Chinese Journal of Tissue Engineering Research ›› 2023, Vol. 27 ›› Issue (24): 3926-3936.doi: 10.12307/2023.701
Extracellular vesicles for acute kidney injury in preclinical research: a meta-analysis
Li Qingru1, Wang Yifan1, Chen Guanting1, Shi Ruoyu1, Zhang Linqi2
- 1Henan University of Chinese Medicine, Zhengzhou 450000, Henan Province, China; 2First Affiliated Hospital of Henan University of Chinese Medicine, Zhengzhou 450000, Henan Province, China
-
Received:2022-09-09Accepted:2022-11-08Online:2023-08-28Published:2023-01-19 -
Contact:Zhang Linqi, Chief physician, Professor, Doctoral supervisor, First Affiliated Hospital of Henan University of Chinese Medicine, Zhengzhou 450000, Henan Province, China -
About author:Li Qingru, Doctoral candidate, Henan University of Chinese Medicine, Zhengzhou 450000, Henan Province, China -
Supported by:General Program of National Natural Science Foundation of China, No. 81973806 (to ZLQ); Major Special Project of Scientific Research on Traditional Chinese Medicine in Henan Province, No. 2019ZYZD05 (to ZLQ)
CLC Number:
Cite this article
Li Qingru, Wang Yifan, Chen Guanting, Shi Ruoyu, Zhang Linqi. Extracellular vesicles for acute kidney injury in preclinical research: a meta-analysis[J]. Chinese Journal of Tissue Engineering Research, 2023, 27(24): 3926-3936.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
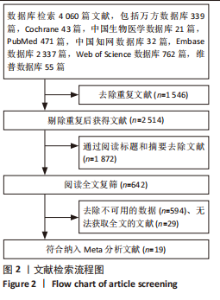
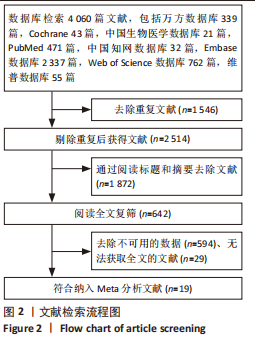
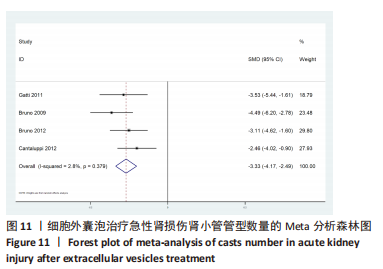
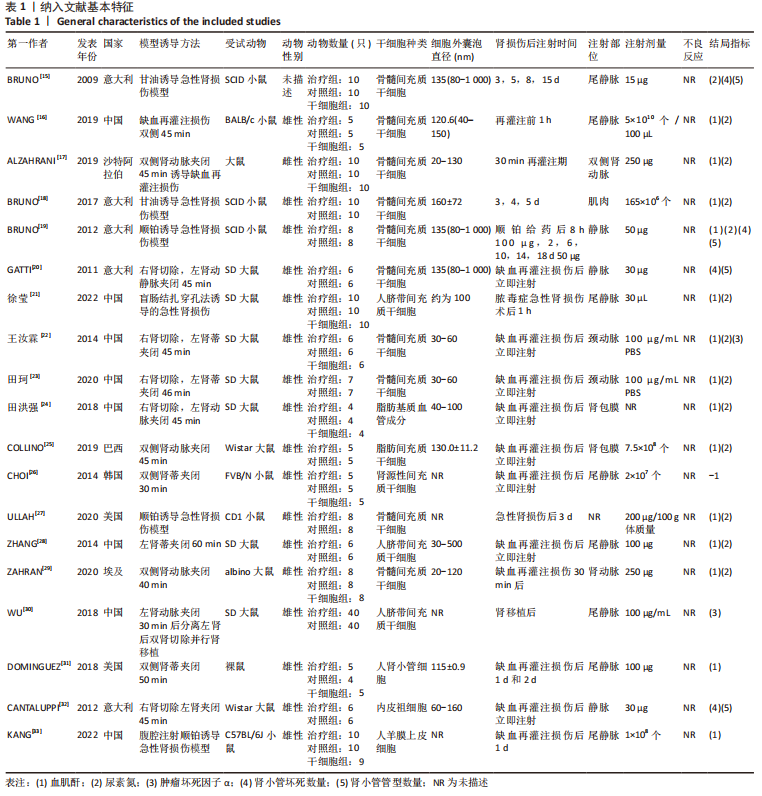
2.2 纳入文献的基本特征 在纳入的19篇文献中[15-33],共包含399只啮齿动物,细胞外囊泡治疗组啮齿动物169只,对照组啮齿动物168只,干细胞治疗组啮齿动物62只,其中18篇期刊文章[15-23,25-33],1篇硕士论文[24];其中有10篇文献使用骨髓来源间充质干细胞[15-20,22-23,27,29],3篇文献使用人脐带间充质干细胞[21,28,30],1篇文献使用脂肪基质血管成分[24],1篇使用脂肪间充质干细胞[25],1篇文献使用肾源性间充质干细胞[26],1篇文献使用人肾小管细胞[31],1篇文献使用内皮祖细胞[32],1篇文献使用人羊膜上皮细胞[33]。共包括3个研究对象为重度联合免疫缺陷(server combined immune-deficiency,SCID)小鼠[15,18-19],1个研究对象为白变种实验室老鼠(BALB/c小鼠) [16],7个研究的对象为SD大鼠[20-24,28,30],2个研究的对象为Wistar 大鼠[25,32],1个研究的对象为FVB/N 小鼠[26],1个研究对象为CD1小鼠[27],1个研究的对象为天竺鼠(albino大鼠)[29],1个研究的对象为裸鼠[31],1个研究的对象为C57BL/6J小鼠[33],1个研究的对象单纯提及为大鼠[17]。文献基线特征见表1。"

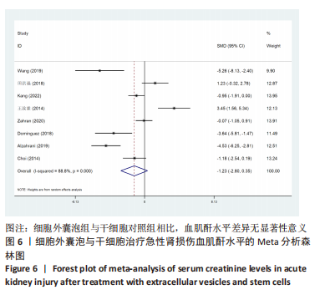
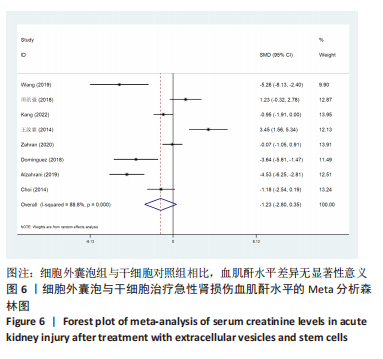
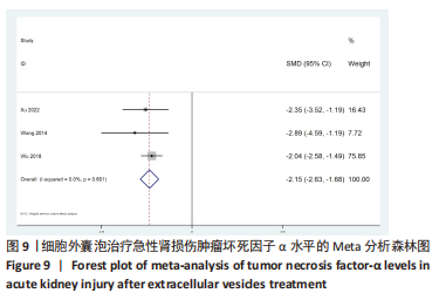
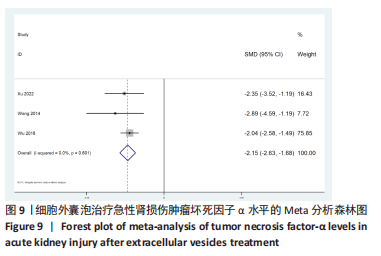
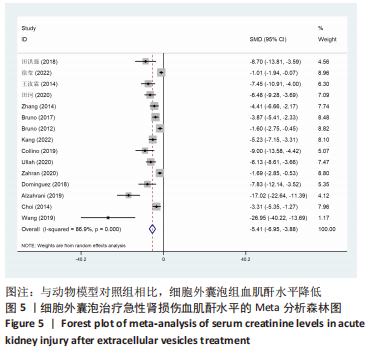
