Chinese Journal of Tissue Engineering Research ›› 2023, Vol. 27 ›› Issue (34): 5477-5482.doi: 10.12307/2023.554
Previous Articles Next Articles
An injectable dextran/gelatin composite hydrogel loaded with exosomes for repair of rat intervertebral disc degeneration
Pan Yujun, Shi Changjiang, Cao Sheng, Mu Huaizhao, Zhang Yilong, Wang Kaihua
- Affiliated Hospital of Chengde Medical College, Chengde 067000, Hebei Province, China
-
Received:2022-09-06Accepted:2022-10-19Online:2023-12-08Published:2023-04-20 -
Contact:Zhang Yilong, Chief physician, MD, Master’s supervisor, Affiliated Hospital of Chengde Medical College, Chengde 067000, Hebei Province, China -
About author:Pan Yujun, Master, Attending physician, Affiliated Hospital of Chengde Medical College, Chengde 067000, Hebei Province, China -
Supported by:Chengde Science and Technology Research and Development Program, No. 201801A050 (to PYJ)
CLC Number:
Cite this article
Pan Yujun, Shi Changjiang, Cao Sheng, Mu Huaizhao, Zhang Yilong, Wang Kaihua. An injectable dextran/gelatin composite hydrogel loaded with exosomes for repair of rat intervertebral disc degeneration[J]. Chinese Journal of Tissue Engineering Research, 2023, 27(34): 5477-5482.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
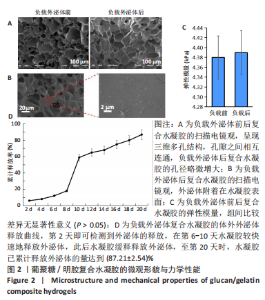
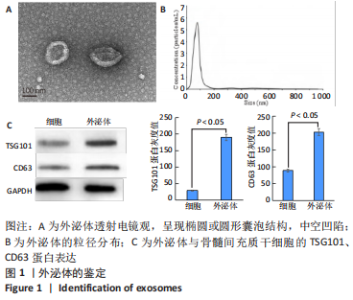
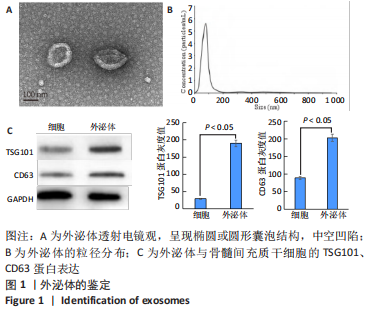
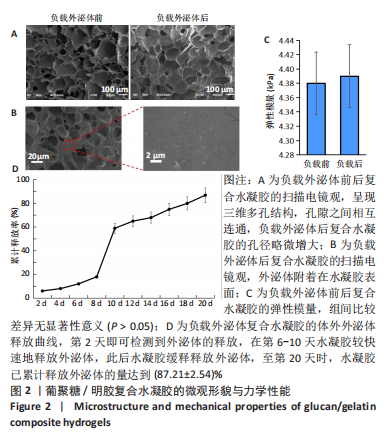
2.2 复合水凝胶的微观形貌、力学性能与体外药物缓释性能 扫描电镜下可见葡聚糖/明胶复合水凝胶呈现三维多孔结构,孔隙之间相互连通,孔径为50-200 μm,负载外泌体后复合水凝胶的孔径略微增大,见图2A,B;负载外泌体前后复合水凝胶的弹性模量分别为(4.38±0.56),(4.41±0.23) kPa,二者比较差异无显著性意义(P > 0.05),见图2C;水凝胶浸泡于PBS中后,第2天即可检测到外泌体的释放,在第6-10天水凝胶较快速地释放外泌体,外泌体累计释放量达到(59.36±9.87)%,此后水凝胶缓释释放外泌体,至第20天时,水凝胶累计释放外泌体的量达到(87.21±2.54)%,见图2D。"
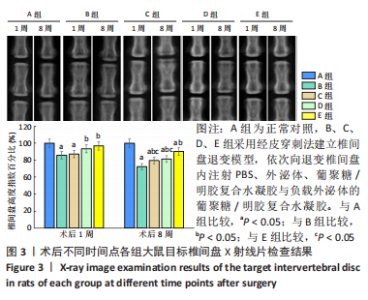
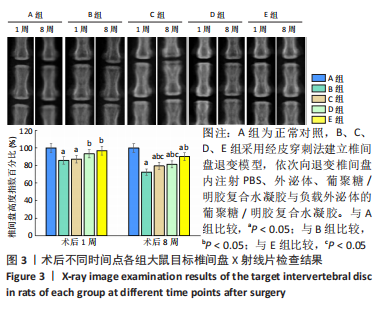
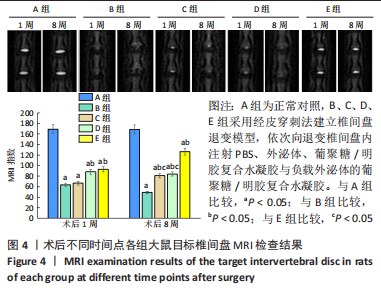
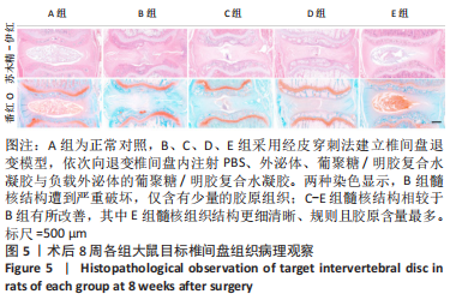
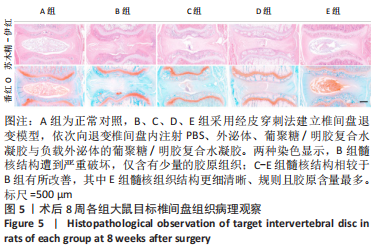
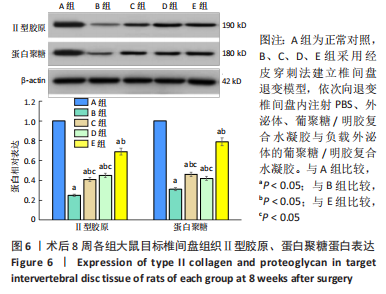
苏木精-伊红染色显示,A组中细胞和细胞外基质组织良好;B组中髓核结构被破坏,纤维环层片状结构紊乱,退变位点被纤维软骨组织和不同细胞形态的细胞所占据,损伤部位存在大量的瘢痕与纤维结缔组织,椎间盘结构无法辨认;C-E组损伤部位不同程度存在瘢痕和纤维结缔组织,部分椎间盘结构无法辨认,其中E组细胞和细胞外基质结构较C、D组清晰、规则。 番红O染色显示,A组髓核组织完整,含有丰富的胶原组织;B组髓核结构遭到严重破坏,仅见少量的胶原组织;C-E组髓核组织结构破坏程度均轻于B组,胶原含量不同程度地增加,其中以E组髓核组织结构更细清晰、规则且胶原含量最多。 椎间盘组织学评分结果显示,A-E组目标椎间盘组织学评分依次为(4.13±0.15),(13.02±0.14),(9.65±0.12),(10.21±0.18),(7.08±0.31)分,B-E组评分均高于A组(P < 0.05),C-E组评分均低于B组(P < 0.05),E组评分低于C、D组(P < 0.05)。 2.3.4 椎间盘组织Western blot检测 检测结果显示,与A组比较,B-E组Ⅱ型胶原、蛋白聚糖蛋白表达均显著减少(P < 0.05);与B组比较,C-E组Ⅱ型胶原、蛋白聚糖蛋白表达均显著增加(P < 0.05);与C、D组比较,E组Ⅱ型胶原、蛋白聚糖蛋白表达均显著增加(P < 0.05),见图6。"
