Chinese Journal of Tissue Engineering Research ›› 2021, Vol. 25 ›› Issue (16): 2575-2581.doi: 10.3969/j.issn.2095-4344.3768
Previous Articles Next Articles
Advantages and characteristics of electrospun aligned nanofibers as scaffolds for tissue engineering
Xie Jian, Su Jiansheng
- Department of Prosthodontics, School and Hospital of Stomatology, Tongji University, Shanghai Engineering Research Center of Tooth Restoration and Regeneration, Shanghai 200072, China
-
Received:2020-03-19Revised:2020-03-24Accepted:2020-05-23Online:2021-06-08Published:2021-01-07 -
Contact:Su Jiansheng, Professor, Department of Prosthodontics, School and Hospital of Stomatology, Tongji University, Shanghai Engineering Research Center of Tooth Restoration and Regeneration, Shanghai 200072, China -
About author:Xie Jian, Doctoral candidate, Department of Prosthodontics, School and Hospital of Stomatology, Tongji University, Shanghai Engineering Research Center of Tooth Restoration and Regeneration, Shanghai 200072, China -
Supported by:the National Natural Science Foundation of China (General Program), No. 81873715, 81572114 (to SJS); the Project of Shanghai Science and Technology Commission, No.18441902100 (to SJS)
CLC Number:
Cite this article
Xie Jian, Su Jiansheng. Advantages and characteristics of electrospun aligned nanofibers as scaffolds for tissue engineering[J]. Chinese Journal of Tissue Engineering Research, 2021, 25(16): 2575-2581.
share this article

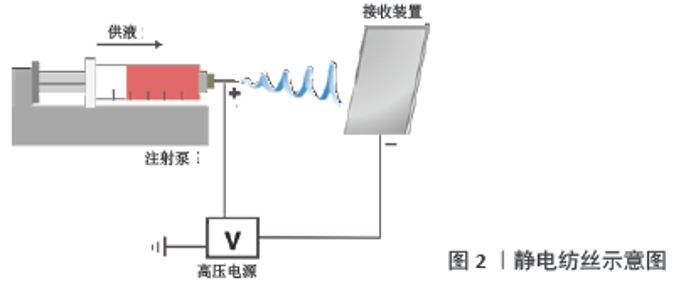
此前已有报道对静电纺丝取向纤维支架的制备方法进行了系统的总结[11],文章着重介绍下列3种主要方法:应用传统滚筒接收装置的静电纺丝;辅助电场或磁场感应的静电纺丝;近场静电纺丝。其中,滚筒接收装置的应用最为广泛(图3A)。当电纺纳米纤维到达旋转轴心时纤维被迅速拉向滚筒,抵消纤维的鞭打运动并形成取向纤维结构,这种方法能够大规模生产连续且高度有序的纳米纤维,但通常要求轴心具有高旋转速度(1 000 r/min或更高),因此难以保证其安全性[12]。此外,如图3B、C所示,笼状滚筒、旋转盘等收集装置也已经用于电纺取向纳米纤维支架的制备,应用笼状滚筒制备的取向纳米纤维取向度可获得较大提升,与普通滚筒相比其对转速要求较低,且纺丝效率较高。此外,应用旋转盘接收装置也可获得取向程度较高的纳米纤维,但其接收面积较小且效率较低,限制了其进一步的发展[1]。 "


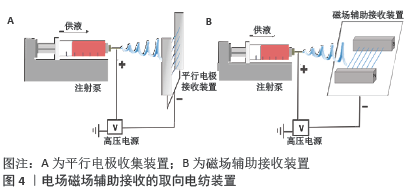
除了改变接收装置,也可应用电场及磁场辅助(图4)控制沉积纤维的排列[13-14],这种方法能够提高纤维取向度,但收集范围较小,且随着纤维沉积厚度增加纤维取向度有所下降。目前这种方法多集中于实验室研究,缺乏大规模生产的能力。此外,近场静电纺丝也是一种在平坦表面上形成高度可控静电纺丝图案的有力手段。在近场电纺中针头与收集器之间的距离非常短(500 μm至3 mm),可防止电纺纤维弯曲不稳定性和断裂,但其价格昂贵且设置复杂,因此限制了其广泛应用[15]。值得注意的是,除上述取向电纺纤维接收装置外,ZHANG等[16]曾报道过多种图案化结构的二维或三维纳米纤维结构。文章聚焦于单纯电纺取向纳米纤维构成的组织工程支架,故图案化结构支架未计入综述范畴。 "

2.2 静电纺丝取向纳米纤维支架在组织工程领域的应用 2.2.1 神经组织工程 周围神经系统直接影响人体的运动和感觉功能,一旦发生损伤可能导致慢性疼痛、感觉或运动功能异常及肌肉萎缩等并发症[17],因此周围神经损伤的治疗对于维持身体功能和生活质量至关重要。近年来,组织工程化神经移植物逐渐受到学者们的关注。组织工程神经导管应当具有可控的降解速率及接近天然组织的强度、良好的生物相容性及对神经细胞的引导作用[17]。其中,静电纺丝取向纳米纤维具有与天然神经纤维束相似的结构[18],并且能够引导神经细胞定向迁移[19]、促进神经干细胞分化[20],已被应用于神经再生的研究中。 HUANG等[21]应用静电纺丝技术制备了表面带有凹槽的取向纳米纤维支架,并证实开槽设计能够协同纳米纤维的取向结构促进大鼠坐骨神经再生。此外,WANG等[22]设计了一种导电取向电纺纳米纤维支架,其研究表明该取向纤维能够引导神经内分泌细胞系PC12细胞与背根神经元沿纤维长轴定向生长。并且,电纺取向纳米纤维与电刺激的协同效应也可增强许旺细胞的黏附、增殖及髓鞘碱性蛋白表达。这些研究证实了电纺取向纳米纤维对神经元再生的引导作用,但开发能够促进神经元生长的三维仿生支架仍然是一项艰巨的挑战。 