2.4 Meta分析结果 2.4.1 各组血肌酐水平差异 细胞外囊泡组vs.动物模型对照组:共15篇文献报道了诱导啮齿动物急性肾损伤后使用细胞外囊泡治疗对动物血肌酐的影响[16-19,21-29,31,33]。经过异质性检验,I2 > 50%,P < 0.001,提示文章所选文献间异质性有显著性意义。为寻求异质性来源,对该研究以动物种类、细胞类型、动物性别及注射部位为变量进行Meta回归分析,结果表明,动物类型(P=0.332)、细胞类型(P=0.302)、动物性别(P=0.409)及注射部位(P=0.807)均无法对该研究异质性作出解释,采用随机效应模型,结果提示,细胞外囊泡组的血肌酐水平显著低于对照组(SMD=-5.41,95%CI:-6.95至-3.88,P < 0.001),见图5。"

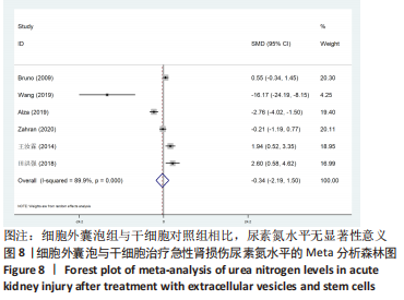
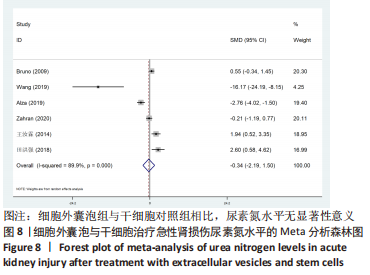
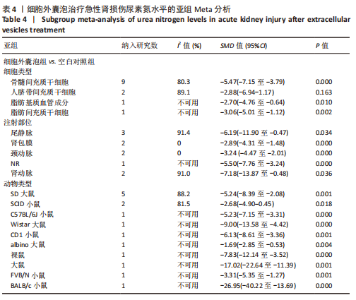
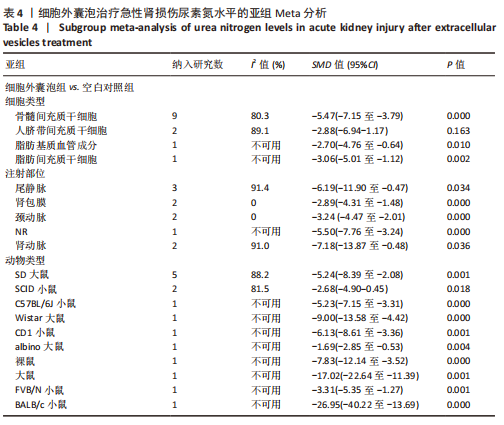
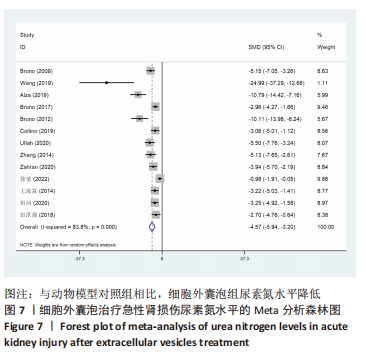
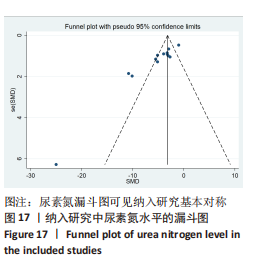
2.4.2 各组尿素氮水平差异 细胞外囊泡组vs.动物模型对照组:共13篇文献报道了诱导啮齿动物急性肾损伤后使用细胞外囊泡治疗对动物尿素氮的影响[15-19,21-25,27-29]。由于各研究结果之间存在异质性(I2 > 50%),且Q检验P < 0.001,提示文章选择文献间异质性有显著性意义。为寻求异质性来源,对该研究以动物种类、细胞类型、动物性别及注射部位为变量进行Meta回归分析,结果表明,Meta回归方程结果显示,细胞类型(P=0.402)、动物性别(P=0.345)及注射部位(P=0.395)均无法对该研究的异质性作出解释,动物类型(P=0.044)可能为异质性来源。采用随机效用模型,结果提示,细胞外囊泡组的尿素氮水平显著低于对照组(SMD=-4.57,95%CI:-5.94至-3.20,P < 0.001),见图7。"
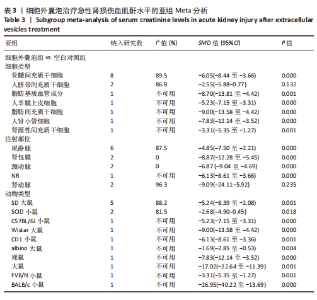
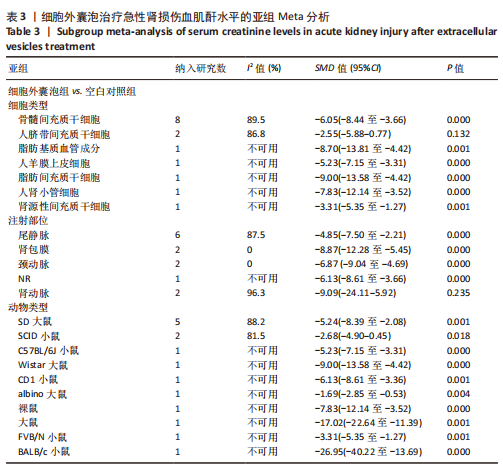
2.4.5 亚组分析结果 血肌酐水平亚组分析:基于细胞类型、注射部位及动物类型进行亚组分析,采用随机效应模型,结果显示,除人脐带间充质干细胞组治疗后的水平与对照组比较差异无显著性意义(SMD=-2.55,95%CI:-5.88-0.77,P=0.132)、注射部位为肾动脉组治疗后水平与对照组比较无显著性意义(SMD=-9.09,95%CI:-24.11-5.92,P=0.235)外,其他亚组治疗后血肌酐水平与对照组相比均有明显下降。其中注射部位为肾包膜及颈动脉的治疗组经异质性分析发现两组无异质性,且注射部位为肾包膜的治疗组(SMD=-8.87,95%CI:-12.28至-5.45,P=0.000)降低血肌酐水平疗效优于注射部位为颈动脉治疗组(SMD=-6.87,95%CI:-9.04至-4.69,P=0.000),见表3。"
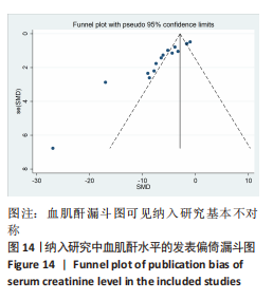
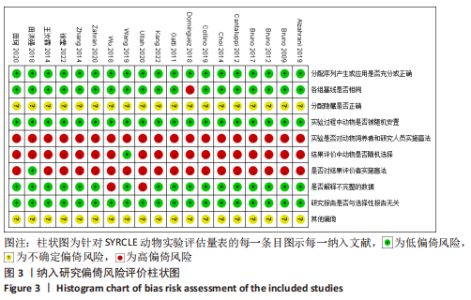
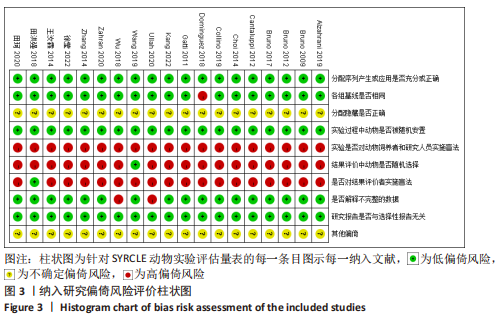
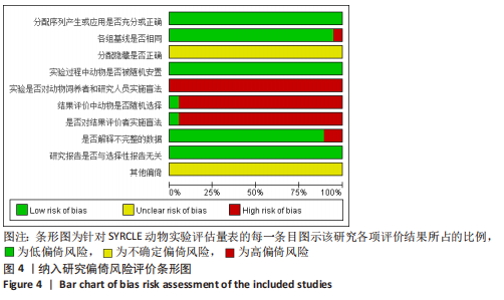
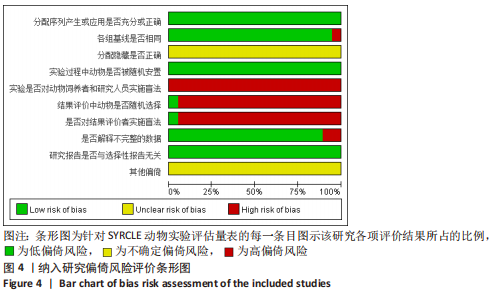
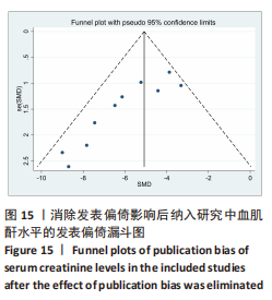
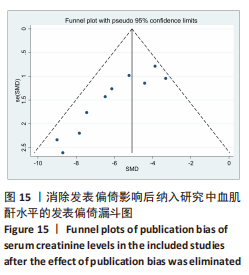
2.6 发表偏倚分析 2.6.1 血肌酐水平发表偏倚分析 对文章纳入的15篇文献进行偏倚性检验,结果发现漏斗图不对称,见图14,更进一步对其进行Egger检验,得出P值小于0.1,提示文章存在发表偏倚 (95%CI:-5.80至-3.85,P < 0.1),剪补前的随机效应模型所得的(SMD=-5.41,95%CI: -6.95至-3.88,P < 0.001),用剪补法修正发表偏倚对结果的影响,结果显示,(SMD=-3.89,95%CI:-5.42至-2.36,P < 0.001),剪补前后95%CI均有显著性意义,结果稳定,补充5个点,消除发表偏倚的影响,漏斗图中心位置变化不大,见图15。发表性偏倚是指有显著性意义的研究结果比无显著性意义的研究更容易投稿和发表,通过对文章纳入的15篇文献进行漏斗图及Egger检验,发现存在发表偏倚,见图14。通过剪补法对漏斗图不对称的部分进行补充,对基于贴补后的漏斗图估计合并效应量的真实值,最后对比发现贴补前后的漏斗图合并效应量变化不大,提示该研究结果稳定,见图15。"
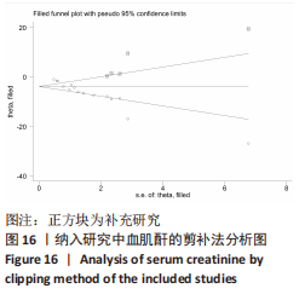
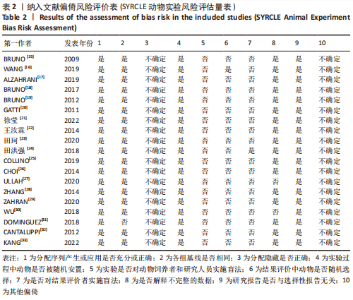
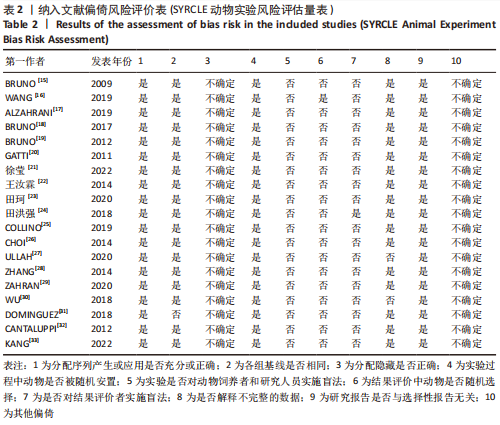
