Chinese Journal of Tissue Engineering Research ›› 2024, Vol. 28 ›› Issue (21): 3385-3392.doi: 10.12307/2024.601
Previous Articles Next Articles
Applicable techniques for subchondral separation of femoral head necrosis treated by tissue engineering
Huang Yixuan, Chen Hao, Xue Peng, Xi Hongzhong, He Shuai, Sun Guangquan, Du Bin, Liu Xin
- Affiliated Hospital of Nanjing University of Chinese Medicine, Nanjing 210029, Jiangsu Province, China
-
Received:2023-05-19Accepted:2023-07-20Online:2024-07-28Published:2023-09-28 -
Contact:Liu Xin, Attending physician, Affiliated Hospital of Nanjing University of Chinese Medicine, Nanjing 210029, Jiangsu Province, China Du Bin, Doctoral supervisor, Chief physician, Affiliated Hospital of Nanjing University of Chinese Medicine, Nanjing 210029, Jiangsu Province, China -
About author:Huang Yixuan, Master candidate, Affiliated Hospital of Nanjing University of Chinese Medicine, Nanjing 210029, Jiangsu Province, China -
Supported by:National Natural Science Foundation of China (General Project), No. 82074471 (to DB); National Natural Science Foundation of China (Youth Fund), No. 81804117 (to LX)
CLC Number:
Cite this article
Huang Yixuan, Chen Hao, Xue Peng, Xi Hongzhong, He Shuai, Sun Guangquan, Du Bin, Liu Xin. Applicable techniques for subchondral separation of femoral head necrosis treated by tissue engineering[J]. Chinese Journal of Tissue Engineering Research, 2024, 28(21): 3385-3392.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
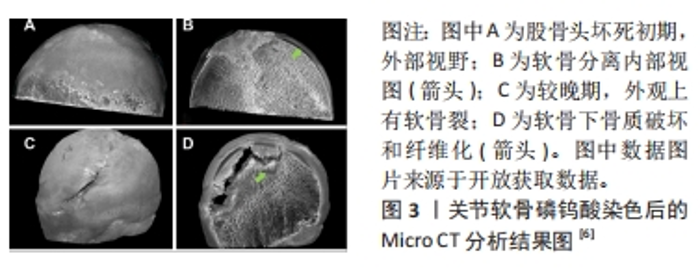
2.1 骨-软骨界面 骨-软骨界面是股骨头坏死中发生软骨下分离直至塌陷过程中的关键组织界面。因此,设计组织工程应用于该界面,必须了解骨软骨组织之间结构与功能的特点。软骨根据软骨细胞形态或细胞外基质成分,在垂直方向上分为了3个区域:浅层(切线层)、中层(过渡区)和深层(基底区),分别占据总体的10%,60%和30%。在细胞形态方面,浅层软骨细胞呈椭圆形,中间层为球形,深层则是由球形软骨细胞和胶原纤维呈垂直柱状排列[17]。随着软骨逐渐加深,软骨细胞数量和外基质的含水量下降,蛋白多糖和胶原纤维含量上升[18]。 软骨下区由软骨下骨板和软骨下松质骨构成,前者更接近皮质骨特点,由垂直于关节面的细小骨小梁和胶原纤维层组成;随着深度的加深,骨小梁之间的间隙明显扩大构成了软骨下松质骨。在组成成分上两者均由Ⅰ型胶原纤维和蛋白多糖等有机物与羟基磷灰石构成[19]。在软骨下区可以看到大量的动脉和静脉的微小分支,可以将营养物质送入钙化软骨层[20]。 软骨与软骨下骨之间存在钙化软骨,属于骨与软骨之间的过渡区域。由肥大的软骨细胞和不同比例的Ⅰ型胶原、透明质酸及羟基磷灰石构成[21],其整合了软骨与软骨下骨的特性,并且通过连续的胶原纤维将不同的界面整合在一起。并且钙化软骨对软骨与软骨下骨之间的应力传导至关重要,机械应力刺激通过钙化软骨传递到软骨下骨[22]。最近几年的研究发现,除了承担软骨与软骨下骨之间生物力学耦合作用之外,钙化软骨可能还有助于两者之间物质传递的作用[23]。潮汐线是深层软骨与钙化软骨之间的三维结构,其中通过纵向的胶原(主要是Ⅱ型胶原)纤维将深层软骨与钙化软骨层束缚在一起,为两者提供栓系机制,并对抗关节面的剪切力[24]。 2.2 股骨头坏死与软骨下分离 目前关于股骨头坏死的病理标本的研究中发现,坏死的股骨头存在软骨下骨的骨折,并且伴随软骨与软骨下骨的分离现象[5-7,25],具体病例见图3。汝强[5]通过收集了25例ARCOⅢ期股骨头坏死标本观察大体形态、冠状剖面和组织学切片,更进一步的指出骨与软骨层面的分离在组织学上发生在钙化软骨与软骨下骨板之间。这与组织学中钙化软骨与软骨下骨板之间没有胶原纤维通过相关[26]。病理学研究也发现股骨头坏死与正常股骨头相比软骨下骨的胶原纤维排列稀疏,胶原含量下降[27]。不同的病理机制可能导致股骨头骨-软骨界面发生分离,而骨-软骨界面的分离又可能进一步加重股骨头塌陷的进展。在血供方面,目前的研究认为ARCOⅠ期的软骨下骨血供与ARCOⅢ期之间存在明显差异[28],这可能会影响到软骨的组织代谢[29],表现在软骨基质与糖胺聚糖含量的下降[30-31]。在病理环境方面,除了血供减少所造成的缺血缺氧环境,ARCOⅢ期软骨退变和软骨下病变与炎症环境中关节腔中炎症因子白细胞介素33和白细胞介素6的升高相关[32]。在生物力学方面,由于骨质的破坏和修复反复进行,坏死区与修复区弹性模量之间的差异导致局部出现应力集中现象,发生软骨下骨小梁的微骨折向下塌陷形成与软骨之间的孔隙[33]。上述对软骨与软骨下骨的影响均可能是出现软骨与软骨下骨分离现象的诱因。"
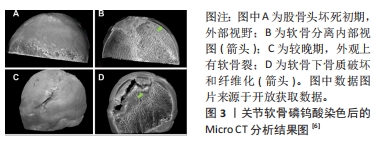

在自然病程中,发生软骨下分离股骨头在应力作用于局部软骨时,由于失去了软骨下骨对应力的分散传导,软骨将出现塌陷,进而发展为撕裂、剥脱,最终损伤到软骨全层[34-35]。在保髋手术的失败案例中同样存在软骨与软骨下分离的现象[13,36],说明植入物并未实现骨-软骨界面的整合,说明保髋手术失败可能与股骨头骨-软骨界面整合不良存在关系。 2.3 股骨头软骨下分离组织工程适应技术的进展 根据股骨头软骨下骨分离机制,利用组织工程技术实现骨-软骨界面的重新整合是解决该问题的可能方法。骨-软骨整合过程包括了骨、软骨细胞的黏附和增殖,间充质干细胞成骨与成软骨分化,骨、软骨基质的沉积,目前组织工程支架在上述过程中均有报道,文章将从支架材料、支架结构、表面修饰和细胞加载4个大方向,比较不同类型技术方式在促进界面整合和骨软骨修复的效果和优缺点,为后续的方法设计提供参考和借鉴。 2.3.1 支架材料 由于涉及到骨与软骨两个不同界面,必须要求支架材料与软骨细胞和骨细胞均具有良好生物相容性和生物活性。天然材料在生物学性能方面具有优势。但是考虑到在股骨头坏死软骨下分离初期,软骨下骨塌陷后原本的力学结构破坏,此时软骨结构相对完好,支架材料必须提供足够的机械支撑和与不同界面整合的能力,单纯的天然材料不能完全满足需求,因此复合材料支架是目前最为理想的材料。目前应用于股骨头坏死的支架复合材料方案主要为生物陶瓷/金属复合材料[37-38]、天然聚合物/金属离子复合材料[39-40]、生物陶瓷/人工有机聚合物[41-42]、生物陶瓷/天然聚合物复合物材料[43]。利用复合材料以实现的生物学和力学双重目的[44-58],不同支架材料的优缺点详见表1。"

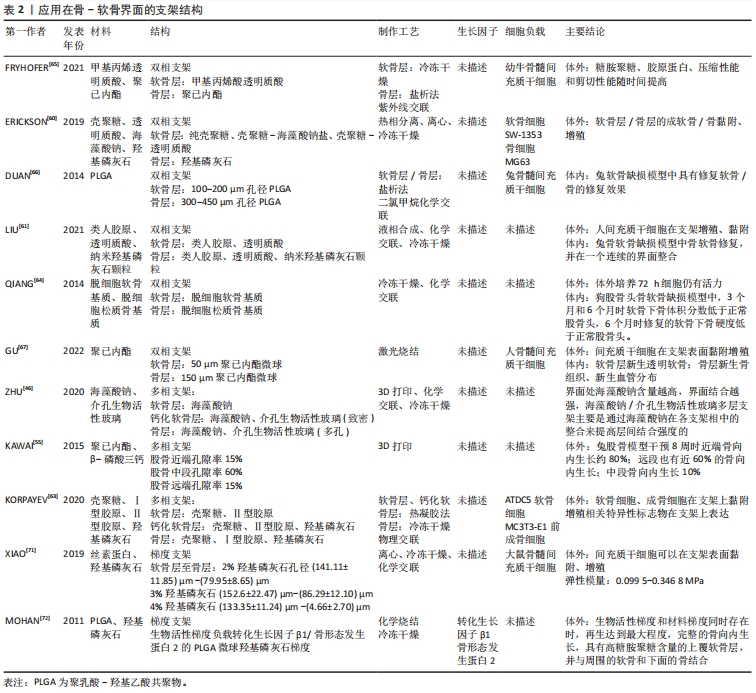
