Chinese Journal of Tissue Engineering Research ›› 2021, Vol. 25 ›› Issue (1): 141-145.doi: 10.3969/j.issn.2095-4344.2133
Previous Articles Next Articles
Future hot spots for cartilage repair: laminin promotes stem cell proliferation
Zhang Xiaobo1, Zhang Jie2, Sun Yu3
- 1Dalian Medical University, Dalian 116044, Liaoning Province, China; 2The Second Xiangya Hospital of Central South University, Changsha 410011, Hunan Province, China; 3Northern Jiangsu People’s Hospital, Yangzhou 225001, Jiangsu Province, China
-
Received:2019-12-18Revised:2019-12-23Accepted:2020-02-14Online:2021-01-08Published:2020-11-20 -
Contact:Sun Yu, MD, Associate chief physician, Associate professor, Master’s supervisor, Northern Jiangsu People’s Hospital, Yangzhou 225001, Jiangsu Province, China -
About author:Zhang Xiaobo, Master candidate, Dalian Medical University, Dalian 116044, Liaoning Province, China Zhang Jie, MD candidate, The Second Xiangya Hospital of Central South University, Changsha 410011, Hunan Province, China -
Supported by:the Social Development Project of Yangzhou Municipal Science and Technology Plan, No. YZ2017073
CLC Number:
Cite this article
Zhang Xiaobo, Zhang Jie, Sun Yu. Future hot spots for cartilage repair: laminin promotes stem cell proliferation[J]. Chinese Journal of Tissue Engineering Research, 2021, 25(1): 141-145.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
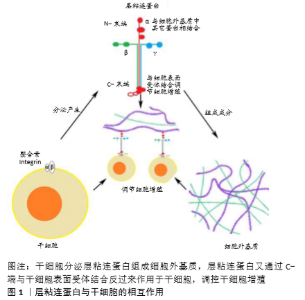
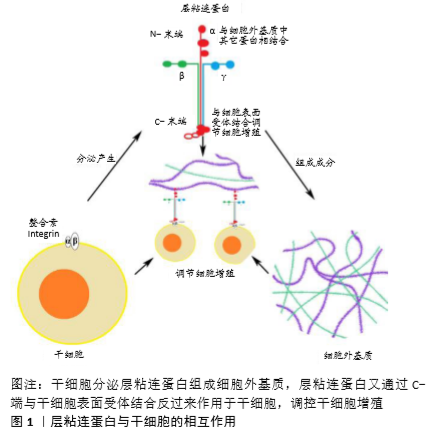
2.1 层粘连蛋白的结构及其表达特征 层粘连蛋白作为大分子糖蛋白,主要存在于细胞外基质中,是由3个二硫键连接而成的多肽,并且存在16种不同的层粘连蛋白异构体[8-9]。层粘连蛋白分子因结构域的数量、大小和表达组织的不同而变化,赋予各个层粘连蛋白家族成员独特的功能,对多种细胞活动和信号传导起着重要的调节作用[8-9]。 位于软骨细胞周基质中的各种层粘连蛋白主要由软骨细胞产生,层粘连蛋白在软骨组织不同发育阶段表达不同且分布各异。KVIST等[14]发现在正常软骨组织的发育阶段层粘连蛋白广泛分布于新生软骨组织中,而在正常软骨组织成熟后则主要分布在软骨细胞周基质中[14-16]。此外,层粘连蛋白也被证实存在于其他组织如半月板和小鼠颞下颌关节纤维软骨中[14,17],此时层粘连蛋白更多的是在周基质中分散表达,与上述层粘连蛋白在确定组分中分布有所不同。以往的研究表明,细胞外基质形成障碍是软骨退化的一个显著特征[18]。在人骨性关节炎的研究中发现,层粘连蛋白在退化关节软骨中的表达明显降低甚至消失;在创伤引起的损伤样软骨组织中层粘连蛋白也有类似的表达情形[19]。由此得出,在软骨组织不同的发育阶段,层粘连蛋白的表达与分布也随时间和空间的不同而变化,并且其表达的降低与软骨组织的退化密切相关。 值得一提的是,有学者在组织工程软骨组织中也发现了层粘连蛋白的表达,例如K?LLMER等[20]利用骨髓来源间充质干细胞形成软骨组织过程中,发现骨髓来源间充质干细胞在体外培养3周后,层粘连蛋白在细胞外基质中呈广泛性弥散式分布;JENG等[21]在山羊软骨缺损修复实验中发现层粘连蛋白在修复组织中广泛性表达,染色分析显示其呈分散性表达,显然有别于正常成熟软骨中层粘连蛋白集中分布在细胞周基质的表达方式。由此可见层粘连蛋白的表达与分布在组织工程软骨组织与体内天然软骨组织中不是完全相同的。 2.2 干细胞与层粘连蛋白 干细胞广义上包括胚胎干细胞、诱导多能干细胞及成体干细胞和祖细胞等。成人间充质干细胞属于成体干细胞,可来源于不同的成人组织,如骨髓、滑膜、肌肉、脂肪或牙齿等,具有与胚胎干细胞及诱导多能干细胞类似的成骨、成软骨以及成脂肪的三系分化潜能[22]。来源于胚胎干细胞或诱导多能干细胞的祖细胞也可以在特殊刺激因子作用下获得软骨细胞分化或其他组织细胞分化的潜能,在软骨组织工程和再生医学领域扮演着重要的角色[23-26]。