进一步对漏斗图分析发现,有5篇文献存在较大的偏倚风险[16-17,19,21,29],经过剔除后,所有研究均分布于漏斗图内,见图16。分析发现,与其他直接注射细胞外囊泡相比,ALZAHRANI等[17]和ZAHRAN等[29]所使用的细胞外囊泡经过了褪黑素预处理,因此可能导致出现偏倚;对比发现,WANG等[16]的注射剂量不同于其他研究,是以每100 μL 5×1010个的浓度输注,可能会导致出现偏倚;在模型诱导方式上,徐莹等[21]不同于其他研究直接诱导模型急性肾损伤,而是通过盲肠结扎穿孔法导致动物模型出现脓毒症间接诱发急性肾损伤,因此可能导致出现偏倚;经研究发现,BRUNO等[19]使用顺铂诱导急性肾损伤,增加了动物模型死亡风险,并且不同于BRUNO其他研究[15,18],此次增加了细胞外囊泡的应用剂量,这或许也是导致偏倚风险增加的因素。对文章进行剪补法分析,剪补前的随机效应模型所得的(SMD=-5.41,95%CI:-6.95至-3.88,P < 0.001),用剪补法修正发表偏倚对结果的影响,结果显示(SMD=-3.89,95%CI:-5.42至-2.36,P < 0.001),从修补图看出,可能是部分阴性结果未发表导致了发表偏倚,剪补前后95%CI均有显著性意义,结果稳定,见图16。"
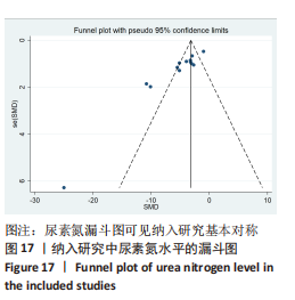
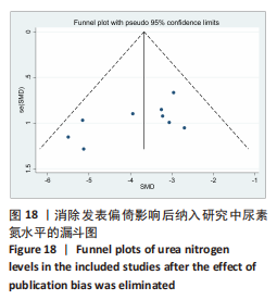
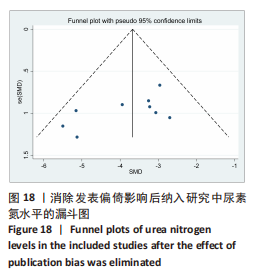
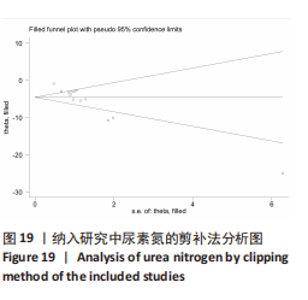
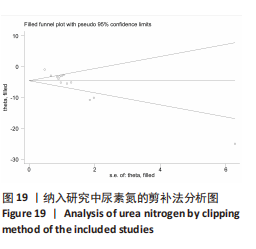
2.6.2 尿素氮水平发表偏倚分析 对此次纳入的13篇文献进行偏倚性检验,漏斗图基本对称,见图17,更进一步对其进行Egger检验,Egger检验提示尿素氮存在发表偏倚(95%CI:-6.64至-3.86,P < 0.1),对漏斗图分析发现,有4篇文献存在较大的偏倚风险[16-17,19,21]。剔除后,所有研究均分布于漏斗图内,见图18。分析发现,与其他直接注射细胞外囊泡相比,ALZAHRANI等[17]、所使用的的细胞外囊泡经过了褪黑素预处理,因此可能导致出现偏倚;对比发现,WANG等[16]的注射剂量不同于其他研究,是以每100 μL 5×1010个的浓度输注,可能会导致出现偏倚;在模型诱导方式上,徐莹等[21]的研究不同于其他研究直接诱导模型急性肾损伤,而是通过盲肠结扎穿孔法导致动物模型出现脓毒症间接诱发急性肾损伤,因此可能导致出现偏倚;经研究发现,BRUNO等[19]使用顺铂诱导急性肾损伤,增加了动物模型死亡风险,并且不同于BRUNO其他研究[15,18],此次增加了细胞外囊泡的应用剂量,这或许也是导致偏倚风险增加的因素。对文章用剪补法分析发表偏倚对结果的影响,剪补前随机效应模型所得(SMD=-5.41,95%CI:-6.95至-3.88,P < 0.001),运用剪补法修正前后效应值无变化(SMD=-4.57,95%CI:-5.94至-3.20,P < 0.001),剪补前后95%CI均有显著性意义,结果稳定,见图19。"
| [1] HAN SJ, LEE HT. Mechanisms and therapeutic targets of ischemic acute kidney injury. Res Clin Pract. 2019;38(4):427-440. [2] AL-JEFRI M, LEE J, JAMES M. Predicting acute kidney injury after surgery. Annu Int Conf IEEE Eng Med Biol Soc. 2020;2020:5606-5609. [3] LEWINGTON AJ, CERDÁ J, MEHTA RL. Raising awareness of acute kidney injury: a global perspective of a silent killer. Kidney Int. 2013; 84(3):457-467. [4] HOSTE EAJ, KELLUM JA, SELBY NM, et al. Global epidemiology and outcomes of acute kidney injury. Nat Rev Nephrol. 2018; 14(10):607-625. [5] RICCI Z, CRUZ DN, RONCO C. Classification and staging of acute kidney injury: beyond the RIFLE and AKIN criteria. Nat Rev Nephrol. 2011;7(4):201-208. [6] JANG HR, RABB H. The innate immune response in ischemic acute kidney injury. Clin Immunol. 