| [1] WANG Y, CHE M, XIN J, et al. The role of IL-1β and TNF-α in intervertebral disc degeneration. Biomed Pharmacother. 2020;131:110660. [2] KIRNAZ S, CAPADONA C, WONG T, et al. Fundamentals of Intervertebral Disc Degeneration. World Neurosurg. 2022;157:264-273. [3] CHAO-YANG G, PENG C, HAI-HONG Z. Roles of NLRP3 inflammasome in intervertebral disc degeneration. Osteoarthritis Cartilage. 2021;29(6):793-801. [4] KOS N, GRADISNIK L, VELNAR T. A Brief Review of the Degenerative Intervertebral Disc Disease. Med Arch. 2019;73(6):421-424. [5] XU P, GUAN J, CHEN Y, et al. Stiffness of photocrosslinkable gelatin hydrogel influences nucleus pulposus cell propertiesin vitro. J Cell Mol Med. 2021;25(2):880-891. [6] CHEN W, CHEN H, ZHENG D, et al. Gene-Hydrogel Microenvironment Regulates Extracellular Matrix Metabolism Balance in Nucleus Pulposus. Adv Sci (Weinh). 2019;7(1):1902099. [7] LI M, LI R, YANG S, et al. Exosomes Derived from Bone Marrow Mesenchymal Stem Cells Prevent Acidic pH-Induced Damage in Human Nucleus Pulposus Cells. Med Sci Monit. 2020;26:e922928. [8] HE L, HE T, XING J, et al. Bone marrow mesenchymal stem cell-derived exosomes protect cartilage damage and relieve knee osteoarthritis pain in a rat model of osteoarthritis. Stem Cell Res Ther. 2020;11(1):276. [9] 甘翼搏,李培,周强,等.不同浓度的葡聚糖/明胶复合水凝胶支架性能表征及其对体外构建组织工程髓核的影响[J].第三军医大学学报,2015,37(8):707-712 [10] LUO L, JIAN X, SUN H, et al. Cartilage endplate stem cells inhibit intervertebral disc degeneration by releasing exosomes to nucleus pulposus cells to activate Akt/autophagy. Stem Cells. 2021;39(4):467-481. [11] WANG Y, WU Y, DENG M, et al. Establishment of a Rabbit Intervertebral Disc Degeneration Model by Percutaneous Posterolateral Puncturing of Lumbar Discs Under Local Anesthesia. World Neurosurg. 2021;154: e830-e837. [12] BAI JY, ZHANG YZ, FAN Q, et al. Reactive Oxygen Species-Scavenging Scaffold with Rapamycin for Treatment of Intervertebral Disk Degeneration. Adv Healthc Mater. 2020; 9(3):e1901186. [13] BOUHSINA N, DECANTE C, HARDEL JB, et al. Correlation between magnetic resonance, X-ray imaging alterations and histological changes in an ovine model of age-related disc degeneration. Eur Cell Mater. 2021;42:166-178. [14] ZHOU J, WU Y, ZHANG X, et al. Enzyme Catalyzed Hydrogel as Versatile Bioadhesive for Tissue Wound Hemostasis, Bonding, and Continuous Repair. Biomacromolecules. 2021; 22(4):1346-1356. [15] ZHANG W, BAO B, JIANG F, et al. Promoting Oral Mucosal Wound Healing with a Hydrogel Adhesive Based on a Phototriggered S-Nitrosylation Coupling Reaction. Adv Mater. 2021; 33(48):e2105667. [16] ZHANG FX, LIU P, DING W, et al. Injectable Mussel-Inspired highly adhesive hydrogel with exosomes for endogenous cell recruitment and cartilage defect regeneration. Biomaterials. 2021;278:121169. [17] LIN J, WANG Z, HUANG J, et al. Microenvironment-Protected Exosome-Hydrogel for Facilitating Endometrial Regeneration, Fertility Restoration, and Live Birth of Offspring. Small. 