目前各类干细胞在再生医学领域的研究还在继续,迄今为止已经发现多种不同的层粘连蛋白异构体对干细胞增殖与分化具有不同的调控作用[12,27-29],对细胞增殖的调控作用尤为显著[27]。 2.3 层粘连蛋白对干细胞增殖作用的调控 2.3.1 胚胎干细胞与诱导多能干细胞 在组织工程应用领域,软骨成功再生的关键一步首先是在诱导分化前通过促进增殖来获得足量的干细胞[7,30-31],而胚胎干细胞和诱导多能干细胞作为经典的多能细胞,都具有分化成软骨细胞的巨大潜能[25-26],被广泛研究用于获取足量干细胞。层粘连蛋白作为细胞外基质的重要组成成分,当其作为基质用于胚胎干细胞或诱导多能干细胞培养时,能够有效地促进相应干细胞的复制、增殖[31-34],见图1。目前人们通过小鼠胚胎干细胞增殖实验发现并比较了不同层粘连蛋白及其异构体对胚胎干细胞增殖的影响。DOMOGATSKAYA等[29]发现不同的层粘连蛋白异构体对胚胎干细胞的影响是多方面的,并利用平均倍增时间把层粘连蛋白异构体对胚胎干细胞增殖所产生的影响做了评估与分析。他们发现在没有其他分化抑制剂的情况下,LM-511仍具有支持小鼠胚胎干细胞增殖长达169 d的强效作用;同时他们分析了促进增殖的可能途径,证明LM-511是通过与整合素β1(主要是α6β1和αVβ1)的相互作用进而增加小鼠胚胎干细胞的黏附能力及其与基质的接触面积来影响小鼠胚胎干细胞的增殖能力;此外,他们还发现LM-332也能促进胚胎干细胞的增殖。由此可以发现,2种层粘连蛋白异构体LM-511与LM-332促进胚胎干细胞增殖的能力与胚胎干细胞对这2种层粘连蛋白异构体极强的黏附性密切相关。MIYAZAKI等[35]证明了LM-E8s作为最小的功能片段,与完整层粘连蛋白分子相比,促进人胚胎干细胞和人诱导多能干细胞增殖作用更加明显;同时应用整合素拮抗剂证明LM-E8片段增加了人胚胎干细胞的黏附性是通过识别整合素α6β1位点;此外,应用分子分析方法证明了促进增殖的可能信号途径是LM-E8通过刺激人胚胎干细胞及诱导多能干细胞的下游AKT、FAK和ERK1/2信号通路,借此影响细胞黏附、存活和迁移,进而促进人胚胎干细胞及诱导多能干细胞在保持未分化状态下的不断增殖。由此可以得出,层粘连蛋白通过调节细胞受体与环境的关系进而对胚胎干细胞和诱导多能干细胞增殖起到明显的促进作用。 目前虽然很多研究表明在上述体外二维环境培养情况下,层粘连蛋白能有效促进干细胞增殖,但毕竟与体内干细胞存在的三维细胞外基质微环境不同[36],干细胞在三维微环境中被大量细胞外基质所围绕。在三维培养系统中,有研究将层粘连蛋白用于支架的修饰以促进培养细胞所用支架的生物活性,进而为干细胞增殖提供有利的微环境[37-39]。HE等[40] 利用层粘连蛋白修饰聚L-丙交酯支架,发现更大量层粘连蛋白的加入可促进小鼠胚胎干细胞更加强效地增殖。由此可见在干细胞生长三维微环境中,层粘连蛋白支架修饰是提高细胞增殖的重要一步,同时细胞增殖跟层粘连蛋白的浓度相关。此外,NIE等[41]在实验中证实应用层粘连蛋白多层涂层培养能够促进人诱导多能干细胞的增殖。在最近的另一项关于层粘连蛋白对大鼠胚胎干细胞向心肌细胞分化的研究中,WANG等[42]发现层粘连蛋白比单层明胶能更加明显地促进大鼠胚胎干细胞增殖。由此可见三维微环境下,层粘连蛋白促进干细胞增殖作用同样显著。总而言之,无论二维还是三维环境中,层粘连蛋白作为细胞外基质中重要的功能成分,对胚胎干细胞及诱导多能干细胞增殖均发挥着重要的调控作用。 2.3.2 成体干细胞 成体干细胞已成为细胞移植、组织再生和组织工程领域重要的细胞来源,在创伤修复、细胞治疗、组织重建以及器官再生等方面具有巨大的应用前景[43]。在脂肪源性干细胞研究中,SANTIAGO等[19]发现利用层粘连蛋白衍生的序列对聚己内酯进行肽表面修饰,能够明显促进成体干细胞快速增殖。类似研究随后也有报道,如LAM等[33]比较了传统的组织培养聚苯乙烯板、基质胶、层粘连蛋白、纤连蛋白和聚L-赖氨酸分别作为附着表面时,发现层粘连蛋白处理组与其他几组相比能明显促进成体干细胞增殖,产生最多的附着细胞和增殖区域。此外,许多研究也报道了层粘连蛋白对间充质干细胞增殖能力的正向调节作用,如HASHIMOTO等[28]证实了层粘连蛋白异构体LM-332具有促进人间充质干细胞增殖的作用。他们通过测定4种不同层粘连蛋白异构体对人间充质干细胞的增殖作用及当整合素α3和α6相应阻断剂分别或共同存在时对干细胞增殖的影响,分析得出LM-332是通过α3β1及α6β1相互作用进而促进人间充质干细胞的有效增殖。MATHEWS等[44]分析了细胞外基质所含不同蛋白成分对人骨髓源性间充质干细胞增殖的影响,通过比较层粘连蛋白组与未处理组对人骨髓源性间充质干细胞的倍增时间,证明层粘连蛋白能促进人骨髓源性间充质干细胞快速增殖。李双星等[26]将鸡骨髓间充质干细胞分别加载在包被层粘连蛋白培养皿和传统培养皿中培养,结果显示层粘连蛋白培养体系对鸡骨髓间充质干细胞扩增能力更强,并且能够快速获得大量良好的未分化鸡骨髓间充质干细胞。HE等[40]研究发现用层粘连蛋白修饰的纳米纤维支架与单纯纳米支架相比能更好地促进小鼠神经干细胞快速增殖。类似结果还有很多,如LUO等[45]发现磷灰石-层粘连蛋白涂层培养能够明显促进神经干细胞增殖;LUO等[45]用共沉淀法制备含有层粘连蛋白的仿生磷灰石生物材料底物,对细胞无毒性且具有良好的生物相容性,同样能促进神经干细胞的增殖。综上所述,在经典的理论中利用层粘连蛋白诱导、促进成体干细胞增殖已经被广泛认可。相比于胚胎干细胞与诱导多能干细胞来源,由此制备干细胞用于解决组织工程细胞来源问题一定程度上减少了取材困难、伦理受限与分化不确定性,仍是未来组织工程或软骨修复研究的重点。 