最近有学者设计了一种“芯-壳”结构三维支架,将导电取向电纺纳米纤维纱线及水凝胶分别作为芯层及壳层,以模拟天然神经组织的分层排列结构,水凝胶外壳能够提供支撑及保护作用,其三维取向结构能够诱导神经细胞迁移及神经突延伸[23]。同样,尹桂波等[24]制备了由高度取向电纺纤维构成的“芯-鞘”结构神经移植物,将其应用于大鼠坐骨神经缺损模型后发现该神经导管有引导神经再生的潜能。由此证实,静电纺丝取向纤维的表面形貌能够刺激周围神经再生,然而其潜在机制尚未完全阐明。JIA等[25]对此进行了探究并发现电纺取向纳米纤维能够诱导愈合前巨噬细胞表型(M2型),而随机纳米纤维则诱导炎症前表型(M1型)。此外,取向纳米纤维诱导的M2型巨噬细胞可显著促进许旺细胞的增殖及迁移,体内实验同样证实其可提高许旺细胞浸润及轴突再生的比例。这项研究表明,取向纤维构成的神经导管能够差异性调节巨噬细胞极化,并通过诱导巨噬细胞的促愈合表型进而促进外周神经再生。因此,以电纺取向纤维为基础的神经导管有希望应用于神经组织工程支架。 2.2.2 肌腱组织工程 肌腱为致密的纤维结缔组织,能够连接骨骼与肌肉,并在应力传递及维持关节稳定中发挥关键作用。肌腱损伤常导致关节不稳定及关节运动异常[26]。由于肌腱组织神经支配有限、血管化较差且细胞含量较少,导致其自然愈合效率极低[27]。目前组织工程学已成为肌腱损伤的一种潜在疗法,因此迫切需要开发能够用于肌腱修复的生物材料[28]。此前的研究强调了生物材料基质结构在干细胞命运调控中的重要性[29]。鉴于肌腱细胞主要位于由平行胶原纤维构成的微环境中,应用纳米/微米级取向电纺纤维作为肌腱组织工程支架可能是一个明智的选择[30]。 ZHANG等[27]应用静电纺丝技术制备了模拟天然肌腱微观结构与机械性能的左旋聚乳酸取向纳米纤维,研究表明该取向纤维能够促进多能干细胞分化为肌腱样细胞,而原位肌腱修复实验则进一步证实该取向纤维支架可显著改善损伤肌腱的结构及力学性能。在此基础上其团队将曲古抑菌素A负载于左旋聚乳酸取向电纺纤维支架,并发现该药物的加载可增强取向支架对肌腱分化的诱导作用,为促进肌腱再生提供了新策略[31]。 考虑到天然肌腱组织复杂的多尺度层次结构,能够更好地模拟肌腱形态结构及力学性能的三维多层取向支架也逐渐进入研究人员的视野[32]。有学者通过静电纺丝制备了多层聚己内酯取向纤维支架,与随机支架相比其可显著促进肌腱相关基因表达,并改善新合成胶原纤维的排列方向[13]。此外,DEEPTHI等[12]开发了一种仿生组织工程肌腱,以电纺取向纳米纤维模拟有序排列的胶原纤维束,壳聚糖-水凝胶外壳模拟鞘层细胞外基质的糖胺聚糖,藻酸盐凝胶为外涂层防止蛋白吸附,研究证实该支架可显著促进肌腱细胞增殖与扩散。因此,肌腱组织工程支架需考虑合适的纤维取向以调控细胞增殖、迁移及分化等功能,而静电纺丝仿生三维支架在改善纤维排列、调控力学性能方面具有不可比拟的优越性,为肌腱组织工程的发展拓展了新思路。 2.2.3 肌肉组织工程 心肌:心肌梗死引发的心力衰竭是导致全世界患者死亡的主要原因之一,通常由冠状动脉阻塞引起,由于缺乏氧气供应导致心肌细胞损伤甚至死亡。成年哺乳动物的心肌细胞自我再生能力有限,在发生心肌梗死时无法自我更新,使得心肌负担过重,最终导致心脏功能受损[33],因此,需要替代治疗以补充内在修复的不足。心脏组织工程结合生物材料支架、细胞及生长因子来改善或恢复心脏功能,提供了非常有前途的治疗方案[34]。 应用静电纺丝技术制备的取向纳米纤维取向结构能够模拟心脏的各向异性结构,并且其微纳结构及优异的机械性能可为增强心肌细胞功能提供指导,因此电纺取向纳米纤维已在心肌组织工程中得到了越来越广泛的应用[35]。据报道,静电纺丝取向纳米纤维结合干细胞或生物活性离子构成的心脏贴片已被应用于心肌组织工程,并证实此类心脏贴片能够促进血管生成、改善梗死心肌组织功能[7,36]。 此前的研究强调了心肌各向异性结构的重要性,这对于心脏组织中适当的机械性能及电耦合至关重要,并且这种复杂的三维各向异性结构可确保其圆形收缩并产生强大的泵功能[37]。然而,目前的心肌组织工程支架厚度有限,导致迁移至纳米纤维结构内部的细胞不足,难以模拟心肌高度组织化的各向异性结构并实现心肌细胞的三维生长[38]。因此,学者们设计了以电纺取向纳米纤维为基础的各向异性三维纱线支架[39]。WU等[40]以电纺取向纳米纤维网格纱线为基础在其外层包覆水凝胶,制备了一种模拟天然心脏组织结构的三维混合支架,研究表明这种支架可促进网格表面取向拉伸排列的心肌细胞成熟,为心肌再生提供了合适的三维微环境,证实其作为心肌组织工程支架具有巨大潜力。 骨骼肌:在人体的正常活动中发挥着重要作用,包括四肢的控制及运动,辅助呼吸、保护脏器等。当前骨骼肌损伤的治疗主要依赖于外科手术,然而这受到供体组织可用性及供体部位肌肉功能丧失的限制[41]。众所周知,骨骼肌组织由高度取向的肌管构成,取向结构的肌管能够伸展或收缩肌肉,并维持肌肉结构[42]。利用组织工程技术制备具有高度取向的生物材料以执行肌肉组织的机械功能,有望成为满足当下医疗需求的潜在手段。生物材料支架的生化及生物物理特性应模拟天然骨骼肌的细胞外基质特征[43]。因此,骨骼肌组织工程支架应具有取向排列的表面形貌,以提供支持肌肉组织再生的环境[44]。 在构建组织工程支架的诸多技术中,静电纺丝技术是一种能够方便快捷地制备取向纳米纤维的有效手段。研究表明,电纺取向纳米纤维对指导细胞有序排列及伸长迁移具有积极作用[45-46]。WANG等[38]制备了模拟天然骨骼肌结构的核-壳结构复合支架,该支架由电纺取向纳米纤维纱线内核及可光固化的水凝胶外壳构成,其研究证实该支架具有良好的生物相容性及诱导细胞三维排列延伸的能力。此外,以C2C12成肌细胞直接进行取向电纺可获得具有高细胞活力、均匀细胞分布的三维生物支架[47],证实此类设计在骨骼肌组织工程领域具有极大潜力。 除形貌线索外,电信号也是重要的刺激因素。CHEN等[14]应用静电纺丝技术开发了可以同时为细胞提供形貌及电学线索的高度取向导电纳米纤维,取向纤维能够指导成肌细胞的排列并促进肌管的形成,其导电性则进一步增强了肌管的成熟度,表明这2种影响因素的组合对骨骼肌再生具有更佳的诱导作用。 2.2.4 骨组织工程 骨组织是人体的主要结构及支撑性结缔组织。创伤、疾病、肿瘤等因素可能引发骨组织损伤或功能障碍等严重并发症,因此受损骨组织的重建仍然是非常具有挑战性的医学难题[48]。近年来,纳米技术的发展使新的骨移植物及支架系统得以出现,从而推动了骨组织工程支架的进步。其中,应用静电纺丝技术构建的新型支架为外科医生提供了恢复组织与器官功能的新选择[49]。 