在天然聚合物中,透明质酸和胶原等作为细胞外基质的主要成分,具有良好的生物相容性和生物活性,在支架材料中加入透明质酸、胶原等可促进细胞外基质和软骨形成基因的表达[59],促进软骨形成[40,59-60]。LIU等[40]设计的多孔钽/胶原膜支架植入兔股骨头坏死骨软骨缺损区,术后12周在缺损边缘附近的钽顶上可见透明软骨。针对天然聚合物材料机械性能和骨传导能力不能满足软骨下力学要求,可加入羟基磷灰石、β-磷酸三钙颗粒以模仿骨-软骨界面。LIU等[61]设计的类人胶原/透明质酸在骨层添加纳米羟基磷灰石颗粒构建的双相支架抗压强度达到(212.11±13.49) kPa,可承受20次以上的压缩,变形量为70%。此外,复合材料中添加天然聚合物如明胶、壳聚糖及海藻酸钠等具有黏附属性,可避免多相支架在不同界面出现分层或结合不良的情况[46,62-63],这种特征可能与胺基和羟基之间形成的氢键以及天然聚合物链之间的疏水相互作用有关[63]。ZHU等[46]设计的分层支架中表明当界面处海藻酸钠含量越高时,界面的结合越强。 2.3.2 支架结构 目前不同结构梯度的支架已经在股骨头坏死领域中被报道,根据股骨头不同部位所需要的力学和骨传导性能,在孔隙率、力学强度和降解速率方面实现支架结构的差异[55],但目前的研究大多聚焦于皮质骨-松质骨的单一骨界面。QIANG等[64]设计的骨-软骨界面支架植入狗股骨头体内实验尚未取得预期效果。要想构建理想的支架实现骨-软骨界面的整合,除了支架材料外,支架结构的选择更加关键。由于骨软骨组织天然的结构分层,单相支架中软骨组织再生程度低,不能完全模拟天然骨-软骨界面,因此无法同时支持软骨与骨的生成[60]。因此双相支架、多相支架和更符合真实生理环境的梯度支架不断被报道。 双相支架:双相支架通常顶部接近软骨端设计为天然聚合物或合成聚合物水凝胶,底部设计为接近骨的生物陶瓷或复合聚合物。FRYHOFER等[65]利用光交联技术制备了软骨层为装载有间充质干细胞的透明质酸水凝胶,软骨下骨层则为多孔聚已内酯支架,在含有转化生长因子β3的培养基中培养长达8周,其结果表明,该制备工艺与细胞活力相容,糖胺聚糖、胶原蛋白含量和力学性能随时间延长而增加。ERICKSON等[60]利用热相分离技术制备了双相支架,其以壳聚糖为主要成分,壳聚糖/透明质酸构成软骨层,羟基磷灰石/钙构成钙化软骨层,体外培养实验中成软骨细胞和成骨细胞相关基因表达说明该支架具有用于骨软骨组织再生的潜力。此外,通过改变孔隙率大小、材料形态或添加生长因子也可以实现双相支架的设计[66-68]。双相支架的出现基本解决了软骨与骨界面组织结构差异的特点,但是由于材料之间的差异,在体内降解速度可能不同,将导致支架的稳定性和生物相容性受到影响[69]。股骨头负重特点是持续重复的,骨层支架降解速度过快,坏死区不能及时修复,极易发生塌陷[64],因此还需要进一步讨论软骨层与骨层的降解速率。 多相支架:钙化软骨层在骨-软骨界面中具有关键的机械和生理作用,充当软骨与软骨下骨的生物化学屏障和机械力学过渡[70]。多相支架尤其是三相支架的出现,更加拟合骨软骨的结构分层。通常中间层相较双相支架正好模拟钙化软骨层的结构。KORPAYEV等[63]采用冷冻干燥法制备由壳聚糖/Ⅰ型胶原/纳米羟基磷灰石颗粒构成的软骨下骨层,通过热凝胶法制备壳聚糖/Ⅱ型胶原的软骨层和壳聚糖/Ⅱ胶原/纳米羟基磷灰石颗粒的钙化软骨层,在培养基培养21 d后,ATDC 5和MC3T3-E细胞增殖都得到了促进,细胞外基质含量也显著增加。但该实验研究只进行了皮下的排异实验,未进一步验证骨软骨缺损的修复能力。虽然多相支架更接近实际骨软骨分层,但是多种材料组合将增加制备技术的难度,不同材料之间的结合能力与降解速率也是必须考虑的问题,否则可能出现分离、脱落的情况,这将影响支架的稳定性[47]。 梯度支架:梯度支架不同于双相支架和多相支架,不同材料之间没有明显的分界面,而是由不同材料按照一定比例和顺序构成,其含量和性质会随着位置的改变而改变。目前KAWAI等[55]报道了应用于股骨头坏死的梯度支架,该支架具有中段低孔隙率(15%)和近端远端高孔隙率(65%)的特点,实现了降解速率和力学强度的功能梯度。虽然目前没有股骨头坏死骨-软骨界面的研究,但在膝关节骨-软骨缺损模型中有许多进展。XIAO等[71]利用冷冻干燥法与离心法组合制备出的丝素蛋白/壳聚糖/纳米羟基磷灰石支架具有渐进变化的孔隙和羟基磷灰石浓度差异。MOHAN等[72]制备了具有生长因子骨形态发生蛋白2和羟基磷灰石有梯度差异的聚乳酸-羟基乙酸共聚物(poly lactic-co-glycolic acid,PLGA)支架,12周的兔缺损体内实验表明,当这两种梯度差异同时存在时具有高糖胺聚糖含量的覆盖软骨层、适当的厚度以及与周围软骨和下层骨的整合。目前梯度支架的性能受到材料性质和制备工艺的限制,尚难达到完全理想的生物学效应。支架结构相关文献详见表2。"
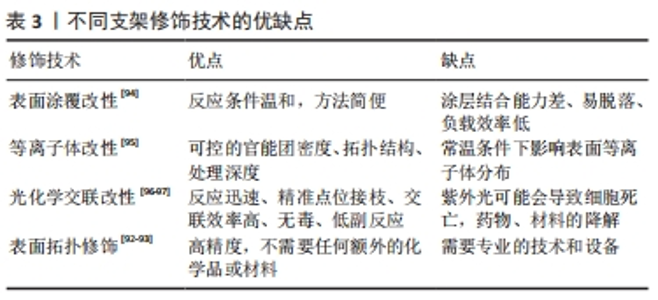
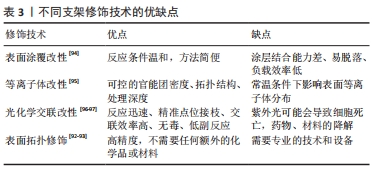
2.3.3 支架材料的修饰 组织工程支架植入体内后,材料表面最先与细胞和组织接触,植物材料的表面形态和化学成分能够直接影响成骨细胞、软骨细胞的黏附、增殖和分化的行为,最终影响植入物对不同界面的整合成功率[73]。支架材料可以通过表面修饰的方式满足不同界面的特性。 表面涂覆改性:该技术是指通过物理吸附的方式在支架材料表面引入新的功能基团,以改善支架表面性能和细胞黏附、增殖和分化等能力。常用的生物活性物质包括纤连蛋白、胶原、层连蛋白和聚赖氨酸等。其中纤连蛋白是一种黏附性多功能蛋白,其参与细胞-基质复合物的组成,并在细胞外基质的组织中发挥作用[74],涉及调节机制的组装、增殖、黏附和组织重塑[75]。目前研究表明纤连蛋白通过表面涂覆修饰组织工程支架具有促进干细胞黏附和分化、促进软骨细胞分化和诱导血管内皮化的作用[76-79]。但是物理吸附的方式存在表面脱落及持久性差的问题,这是未来需要解决的问题。 等离子体改性:该技术通过足够高的能量使反应物激发、分离,并保持反应体系低温甚至室温,能够将聚合物表面各种化学键断裂、重组或引入新的官能团,实现改性。聚合物表面的官能团对于材料与界面的整合具有重要意义。例如羟基、氨基、羧基及羰基等可以调节支架表面的亲水键和电荷促进细胞的黏附与增殖[80-83]。ESBAH等[82]利用等离子体技术将羧基添加到壳聚糖/聚氧乙烯静电纺丝纤维支架中,通过增加表面湿润性来增强MC3T3成骨细胞在等离子体激活纤维上的黏附和增殖。XUE等[84]利用等离子改性技术将淫羊藿苷和镁离子修饰β-磷酸三钙支架表面,在兔股骨头坏死模型中表现出良好的成骨和成血管能力。 光化学交联:该方法通常将目标分子与光偶联剂进行化学反应,生成有光活性基团的衍生物,然后进行光化学反应使目标分子共价偶联到高分子材料表面,通过调节光的强度和曝光时间来精准控制交联密度和物理化学性质,以促进细胞增殖和分化[85]。此外,为了功能化支架,可以将许多添加物如纳米颗粒、功能性细胞、药物和细胞因子引入支架系统中[86-89],改善支架物理和化学性质。HAM等[89]设计的偶氮二苯/壳聚糖/胶原支架通过光交联将骨形态发生蛋白2固定在支架表面,在大鼠颅骨缺损模型中表现出较好的成骨能力。HUA等[90]研制了一种基于光交联软骨/骨衍生细胞外基质的双相骨-软骨整合支架,能够实现对双相骨-软骨整合微环境的仿生和重建。 表面拓扑结构修饰:通过对材料表面的拓扑结构进行修饰,例如修饰材料表面的孔隙大小及分布影响细胞的黏附、增殖和分化,从而影响支架与界面之间的整合[91]。激光处理的支架提供了足够的表面用于细胞黏附和增殖,并且可以具有与植入体内的活细胞直接结合的能力。DASKALOVA等[92]利用激光飞秒技术在3D打印聚已内酯支架增加孔隙率改善亲水性,成骨细胞在改性后支架上活性增强。此外,利用激光消融可以在电纺纳米纤维支架中形成微米级孔隙促进胶原凝胶渗透来改善界面整合,并且有利于避免多相支架之间的分层[93]。 文章总结了支架材料不同的表面修饰优缺点[92-97],见表3。"

2.3.4 种子细胞的加载 在骨软骨工程支架中对修复细胞培养是骨-软骨界面整合的一种方法,最常用于组织工程的是干细胞。干细胞因其强大的扩增能力、多向分化潜能以及可自体取材的特征,已逐渐成为种子细胞的首选。骨髓间充质干细胞是第一个被发现的间充质干细胞,也是目前在治疗股骨头坏死中常用的间充质干细胞[98]。目前的研究发现在缺氧条件下的骨髓间充质干细胞增殖能力、集落形成能力和成骨基因表达能力均显著增强[99]。此外骨髓间充质干细胞还具有向软骨细胞分化的能力,LIU等[40]设计的Bio-Gide胶原/多孔钽支架加载骨髓间充质干细胞植入兔股骨头坏死模型,在术后12周支架顶部出现透明软骨,表明骨髓间充质干细胞在股骨头坏死中具有骨软骨修复能力。 脂肪间充质干细胞来源于脂肪,其来源丰富、容易获得,且具有分化为软骨、肌腱、骨骼肌和脂肪的潜力[43]。在兔股骨头坏死模型中,加载脂肪间充质干细胞的支架表现出了骨、软骨修复和成血管能力[43]。但是与骨髓间充质干细胞相比,脂肪间充质干细胞的成软骨能力较低,但可以通过转化生长因子、骨形态发生蛋白或者胰岛素样生长因子联合使用来克服这一缺点[100]。QIN等[101]设计的支架在骨形态发生蛋白2和胰岛素样生长因子1诱导下,放置于软骨区,支持脂肪干细胞球体的形成和融合,导致软骨分化。体外培养7 d后脂肪干细胞出现向骨区的迁移,28 d后开始诱导成骨。另有报道不借助生长因子,仅通过一种钙梯度支架也实现了同时诱导脂肪干细胞的成骨和软骨形成[70]。 滑膜间充质干细胞易获得,不易受到疾病、年龄等因素影响,比骨髓间充质干细胞和脂肪间充质干细胞具有更强的移植存活率、快速增殖和成软骨能力[102]。目前已经报道使用滑膜间充质干细胞治疗股骨头坏死[103-104],但滑膜间充质干细胞的分离产量极低,难以从有限的滑膜中获得所需的临床用量。KIM等[105]首次尝试通过采用基于重演组织特异性离体稳态的微器官培养系统来保护生态位的非酶促分离滑膜间充质干细胞;研究表明与骨髓间充质干细胞相比,其软骨微组织形成增加了9倍,硫酸化蛋白聚糖沉积增加了13倍,其设计的PLGA纤维支架将滑膜间充质干细胞与骨形态发生蛋白7联合植入兔模型关节内,研究中其展现出了良好的透明软骨再生的能力。 脐带间充质干细胞具有易获取、免疫原性低、产量高及扩增稳定等明显优势,在许多疾病中作为治疗手段,无免疫排斥反应或毒性反应。研究表明,脐带间充质干细胞能够在缺血缺氧微环境中存活,细胞具有分泌功能,对股骨头坏死的病理过程和组织代谢有积极影响[106]。在骨软骨组织工程支架中,通过负载脐带间充质干细胞可以向成骨与成软骨分化,增强对骨软骨缺损的修复能力[107-108]。但目前对股骨头坏死中修复骨-软骨界面目前还尚未见报道。 上述间充质干细胞均表现出向成骨、成软骨细胞分化的潜力,为解决骨-软骨界面整合,同一种间充质干细胞同时向成骨和成软骨细胞分化仍然是目前的难点。目前报道通过负载不同生长因子和不同浓度Ca2+可以实现间充质干细胞差异分化[70,109],为后续的研究提供了思路。"