2009;130(1):41-50. [7] LI JS, LI B. Renal injury repair: how about the role of stem cells. Adv Exp Med Biol. 2019;1165:661-670. [8] BLAU HM, DALEY GQ. Stem cells in the treatment of disease. N Engl J Med. 2019; 380(18):1748-1760. [9] YUN CW, LEE SH. Potential and therapeutic efficacy of cell-based therapy using mesenchymal stem cells for acute/chronic kidney disease. Int J Mol Sci. 2019;20(7):1619. [10] CALDAS HC, LOJUDICE FH, DIAS C,et al. Induced pluripotent stem cells reduce progression of experimental chronic kidney disease but develop Wilms’ tumors. Stem Cells Int. 2017;2017:7428316. [11] XUNIAN Z, KALLURI R. Biology and therapeutic potential of mesenchymal stem cell-derived exosomes. Cancer Sci. 2020; 111(9):3100-3110. [12] CAO JY, WANG B, TANG TT, et al. Exosomal miR-125b-5p deriving from mesenchymal stem cells promotes tubular repair by suppression of p53 in ischemic acute kidney injury. Theranostics. 2021;11(11):5248-5266. [13] EIRIN A, ZHU XY, PURANIK AS, et al. Mesenchymal stem cell-derived extracellular vesicles attenuate kidney inflammation. Kidney Int. 2016;92(1):114-124. [14] HOOIJMANS CR, ROVERS MM, DE VRIES RB, et al. SYRCLE’s risk of bias tool for animal studies. BMC Med Res Methodol. 2014;26;14:43. [15] BRUNO S, GRANGE C, DEREGIBUS MC, et al. Mesenchymal stem cell-derived microvesicles protect against acute tubular injury. J Am Soc Nephrol. 2009;20:1053-1067. [16] WANG C, ZHU G, HE W, et al. BMSCs protect against renal ischemia-reperfusion injury by secreting exosomes loaded with miR-199a-5p that target BIP to inhibit endoplasmic reticulum stress at the very early reperfusion stages. FASEB J. 2019;33: 5440-5456. [17] ALZAHRANI FA. Melatonin improves therapeutic potential of mesenchymal stem cells-derived exosomes against renal ischemia-reperfusion injury in rats. Am J Transl Res. 2019;11:2887-2907. [18] BRUNO S, TAPPARO M, COLLINO F, et al. Renal regenerative potential of different extracellular vesicle populations derived from bone marrow mesenchymal stromal cells. Tissue Eng Part A. 2017;23:1262-1273. [19] BRUNO S, GRANGE C, COLLINO F, et al. Microvesicles derived from mesenchymal stem cells enhance survival in a lethal model of acute kidney injury. PLoS One. 2012;7(3):e33115. [20] GATTI S, BRUNO S, DEREGIBUS MC, et al. Microvesicles derived from human adult mesenchymal stem cells protect against ischaemia-reperfusion-induced acute and chronic kidney injury. Nephrol Dial Transplant. 