有趣的是层粘连蛋白的负性增殖作用也有部分报道,如ABAY等[46]研究发现层粘连蛋白通过减少细胞扩增、诱导细胞在支架上分化而抑制猪牙胚干细胞增殖;SEEGER等[11]将人间充质细胞培养在含或不含层粘连蛋白异构体的扩增培养基中7 d,发现LM-211,LM-411,LM-511以及LM-521均具有抑制未分化间充质细胞增殖的作用,在成肌培养基中也只有LM-521增强了间充质细胞的增殖速率。因此对于层粘连蛋白不同异构体对成体干细胞的确切作用与作用机制还有待更进一步研究。 2.3.3 祖细胞 由于祖细胞也具有一定程度的分化潜能,使其成为组织工程的另一个重要的细胞来源,因此通过层粘连蛋白调节祖细胞增殖进而制备软骨细胞、促进软骨形成成为了另一个用于软骨再生的有效方法[10,47]。在小鼠畸胎癌来源的软骨细胞系(ATDC5)的研究中,CHOI等[27]发现小鼠软骨祖细胞在层粘连蛋白衍生肽涂覆的杂交贻贝黏附蛋白表面(fp-151)培养时,层粘连蛋白能明显增加小鼠ATDC5细胞增殖。此外,KOMURA等[48]发现固定化LM-111,LM-332的底物可有效促进源自诱导多能干细胞的小鼠神经祖细胞的增殖。虽然上述研究提示层粘连蛋白具有促进祖细胞增殖的能力,但是在其他一些研究中也有相反的结果报道,认为层粘连蛋白通过一定程度减少细胞数量和扩增期细胞活力,对祖细胞起到抑制作用[37,46]。ARULMOLI等[49]发现人神经祖细胞在纤维蛋白和组合有透明质酸的支架中生长良好,但当加入层粘连蛋白时,通过免疫染色Ki-67定量方法分析结果显示层粘连蛋白不能显著增加细胞增殖。相同的结论在ADDINGTON等[50]所做的部分实验中得到证实,他们发现小鼠神经祖细胞在涂有层粘连蛋白的表面培养,其数量并没有明显增加。这使得层粘连蛋白对祖细胞增殖的影响似乎并不那么确切。然而,ADDINGTON等[50]所做的另一部分研究中发现含有层粘连蛋白、胶原蛋白、硫酸肝素蛋白多糖以及生长因子的基底膜基质却能够显著诱导细胞增殖,这一定程度上说明当层粘连蛋白与内在生长因子结合时能够更好地促进祖细胞的有效增殖。ADDINGTON等[50]探测神经损伤修复机制后发现添加基质细胞因子1α的层粘连蛋白组与未添加基质细胞因子1α组相比,神经祖细胞增殖明显更加强烈,推测神经祖细胞的强烈增殖是层粘连蛋白与基质细胞因子1α共同相互作用所产生的结果。虽然关于层粘连蛋白对祖细胞的研究并不像干细胞那样常见,但这些都说明层粘连蛋白在细胞因子的共同作用下,才能充分发挥其促进祖细胞增殖的作用,也提示在三维培养或体内时层粘连蛋白对干细胞的增殖作用也许并不是单纯的单一通路直接作用,还有复杂的多因素调控有待阐明。"
| [1] MADEIRA C, SANTHAGUNAM A, SALGUEIRO JB, et al. Advanced cell therapies for articular cartilage regeneration. Trends Biotechnol. 2015;33(1):35-42. [2] FELSON DT. Clinical practice. Osteoarthritis of the knee. N Engl J Med. 2006; 354(8):841-848. [3] MAMIDI MK, DAS AK, ZAKARIA Z, et al. Mesenchymal stromal cells for cartilage repair in osteoarthritis. Osteoarthritis Cartilage. 2016;24(8): 1307-1316. [4] FLANAGAN LA, REBAZA LM, DERZIC S, et al. Regulation of human neural precursor cells by laminin and integrins. J Neurosci Res. 2006; 83(5):845-856. [5] SUH HN, KIM MO, HAN HJ. Laminin-111 stimulates proliferation of mouse embryonic stem cells through a reduction of gap junctional intercellular communication via RhoA-mediated Cx43 phosphorylation and dissociation of Cx43/ZO-1/drebrin complex. Stem Cells Dev. 2012; 21(11):2058-2070. [6] LYNCH K, PEI M. Age associated communication between cells and matrix: a potential impact on stem cell-based tissue regeneration strategies. Organogenesis. 2014;10(3):289-298. [7] ABDAL DAYEM A, LEE S, CHOI HY, et al. The Impact of Adhesion Molecules on the In Vitro Culture and Differentiation of Stem Cells. Biotechnol J. 2018;13(2). [8] AUMAILLEY M. The laminin family. Cell Adh Migr. 2013;7(1):48-55. [9] AUMAILLEY M, BRUCKNER-TUDERMAN L, CARTER WG, et al. A simplified laminin nomenclature. Matrix Biol. 2005;24(5):326-332. [10] LEITON CV, ARANMOLATE A, EYERMANN C, et al. Laminin promotes metalloproteinase-mediated dystroglycan processing to regulate oligodendrocyte progenitor cell proliferation. J Neurochem. 