细胞。张静等[49]研究发现脂肪干细胞外泌体可通过促进人脐静脉内皮细胞增殖和迁移从而促进血管新生。过表达Nrf2的脂肪干细胞外泌体可促进血管内皮祖细胞增殖、降低炎症蛋白的表达,从而促进糖尿病大鼠足部创面愈合[50]。此外,作为外泌体固定剂和伤口敷料的透明质酸与外泌体联合治疗更能促进小鼠皮肤创面愈合,组织学染色结果显示脂肪干细胞外泌体+透明质酸联合组可见完整的皮肤全层再生和更显著的血管新生[51]。因此,脂肪干细胞外泌体可影响创面修复的主要功能细胞,包括成纤维细胞、角质形成细胞和血管内皮细胞等,从而加速创面愈合,机制可能在于脂肪干细胞外泌体中含有的活性RNA物质。 2.2.3 脂肪干细胞外泌体与心肌保护 尽快恢复缺血心肌的血流灌注是急性心肌梗死的有效治疗方法,但缺血再灌注可能会引起氧化应激和炎症反应,导致心肌细胞进一步损伤和凋亡。近年来,越来越多的研究证明来自不同干细胞的外泌体可保护心肌细胞,增强心肌功能[52-53]。LIU等[54]研究报道了脂肪干细胞外泌体对心肌细胞缺血再灌注损伤的治疗效果。研究发现,脂肪干细胞外泌体不仅能被小鼠心肌细胞摄取,而且能明显抑制过氧化氢诱导的小鼠心肌细胞凋亡,说明外泌体能保护小鼠心肌细胞免受氧化应激所致的细胞凋亡。此外,CUI等[55]体外研究发现脂肪干细胞外泌体通过激活Wnt/β‐catenin信号通路具有心肌保护作用;在小鼠缺血再灌注损伤模型中,脂肪干细胞外泌体治疗可显著减少小鼠心肌梗死面积,且血清CK-MB、LDH和cTnI水平以及Caspase 3活化显著降低;在大鼠急性心肌梗死模型中,富含miR-146a的外泌体组通过抑制早期生长反应因子的表达更能有效抑制心肌细胞凋亡、降低炎症反应水平及减轻心肌纤维化[56]。综上所述,脂肪干细胞外泌体通过抑制心肌细胞凋亡具有明显的心肌保护作用,而含有miRNA的外泌体增强了此种作用。 2.2.4 脂肪干细胞外泌体与特应性皮炎 特应性皮炎又名特应性湿疹,以血清IgE升高及外周血嗜酸性粒细胞增多为特点,常表现为皮肤干燥、湿疹样皮疹、剧烈瘙痒。最近有研究证实了脂肪干细胞外泌体可通过调节炎症及炎性细胞因子的表达从而改善特应性皮炎的发生发展。在该研究中,脂肪干细胞外泌体被应用于含尘螨抗原的Biostir?-AD霜诱导的小鼠特应性皮炎模型,发现与口服强的松相比,连续4周每周注射脂肪干细胞外泌体3次可显著减轻特应性皮炎的症状,进一步研究发现脂肪干细胞外泌体注射后显著减少了皮损中炎性细胞如CD86+和CD206+的数量以及血清IgE介导的嗜酸性粒细胞水平,并以剂量依赖性的方式降低皮损中白细胞介素4、白细胞介素23、白细胞介素31和肿瘤坏死因子α mRNA水平[57]。 "
| [1] SARKANEN JR, KAILA V, MANNERSTRÖM B, et al. Human adipose tissue extract induces angiogenesis and adipogenesis in vitro. Tissue Eng Part A. 2012;18(1-2):17-25. [2] ZAREI F, ABBASZADEH A. Stem cell and skin rejuvenation. J Cosmet Laser Ther. 2018;20(3):193-197. [3] WOLF DA, BEESON W, RACHEL JD, et al. Mesothelial Stem Cells and Stromal Vascular Fraction for Skin Rejuvenation. Facial Plast Surg Clin North Am. 2018;26(4):513-532. [4] 陈筑昕.脂肪干细胞来源外泌体促进组织修复的应用进展[J].组织工程与重建外科杂志,2018,14(4):219-222. [5] KAUR B, KHWAJA FW, SEVERSON EA, et al. Hypoxia and the hypoxia-inducible-factor pathway in glioma growth and angiogenesis. Neuro Oncol. 2005;7(2):134-153. [6] LEE EY, XIA Y, KIM WS, et al. Hypoxia-enhanced wound-healing function of adipose-derived stem cells: increase in stem cell proliferation and up-regulation of VEGF and bFGF. Wound Repair Regen. 2009;17(4):540-547. [7] HE J, CAI Y, LUO LM, et al. Hypoxic adipose mesenchymal stem cells derived conditioned medium protects myocardial infarct in rat. Eur Rev Med Pharmacol Sci. 2015;19(22):4397-4406. [8] CHO YJ, SONG HS, BHANG S, et al. Therapeutic effects of human adipose stem cell-conditioned medium on stroke. J Neurosci Res. 2012;90(9): 1794-1802. [9] YLOSTALO JH, BARTOSH TJ, TIBLOW A, et al. Unique characteristics of human mesenchymal stromal/progenitor cells pre-activated in 3-dimensional cultures under different conditions. Cytotherapy. 2014;16(11):1486-1500. [10] HU S, LI Z, CORES J, et al. Needle-Free Injection of Exosomes Derived from Human Dermal Fibroblast Spheroids Ameliorates Skin Photoaging. ACS Nano. 2019;13(10):11273-11282. [11] YANG JA, CHUNG HM, WON CH, et al. Potential application of adipose-derived stem cells and their secretory factors to skin: discussion from both clinical and industrial viewpoints. Expert Opin Biol Ther. 2010;10(4):495-503. [12] 王婷,郭澍,陈鹏,等. 脂肪干细胞条件培养基对不同衰老程度成纤维细胞中波紫外线损伤的修复效应[J]. 中华医学美学美容杂志,2018, 24(2):79-83. [13] 张华,郭澍,王婷,等.脂肪干细胞条件培养基修复人成纤维细胞衰老损伤的研究[J].中华整形外科, 2018,34(10):856-861. [14] 张坤,延冰,李芳,等.