研究表明,组织工程支架的表面形貌对干细胞形态及差异性分化具有重要影响[50-51](图5)。应用静电纺丝制备的骨组织再生支架具有模拟天然细胞外基质的多孔微观结构,可为骨骼向内生长提供机械支持[52]。与相同诱导条件下的随机纤维相比,取向电纺纤维支架可显著增强骨髓间充质干细胞的成骨分化[53]。此外,取向电纺纤维与Ⅰ型胶原蛋白的结合也可增强其定向诱导成骨的能力[54]。 此前的研究证实了骨髓间充质干细胞成骨分化的潜力,适当的生长因子是调节其定向分化的关键。研究表明,电纺取向纳米纤维与生长因子的结合能够显著影响骨髓间充质干细胞的分化过程[55]。骨形态发生蛋白2作为正常骨骼愈合过程中成骨和成血管的有效诱导剂,引起了研究人员的广泛关注。负载骨形态发生蛋白2的电纺取向纳米纤维也已被应用于骨组织工程支架[56],通过接触指导,纳米纤维的取向结构能够引导骨髓间充质干细胞的黏附和生长,骨形态发生蛋白2的加载则将骨髓间充质干细胞主动转化为成骨细胞。成骨细胞沿纤维延伸方向排列并分泌胶原蛋白,随后胶原蛋白沿纤维方向沉积并形成胶原蛋白原纤维束,最终发生矿化实现骨骼再生[56-57]。因此,这种结合生长因子的电纺取向纳米纤维支架可能具有引导骨组织再生的潜力。 2.2.5 伤口愈合 慢性伤口愈合是一个重要的医学问题,创伤部位细胞因子的失衡常导致慢性伤口的炎症阶段延长。目前,研究人员已开发出多种支架材料以促进慢性伤口愈合。静电纺丝纳米纤维能够在形态及尺寸上模拟天然细胞外基质纤维,因此具有很大的应用潜力。研究表明,纳米纤维基质对于减轻细菌定植并刺激感染伤口的闭合具有重要重要意义。纤维支架能够支持成纤维细胞的附着、促进成纤维细胞的迁移,因此具有作为伤口愈合材料的天然优势[58]。据报道,纤维排列方向高度一致的电纺取向纳米纤维能够进一步促进细胞黏附及迁移[59-60]。LEE等[61]以静电纺丝纳米纤维支架模拟伤口愈合模型,探究纤维支架的取向度对伤口愈合的影响情况,研究表明电纺取向纳米纤维能够促进细胞早期的排列,同时从长远来看减少了过度的细胞增殖及胶原蛋白产生。这意味着取向结构在纳米纤维模型中的引入可能会改变成纤维细胞从修复到愈合的反应,暗示纤维基质排列是避免瘢痕形成及促进软组织损伤修复的关键设计因素。ZHU等[62]设计了一种负载他扎罗汀(促进血管生成的活性药物)的电纺取向聚己内酯纳米纤维支架,他扎罗汀持续从纳米纤维中释放出来,该支架可刺激人脐静脉内皮细胞增殖、迁移并促进血管内皮生长因子的分泌;此外,体内实验证实其可改善新生血管形成并抑制伤口区域的炎症反应。这些实验表明电纺取向纳米纤维支架有望成为潜在的伤口护理材料。 "

| [1] JIN G, HE R, SHA B, et al. Electrospun three-dimensional aligned nanofibrous scaffolds for tissue engineering. Mater Sci Eng C Mater Biol Appl. 2018;92:995-1005. [2] STEFFENS D, BRAGHIROLLI DI, MAURMANN N, et al. Update on the main use of biomaterials and techniques associated with tissue engineering. Drug Discovery Today. 2018;23(8):1474-1488. [3] MONDSCHEIN RJ, KANITKAR A, WILLIAMS CB, et al. Polymer structure-property requirements for stereolithographic 3D printing of soft tissue engineering scaffolds. Biomaterials. 2017;140:170-188. [4] GAO B, YANG Q, ZHAO X, et al. 4D Bioprinting for Biomedical Applications. Trends Biotechnol. 2016;34(9):746-756. [5] CHENG J, JUN Y, QIN J, et al. Electrospinning versus microfluidic spinning of functional fibers for biomedical applications. Biomaterials. 2017;114:121-143. [6] LUO BW, TIAN LL, CHEN N, et al. Electrospun nanofibers facilitate better alignment, differentiation, and long-term culture in an in vitro model of the neuromuscular junction (NMJ). Biomater Sci-Uk. 2018;6(12):3262-3272. [7] WANG X, WANG L, WU Q, et al. Chitosan/Calcium Silicate Cardiac Patch Stimulates Cardiomyocyte Activity and Myocardial Performance after Infarction by Synergistic Effect of Bioactive Ions and Aligned Nanostructure. ACS Appl Mater Interfaces. 2019;11(1):1449-1468. [8] JIN G, LI J, LI K. Photosensitive semiconducting polymer-incorporated nanofibers for promoting the regeneration of skin wound. Mater Sci Eng C Mater Biol Appl. 2017;70(Pt 2):1176-1181. [9] JIN G, LI K. The electrically conductive scaffold as the skeleton of stem cell niche in regenerative medicine. Mater Sci Eng C Mater Biol Appl. 2014;45:671-681. [10] QI S, CRAIG D. Recent developments in micro- and nanofabrication techniques for the preparation of amorphous pharmaceutical dosage forms. Adv Drug Deliv Rev. 2016;100:67-84. [11] 蒋敏,王敏,魏仕勇,等.