Chinese Journal of Tissue Engineering Research ›› 2013, Vol. 17 ›› Issue (8): 1465-1472.doi: 10.3969/j.issn.2095-4344.2013.08.023
Previous Articles Next Articles
Bone cement is required for multi-segment osteoporotic compression fractures
Zeng Yi-wen1, 2, Xu Jian-an1, Gui Jian-chao2, Ma Yong1
- 1 Nanjing University of Chinese Medicine, Nanjing 210029, Jiangsu Province, China
2 Nanjing Hospital Affiliated to Nanjing Medical University, Nanjing 210006, Jiangsu Province, China
-
Received:2012-05-12Revised:2013-01-31Online:2013-02-19Published:2013-02-19 -
Contact:Gui Jian-chao, Doctor, Professor, Master’s supervisor, Chief physician, Nanjing Hospital Affiliated to Nanjing Medical University, Nanjing 210006, Jiangsu Province, China -
About author:Zeng Yi-wen☆, Studying for doctorate, Chief physician, Nanjing University of Chinese Medicine, Nanjing 210029, Jiangsu Province, China; Nanjing Hospital Affiliated to Nanjing Medical University, Nanjing 210006, Jiangsu Province, China zenyiwen@163.com
CLC Number:
Cite this article
Zeng Yi-wen1,,Xu Jian-an, Gui Jian-chao, Ma Yong. Bone cement is required for multi-segment osteoporotic compression fractures[J]. Chinese Journal of Tissue Engineering Research, 2013, 17(8): 1465-1472.
share this article
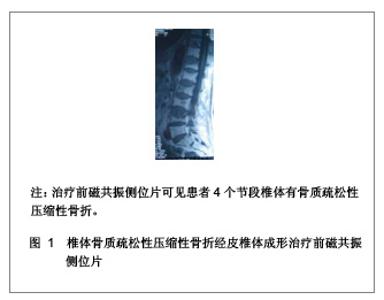
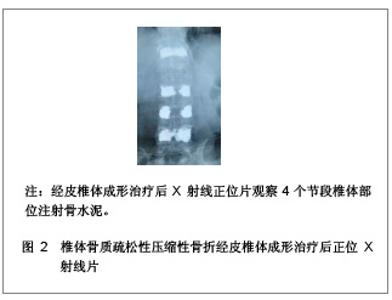
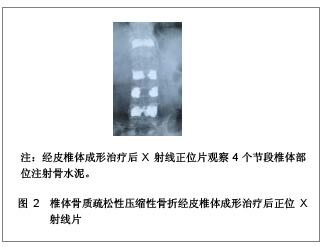
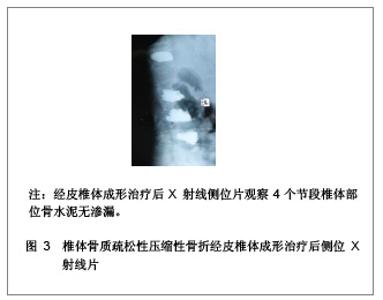
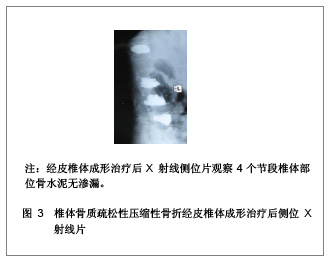
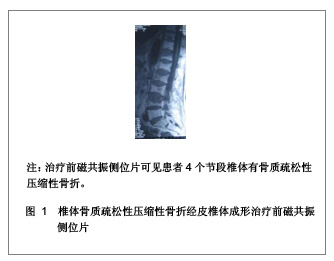
1 骨水泥材料充填经皮椎体成形治疗多节段骨质疏松性压缩性骨折的病例分析 对象:选取2006年1月至2011年1月在南京医科大学附属南京医院骨科就诊的多椎体(3个节段及以上)骨质疏松性压缩性骨折患者32例,共158个椎体,采用经皮椎体成形治疗方法。男13例,女19例,患者年龄在64-87岁,平均年龄为74岁。骨折部位在T2-L4,胸椎骨折椎体有96个,腰椎骨折椎体有62个。骨折压缩程度为20%-90%。治疗前疼痛强度评价的目测类比评分为(8.3±1.6)分。治疗前进行常规血常规、凝血功能、血液化生化、心电图、胸部透视等各项检查。 纳入标准:①所以患者均无明显外伤史或者仅有轻微的外伤史,患者病程在2 d-10年,服用非甾体类抗炎药或止痛药效果不明显,腰背部明显胀痛,患者在坐立和行走过程中疼痛症状加重,以至于患者被迫卧床休息。②病变椎体棘突会有明显压痛和叩击痛。③治疗前所有患者均行X射线及磁共振检查,均被确诊为骨质疏松性压缩性骨折的患者。④治疗前CT结果显示椎管无塌陷和狭窄,且周围骨壁完整。 排除标准:①患者年龄小于55岁。②椎体骨髓炎患者。③非骨质疏松性椎体急性创伤性骨折。④对治疗药物有过敏反应或有凝血机制障碍的患者。 主要仪器:微创器械及特制球囊,来源为Kyphon公司。flexiveiw8800C型臂X射线机来源为GE公司产品。 治疗方法:采用微创器械及特制球囊在flexiveiw8800C型臂X线机下行经皮椎体成形治疗。35个椎体行经皮椎体成形治疗,123个椎体行经皮球囊椎体后凸成形治疗。114个椎体单侧椎弓根注射骨水泥,34个椎体双侧椎弓根注射骨水泥。治疗后进行透视检查,若注射的骨水泥未超越椎体中线,再行对侧穿刺,以防止椎体受力不均匀。