脂肪源性干细胞对内源性皮肤老化的治疗作用[J].山东大学学报(医学版),2018,56(12):19-25,46. [15] ZHOU BR, ZHANG T, BIN JAMEEL AA, et al. The efficacy of conditioned media of adipose-derived stem cells combined with ablative carbon dioxide fractional resurfacing for atrophic acne scars and skin rejuvenation. J Cosmet Laser Ther. 2016;18(3):138-148. [16] WANG X, SHU X, HUO W, et al. Efficacy of protein extracts from medium of Adipose-derived stem cells via microneedles on Asian skin. J Cosmet Laser Ther. 2018;20(4):237-244. [17] LEE CM. Fifty years of research and development of cosmeceuticals: a contemporary review. J Cosmet Dermatol. 2016;15(4):527-539. [18] GONG M, ZHANG P, LI C, et al. Protective Mechanism of Adipose-Derived Stem Cells in Remodelling of the Skin Stem Cell Niche During Photoaging. Cell Physiol Biochem. 2018;51(5):2456-2471. [19] LEE SE, KWON TR, KIM JH, et al. Anti‑photoaging and anti‑oxidative activities of natural killer cell conditioned medium following UV‑B irradiation of human dermal fibroblasts and a reconstructed skin model. Int J Mol Med. 2019;44(5):1641-1652. [20] SON WC, YUN JW, KIM BH. Adipose-derived mesenchymal stem cells reduce MMP-1 expression in UV-irradiated human dermal fibroblasts: therapeutic potential in skin wrinkling. Biosci Biotechnol Biochem. 2015;79(6):919-925. [21] GUO S, WANG T, ZHANG S, et al. Adipose-derived stem cell-conditioned medium protects fibroblasts at different senescent degrees from UVB irradiation damages. Mol Cell Biochem. 2020;463(1-2):67-78. [22] LI L, NGO HTT, HWANG E, et al. Conditioned Medium from Human Adipose-Derived Mesenchymal Stem Cell Culture Prevents UVB-Induced Skin Aging in Human Keratinocytes and Dermal Fibroblasts. Int J Mol Sci. 2019;21(1):49. [23] COOPER DR, WANG C, PATEL R, et al. Human Adipose-Derived Stem Cell Conditioned Media and Exosomes Containing MALAT1 Promote Human Dermal Fibroblast Migration and Ischemic Wound Healing. Adv Wound Care (New Rochelle). 2018;7(9):299-308. [24] SEO E, LIM JS, JUN JB, et al. Exendin-4 in combination with adipose-derived stem cells promotes angiogenesis and improves diabetic wound healing. J Transl Med. 2017;15(1):35. [25] 黄敏,颜洪.脂肪间充质干细胞条件培养液联合富血小板纤维蛋白修复小鼠皮肤损伤[J].中国组织工程研究,2019,23(1):18-23. [26] ZHAO J, HU L, LIU J, et al. The effects of cytokines in adipose stem cell-conditioned medium on the migration and proliferation of skin fibroblasts in vitro. Biomed Res Int. 2013;2013:578479. [27] WANG X, MA Y, GAO Z, et al. Human adipose-derived stem cells inhibit bioactivity of keloid fibroblasts. Stem Cell Res Ther. 2018;9(1):40. [28] 宋菲,原博.脂肪干细胞来源的条件培养基对TGF-β1共同干预下的真皮成纤维细胞致纤维化能力的影响[J].上海交通大学学报(医学版), 2017,37(5):588-594. [29] MA J, YAN X, LIN Y, et al. Hepatocyte growth factor secreted from human adipose-derived stem cells inhibits fibrosis in hypertrophic scar fibroblasts. Curr Mol Med. 2020;20(7):558-571. [30] LI Y, ZHANG W, GAO J, et al. Adipose tissue-derived stem cells suppress hypertrophic scar fibrosis via the p38/MAPK signaling pathway. Stem Cell Res Ther. 2016;7(1):102. [31] ZHANG Q, LIU LN, YONG Q, et al. Intralesional injection of adipose-derived stem cells reduces hypertrophic scarring in a rabbit ear model. Stem Cell Res Ther. 2015;6(1):145. [32] 张琪,刘李娜,邓景成,等.