基于静电纺丝技术的取向纳米纤维[J].化学进展,2016,28(5):115-130. [12] DEEPTHI S, NIVEDHITHA SUNDARAM M, DEEPTI KADAVAN J, et al. Layered chitosan-collagen hydrogel/aligned PLLA nanofiber construct for flexor tendon regeneration. Carbohydr Polym. 2016;153:492-500. [13] ORR SB, CHAINANI A, HIPPENSTEEL KJ, et al. Aligned multilayered electrospun scaffolds for rotator cuff tendon tissue engineering. Acta Biomater. 2015;24:117-126. [14] CHEN MC, SUN YC, CHEN YH. Electrically conductive nanofibers with highly oriented structures and their potential application in skeletal muscle tissue engineering. Acta Biomater. 2013;9(3):5562-5572. [15] DI CAMILLO D, FASANO V, RUGGIERI F, et al. Near-field electrospinning of light-emitting conjugated polymer nanofibers. Nanoscale. 2013; 5(23):11637-11642. [16] ZHANG D, CHANG J. Patterning of Electrospun Fibers Using Electroconductive Templates. Adv Mater. 2007;19(21):3664-3667. [17] CHEONG H, KIM J, KIM BJ, et al. Multi-dimensional bioinspired tactics using an engineered mussel protein glue-based nanofiber conduit for accelerated functional nerve regeneration.Acta Biomater.2019;90:87-99. [18] NUNE M, SUBRAMANIAN A, KRISHNAN UM, et al. Self-assembling peptide nanostructures on aligned poly(lactide-co-glycolide) nanofibers for the functional regeneration of sciatic nerve. Nanomedicine (Lond). 2017;12(3):219-235. [19] ALVAREZ Z, CASTANO O, CASTELLS AA, et al. Neurogenesis and vascularization of the damaged brain using a lactate-releasing biomimetic scaffold. Biomaterials. 2014;35(17):4769-4781. [20] SILANTYEVA EA, NASIR W, CARPENTER J, et al. Accelerated neural differentiation of mouse embryonic stem cells on aligned GYIGSR-functionalized nanofibers. Acta Biomater. 2018;75:129-139. [21] HUANG C, OUYANG Y, NIU H, et al. Nerve Guidance Conduits from Aligned Nanofibers: Improvement of Nerve Regeneration through Longitudinal Nanogrooves on a Fiber Surface. Acs Appl Mater Inter. 2015;7(13):7189-7196. [22] WANG J, TIAN L, CHEN N, et al. The cellular response of nerve cells on poly-l-lysine coated PLGA-MWCNTs aligned nanofibers under electrical stimulation. Mater Sci Eng C Mater Biol Appl. 2018;91:715-726. [23] WANG L, WU Y, HU T, et al. Aligned conductive core-shell biomimetic scaffolds based on nanofiber yarns/hydrogel for enhanced 3D neurite outgrowth alignment and elongation.Acta Biomater. 2019;96:175-187. [24] 尹桂波,VICTORLEUNG, FRANK KKO,等.含有取向纳米纤维束的神经移植物构建与神经修复应用研究[J].现代纺织技术,2017,25(6):1-6. [25] JIA Y, YANG W, ZHANG K, et al. Nanofiber arrangement regulates peripheral nerve regeneration through differential modulation of macrophage phenotypes. Acta Biomater. 