2015;135(3):522-538. [11] SEEGER T, HART M, PATARROYO M, et al. Mesenchymal Stromal Cells for Sphincter Regeneration: Role of Laminin Isoforms upon Myogenic Differentiation. PLoS One. 2015;10(9):e0137419. [12] YAP L, TAY HG, NGUYEN MTX, et al. Laminins in Cellular Differentiation. Trends Cell Biol. 2019;29(12):987-1000. [13] GANDHI JK, KNUDSEN T, HILL M, et al. Human Fibrinogen for Maintenance and Differentiation of Induced Pluripotent Stem Cells in Two Dimensions and Three Dimensions. Stem Cells Transl Med. 2019;8(6):512-521. [14] KVIST AJ, NYSTRÖM A, HULTENBY K, et al. The major basement membrane components localize to the chondrocyte pericellular matrix--a cartilage basement membrane equivalent? Matrix Biol. 2008;27(1):22-33. [15] BARRY F, MURPHY M. Mesenchymal stem cells in joint disease and repair. Nat Rev Rheumatol. 2013;9(10):584-594. [16] FOLDAGER CB, TOH WS, CHRISTENSEN BB, et al. Collagen Type IV and Laminin Expressions during Cartilage Repair and in Late Clinically Failed Repair Tissues from Human Subjects. Cartilage. 2016;7(1):52-61. [17] CHU WC, ZHANG S, SNG TJ, et al. Distribution of pericellular matrix molecules in the temporomandibular joint and their chondroprotective effects against inflammation. Int J Oral Sci. 2017;9(1):43-52. [18] MOAZEDI-FUERST FC, GRUBER G, STRADNER MH, et al. Effect of Laminin-A4 inhibition on cluster formation of human osteoarthritic chondrocytes. J Orthop Res. 2016;34(3):419-426. [19] SANTIAGO LY, NOWAK RW, PETER RUBIN J, et al. Peptide-surface modification of poly(caprolactone) with laminin-derived sequences for adipose-derived stem cell applications. Biomaterials. 2006;27(15): 2962-2969. [20] KÖLLMER M, KESKAR V, HAUK TG, et al. Stem cell-derived extracellular matrix enables survival and multilineage differentiation within superporous hydrogels. Biomacromolecules. 2012;13(4): 963-973. [21] JENG L, OLSEN BR, SPECTOR M. Engineering endostatin-expressing cartilaginous constructs using injectable biopolymer hydrogels. Acta Biomater. 2012;8(6): 2203-2212. [22] TOH WS, FOLDAGER CB, PEI M, et al. Advances in mesenchymal stem cell-based strategies for cartilage repair and regeneration. Stem Cell Rev Rep. 2014; 10(5):686-696. [23] LI J, HANSEN KC, ZHANG Y, et al. Rejuvenation of chondrogenic potential in a young stem cell microenvironment. Biomaterials. 