| [1] HINES JT, JO WL, CUI Q, et al. Osteonecrosis of the femoral head: an updated review of arco on pathogenesis, staging and treatment. J Korean Med Sci. 2021;36(24):e177. [2] GRIESER T. Atraumatic and aseptic osteonecrosis of large joints. Radiologe. 2019;59(7):647-662. [3] ZHANG QY, LI ZR, GAO FQ, et al. Pericollapse stage of osteonecrosis of the femoral head: a last chance for joint preservation. Chin Med J (Engl). 2018;131(21):2589-2598. [4] SUGANO N, KUBO T, TAKAOKA K, et al. Diagnostic criteria for non-traumatic osteonecrosis of the femoral head: a multicentre study. J Bone Joint Surg Br. 1999;81-B(4):590-595. [5] 汝强. 股骨头坏死塌陷后软骨下骨的组织学观察[D].郑州:郑州大学, 2013. [6] RONY L, CHAPPARD D. Necrosis of the femoral head, X-ray microtomography (microCT) and histology of retrieved human femoral heads. Morphologie. 2021;105(349):134-142. [7] YAMAMOTO N, MOTOMURA G, IKEMURA S, et al. Relationship between the degree of subchondral collapse and articular surface irregularities in osteonecrosis of the femoral head. J Orthop Res. 2023. doi:10.1002/jor.25539. [8] CONZEMIUS MG, BROWN TD, ZHANG Y, et al. A new animal model of femoral head osteonecrosis: one that progresses to human-like mechanical failure. J Orthop Res. 2002;20(2):303-309. [9] MA C, ANDRE G, EDWARDS D, et al. A rat model of ischemic osteonecrosis for investigating local therapeutics using biomaterials. Acta Biomaterialia. 2021;132:260-271. [10] ZHAO D, ZHANG F, WANG B, et al. Guidelines for clinical diagnosis and treatment of osteonecrosis of the femoral head in adults (2019 version). J Orthop Translat. 2020;21:100-110. [11] SONG WS, YOO JJ, KIM YM, et al. Results of multiple drilling compared with those of conventional methods of core decompression. Clin Orthop Relat Res. 2007;454:139-146. [12] 向富州,殷文靖,朱振中,等.带血管腓骨移植治疗股骨头坏死后股骨头塌陷的危险因素[J].中华关节外科杂志(电子版),2018,12(5):626-630. [13] TANZER M, BOBYN JD, KRYGIER JJ, et al. Histopathologic retrieval analysis of clinically failed porous tantalum osteonecrosis implants. JBJS. 2008; 90(6):1282. [14] 林苗远,杨继滨,闫文强,等.组织工程技术促进股骨头坏死骨组织再血管化的研究进展[J].中国修复重建外科杂志,2021,35(11):1479-1485. [15] 薛鹏,杜斌,刘锌,等.镀膜工艺结合3D打印制备Mg-F膜/淫羊藿素膜/β-磷酸三钙支架的表征及成骨能力[J].中国组织工程研究,2023, 27(16):2480-2487. [16] 吴勰星.用于治疗股骨头坏死的含锶磷酸钙纳米复合材料的制备及其性能研究[D]. 苏州:苏州大学,2021. [17] KARIM A, AMIN AK, HALL AC. The clustering and morphology of chondrocytes in normal and mildly degenerate human femoral head cartilage studied by confocal laser scanning microscopy. J Anat. 2018;232(4): 686-698. [18] KHANARIAN NT, BOUSHELL MK, SPALAZZI JP, et al. FTIR-I compositional mapping of the cartilage-to-bone interface as a function of tissue region and age. J Bone Miner Res. 2014;29(12):2643-2652. [19] MADRY H, VAN DIJK CN, MUELLER-GERBL M. The basic science of the subchondral bone. Knee Surg Sports Traumatol Arthrosc. 2010;18(4):419-433. [20] MILZ S, PUTZ R. Quantitative morphology of the subchondral plate of the tibial plateau. J Anat. 1994;185(Pt 1):103-110. [21] ZHOU H, YUAN L, XU Z, et al. Mimicking the composition and structure of the osteochondral tissue to fabricate a heterogeneous three-layer scaffold for the repair of osteochondral defects. ACS Appl. Bio Mater. 2022;5(2):734-746. [22] WANG X, LIN J, LI Z, et al. Identification of an ultrathin osteochondral interface tissue with specific nanostructure at the human knee joint. Nano Lett. 2022;22(6):2309-2319. [23] TANG T, LANDIS W, RAGUIN E, et al. A 3D network of nanochannels for possible ion and molecule transit in mineralizing bone and cartilage. Adv Nanobiomed Res. 2022;2(8):2100162. [24] REDLER I, MOW VC, ZIMNY ML, et al. The ultrastructure and biomechanical significance of the tidemark of articular cartilage. Clin Orthop Relat Res. 1975(112):357-362. [25] 高润子,夏天卫,刘金柱,等.基于“三柱结构”的中日友好医院分型L2型及L3型股骨头坏死标本骨微结构及病理学研究[J].中国修复重建外科杂志,2022,36(8):1003-1010. [26] 赵晋,张立智,闫振宇.骨性关节炎中软骨和软骨下骨微结构病变的研究进展[J].中华实验外科杂志,2017,34(2):355-358. [27] WU Z, WANG B, TANG J, et al. Degradation of subchondral bone collagen in the weight-bearing area of femoral head is associated with osteoarthritis and osteonecrosis. J Orthop Surg Res. 2020;15(1):526. [28] YAN M, LI R, HU D, et al. Detection of subchondral bone microcirculatory perfusion in adults with early osteonecrosis of the femoral head using contrast-enhanced ultrasound: a prospective study. Ultrasound Med Biol. 2023;49(2):635-644. [29] 赵伦华,程少华,汤显斌,等.