瘢痕内注射脂肪来源干细胞对兔耳增生性瘢痕的抑制作用研究[J].组织工程与重建外科,2015,11(3):139-143. [33] WEI X, DU Z, ZHAO L, et al. IFATS collection: The conditioned media of adipose stromal cells protect against hypoxia-ischemia-induced brain damage in neonatal rats. Stem Cells. 2009;27(2):478-488. [34] SALGADO AJ, REIS RL, SOUSA NJ, et al. Adipose tissue derived stem cells secretome: soluble factors and their roles in regenerative medicine. Curr Stem Cell Res Ther. 2010;5(2):103-110. [35] LU H, POIRIER C, COOK T, et al. Conditioned media from adipose stromal cells limit lipopolysaccharide-induced lung injury, endothelial hyperpermeability and apoptosis. J Transl Med. 2015;13:67. [36] RATHINASABAPATHY A, BRUCE E, ESPEJO A, et al. Therapeutic potential of adipose stem cell-derived conditioned medium against pulmonary hypertension and lung fibrosis. Br J Pharmacol. 2016;173(19):2859-2879. [37] FERREIRA ADF, GOMES DA. Stem Cell Extracellular Vesicles in Skin Repair. Bioengineering (Basel). 2018;6(1):4. [38] XU R, GREENING DW, ZHU HJ, et al. Extracellular vesicle isolation and characterization: toward clinical application. J Clin Invest. 2016;126(4): 1152-1162. [39] KONOSHENKO MY, LEKCHNOV EA, VLASSOV AV, et al. Isolation of Extracellular Vesicles: General Methodologies and Latest Trends. Biomed Res Int. 2018;2018:8545347. [40] JEYARAM A, JAY SM. Preservation and Storage Stability of Extracellular Vesicles for Therapeutic Applications. AAPS J. 2017;20(1):1. [41] NAKAI W, YOSHIDA T, DIEZ D, et al. A novel affinity-based method for the isolation of highly purified extracellular vesicles. Sci Rep. 2016;6:33935. [42] WANG C, SONG W, CHEN B, et al. Exosomes Isolated From Adipose-Derived Stem Cells: A New Cell-Free Approach to Prevent the Muscle Degeneration Associated With Torn Rotator Cuffs. Am J Sports Med. 2019;47(13): 3247-3255. [43] HU L, WANG J, ZHOU X, et al. Exosomes derived from human adipose mensenchymal stem cells accelerates cutaneous wound healing via optimizing the characteristics of fibroblasts. Sci Rep. 2016;6:32993. [44] ZHANG W, BAI X, ZHAO B, et al. Cell-free therapy based on adipose tissue stem cell-derived exosomes promotes wound healing via the PI3K/Akt signaling pathway. Exp Cell Res. 2018;370(2):333-342. [45] 张远远,曾悦,朱艳霞,等.人脂肪干细胞源外泌体对HaCaT细胞损伤修复及迁移功能影响的初步实验研究[J].中华皮肤科杂志,2019,52(9): 616-623. [46] 田新立,江波,颜洪.脂肪间充质干细胞来源外泌体对角质形成细胞增殖和迁移的影响与机制[J].中国组织工程研究,2019,23(1):68-73. [47] MA T, FU B, YANG X, et al. Adipose mesenchymal stem cell-derived exosomes promote cell proliferation, migration, and inhibit cell apoptosis via Wnt/beta-catenin signaling in cutaneous wound healing. J Cell Biochem. 2019;120(6):10847-10854. [48] YANG C, LUO L, BAI X, et al. Highly-expressed micoRNA-21 in adipose derived stem cell exosomes can enhance the migration and proliferation of the HaCaT cells by increasing the MMP-9 expression through the PI3K/AKT pathway. Arch Biochem Biophys. 2020;681:108259. [49] 张静,易阳艳,阳水发,等.脂肪干细胞来源外泌体对人脐静脉血管内皮 细胞增殖、迁移及管样分化的影响[J].