2019;83:291-301. [26] WU S, PENG H, LI X, et al. Effect of scaffold morphology and cell co-culture on tenogenic differentiation of HADMSC on centrifugal melt electrospun poly (Llactic acid) fibrous meshes. Biofabrication. 2017;9(4):044106. [27] ZHANG C, YUAN H, LIU H, et al. Well-aligned chitosan-based ultrafine fibers committed teno-lineage differentiation of human induced pluripotent stem cells for Achilles tendon regeneration.Biomaterials. 2015;53:716-730. [28] TONG WY, SHEN W, YEUNG CW, et al. Functional replication of the tendon tissue microenvironment by a bioimprinted substrate and the support of tenocytic differentiation of mesenchymal stem cells.Biomaterials. 2012;33(31):7686-7698. [29] EICHHOLZ KF, HOEY DA. Mediating human stem cell behaviour via defined fibrous architectures by melt electrospinning writing. Acta Biomater. 2018;75:140-151. [30] ERISKEN C, ZHANG X, MOFFAT KL, et al. Scaffold fiber diameter regulates human tendon fibroblast growth and differentiation.Tissue Eng Part A. 2013;19(3-4):519-528. [31] ZHANG C, WANG XL, ZHANG EC, et al. An epigenetic bioactive composite scaffold with well-aligned nanofibers for functional tendon tissue engineering.Acta Biomater.2018;66:141-156. [32] SENSINI A, GUALANDI C, FOCARETE ML, et al. Multiscale hierarchical bioresorbable scaffolds for the regeneration of tendons and ligaments.Biofabrication. 2019;11(3):035026. [33] KANG BJ, KIM H, LEE SK, et al. Umbilical-cord-blood-derived mesenchymal stem cells seeded onto fibronectin-immobilized polycaprolactone nanofiber improve cardiac function.Acta Biomater. 2014;10(7):3007-3017. [34] MOHAMMADI AMIRABAD L, MASSUMI M, SHAMSARA M, et al. Enhanced Cardiac Differentiation of Human Cardiovascular Disease Patient-Specific Induced Pluripotent Stem Cells by Applying Unidirectional Electrical Pulses Using Aligned Electroactive Nanofibrous Scaffolds. Acs Appl Mater Inter. 2017;9(8):6849-6864. [35] SUHAERI M, SUBBIAH R, KIM SH, et al. Novel Platform of Cardiomyocyte Culture and Coculture via Fibroblast-Derived Matrix-Coupled Aligned Electrospun Nanofiber.ACS Appl Mater Interfaces. 2017;9(1):224-235. [36] STREETER BW, XUE J, XIA Y, et al. Electrospun Nanofiber-Based Patches for the Delivery of Cardiac Progenitor Cells.ACS Appl Mater Interfaces. 2019;11(20):18242-18253. [37] BIAN W, BADIE N, HIMEL HDT, et al. Robust T-tubulation and maturation of cardiomyocytes using tissue-engineered epicardial mimetics.Biomaterials. 2014;35(12):3819-3828. [38] WANG L, WU Y, GUO B, et al. Nanofiber Yarn/Hydrogel Core-Shell Scaffolds Mimicking Native Skeletal Muscle Tissue for Guiding 3D Myoblast Alignment, Elongation, and Differentiation. Acs Nano. 2015; 9(9):9167-9179. [39] WU S, DUAN B, LIU P, et al. Fabrication of Aligned Nanofiber Polymer Yarn Networks for Anisotropic Soft Tissue Scaffolds.Acs Appl Mater Inter. 