2011;26:1474-1483. [21] 徐莹,周茹,张欣洲,等.间充质干细胞外泌体对CLP大鼠急性肾损伤作用研究[J].湖北医药学院学报,2022,41(2):116-120. [22] 王汝霖,林淼,黎力平,等.骨髓间充质干细胞来源exosome对大鼠肾缺血再灌注损伤的保护作用[J].中华医学杂志, 2014,94(42):3298-3303. [23] 田珂.骨髓间充质干细胞来源外泌体对大鼠肾脏缺血再灌注损伤保护作用研究[J]. 医药前沿,2020,10(18):99-100. [24] 田洪强. Ad-SVF来源的外泌体对大鼠肾脏缺血再灌注损伤的影响及机制[D].南京:南京医科大学,2018. [25] COLLINO F, LOPES JA, CORRÊA S, et al. Adipose-derived mesenchymal stromal cells under hypoxia: changes in extracellular vesicles secretion and improvement of renal recovery after ischemic injury. Cell Physiol Biochem. 2019;52:1463-1483. [26] CHOI HY, MOON SJ, RATLIFF BB, et al. Microparticles from kidney-derived mesenchymal stem cells act as carriers of proangiogenic signals and contribute to recovery from acute kidney injury. PLoS One. 2014;9(2):e87853. [27] ULLAH M, LIU DD, RAI S, et al. HSP70-mediated NLRP3 inflammasome suppression underlies reversal of acute kidney injury following extracellular vesicle and focused ultrasound combination therapy. Int J Mol Sci. 2020;21(11):4085. [28] ZHANG G, ZOU X, MIAO S, et al. The anti-oxidative role of micro-vesicles derived from human Wharton-Jelly mesenchymal stromal cells through NOX2/gp91(phox) suppression in alleviating renal ischemia-reperfusion injury in rats. PLoS One. 2014; 9:e92129. [29] ZAHRAN R, GHOZY A, ELKHOLY SS, et al. Combination therapy with melatonin, stem cells and extracellular vesicles is effective in limiting renal ischemia-reperfusion injury in a rat model. Int J Urol. 2020;27(11):1039-1049. [30] WU X, YAN T, WANG Z, et al. Micro-vesicles derived from human Wharton’s Jelly mesenchymal stromal cells mitigate renal ischemia-reperfusion injury in rats after cardiac death renal transplantation. J Cell Biochem. 2018;119(2):1879-1888. [31] DOMINGUEZ JM 2ND, DOMINGUEZ JH, XIE D, et al. Human extracellular microvesicles from renal tubules reverse kidney ischemia-reperfusion injury in rats. PLoS One. 2018; 13:e0202550. [32] CANTALUPPI V, GATTI S, MEDICA D, et al. Microvesicles derived from endothelial progenitor cells protect the kidney from ischemia-reperfusion injury by microRNA-dependent reprogramming of resident renal cells. Kidney Int. 2012;82(4):412-427. [33] KANG X, CHEN Y, XIN X, et al. Human amniotic epithelial cells and their derived exosomes protect against cisplatin-induced acute kidney injury without compromising its antitumor activity in mice. Front Cell Dev Biol. 2021;9:752053. [34] GREENBERG JH, COCA S, PARIKH CR. Long-term risk of chronic kidney disease and mortality in children after acute kidney injury: a systematic review. BMC Nephrol. 