治疗后观察患者的双下肢感觉和运动情况,同时监测患者的生命体征,并进行血常规及血液大生化复查,患者在平卧 4 h 后即可自由翻身,给予抗生素治疗1 d。 疗效评估:治疗后患者行X 射线和CT检查,了解患者骨折部位椎体的复位情况、骨水泥的分布情况以及是否出现骨水泥外漏。检查患者治疗前后血常规和大生化指标的变化,以及对患者的影响。采用CT容量分析法检测骨水泥注射治疗前后椎体的容积变化,患者在治疗前后疼痛强度评价的目测类比评分变化,并进行组间t检验比较,P < 0.05为差异有显著性意义。目测类比评分[3]:0分为无痛,1-3分为轻度疼痛,4-6分为中度疼痛,7-9分为重度疼痛,10分为剧烈疼痛。 治疗结果:32例患者对158个椎体进行骨水泥注射,治疗过程中的失血量忽略不计,所有患者均纳入结果分析,无神经根及脊髓损伤,未出现肺栓塞和心脑血管系统的急性反应。治疗前后血常规、大生化无明显变化,治疗对人体机能无影响。每个椎体的手术时间为(19.4±3.6) min,注射骨水泥(4.2±2.1) mL。每个患者的手术时间为(88.5±23.4) min,骨水泥注射量为(20.4±8.6) mL。治疗后X射线片检查显示,注射的骨水泥呈斑片状、团块状或弥散状分布。 典型病例分析:患者女,69岁,4个节段椎体骨质疏松性压缩性骨折一期经皮椎体成形治疗的前后磁共振和X射线片,见图1-3。"

注射骨水泥治疗前椎体体积是(22.2±8.6) cm3,治疗后椎体体积增加到(24.8±6.9) cm3,椎体体积变化差异有统计学意义(P < 0.05)。充填治疗后发现有6个椎体出现骨水泥外溢现象,其中椎管内硬膜外有2个外溢,椎旁静脉4个外溢,但椎弓根和椎管的结构保持完整。 治疗后48 h目测类比评分为(2.2±3.7)分,较骨水泥充填治疗前的(8.3±1.6)分有明显降低(t=25.2, P < 0.05),26例患者在2 d后可以佩戴腰围下床活动,28例患者在2个月后腰部疼痛症状消失。15例患者治疗后3 d即可出院,5例患者治疗后7 d出院,2例患者治疗后14 d出院。治疗后随访时间6-16个月,平均随访时间为10个月,均无椎体塌陷、疼痛复发以及神经症状。"


2 经皮椎体成形治疗多节段骨质疏松性压缩性骨折的充填材料 用于经皮椎体成形治疗的理想充填材料应具备以下特点:①具有良好的显影性能。②治疗中调制的操作简便,易于注射,黏度适宜。③聚合温度适宜。④治疗中的操作时间在6-10 min,凝固时间15 min左右。⑤具有良好的生物力学特性。⑥无毒,使用安全。⑦具有良好的骨传导性和骨诱导性。⑧良好的重吸收率。⑨良好的生物相容性及生物活性。⑩价格合理,能被大部分患者所接受。此外,骨水泥填充材料还应该适合于作为一些药物和生物活性因子的载体,在治疗的过程中具有缓释作用。 聚甲基丙烯酸甲酯是目前应用最为广泛的椎体成形治疗充填材料,聚甲基丙烯酸甲酯由液态的单体和粉状的多聚体通过聚合反应凝结而形成,瞬间具有较高的黏滞性,注射需要时间长,具有低聚合温度的特点,降低骨水泥渗漏的发生率[4-6]。目前,用于经皮椎体成形治疗的骨水泥主要有硫酸钙骨水泥、磷酸钙骨水泥、珍珠母粉末、石珊瑚颗粒、复合骨水泥。 2.1 硫酸钙骨水泥用于经皮椎体成形治疗骨质疏松性压缩性骨折 硫酸钙骨水泥是一种新型的骨充填材料,能够充分满足椎体充填材料的生物力学要求,硫酸钙骨水泥的特点是可以在骨缺损部位快速的硬化,在局部形成微酸性的环境,使破骨细胞将硫酸钙骨水泥吸收,使成骨细胞吸附在硫酸钙骨水泥上,有利于血管和成骨细胞的长入。目前,以高纯度α晶体结构硫酸钙为原料制成的MIIG X 3(美国WRIGHT 医疗技术公司),由特制硫酸钙粉剂和相应的稀释剂构成,已在临床得到应用,固化时间约在5 min,固化后形成的微孔样结构,有利于新骨的长入,材料的强度与松质骨结构相似,对骨骼临时的内部支撑起重要作用,而且可以在10-14周的时间里100%被吸收[7-9]。硫酸钙骨水泥用于经皮椎体成形治疗骨质疏松性压缩性骨折的文献分析,见表1。"


此外,磷酸钙骨水泥可以减少椎体成形治疗后与相邻椎体之间的应力梯度问题,延缓椎体的退变和相邻椎体骨折的发生率[24]。磷酸钙骨水泥充填后椎体具有良好的生物力学强度[25]。在体内自固化过程中不会产生热量,具有骨传导性和生物降解性等特点,可以克服界面松动和生物相容性差的不足[26]。老年骨质疏松患者在进行椎体成形治疗后,疼痛缓解效果良好,椎体塌陷及假关节形成的危险有所降低[27]。但磷酸钙骨水泥材料也存在一些问题,如无成骨细胞活性,遇到血液和体液固化时比较困难,材料黏稠度不够、显影效果欠佳、力学性能不足、材料脆性较大、生物降解速度很难调控等缺点。随着磷酸钙骨水泥性状研究的改进,会研制出更符合临床应用要求的材料。 刘兴炎等[28]研究表明应用复合重组人类骨形态发生蛋白2的磷酸钙骨水泥可以加速骨质疏松性骨缺损的愈合,是治疗骨质疏松性骨缺损的良好骨科修复重建材料。赵刚等[29]对山羊标本电镜观察中发现,聚甲基丙烯酸甲酯材料与宿主骨组织之间不能够紧密结合,复合重组人类骨形态发生蛋白2的磷酸钙骨水泥能够与宿主骨组织紧密结合,而且未见界膜形成,复合重组人类骨形态发生蛋白2的磷酸钙骨水泥具有可操作性,骨替代时间较短的特点,有利于永久性的重建椎体的稳定性。周英杰等[30]使用复合重组人类骨形态发生蛋白2的磷酸钙骨水泥充填治疗15例胸椎和腰椎压缩性骨折的患者,患者经治疗后活动能力较治疗前得到明显增加,且疼痛症状得到明显改善。 