2014;35(2):642-653. [24] PIZZUTE T, LI J, ZHANG Y, et al. Fibroblast Growth Factor Ligand Dependent Proliferation and Chondrogenic Differentiation of Synovium-Derived Stem Cells and Concomitant Adaptation of Wnt/Mitogen-Activated Protein Kinase Signals. Tissue Eng Part A. 2016;22(15-16):1036-1046. [25] KO JY, KIM KI, PARK S, et al. In vitro chondrogenesis and in vivo repair of osteochondral defect with human induced pluripotent stem cells. Biomaterials. 2014;35(11):3571-3581. [26] 李双星,戚媛,朴丰源,等.利用层粘连蛋白扩增罗曼鹤鸡骨髓间充质干细胞[J].中国组织工程研究,2014,18(45):7222-7226. [27] CHOI BH, CHOI YS, KANG DG, et al. Cell behavior on extracellular matrix mimic materials based on mussel adhesive protein fused with functional peptides. Biomaterials. 2010;31(34):8980-8988. [28] HASHIMOTO J, KARIYA Y, MIYAZAKI K. Regulation of proliferation and chondrogenic differentiation of human mesenchymal stem cells by laminin-5 (laminin-332). Stem Cells. 2006;24(11):2346-2354. [29] DOMOGATSKAYA A, RODIN S, BOUTAUD A, et al. Laminin-511 but not -332, -111, or -411 enables mouse embryonic stem cell self-renewal in vitro. Stem Cells. 2008;26(11):2800-2809. [30] PEI M. Environmental preconditioning rejuvenates adult stem cells’ proliferation and chondrogenic potential. Biomaterials. 2017;117: 10-23. [31] PACCOLA MESQUITA FC, HOCHMAN-MENDEZ C, MORRISSEY J, et al. Laminin as a Potent Substrate for Large-Scale Expansion of Human Induced Pluripotent Stem Cells in a Closed Cell Expansion System. Stem Cells Int. 2019;2019:9704945. [32] LAPERLE A, HSIAO C, LAMPE M, et al. α-5 Laminin Synthesized by Human Pluripotent Stem Cells Promotes Self-Renewal. Stem Cell Reports. 2015;5(2): 195-206. [33] LAM MT, LONGAKER MT. Comparison of several attachment methods for human iPS, embryonic and adipose-derived stem cells for tissue engineering. J Tissue Eng Regen Med. 2012;6 Suppl 3:s80-86. [34] RODIN S, ANTONSSON L, NIAUDET C, et al. Clonal culturing of human embryonic stem cells on laminin-521/E-cadherin matrix in defined and xeno-free environment. Nat Commun. 2014;5:3195. [35] MIYAZAKI T, FUTAKI S, SUEMORI H, et al. Laminin E8 fragments support efficient adhesion and expansion of dissociated human pluripotent stem cells. Nat Commun. 2012;3:1236. [36] PEI M, LI JT, SHOUKRY M, et al. A review of decellularized stem cell matrix: a novel cell expansion system for cartilage tissue engineering. Eur Cell Mater. 