兔激素性股骨头缺血性坏死血管变化及对关节软骨的影响[J].陕西医学杂志,2008,37(3):276-277. [30] SONG J, WU J, POULET B, et al. Proteomics analysis of hip articular cartilage identifies differentially expressed proteins associated with osteonecrosis of the femoral head. Osteoarthritis Cartilage. 2021;29(7):1081-1092. [31] ZHANG J, NISHIDA Y, KOIKE H, et al. Hyaluronan in articular cartilage: Analysis of hip osteoarthritis and osteonecrosis of femoral head. J Orthop Res. 2023;41(2):307-315. [32] GAO Y H, DONG N, YANG C, et al. Elevated synovial fluid IL-33 and IL-6 levels and cartilage degeneration in stage III osteonecrosis of the femoral head. THC. 2020;28(2):203-212. [33] 杨宾宾,刘耀升,刘蜀彬.钽金属棒置入修复股骨头坏死的三维有限元分析[J].中国组织工程研究,2016,20(9):1295-1301. [34] BROWN TD, HILD GL. Pre-collapse stress redistributions in femoral head osteonecrosis--a three-dimensional finite element analysis. J Biomech Eng. 1983;105(2):171-176. [35] WANG L, ZHANG L, PAN H, et al. Abnormal subchondral bone microstructure following steroid administration is involved in the early pathogenesis of steroid-induced osteonecrosis. Osteoporos Int. 2016;27(1):153-159. [36] 庞智晖,何伟,欧志学,等.钽棒治疗早期股骨头坏死失败原因分析与对策[J].广东医学,2012,33(1):88-91. [37] LEE HJ, JEON JH, KIM CH, et al. Photosynthetic microporous bioactive glass ceramic beads for treating avascular osteonecrosis. J Indust Eng Chem. 2023;122:551-561. [38] LI B, LEI Y, HU Q, et al. Porous copper- and lithium-doped nano-hydroxyapatite composite scaffold promotes angiogenesis and bone regeneration in the repair of glucocorticoids-induced osteonecrosis of the femoral head. Biomed Mater. 2021;16(6):065012. [39] LU X, GUO H, LI J, et al. Recombinant human bone morphogenic protein-2 immobilized fabrication of magnesium functionalized injectable hydrogels for controlled-delivery and osteogenic differentiation of rat bone marrow-derived mesenchymal stem cells in femoral head necrosis repair. Front Cell Dev Biol. 2021;9:723789. [40] LIU B, YANG F, WEI X, et al. An exploratory study of articular cartilage and subchondral bone reconstruction with bone marrow mesenchymal stem cells combined with porous tantalum/Bio-Gide collagen membrane in osteonecrosis of the femoral head. Mater Sci Eng C Mater Biol Appl. 2019;99:1123-1132. [41] LOU P, DENG X, HOU D. The effects of nano-hydroxyapatite/polyamide 66 scaffold on dog femoral head osteonecrosis model: a preclinical study. Biomed Mater. 2023;18(2):025011. [42] ZHU T, JIANG M, ZHANG M, et al. Biofunctionalized composite scaffold to potentiate osteoconduction, angiogenesis, and favorable metabolic microenvironment for osteonecrosis therapy. Bioact Mater. 2022;9:446-460. [43] ZHU H, HU B, ZHANG X, et al. Repair of osteonecrosis of the femoral head with adipose-derived stem cell composite with new nanomaterial. J Nanomater. 2022;2022:e5118455. [44] WANG L, XU L, PENG C, et al. The effect of bone marrow mesenchymal stem cell and nano-hydroxyapatite/collagen I/poly-L-lactic acid scaffold implantation on the treatment of avascular necrosis of the femoral head in rabbits. Exp Ther Med. 2019;18(3):2021-2028. [45] XU H, WANG C, LIU C, et al. Cotransplantation of mesenchymal stem cells and endothelial progenitor cells for treating steroid-induced osteonecrosis of the femoral head. Stem Cells Transl Med. 2021;10(5):781-796. [46] ZHU M, HE X, XIN C, et al. 3D printing of an integrated triphasic MBG-alginate scaffold with enhanced interface bonding for hard tissue applications. J Mater Sci Mater Med. 2020;31(12):113. [47] MODRÁK M, TREBUŇOVÁ M, BALOGOVÁ A F, et al. Biodegradable materials for tissue engineering: development, classification and current applications. J Funct Biomater. 