2.3 珍珠母粉末、石珊瑚颗粒用于经皮椎体成形治疗骨质疏松性压缩性骨折 珍珠母的成分可以被成骨细胞所识别,并可以诱导成骨,具有良好的生物相容性。Lamghari等[31]通过对动物实验发现珍珠母粉末与自体的血液相混合制成糊状后可以容易注射,并可以释放刺激因子,诱导成骨细胞的分化,具有良好的骨诱导性。 石珊瑚的主要成分是碳酸钙,材料的空隙率与人的松质骨结构相似,孔径大小在190-260 μm,适宜新骨长入的同时具有骨传导作用。严宁等[32]通过基础实验发现天然珊瑚颗粒和复合纤维蛋白的天然珊瑚颗粒均能够促进骨的形成。Cunin等[33]发现加入骨形态发生蛋白的天然珊瑚颗粒能够稳定骨质疏松性椎体骨折,具有良好的生物相容性、可生物降解性以及骨传导性,在X射线片显影方面还具有良好的对比性。 目前,珍珠母粉末和石珊瑚颗粒仅作为一种骨科替代充填材料来使用,其材料特性和临床应用还需深入研究。 2.4 复合骨水泥用于经皮椎体成形治疗骨质疏松性压缩性骨折 对于复合骨水泥的研究目前还处于探索研究阶段。如玻璃陶瓷强化复合骨水泥Orthocomp、Cortoss、Hydroxy apatite composite resin以及可生物降解的骨水泥,如天然珊瑚骨替代物和磷酸钙骨等。复合骨水泥材料与聚甲基丙烯酸甲酯具有相似的基本性质,但较聚甲基丙烯酸甲酯有更合适的黏稠度、X射线片的不透射性、硬化快、产热低、具有更好的力学性能、生物活性及骨诱导性等优点。Orthocomp是以甲基丙烯酸甲酯的衍生物为基质,具有生物活性的骨水泥,其表面具有亲水性,可以使骨水泥通过表面的化学键与松质骨连结,具有良好的骨传导作用,缺点是不能诱导骨组织的生长,也不能生物降解[34]。Cortoss是一种新型的合成填充物,容易弥散进入松质骨,并且弹性模量与骨结果非常相似。钱明等[35]将Cortoss新型生物骨水泥与传统的聚甲基丙烯酸甲酯骨水泥进行生物力学和聚合温度比较,将聚甲基丙烯酸甲酯与新型骨水泥Cortoss按照ISO5833:2002标准分别制作成抗压及抗弯模型,将制作的模型使用生物力学机测试两种骨水泥的抗弯及抗压模量,同时在制模过程中测量骨水泥聚合温度。结果发现,与聚甲基丙烯酸甲酯骨水泥比较,Cortoss骨水泥在聚合过程中最高温度相对较低、抗压强度较强、抗弯模量降低,差异均具有显著性意义(P < 0.05),抗弯强度间差异无显著性意义(P > 0.05)。说明Cortoss生物力学强度优于聚甲基丙烯酸甲酯骨水泥,由于Cortoss在聚合时放热较少,所以其用于外科治疗时有较好的安全性。"

| [1] Galibert P, Deramond H, Rosat P, et al.Preliminary note on the treatment of vertebral angioma by percutaneous acrylic vertebroplasty.Neurochirurgie.1987;33(2):166-168.[2] Deramond H, Depriester C, Galibert P, et al.Percutaneous vertebroplasty with polymethylmethacrylate. Technique, indications, and results.Radiol Clin North Am.1998;36(3): 533-546.[3] Lieberman IH, Dudeney S, Reinhardt MK, et al.Initial outcome and efficacy of "kyphoplasty" in the treatment of painful osteoporotic vertebral compression fractures.Spine (Phila Pa 1976).2001;26(14):1631-1638.[4] Georgy BA.Clinical experience with high-viscosity cements for percutaneous vertebral body augmentation: occurrence, degree, and location of cement leakage compared with kyphoplasty.AJNR Am J Neuroradiol.2010;31(3):504-508. [5] 蔺福辉,易小波,任志宏,等.高粘度骨水泥在椎体成形术中的应用[J].中国骨与关节损伤杂志,2009,24(1):66-67.[6] Anselmetti GC, Zoarski G, Manca A, et al.Percutaneous vertebroplasty and bone cement leakage: clinical experience with a new high-viscosity bone cement and delivery system for vertebral augmentation in benign and malignant compression fractures.Cardiovasc Intervent Radiol.2008; 31(5):937-947.