2011; 22:333-343. [37] KANG BJ, RYU HH, PARK SS, et al. Effect of matrigel on the osteogenic potential of canine adipose tissue-derived mesenchymal stem cells. J Vet Med Sci. 2012; 74(7):827-836. [38] HEYDARKHAN-HAGVALL S, GLUCK JM, DELMAN C, et al. The effect of vitronectin on the differentiation of embryonic stem cells in a 3D culture system. Biomaterials. 2012;33(7):2032-2040. [39] BARROS D, CONDE-SOUSA E, GONÇALVES AM, et al. Engineering hydrogels with affinity-bound laminin as 3D neural stem cell culture systems. Biomater Sci. 2019; 7(12):5338-5349. [40] HE L, TANG S, PRABHAKARAN MP, et al. Surface modification of PLLA nano-scaffolds with laminin multilayer by LbL assembly for enhancing neurite outgrowth. Macromol Biosci. 2013;13(11):1601-1609. [41] NIE Y, WANG W, XU X, et al. Enhancement of human induced pluripotent stem cells adhesion through multilayer laminin coating. Clin Hemorheol Microcirc. 2018;70(4):531-542. [42] WANG D, WANG Y, LIU H, et al. Laminin promotes differentiation of rat embryonic stem cells into cardiomyocytes by activating the integrin/FAK/PI3K p85 pathway. J Cell Mol Med. 2019;23(5):3629-3640. [43] CELEBI B, MANTOVANI D, PINEAULT N. Effects of extracellular matrix proteins on the growth of haematopoietic progenitor cells. Biomed Mater. 2011;6(5):055011. [44] MATHEWS S, BHONDE R, GUPTA PK, et al. Extracellular matrix protein mediated regulation of the osteoblast differentiation of bone marrow derived human mesenchymal stem cells. Differentiation. 2012;84(2):185-192. [45] LUO D, RUAN S, LIU A, et al. Laminin functionalized biomimetic apatite to regulate the adhesion and proliferation behaviors of neural stem cells. Int J Nanomedicine. 2018;13:6223-6233. [46] ABAY N, GUREL PEKOZER G, RAMAZANOGLU M, et al. Bone Formation from Porcine Dental Germ Stem Cells on Surface Modified Polybutylene Succinate Scaffolds. Stem Cells Int. 2016;2016:8792191. [47] ORTINAU S, SCHMICH J, BLOCK S, et al. Effect of 3D-scaffold formation on differentiation and survival in human neural progenitor cells. Biomed Eng Online. 2010;9:70. [48] KOMURA T, KATO K, KONAGAYA S, et al. Optimization of surface-immobilized extracellular matrices for the proliferation of neural progenitor cells derived from induced pluripotent stem cells. Biotechnol Bioeng. 2015;112(11):2388-2396. [49] ARULMOLI J, WRIGHT HJ, PHAN DTT, et al. Combination scaffolds of salmon fibrin, hyaluronic acid, and laminin for human neural stem cell and vascular tissue engineering. Acta Biomater. 