2023;14(3):159. [48] ABARRATEGI A, LÓPIZ-MORALES Y, RAMOS V, et al. Chitosan scaffolds for osteochondral tissue regeneration. J Biomed Mater Res A. 2010;95(4): 1132-1141. [49] WANG J, MI R, ZHAO G, et al. A silk-based high impact composite for the core decompression of the femoral head. J Mater Chem B. 2020;8(42): 9734-9743. [50] LIU J, FANG Q, YU X, et al. Chitosan-based nanofibrous membrane unit with gradient compositional and structural features for mimicking calcified layer in osteochondral matrix. Int J Mol Sci. 2018;19(8):2330. [51] ZHU W, ZHAO Y, MA Q, et al. 3D-printed porous titanium changed femoral head repair growth patterns: osteogenesis and vascularisation in porous titanium. J Mater Sci Mater Med. 2017;28(4):62. [52] MAZAKI T, SHIOZAKI Y, YAMANE K, et al. A novel, visible light-induced, rapidly cross-linkable gelatin scaffold for osteochondral tissue engineering. Sci Rep. 2014;4:4457. [53] QIAO Z, LIAN M, HAN Y, et al. Bioinspired stratified electrowritten fiber-reinforced hydrogel constructs with layer-specific induction capacity for functional osteochondral regeneration. Biomaterials. 2021;266:120385. [54] XIE X H, WANG X L, ZHANG G, et al. Structural and degradation characteristics of an innovative porous PLGA/TCP scaffold incorporated with bioactive molecular icaritin. Biomed Mater. 2010;5(5):054109. [55] KAWAI T, SHANJANI Y, FAZELI S, et al. Customized, degradable, functionally graded scaffold for potential treatment of early stage osteonecrosis of the femoral head. J Orthop Res. 2018;36(3):1002-1011. [56] ZHU W, MA Q, BORG S, et al. Cemented injectable multi-phased porous bone grafts for the treatment of femoral head necrosis. J Mat Chem B. 2019;7(18):2997-3006. [57] ZHANG W, LIAN Q, LI D, et al. The effect of interface microstructure on interfacial shear strength for osteochondral scaffolds based on biomimetic design and 3D printing. Mater Sci Eng C Mater Biol Appl. 2015;46:10-15. [58] WANG G, XIN H, TIAN G, et al. Core decompression combined with implantation of β-tricalcium phosphate modified by a BMSC affinity cyclic peptide for the treatment of early osteonecrosis of the femoral head. Am J Transl Res. 2021;13(3):967-978. [59] YANG Z, WANG B, LIU W, et al. In situ self-assembled organoid for osteochondral tissue regeneration with dual functional units. Bioact Mater. 2023;27:200-215. [60] ERICKSON AE, SUN J, LAN LEVENGOOD SK, et al. Chitosan-based composite bilayer scaffold as an in vitro osteochondral defect regeneration model. Biomed Microdevices. 2019;21(2):34. [61] LIU K, LIU Y, DUAN Z, et al. A biomimetic bi-layered tissue engineering scaffolds for osteochondral defects repair. Sci. China Technol Sci. 2021; 64(4):793-805. [62] 邓天政,吕晶,杨捷绯,等.体外构建组织工程骨-软骨复合组织[J].中国组织工程研究,2013,17(8):1354-1361. [63] KORPAYEV S, KAYGUSUZ G, ŞEN M, et al. Chitosan/collagen based biomimetic osteochondral tissue constructs: a growth factor-free approach. Int J Biol Macromol. 2020;156:681-690. [64] QIANG Y, YANHONG Z, JIANG P, et al. Xenoimplantation of an extracellular-matrix-derived, biphasic, cell-scaffold construct for repairing a large femoral-head high-load-bearing osteochondral defect in a canine model. ScientificWorldJournal. 2014;2014:127084. [65] FRYHOFER GW, ZLOTNICK HM, STOECKL BD, et al. Fabrication and maturation of integrated biphasic anatomic mesenchymal stromal cell-laden composite scaffolds for osteochondral repair and joint resurfacing. J Orthop Res. 2021;39(11):2323-2332. [66] DUAN P, PAN Z, CAO L, et al. The effects of pore size in bilayered poly(lactide-co-glycolide) scaffolds on restoring osteochondral defects in rabbits. J Biomed Mater Res A. 2014;102(1):180-192. [67] GU X, ZHA Y, LI Y, et al. Integrated polycaprolactone microsphere-based scaffolds with biomimetic hierarchy and tunable vascularization for osteochondral repair. Acta Biomater. 2022;141:190-197. [68] HOLLAND TA, BODDE EWH, BAGGETT LS, et al. Osteochondral repair in the rabbit model utilizing bilayered, degradable oligo(poly(ethylene glycol) fumarate) hydrogel scaffolds. J Biomed Mater Res A. 2005;75(1):156-167. [69] DANG W, WANG X, LI J, et al. 3D printing of Mo-containing scaffolds with activated anabolic responses and bi-lineage bioactivities. Theranostics. 2018;8(16):4372-4392. [70] MELLOR LF, MOHITI-ASLI M, WILLIAMS J, et al. Extracellular calcium modulates chondrogenic and osteogenic differentiation of human adipose-derived stem cells: a novel approach for osteochondral tissue engineering using a single stem cell source. Tissue Eng Part A. 2015;21(17-18):2323-2333. [71] XIAO H, HUANG W, XIONG K, et al. Osteochondral repair using scaffolds with gradient pore sizes constructed with silk fibroin, chitosan, and nano-hydroxyapatite. Int J Nanomedicine. 