[7] 姜星杰,吴小涛,张绍东,等.胸腰椎爆裂骨折模型中硫酸钙骨水泥椎体成形术的生物力学性能[J].中国矫形外科杂志,2008,16(16): 1248-1258.[8] 张绍东,王宸,姜星杰,等.硫酸钙骨水泥在胸腰椎爆裂骨折椎体成形术的生物力学性能[J]. 中国矫形外科杂志,2009,17(4): 288-292.[9] Perry A, Mahar A, Massie J, et al.Biomechanical evaluation of kyphoplasty with calcium sulfate cement in a cadaveric osteoporotic vertebral compression fracture model.Spine J. 2005;5(5):489-493.[10] Kelly CM, Wilkins RM.Treatment of benign bone lesions with an injectable calcium sulfate-based bone graft substitute. Orthopedics.2004;27(1):131-135.[11] 赵磊,王黎明,王钢锐,等.经皮椎体后凸成形术(PKP)结合可注射硫酸钙(MIIG X3)治疗胸腰椎骨质疏松性压缩骨折[J].中国骨质疏松杂志,2007,13(6):424-428.[12] Turner TM, Urban RM, Singh K, et al.Vertebroplasty comparing injectable calcium phosphate cement compared with polymethylmethacrylate in a unique canine vertebral body large defect model.Spine J.2008;8(3):482-487.[13] Grafe IA, Baier M, Nöldge G, et al.Calcium-phosphate and polymethylmethacrylate cement in long-term outcome after kyphoplasty of painful osteoporotic vertebral fractures. Spine. 2008;33(11):1284-1290.[14] Link DP, van den Dolder J, Jurgens WJ, et al.Mechanical evaluation of implanted calcium phosphate cement incorporated with PLGA microparticles.Biomaterials. 2006; 27(28):4941-4947.[15] Wang X, Ye J, Wang Y, et al.Self-setting properties of a beta-dicalcium silicate reinforced calcium phosphate cement.J Biomed Mater Res B Appl Biomater.2007;82(1): 93-99.[16] Lin J, Zhang S, Chen T, et al.Calcium phosphate cement reinforced by polypeptide copolymers.J Biomed Mater Res B Appl Biomater.2006;76(2):432-439.[17] 郑召民,郭家伟,崔力扬,等.磷酸钙骨水泥的显影性及生物力学实验研究[J].中国脊柱脊髓杂志,2006,16(9):696-699. [18] Pan Z, Jiang P, Fan Q, et al.Mechanical and biocompatible influences of chitosan fiber and gelatin on calcium phosphate cement.J Biomed Mater Res B Appl Biomater.2007;82(1): 246-252. [19] Seeherman H, Li R, Bouxsein M, et al.rhBMP-2/calcium phosphate matrix accelerates osteotomy-site healing in a nonhuman primate model at multiple treatment times and concentrations.J Bone Joint Surg Am.2006;88(1):144-160.[20] Wang X, Ye J, Wang Y.Influence of a novel radiopacifier on the properties of an injectable calcium phosphate cement. Acta Biomater.2007;3(5):757-763.[21] 巫洪波,武明鑫,邓兰英,等.复合生物型自体红骨髓磷酸钙水泥加可吸收颗粒治疗骨质疏松性脊柱骨折[J].中国医药导报,2009,6(8):26-27. [22] 董伟强,白波,张妹江,等.