中国修复重建外科杂志, 2018,32(10):1351-1357. [50] LI X, XIE X, LIAN W, et al. Exosomes from adipose-derived stem cells overexpressing Nrf2 accelerate cutaneous wound healing by promoting vascularization in a diabetic foot ulcer rat model. Exp Mol Med. 2018; 50(4):29. [51] LIU K, CHEN C, ZHANG H, et al. Adipose stem cell-derived exosomes in combination with hyaluronic acid accelerate wound healing through enhancing re-epithelialization and vascularization. Br J Dermatol. 2019; 181(4):854-856. [52] CHEN L, WANG Y, PAN Y, et al. Cardiac progenitor-derived exosomes protect ischemic myocardium from acute ischemia/reperfusion injury. Biochem Biophys Res Commun. 2013;431(3):566-571. [53] ARSLAN F, LAI RC, SMEETS MB, et al. Mesenchymal stem cell-derived exosomes increase ATP levels, decrease oxidative stress and activate PI3K/Akt pathway to enhance myocardial viability and prevent adverse remodeling after myocardial ischemia/reperfusion injury. Stem Cell Res. 2013;10(3):301-312. [54] LIU Z, XU Y, WAN Y, et al. Exosomes from adipose-derived mesenchymal stem cells prevent cardiomyocyte apoptosis induced by oxidative stress. Cell Death Discov. 2019;5:79. [55] CUI X, HE Z, LIANG Z, et al. Exosomes From Adipose-derived Mesenchymal Stem Cells Protect the Myocardium Against Ischemia/Reperfusion Injury Through Wnt/β-Catenin Signaling Pathway. J Cardiovasc Pharmacol. 2017; 70(4):225-231. [56] PAN J, ALIMUJIANG M, CHEN Q, et al. Exosomes derived from miR-146a-modified adipose-derived stem cells attenuate acute myocardial infarction-induced myocardial damage via downregulation of early growth response factor 1. J Cell Biochem. 2019;120(3):4433-4443. [57] CHO BS, KIM JO, HA DH,et al. Exosomes derived from human adipose tissue-derived mesenchymal stem cells alleviate atopic dermatitis. Stem Cell Res Ther. 2018;9(1):187. [58] HE Y, XIA J, CHEN H, et al. Human adipose liquid extract induces angiogenesis and adipogenesis: a novel cell-free therapeutic agent. Stem Cell Res Ther. 2019;10(1):252. [59] 郑洋洋,刘毅.纳米脂肪衍生物的研究进展[J].中国美容整形外科,2019, 30(10):631-633. [60] YAO Y, DONG Z, LIAO Y, et al. Adipose Extracellular Matrix/Stromal Vascular Fraction Gel: A Novel Adipose Tissue-Derived Injectable for Stem Cell Therapy. Plast Reconstr Surg. 2017;139(4):867-879. [61] DENG C, HE Y, FENG J, et al. Conditioned medium from 3D culture system of stromal vascular fraction cells accelerates wound healing in diabetic rats. Regen Med. 2019;14(10):925-937. [62] DENG C, HE Y, FENG J, et al. Extracellular matrix/stromal vascular fraction gel conditioned medium accelerates wound healing in a murine model. Wound Repair Regen. 2017;25(6):923-932. |
| [1] | Pu Rui, Chen Ziyang, Yuan Lingyan. Characteristics and effects of exosomes from different cell sources in cardioprotection [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(在线): 1-. |
| [2] | Lin Qingfan, Xie Yixin, Chen Wanqing, Ye Zhenzhong, Chen Youfang. Human placenta-derived mesenchymal stem cell conditioned medium can upregulate BeWo cell viability and zonula occludens expression under hypoxia [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(在线): 4970-4975. |
| [3] | Zhang Chao, Lü Xin. Heterotopic ossification after acetabular fracture fixation: risk factors, prevention and treatment progress [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1434-1439. |