2019;14:2011-2027. [72] MOHAN N, DORMER NH, CALDWELL KL, et al. Continuous gradients of material composition and growth factors for effective regeneration of the osteochondral interface. Tissue Engineering Part A. 2011;17(21-22): 2845-2855. [73] HUANG L, CAI B, HUANG Y, et al. Comparative study on 3D printed Ti6Al4V scaffolds with surface modifications using hydrothermal treatment and microarc oxidation to enhance osteogenic activity. ACS Omega. 2021;6(2): 1465-1476. [74] CHEVALIER X. Fibronectin, cartilage, and osteoarthritis. Semin Arthritis Rheum. 1993;22(5):307-318. [75] VAN VLIET A, BAELDE HJ, VLEMING LJ, et al. Distribution of fibronectin isoforms in human renal disease. J Pathol. 2001;193(2):256-262. [76] FANAEE S, LABBAF S, ENAYATI MH, et al. A nano approach towards the creation of a biointerface as stimulator of osteogenic differentiation. Mater Sci Eng C Mater Biol Appl. 2021;120:111746. [77] YAMAZAKI M, ONODERA K, IIJIMA K. Surface modification of silica nonwoven fabrics for osteogenesis of bone marrow-derived mesenchymal stem cells. J Biosci Bioeng. 2022;134(6):541-548. [78] ALADAL M, YOU W, HUANG R, et al. Insights into the implementation of Fibronectin 1 in the cartilage tissue engineering. Biomed Pharmacother. 2022;148:112782. [79] DAUM R, VISSER D, WILD C, et al. Fibronectin adsorption on electrospun synthetic vascular grafts attracts endothelial progenitor cells and promotes endothelialization in dynamic in vitro culture. Cells. 2020;9(3):778. [80] LIU X, FENG Q, BACHHUKA A, et al. Surface modification by allylamine plasma polymerization promotes osteogenic differentiation of human adipose-derived stem cells. ACS Appl Mater Interfaces. 2014;6(12):9733-9741. [81] JIAO Y, XU J, ZHOU C. Effect of ammonia plasma treatment on the properties and cytocompatibility of a poly (L-lactic acid) film surface. J Biomater Sci Polym Ed. 2012;23(6):763-777. [82] ESBAH TABAEI PS, ASADIAN M, GHOBEIRA R, et al. Combinatorial effects of coral addition and plasma treatment on the properties of chitosan/polyethylene oxide nanofibers intended for bone tissue engineering. Carbohydr Polym. 2021;253:117211. [83] LERMAN MJ, SMITH BT, GERALD AG, et al. Aminated 3D printed polystyrene maintains stem cell proliferation and osteogenic differentiation. Tissue Eng Part C Methods. 2020;26(2):118-131. [84] XUE P, CHEN H, XI H, et al. Magnesium dopped calcium-fluoride/icaritin composite multi-layer coating functionalized 3D printed β-TCP scaffold inducessustained bone regeneration in a rabbit model. Mater Design. 2022;223:111156. [85] VIJAYAVENKATARAMAN S, YAN W C, LU W F, et al. 3D bioprinting of tissues and organs for regenerative medicine. Adv Drug Deliv Rev. 2018;132:296-332. [86] ZHANG X, HUANG P, JIANG G, et al. A novel magnesium ion-incorporating dual-crosslinked hydrogel to improve bone scaffold-mediated osteogenesis and angiogenesis. Mater Sci Eng C Mater Biol Appl. 2021;121:111868. [87] CHAI S, HUANG J, MAHMUT A, et al. Injectable photo-crosslinked bioactive BMSCs-BMP2-GelMA scaffolds for bone defect repair. Front Bioeng Biotechnol. 2022;10:875363. [88] HUANG B, CHEN M, TIAN J, et al. Oxygen-carrying and antibacterial fluorinated nano-hydroxyapatite incorporated hydrogels for enhanced bone regeneration. Adv Healthc Mater. 2022;11(12):e2102540. [89] HAM DW, SON TI, LEE TJ, et al. Osteogenic effectiveness of photo-immobilized bone morphogenetic protein-2 using different azidophenyl-natural polymer carriers in rat calvarial defect model. Int J Biol Macromol. 2019;121:333-341. [90] HUA Y, HUO Y, BAI B, et al. Fabrication of biphasic cartilage-bone integrated scaffolds based on tissue-specific photo-crosslinkable acellular matrix hydrogels. Mater Today Bio. 2022;17:100489. [91] BARRON V, NEARY M, MOHAMED KMS, et al. Evaluation of the early in vivo response of a functionally graded macroporous scaffold in an osteochondral defect in a rabbit model. Ann Biomed Eng. 2016;44(5):1832-1844. [92] DASKALOVA A, OSTROWSKA B, ZHELYAZKOVA A, et al. Improving osteoblasts cells proliferation via femtosecond laser surface modification of 3D-printed poly-ε-caprolactone scaffolds for bone tissue engineering applications. Appl Phys A. 2018;124(6):413. [93] MCCULLEN SD, MILLER PR, GITTARD SD, et al. In situ collagen polymerization of layered cell-seeded electrospun scaffolds for bone tissue engineering applications. Tissue Eng Part C Methods. 