2014;15:184. [35] VANDENBERGHE W, GEVAERT S, KELLUM JA, et al. Acute kidney injury in cardiorenal syndrome type 1 patients: a systematic review and meta-analysis. Cardiorenal Med. 2016;6(2):116-128. [36] ELEFTHERIADIS T, PISSAS G, CRESPO M, et al. A role for human renal tubular epithelial cells indirect allo-recognition by CD4+T-cells and the effect of ischemia-reperfusion. Int J Mol Sci. 2021;22(4):1733. [37] FAZEKAS B, GRIFFIN MD. Mesenchymal stromal cell-based therapies for acute kidney injury: progress in the last decade. Kidney Int . 2020;97(6):1130-1140. [38] SHEN B, LIU J, ZHANG F, et al. CCR2 positive exosome released by mesenchymal stem cells suppresses macrophage functions and alleviates ischemia/reperfusion-induced renal injury. Stem Cells Int. 2016; 2016:1240301. [39] RAGNI E, BANFI F, BARILANI M, et al. Extracellular vesicle-shuttled mRNA in mesenchymal stem cell communication. Stem Cells. 2017;35(4):1093-1105. [40] ZHAO L, HU C, HAN F, et al. Regenerative abilities of mesenchymal stem cells via acting as an ideal vehicle for subcellular component delivery in acute kidney injury. J Cell Mol Med. 2020;24(9):4882-4891. [41] YUAN X, LI D, CHEN X, et al. Extracellular vesicles from human-induced pluripotent stem cell-derived mesenchymal stromal cells (hiPSC-MSCs) protect against renal ischemia/reperfusion injury via delivering specificity protein (SP1) and transcriptional activating of sphingosine kinase 1 and inhibiting necroptosis. Cell Death Dis. 2017;8(12):3200. [42] THÉRY C, WITWER KW, AIKAWA E, et al. Minimal information for studies of extracellular vesicles 2018 (MISEV2018): a position statement of the International Society for Extracellular Vesicles and update of the MISEV2014 guidelines. J Extracell Vesicles. 2018;7(1):1535750. [43] REIS LA, BORGES FT, SIMÕES MJ, et al. Bone marrow-derived mesenchymal stem cells repaired but did not prevent gentamicin-induced acute kidney injury through paracrine effects in rats. PLoS One. 2012;7(9):e44092. [44] 李清茹,张琳琪,陈旭,等.间充质干细胞来源细胞外囊泡治疗和修复急慢性肾损伤[J].