| [4] | Zhou Jihui, Li Xinzhi, Zhou You, Huang Wei, Chen Wenyao. Multiple problems in the selection of implants for patellar fracture [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1440-1445. |
| [5] | Wang Debin, Bi Zhenggang. Related problems in anatomy mechanics, injury characteristics, fixed repair and three-dimensional technology application for olecranon fracture-dislocations [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1446-1451. |
| [6] | Ji Zhixiang, Lan Changgong. Polymorphism of urate transporter in gout and its correlation with gout treatment [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1290-1298. |
| [7] | Yuan Mei, Zhang Xinxin, Guo Yisha, Bi Xia. Diagnostic potential of circulating microRNA in vascular cognitive impairment [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1299-1304. |
| [8] | Zhang Xiumei, Zhai Yunkai, Zhao Jie, Zhao Meng. Research hotspots of organoid models in recent 10 years: a search in domestic and foreign databases [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1249-1255. |
| [9] | Hou Jingying, Yu Menglei, Guo Tianzhu, Long Huibao, Wu Hao. Hypoxia preconditioning promotes bone marrow mesenchymal stem cells survival and vascularization through the activation of HIF-1α/MALAT1/VEGFA pathway [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 985-990. |
| [10] | Shi Yangyang, Qin Yingfei, Wu Fuling, He Xiao, Zhang Xuejing. Pretreatment of placental mesenchymal stem cells to prevent bronchiolitis in mice [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 991-995. |
| [11] | Liang Xueqi, Guo Lijiao, Chen Hejie, Wu Jie, Sun Yaqi, Xing Zhikun, Zou Hailiang, Chen Xueling, Wu Xiangwei. Alveolar echinococcosis protoscolices inhibits the differentiation of bone marrow mesenchymal stem cells into fibroblasts [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 996-1001. |
| [12] | Fan Quanbao, Luo Huina, Wang Bingyun, Chen Shengfeng, Cui Lianxu, Jiang Wenkang, Zhao Mingming, Wang Jingjing, Luo Dongzhang, Chen Zhisheng, Bai Yinshan, Liu Canying, Zhang Hui. Biological characteristics of canine adipose-derived mesenchymal stem cells cultured in hypoxia [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1002-1007. |
| [13] | Geng Yao, Yin Zhiliang, Li Xingping, Xiao Dongqin, Hou Weiguang. Role of hsa-miRNA-223-3p in regulating osteogenic differentiation of human bone marrow mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1008-1013. |
| [14] | Lun Zhigang, Jin Jing, Wang Tianyan, Li Aimin. Effect of peroxiredoxin 6 on proliferation and differentiation of bone marrow mesenchymal stem cells into neural lineage in vitro [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1014-1018. |
| [15] | Zhu Xuefen, Huang Cheng, Ding Jian, Dai Yongping, Liu Yuanbing, Le Lixiang, Wang Liangliang, Yang Jiandong. Mechanism of bone marrow mesenchymal stem cells differentiation into functional neurons induced by glial cell line derived neurotrophic factor [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1019-1025. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||