2010;16(5):1095-1105. [94] 董少杰,王旭东,沈国芳,等.生物陶瓷支架的功能改性及应用研究进展[J].无机材料学报,2020,35(8):867-881. [95] BU Y, MA J, BEI J, et al. Surface modification of aliphatic polyester to enhance biocompatibility. Front Bioeng Biotechnol. 2019;7:98. [96] MOON SH, HWANG HJ, JEON HR, et al. Photocrosslinkable natural polymers in tissue engineering. Front Bioeng Biotechnol. 2023;11:1127757. [97] FENN SL, OLDINSKI RA. Visible light crosslinking of methacrylated hyaluronan hydrogels for injectable tissue repair. J Biomed Mater Res B Appl Biomater. 2016;104(6):1229-1236. [98] LI R, LIN QX, LIANG XZ, et al. Stem cell therapy for treating osteonecrosis of the femoral head: From clinical applications to related basic research. Stem Cell Res Ther. 2018;9(1):291. [99] CIAPETTI G, GRANCHI D, FOTIA C, et al. Effects of hypoxia on osteogenic differentiation of mesenchymal stromal cells used as a cell therapy for avascular necrosis of the femoral head. Cytotherapy. 2016;18(9):1087-1099. [100] PINTUS E, BALDASSARRI M, PERAZZO L, et al. Stem cells in osteochondral tissue engineering. Adv Exp Med Biol. 2018;1058:359-372. [101] QIN L, YAO D, ZHENG L, et al. Phytomolecule icaritin incorporated PLGA/TCP scaffold for steroid-associated osteonecrosis: Proof-of-concept for prevention of hip joint collapse in bipedal emus and mechanistic study in quadrupedal rabbits. Biomaterials. 2015;59:125-143. [102] DIAZ-RODRIGUEZ P, ERNDT-MARINO JD, GHARAT T, et al. Toward zonally tailored scaffolds for osteochondral differentiation of synovial mesenchymal stem cells. J Biomed Mater Res B Appl Biomater. 2019; 107(6):2019-2029. [103] CHEN YP, CHEN WC, WANG KC, et al. Effectiveness of synovial fluid mesenchymal stem cells embedded in alginate beads for treatment of steroid-induced avascular necrosis of the femoral head. J Orthop Sci. 2014; 19(4):657-666. [104] GUO SC, TAO SC, YIN WJ, et al. Exosomes from human synovial-derived mesenchymal stem cells prevent glucocorticoid-induced osteonecrosis of the femoral head in the rat. Int J Biol Sci. 2016;12(10):1262-1272. [105] KIM HJ, HAN MA, SHIN JY, et al. Intra-articular delivery of synovium-resident mesenchymal stem cells via BMP-7-loaded fibrous PLGA scaffolds for cartilage repair. J Control Release. 2019;302:169-180. [106] ZHAO J, MENG H, LIAO S, et al. Therapeutic effect of human umbilical cord mesenchymal stem cells in early traumatic osteonecrosis of the femoral head. J Orthop Transl. 2022;37:126-142. [107] ZHENG P, HU X, LOU Y, et al. A rabbit model of osteochondral regeneration using three-dimensional printed polycaprolactone-hydroxyapatite scaffolds coated with umbilical cord blood mesenchymal stem cells and chondrocytes. Med Sci Monit. 2019;25:7361-7369. [108] CUI X, ALCALA-OROZCO CR, BAER K, et al. 3D bioassembly of cell-instructive chondrogenic and osteogenic hydrogel microspheres containing allogeneic stem cells for hybrid biofabrication of osteochondral constructs. Biofabrication. 2022;14(3):034101. [109] LU S, LAM J, TRACHTENBERG JE, et al. Dual growth factor delivery from bilayered, biodegradable hydrogel composites for spatially-guided osteochondral tissue repair. Biomaterials. 2014;35(31):8829-8839. [110] CHOE R, DEVOY E, JABARI E, et al. Biomechanical aspects of osteochondral regeneration: implications and strategies for three-dimensional bioprinting. Tissue Eng Part B Rev. 2022;28(4):766-788. [111] HUNZIKER EB. Articular cartilage repair: basic science and clinical progress. A review of the current status and prospects. Osteoarthritis Cartilage. 2002;10(6):432-463. [112] MANCINI I AD, SCHMIDT S, BROMMER H, et al. A composite hydrogel-3D printed thermoplast osteochondral anchor as example for a zonal approach to cartilage repair: in vivo performance in a long-term equine model. Biofabrication. 2020;12(3):035028. [113] HSIEH YH, SHEN BY, WANG YH, et al. Healing of osteochondral defects implanted with biomimetic scaffolds of poly(ε-caprolactone)/hydroxyapatite and glycidyl-methacrylate-modified hyaluronic acid in a minipig. Int J Mol Sci. 2018;19(4):1125. [114] MELTON JT, WILSON AJ, CHAPMAN-SHEATH P, et al. TruFit CB® bone plug: chondral repair, scaffold design, surgical technique and early experiences. Exp Rev Med Dev. 2010;7(3):333-341. |