2016;8(26):16950-16960. [40] WU Y, WANG L, GUO B, et al. Interwoven Aligned Conductive Nanofiber Yarn/Hydrogel Composite Scaffolds for Engineered 3D Cardiac Anisotropy. Acs Nano. 2017;11(6):5646-5659. [41] JANA S, LEUNG M, CHANG J, et al. Effect of nano- and micro-scale topological features on alignment of muscle cells and commitment of myogenic differentiation.Biofabrication. 2014; 6(3):035012. [42] YEO M, LEE H, KIM GH .Combining a micro/nano-hierarchical scaffold with cell-printing of myoblasts induces cell alignment and differentiation favorable to skeletal muscle tissue regeneration.Biofabrication. 2016;8(3):035021. [43] OSTROVIDOV S, SHI X, ZHANG L, et al. Myotube formation on gelatin nanofibers - multi-walled carbon nanotubes hybrid scaffolds.Biomaterials. 2014;35(24):6268-6277. [44] LEE H, KIM W, LEE J, et al. Effect of Hierarchical Scaffold Consisting of Aligned dECM Nanofibers and Poly(lactide-co-glycolide) Struts on the Orientation and Maturation of Human Muscle Progenitor Cells.ACS Appl Mater Interfaces. 2019;11(43):39449-39458. [45] PADHI A, THOMSON AH, PERRY JB, et al. Bioenergetics underlying single-cell migration on aligned nanofiber scaffolds.Am J Physiol-Cell Ph. 2020;318(3):C476-C485. [46] SU N, GAO PL, WANG K, et al. Fibrous scaffolds potentiate the paracrine function of mesenchymal stem cells: A new dimension in cell-material interaction. Biomaterials. 2017;141:74-85. [47] YEO M, KIM GH. Anisotropically Aligned Cell-Laden Nanofibrous Bundle Fabricated via Cell Electrospinning to Regenerate Skeletal Muscle Tissue. Small. 2018;14(48):e1803491. [48] CRISTOFARO F, GIGLI M, BLOISE N, et al. Influence of the nanofiber chemistry and orientation of biodegradable poly(butylene succinate)-based scaffolds on osteoblast differentiation for bone tissue regeneration. Nanoscale. 2018;10(18):8689-8703. [49] WALMSLEY GG, MCARDLE A, TEVLIN R, et al. Nanotechnology in bone tissue engineering. Nanomedicine. 2015;11(5):1253-1263. [50] 贺云飞,王爽,马俊,等. P38/AKT通路调控骨髓间充质干细胞在不同空间结构纳米纤维环支架中的定向分化[J].中国组织工程研究,2020,24(10):1540-1546. [51] LIU W, WEI Y, ZHANG X, et al. Lower extent but similar rhythm of osteogenic behavior in hBMSCs cultured on nanofibrous scaffolds versus induced with osteogenic supplement.Acs Nano. 2013;7(8): 6928-6938. [52] MOHAMMADI M, MOUSAVI SHAEGH SA, ALIBOLANDI M, et al. Micro and nanotechnologies for bone regeneration: Recent advances and emerging designs. J Control Release. 2018;274:35-55. [53] GAO X, SONG J, ZHANG Y, et al. Bioinspired Design of Polycaprolactone Composite Nanofibers as Artificial Bone Extracellular Matrix for Bone Regeneration Application. ACS Appl Mater Interfaces. 2016;8(41): 27594-27610. [54] CHEN H, QIAN Y, XIA Y, et al. Enhanced Osteogenesis of ADSCs by the Synergistic Effect of Aligned Fibers Containing Collagen I. Acs Appl Mater Inter. 