纳米碳管处理磷酸钙骨水泥的生物相容性及其强度和韧性[J]. 中国组织工程研究与临床康复,2010, 14(29):5363-5366. [23] Pina S, Torres PM, Ferreira JM.Injectability of brushite-forming Mg-substituted and Sr-substituted alpha-TCP bone cements.J Mater Sci Mater Med.2010; 21(2):431-438.[24] 白楚杰,张烽,董启榕,等.应用三维有限元法评估聚甲基丙烯酸甲酯和磷酸钙骨水泥铸件的力学特征[J].中国组织工程研究与临床康复,2007,11(12):4313-4317.[25] 朱雪松,张志明,毛海青,等.磷酸钙骨水泥填充椎体成形过程中椎体生物力学评价[J].中国组织工程研究与临床康复,2008, 12(41): 8071-8074. [26] 陈艺,白波,孙辉,等.可注射磷酸钙骨水泥在经皮椎体后凸成形术中的体外生物力学评价[J].中华生物医学工程杂志,2007,13(4): 216. [27] Ishiguro S, Kasai Y, Sudo A, et al.Percutaneous vertebroplasty for osteoporotic compression fractures using calcium phosphate cement.J Orthop Surg (Hong Kong).2010; 18(3):346-351.[28] 刘兴炎,历孟,刘旭东,等.rhBMP-2/CPC 对骨质疏松骨缺损的治疗作用的研究[J].中国骨质疏松杂志,2009,15(1):16-19.[29] 赵刚,刘宏建,周英杰,等.不同充填材料应用于经皮椎体成形术的效果及影像特征[J].中国组织工程研究与临床康复,2010,14(16): 3007-3010. [30] 周英杰,赵刚,刘宏建,等.复合 BMP 的磷酸钙骨水泥应用于椎体成形术治疗骨质疏松性胸腰椎压缩骨折15例[J].郑州大学学报:医学版,2009,44(5):1101-1103. [31] Lamghari M, Berland S, Laurent A, et al.Bone reactions to nacre injected percutaneously into the vertebrae of sheep. Biomaterials.2001;22(6):555-562. [32] 严宁,张汉东,周磊,等.Y500R可吸收珊瑚羟基磷灰石人工骨体外成骨细胞相容性研究[J].临床口腔医学杂志,2006,22(8): 456-459.[33] Cunin G, Boissonnet H, Petite H, et al.Experimental vertebroplasty using osteoconductive granular material.Spine (Phila Pa 1976).2000;25(9):1070-1076.[34] Togawa D, Kovacic JJ, Bauer TW, et al.Radiographic and histologic findings of vertebral augmentation using polymethylmethacrylate in the primate spine: percutaneous vertebroplasty versus kyphoplasty.Spine (Phila Pa 1976). 2006;31(1):4-10.[35] 钱明,潘俊,孟斌,等.聚甲基丙烯酸甲酯骨水泥与Cortoss骨水泥生物力学性能及聚合温度比较[J].中国组织工程研究与临床康复,2012,16(16):2935-2938. [36] Cotten A, Boutry N, Cortet B, et al.Percutaneous vertebroplasty: state of the art.Radiographics.1998;18(2): 311-320.[37] Muto M, Muto E, Izzo R, et al.Vertebroplasty in the treatment of back pain.Radiol Med.2005;109(3):208-219.[38] Cotten A, Dewatre F, Cortet B, et al.Percutaneous vertebroplasty for osteolytic metastases and myeloma: effects of the percentage of lesion filling and the leakage of methyl methacrylate at clinical follow-up.Radiology.1996;200(2): 525-530.[39] Liebschner MA, Rosenberg WS, Keaveny TM.Effects of bone cement volume and distribution on vertebral stiffness after vertebroplasty.Spine (Phila Pa 1976).2001;26(14):1547-1554.