| [1] | Chen Kaijia, Liu Jingyun, Cao Ning, Sun Jianbo, Zhou Yan, Mei Jianguo, Ren Qiang. Application and prospect of tissue engineering in treatment of osteonecrosis of the femoral head [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(9): 1450-1456. |
| [2] | Feng Ruiqin, Han Na, Zhang Meng, Gu Xinyi, Zhang Fengshi. Combination of 1% platelet-rich plasma and bone marrow mesenchymal stem cells improves the recovery of peripheral nerve injury [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(7): 985-992. |
| [3] | Qiu Xiaoyan, Li Bixin, Li Jingdi, Fan Chuiqin, Ma Lian, Wang Hongwu. Differentiation of insulin-producing cells from human umbilical cord mesenchymal stem cells infected by MAFA-PDX1 overexpressed lentivirus [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(7): 1000-1006. |
| [4] | Liu Qiwei, Zhang Junhui, Yang Yuan, Wang Jinjuan. Role and mechanism of umbilical cord mesenchymal stem cells on polycystic ovary syndrome [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(7): 1015-1020. |
| [5] | Pan Xiaolong, Fan Feiyan, Ying Chunmiao, Liu Feixiang, Zhang Yunke. Effect and mechanism of traditional Chinese medicine on inhibiting the aging of mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(7): 1091-1098. |
| [6] | Ma Shuwei, He Sheng, Han Bing, Zhang Liaoyun. Exosomes derived from mesenchymal stem cells in treatment of animals with acute liver failure: a meta-analysis [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(7): 1137-1142. |
| [7] | Xue Xiaofeng, Wei Yongkang, Qiao Xiaohong, Du Yuyong, Niu Jianjun, Ren Lixin, Yang Huifeng, Zhang Zhimin, Guo Yuan, Chen Weiyi. Finite element analysis of osteoporosis in proximal femur after cannulated screw fixation for femoral neck fracture [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(6): 862-867. |
| [8] | Zhang Kefan, Shi Hui. Research status and application prospect of cytokine therapy for osteoarthritis [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(6): 961-967. |
| [9] | Yin Kaiwen, Li Yunfeng. Application of metal-organic frameworks in implant surface modification [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(5): 783-788. |
| [10] | Wei Yuanxun, Chen Feng, Lin Zonghan, Zhang Chi, Pan Chengzhen, Wei Zongbo. The mechanism of Notch signaling pathway in osteoporosis and its prevention and treatment with traditional Chinese medicine [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(4): 587-593. |
| [11] | Lin Feng, Cheng Ling, Gao Yong, Zhou Jianye, Shang Qingqing. Hyaluronic acid hydrogel-encapsulated bone marrow mesenchymal stem cells promote cardiac function in myocardial infarction rats (III) [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(3): 355-359. |
| [12] | Bi Yujie, Ma Dujun, Peng Liping, Zhou Ziqiong, Zhao Jing, Zhu Houjun, Zhong Qiuhui, Yang Yuxin. Strategy and significance of Chinese medicine combined with medical hydrogel for disease treatment [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(3): 419-425. |
| [13] | Yang Jie, Hu Haolei, Li Shuo, Yue Wei, Xu Tao, Li Yi. Application of bio-inks for 3D printing in tissue repair and regenerative medicine [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(3): 445-451. |
| [14] | Long Zhisheng, Gong Feipeng, Wen Jiabin, Min Huan, Shu Yang, Lai Zhuoxi, Chen Gang. Bone marrow mesenchymal stem cell exosomes combined with epigallocatechin-3-gallate in treatment of spinal cord ischemia/reperfusion injury in rats [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(19): 2953-2959. |
| [15] | Chen Na, Wang Yanlin, Sun Huifang, Fan Feiyan, Li Donghong, Zhang Yunke. Shexiang Huangqi compound dripping pills-containing serum promotes proliferation and differentiation of bone marrow mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(19): 2960-2966. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||