中国组织工程研究,2022,26(31): 5069-5075. [45] SUN X, MENG H, WAN W, et al. Application potential of stem/progenitor cell-derived extracellular vesicles in renal diseases.Stem Cell Res Ther. 2019;10(1):8. [46] LIU C, WANG J, HU J, et al. Extracellular vesicles for acute kidney injury in preclinical rodent models: a meta-analysis. Stem Cell Res Ther. 2020;11(1):11. [47] CHEN TS, ARSLAN F, YIN Y, et al. Enabling a robust scalable manufacturing process for therapeutic exosomes through oncogenic immortalization of human ESC‐derived MSCs. J Transl Med. 2011;9:47. [48] GREENING DW, GOPAL SK, XU R, et al. Exosomes and their roles in immune regulation and cancer. Semin Cell Dev Biol. 2015;40:72-81. [49] 徐同成,李霞,王文亮,等.分类变量Meta分析中偏倚的检测—Egger法和Begg法[J].循证医学,2009,9(3):181-184. |
| [1] | Wang Yanjin, Zhou Yingjie, Chai Xubin, Zhuo Hanjie. Meta-analysis of the efficacy and safety of 3D printed porous titanium alloy fusion cage in anterior cervical discectomy and fusion [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(9): 1434-1440. |
| [2] | Jiang Xiaocheng, Shi Lu, Wang Yinbin, Li Qiujiang, Xi Chuangzhen, Ma Zefeng, Cai Lijun. Systematical evaluation of bone fusion rate after interbody fusion in patients with osteoporosis and lumbar degenerative disease treated with teriparatide [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(9): 1427-1433. |
| [3] | Li Wenjie, You Aijia, Zhou Junli, Fang Sujuan, Li Chun. Effects of different dressings in the treatment of burn wounds: a network meta-analysis [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1141-1148. |
| [4] | Chen Guanting, Zhang Linqi, Li Qingru. Meta-analysis of the value of exosomal miRNA for the diagnosis of chronic kidney disease [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(6): 970-976. |
| [5] | Yan Le, Zhang Huiping, Dai Lintong. Mesenchymal stem cells for allergic rhinitis: a meta-analysis based on animal experiments [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(6): 977-984. |
| [6] | Feng Liang, Gong Shuhui, Huo Hongfeng. Effect of short-foot training on foot and ankle function in patients with flat feet: Meta-analysis and systematic review [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(5): 799-804. |