2021;17(11):e2007235. [18] HU J, LU Y, CAI L, et al. Functional compressive mechanics and tissue biocompatibility of an injectable SF/PU hydrogel for nucleus pulposus replacement. Sci Rep. 2017;7(1):2347. [19] CHRISTIANI T, MYS K, DYER K, et al. Using embedded alginate microparticles to tune the properties of in situ forming poly(N-isopropylacrylamide)-graft-chondroitin sulfate bioadhesive hydrogels for replacement and repair of the nucleus pulposus of the intervertebral disc. JOR Spine. 2021;4(3):e1161. [20] YU L, SUN ZJ, TAN QC, et al. Thermosensitive injectable decellularized nucleus pulposus hydrogel as an ideal biomaterial for nucleus pulposus regeneration. J Biomater Appl. 2020; 35(2):182-192. [21] ZHU Y, TAN J, ZHU H, et al. Development of kartogenin-conjugated chitosan-hyaluronic acid hydrogel for nucleus pulposus regeneration. Biomater Sci. 2017;5(4):784-791. [22] GENG X, MO X, FAN L, et al. Hierarchically designed injectable hydrogel from oxidized dextran, amino gelatin and 4-arm poly(ethylene glycol)-acrylate for tissue engineering application. J Mater Chem. 2012;22(48):25130-25139. [23] UNAL S, ARSLAN S, KARADEMIR YILMAZ B, et al.Glioblastoma cell adhesion properties through bacterial cellulose nanocrystals in polycaprolactone/gelatin electrospun nanofibers. Carbohydr Polym. 2020;233:115820. [24] CAZZANELLI P, WUERTZ-KOZAK K. MicroRNAs in Intervertebral Disc Degeneration, Apoptosis, Inflammation, and Mechanobiology. Int J Mol Sci. 2020;21(10):3601. [25] ZHANG Y, HAN S, KONG M, et al. Single-cell RNA-seq analysis identifies unique chondrocyte subsets and reveals involvement of ferroptosis in human intervertebral disc degeneration. Osteoarthritis Cartilage. 2021;29(9):1324-1334. [26] SHIM EK, LEE JS, KIM DE, et al. Autogenous Mesenchymal Stem Cells from the Vertebral Body Enhance Intervertebral Disc Regeneration via Paracrine Interaction: An in Vitro Pilot Study. Cell Transplant. 2016;25(10):1819-1832. [27] Bhujel B, Shin HE, Choi DJ, et al. Mesenchymal Stem Cell-Derived Exosomes and Intervertebral Disc Regeneration: Review.Int J Mol Sci. 2022;23(13):7306. [28] FOO JB, LOOI QH, CHONG PP, et al. Comparing the therapeutic potential of stem cells and their secretory products in regenerative medicine. Stem Cells Int. 2021;2021:2616807. [29] ONO M, KOSAKA N, TOMINAGA N, et al. Exosomes from bone marrow mesenchymal stem cells contain a micro RNA that promotes dormancy in metastatic breast cancer cells. Science signaling. 2014;7(332):ra63. [30] LIAO Z, LUO R, LI G, et al. Exosomes from mesenchymal stem cells modulate endoplasmic reticulum stress to protect against nucleus pulposus cell death and ameliorate intervertebral disc degeneration in vivo. Theranostics. 2019;9(14):4084-4100. [31] HU Y, TAO R, WANG L, et al. Exosomes Derived from Bone Mesenchymal Stem Cells Alleviate Compression-Induced Nucleus Pulposus Cell Apoptosis by Inhibiting Oxidative Stress. Oxid Med Cell Longev. 