2016;8(43):29289-29297. [55] OLVERA D, SATHY BN, CARROLL SF, et al. Modulating microfibrillar alignment and growth factor stimulation to regulate mesenchymal stem cell differentiation. Acta Biomater. 2017;64:148-160. [56] MADHURAKKAT PERIKAMANA SK, LEE J, AHMAD T, et al .Effects of Immobilized BMP-2 and Nanofiber Morphology on In Vitro Osteogenic Differentiation of hMSCs and In Vivo Collagen Assembly of Regenerated Bone. Acs Appl Mater Inter. 2015;7(16):8798-8808. [57] ZHANG X, WANG C, LIAO M, et al. Aligned electrospun cellulose scaffolds coated with rhBMP-2 for both in vitro and in vivo bone tissue engineering. Carbohyd Polym. 2019;213:27-38. [58] ALBRIGHT V, XU M, PALANISAMY A, et al. Micelle-Coated, Hierarchically Structured Nanofibers with Dual-Release Capability for Accelerated Wound Healing and Infection Control. Adv Healthc Mater. 2018;7(11):e1800132. [59] WANG K, LIU LP, XIE J, et al. Facile Strategy to Generate Aligned Polymer Nanofibers: Effects on Cell Adhesion. Acs Appl Mater Inter. 2018;10(2):1566-1574. [60] XUE JJ, WU T, XIA YN. Perspective: Aligned arrays of electrospun nanofibers for directing cell migration. Apl Materials. 2018);6(12): 120902. [61] LEE NM, ERISKEN C, ISKRATSCH T, et al. Polymer fiber-based models of connective tissue repair and healing. Biomaterials. 2017;112:303-312. [62] ZHU Z, LIU Y, XUE Y, et al. Tazarotene Released from Aligned Electrospun Membrane Facilitates Cutaneous Wound Healing by Promoting Angiogenesis. ACS Appl Mater Interfaces. 2019;11(39):36141-36153. [63] QING H, JIN G, ZHAO G, et al. Heterostructured Silk-Nanofiber-Reduced Graphene Oxide Composite Scaffold for SH-SY5Y Cell Alignment and Differentiation. Acs Appl Mater Inter. 2018;10(45):39228-39237. [64] 王丹丹,钟惠湘,轩留洋,等.制备可促进干细胞增殖的三维取向电纺纤维膜[J].中山大学学报(自然科学版),2017,56(6):76-82. [65] HUANG R, GAO X, WANG J, et al. Triple-Layer Vascular Grafts Fabricated by Combined E-Jet 3D Printing and Electrospinning.Ann Biomed Eng. 2018;46(9):1254-1266. [66] WADE RJ, BASSIN EJ, GRAMLICH WM, et al. Nanofibrous hydrogels with spatially patterned biochemical signals to control cell behavior. Adv Mater. 2015;27(8):1356-1362. [67] DONG Y, JIN G, HONG Y, et al. Engineering the Cell Microenvironment Using Novel Photoresponsive Hydrogels.ACS Appl Mater Interfaces. 2018;10(15):12374-12389. |
| [1] | Pu Rui, Chen Ziyang, Yuan Lingyan. Characteristics and effects of exosomes from different cell sources in cardioprotection [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(在线): 1-. |
| [2] | Zhang Tongtong, Wang Zhonghua, Wen Jie, Song Yuxin, Liu Lin. Application of three-dimensional printing model in surgical resection and reconstruction of cervical tumor [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1335-1339. |