| [7] | Pan Weimin, Wang Bing, Han Yabing, Li Ting, Song Jiaqi, Qin Huasheng, Liu Yang. Effects of blood flow restriction training on muscle strength, muscle mass and physical performance in older adults: a Meta-analysis [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(5): 805-812. |
| [8] | Yu Jiaan, Liu Xinwei, Lian Hongyu, Liu Kexin, Li Zitao. Medial open-wedge tibial osteotomy versus lateral closed-wedge tibial osteotomy for unicompartmental knee osteoarthritis: a meta-analysis [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(4): 632-639. |
| [9] | Xu Yangyang, He Peiliang, Meng Qingqi, Li Siming. A meta-analysis of the effects of continuous adductor canal block and continuous femoral nerve block on early activity after knee arthroplasty [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(4): 640-645. |
| [10] | Chai Hao, Yang Deyong, Zhang Lei, Shu Li. 3D printing personalized osteotomy guide technology versus conventional total knee arthroplasty on the accuracy of lower limb force alignment: a meta-analysis [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(4): 646-654. |
| [11] | You Aijia, Li Wenjie, Zhou Junli, Li Chun. Systematic evaluation of six dressings on wound safety following total hip and knee arthroplasty [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(3): 486-492. |
| [12] | Cai Chuang, Huang Yonghui, Cao Xingbing, Sun Haitao, Guan Shihao, Huang Wenkang, Han Bo. Astrocyte-derived extracellular vesicles promote the differentiation of rat neural stem cells into neurons [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(24): 3845-3851. |
| [13] | Zhu Junjie, Wang Haihuang, Li Jianmin, Zhang Yuxin, Liu Junjie. Meta-analysis of the association between methylenetetrahydrofolate reductase gene polymorphisms and hemorrhagic stroke [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(23): 3763-3772. |
| [14] | Ma Maoxiao, Cui Guofeng, Zhang Xue, Zhang Hong, Liu Youwen, Yue Chen. Complications after spinal fusion in patients with metabolic syndrome: a systematic review and meta-analysis [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(22): 3602-3608. |
| [15] | Yin Yikun, Wang Jialin, Sun Junzhi. Therapeutic effect of different-frequency repetitive transcranial magnetic stimulations on post-stroke cognitive impairment: a Meta-analysis [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(20): 3274-3280. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||