[40] Komemushi A, Tanigawa N, Kariya S, et al.Percutaneous vertebroplasty for compression fracture: analysis of vertebral body volume by CT volumetry.Acta Radiol.2005;46(3): 276-279.[41] Brown DB, Glaiberman CB, Gilula LA, et al.Correlation between preprocedural MRI findings and clinical outcomes in the treatment of chronic symptomatic vertebral compression fractures with percutaneous vertebroplasty.AJR Am J Roentgenol.2005;184(6):1951-1955.[42] Syed MI, Patel NA, Jan S, et al.New symptomatic vertebral compression fractures within a year following vertebroplasty in osteoporotic women.AJNR Am J Neuroradiol.2005;26(6): 1601-1604. |
| [1] | Li Li, Ma Li. Immobilization of lactase on magnetic chitosan microspheres and its effect on enzymatic properties [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 576-581. |
| [2] | Zhou Anqi, Tang Yufei, Wu Bingfeng, Xiang Lin. Designing of periosteum tissue engineering: combination of generality and individuality [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3551-3557. |
| [3] | Lang Limin, He Sheng, Jiang Zengyu, Hu Yiyi, Zhang Zhixing, Liang Minqian. Application progress of conductive composite materials in the field of tissue engineering treatment of myocardial infarction [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3584-3590. |
| [4] | Xie Jian, Su Jiansheng. Advantages and characteristics of electrospun aligned nanofibers as scaffolds for tissue engineering [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(16): 2575-2581. |
| [5] | Ji Qi, Yu Zhengwen, Zhang Jian. Problems and trends of technique and clinical application of metallic biomaterials prepared by three-dimensional printing technology [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(16): 2597-2604. |
| [6] | Zhang Le, Pan Bin, Ruan Ruxin, Guo Kaijin. Effect of lumbopelvic sagittal parameters on secondary adjacent vertebral fracture after percutaneous kyphoplasty for osteoporotic vertebral compression fracture [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(15): 2370-2374. |
| [7] | Qian Nannan, Zhang Qian, Yang Rui, Ao Jun, Zhang Tao. Mesenchymal stem cells in the treatment of spinal cord injury: cell therapy and combination of new drugs and biomaterials [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(13): 2114-2120. |