| [3] | Zhang Chao, Lü Xin. Heterotopic ossification after acetabular fracture fixation: risk factors, prevention and treatment progress [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1434-1439. |
| [4] | Zhou Jihui, Li Xinzhi, Zhou You, Huang Wei, Chen Wenyao. Multiple problems in the selection of implants for patellar fracture [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1440-1445. |
| [5] | Wang Debin, Bi Zhenggang. Related problems in anatomy mechanics, injury characteristics, fixed repair and three-dimensional technology application for olecranon fracture-dislocations [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1446-1451. |
| [6] | Ji Zhixiang, Lan Changgong. Polymorphism of urate transporter in gout and its correlation with gout treatment [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1290-1298. |
| [7] | Yuan Mei, Zhang Xinxin, Guo Yisha, Bi Xia. Diagnostic potential of circulating microRNA in vascular cognitive impairment [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1299-1304. |
| [8] | Wan Ran, Shi Xu, Liu Jingsong, Wang Yansong. Research progress in the treatment of spinal cord injury with mesenchymal stem cell secretome [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1088-1095. |
| [9] | Liao Chengcheng, An Jiaxing, Tan Zhangxue, Wang Qian, Liu Jianguo. Therapeutic target and application prospects of oral squamous cell carcinoma stem cells [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1096-1103. |
| [10] | Zhao Min, Feng Liuxiang, Chen Yao, Gu Xia, Wang Pingyi, Li Yimei, Li Wenhua. Exosomes as a disease marker under hypoxic conditions [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1104-1108. |
| [11] | Xie Wenjia, Xia Tianjiao, Zhou Qingyun, Liu Yujia, Gu Xiaoping. Role of microglia-mediated neuronal injury in neurodegenerative diseases [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1109-1115. |
| [12] | Li Shanshan, Guo Xiaoxiao, You Ran, Yang Xiufen, Zhao Lu, Chen Xi, Wang Yanling. Photoreceptor cell replacement therapy for retinal degeneration diseases [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1116-1121. |
| [13] | Jiao Hui, Zhang Yining, Song Yuqing, Lin Yu, Wang Xiuli. Advances in research and application of breast cancer organoids [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1122-1128. |
| [14] | Wang Shiqi, Zhang Jinsheng. Effects of Chinese medicine on proliferation, differentiation and aging of bone marrow mesenchymal stem cells regulating ischemia-hypoxia microenvironment [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1129-1134. |
| [15] | Zeng Yanhua, Hao Yanlei. In vitro culture and purification of Schwann cells: a systematic review [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1135-1141. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||