2016;43:122-138. [50] ADDINGTON CP, PAUKEN CM, CAPLAN MR, et al. The role of SDF-1α-ECM crosstalk in determining neural stem cell fate. Biomaterials. 2014;35(10):3263-3272. [51] TOH WS, FOLDAGER CB, HUI JH, et al. Exploiting Stem Cell-Extracellular Matrix Interactions for Cartilage Regeneration: A Focus on Basement Membrane Molecules. Curr Stem Cell Res Ther. 2016;11(8):618-625. [52] RAZAVI M, QIAO Y, THAKOR AS. Three-dimensional cryogels for biomedical applications. J Biomed Mater Res A. 2019;107(12): 2736-2755. [53] CHUN SY, LIM JO, LEE EH, et al. Preparation and Characterization of Human Adipose Tissue-Derived Extracellular Matrix, Growth Factors, and Stem Cells: A Concise Review. Tissue Eng Regen Med. 2019; 16(4):385-393. [54] SUN Y, CHEN S, PEI M. Comparative advantages of infrapatellar fat pad: an emerging stem cell source for regenerative medicine. Rheumatology (Oxford). 2018;57(12):2072-2086. [55] SCHREIBER RE, ILTEN-KIRBY BM, DUNKELMAN NS, et al. Repair of osteochondral defects with allogeneic tissue engineered cartilage implants. Clin Orthop Relat Res. 1999;(367 Suppl):S382-395. [56] KIYOTAKE EA, BECK EC, DETAMORE MS. Cartilage extracellular matrix as a biomaterial for cartilage regeneration. Ann N Y Acad Sci. 2016;1383(1):139-159. [57] SUN Y, YAN L, CHEN S, et al. Functionality of decellularized matrix in cartilage regeneration: A comparison of tissue versus cell sources. Acta Biomater. 2018; 74:56-73. |
| [1] | Lin Qingfan, Xie Yixin, Chen Wanqing, Ye Zhenzhong, Chen Youfang. Human placenta-derived mesenchymal stem cell conditioned medium can upregulate BeWo cell viability and zonula occludens expression under hypoxia [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(在线): 4970-4975. |
| [2] | Pu Rui, Chen Ziyang, Yuan Lingyan. Characteristics and effects of exosomes from different cell sources in cardioprotection [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(在线): 1-. |
| [3] | Zhang Yu, Tian Shaoqi, Zeng Guobo, Hu Chuan. Risk factors for myocardial infarction following primary total joint arthroplasty [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1340-1345. |
| [4] | Xiao Guoqing, Liu Xuanze, Yan Yuhao, Zhong Xihong. Influencing factors of knee flexion limitation after total knee arthroplasty with posterior stabilized prostheses [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1362-1367. |
| [5] | Wang Haiying, Lü Bing, Li Hui, Wang Shunyi. Posterior lumbar interbody fusion for degenerative lumbar spondylolisthesis: prediction of functional prognosis of patients based on spinopelvic parameters [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1393-1397. |