| [8] | Jia Wei, Zhang Mandong, Chen Weiyi, Wang Chenyan, Guo Yuan. Effects of femoral prosthetic materials on artificial knee arthroplasty performance [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(10): 1477-1481. |
| [9] | Wang Qian, Li Lu, Shu Jingyuan, Dong Zhiheng, Jin Youshi, Wang Qingshan. Micro-morphology and phase of zirconia-based nano-hydroxyapatite functional gradient biomaterials [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(10): 1517-1521. |
| [10] | Jing Wanli, Zhang Tao, Teng Donghui, Shi Tao, Zhou Qiang. Poor outcomes of bone filling mesh container vertebroplasty for the treatment of osteoporotic vertebral compression fractures with vertebral body wall incompetence [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(10): 1522-1527. |
| [11] | Zhang Zhiwei, Li Li, Huang Ziyu, Wu Duoyi, Gan Farong, Ye Baofei, Zhang Yan, Zhang Taibiao, Hu Wanjun. Percutaneous vertebroplasty through unilateral and bilateral pedicle approaches and unilateral pedicle extrapedicle approach for the treatment of thoracolumbar vertebral compression fractures: bone cement perfusion volume and cement leakage rate [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(9): 1353-1358. |
| [12] | Wang Yiya, Zhang Han, Lan Hai. Digital evaluation of finite element model for percutaneous kyphoplasty with bone cement injection [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(9): 1378-1383. |
| [13] | Liu Qun, Sun Dongdong, Gao Lilan, He Zhijiang, Sun Minglin. Risk factors for fractures secondary to percutaneous kyphoplasty: a meta-analysis [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(6): 976-984. |
| [14] | Li Li, Ma Li, Li He. Preparation and characterization of magnetic chitosan microspheres [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(4): 577-582. |
| [15] | Tang Mengmeng, Chen Hechun, Xie Hongchen, Zhang Yu, Tan Xiaoshuang, Sun Yixuan, Huang Yina. Histocompatibility of poly(L-lactide-co-ε-caprolactone)/cross-linked polyvinylpyrrolidone ureteral stent grafted into the rat bladder [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(4): 583-588. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||