| [6] | Zhang Chao, Lü Xin. Heterotopic ossification after acetabular fracture fixation: risk factors, prevention and treatment progress [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1434-1439. |
| [7] | Gu Xia, Zhao Min, Wang Pingyi, Li Yimei, Li Wenhua. Relationship between hypoxia inducible factor 1 alpha and hypoxia signaling pathway [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1284-1289. |
| [8] | Wu Xun, Meng Juanhong, Zhang Jianyun, Wang Liang. Concentrated growth factors in the repair of a full-thickness condylar cartilage defect in a rabbit [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1166-1171. |
| [9] | Shen Jinbo, Zhang Lin. Micro-injury of the Achilles tendon caused by acute exhaustive exercise in rats: ultrastructural changes and mechanism [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1190-1195. |
| [10] | Chai Le, Lü Jianlan, Hu Jintao, Hu Huahui, Xu Qingjun, Yu Jinwei, Quan Renfu. Signal pathway variation after induction of inflammatory response in rats with acute spinal cord injury [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1218-1223. |
| [11] | Zhang Xiumei, Zhai Yunkai, Zhao Jie, Zhao Meng. Research hotspots of organoid models in recent 10 years: a search in domestic and foreign databases [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1249-1255. |
| [12] | Liu Cong, Liu Su. Molecular mechanism of miR-17-5p regulation of hypoxia inducible factor-1α mediated adipocyte differentiation and angiogenesis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1069-1074. |
| [13] | Wang Zhengdong, Huang Na, Chen Jingxian, Zheng Zuobing, Hu Xinyu, Li Mei, Su Xiao, Su Xuesen, Yan Nan. Inhibitory effects of sodium butyrate on microglial activation and expression of inflammatory factors induced by fluorosis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1075-1080. |
| [14] | Wang Xianyao, Guan Yalin, Liu Zhongshan. Strategies for improving the therapeutic efficacy of mesenchymal stem cells in the treatment of nonhealing wounds [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1081-1087. |
| [15] | Liao Chengcheng, An Jiaxing, Tan Zhangxue, Wang Qian, Liu Jianguo. Therapeutic target and application prospects of oral squamous cell carcinoma stem cells [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1096-1103. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||