2021;2021:2310025. [32] XIAO Q, ZHAO Z, TENG Y, et al. BMSC-Derived Exosomes Alleviate Intervertebral Disc Degeneration by Modulating AKT/mTOR-Mediated Autophagy of Nucleus Pulposus Cells. Stem Cells Int. 2022;2022:9896444. [33] ZHU L, SHI Y, LIU L, et al. Mesenchymal stem cells-derived exosomes ameliorate nucleus pulposus cells apoptosis via delivering miR-142-3p: therapeutic potential for intervertebral disc degenerative diseases. Cell Cycle. 2020;19(14):1727-1739. [34] QIAN J, WANG X, SU G, et al. Platelet-rich plasma-derived exosomes attenuate intervertebral disc degeneration by promoting NLRP3 autophagic degradation in macrophages. Int Immunopharmacol. 2022;110:108962. |
| [1] | Nong Fuxiang, Jiang Zhixiong, Li Yinghao, Xu Wencong, Shi Zhilan, Luo Hui, Zhang Qinglang, Zhong Shuang, Tang Meiwen. Bone cement augmented proximal femoral nail antirotation for type A3.3 intertrochanteric femoral fracturalysis [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(在线): 1-10. |
| [2] | Cao Sheng, Kong Lingwei, Xu Kun, Sun Zhijie. Correlation of cervical sagittal force line parameters with degenerative segment and Pfirrmann classification in patients with cervical intervertebral disc degeneration [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(9): 1319-1324. |
| [3] | Pan Zhongjie, Qin Zhihong, Zheng Tiejun, Ding Xiaofei, Liao Shijie. Targeting of non-coding RNAs in the pathogenesis of the osteonecrosis of the femoral head [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(9): 1441-1447. |
| [4] | Lian Shilin, Zhang Yan, Jiang Qiang, Zhang Hanshuo, Li Tusheng, Ding Yu. Interventional effects of whole blood and platelet-rich plasma with different preparation methods on nucleus pulposus cells [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(8): 1199-1204. |
| [5] | Liu Xiaolin, Mu Xinyue, Ma Ziyu, Liu Shutai, Wang Wenlong, Han Xiaoqian, Dong Zhiheng. Effect of hydrogel-loaded simvastatin microspheres on osteoblast proliferation and differentiation [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 998-1003. |
| [6] | Li Cheng, Zheng Guoshuang, Kuai Xiandong, Yu Weiting. Alginate scaffold in articular cartilage repair [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1080-1088. |
| [7] | Liu Wentao, Feng Xingchao, Yang Yi, Bai Shengbin. Effect of M2 macrophage-derived exosomes on osteogenic differentiation of bone marrow mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(6): 840-845. |
| [8] | Li Qicheng, Deng Jin, Fu Xiaoyang, Han Na. Effects of bone marrow mesenchymal stem cells-derived exosomes on hypoxia-treated myoblasts [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(6): 853-859. |
| [9] | Wang Min, Yin Xiushan, Wang Yingxi, Zhang Yan, Zhao Long, Xia Shuyue. Inhalation of bone marrow mesenchymal stem cells-derived exosomes alleviates inflammatory injury in chronic obstructive pulmonary disease [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(6): 827-834. |
| [10] | Zhang Houjun, Deng Bowen, Jiang Shengyuan, Zhao Yi, Ren Jingpei, Xu Lin, Mu Xiaohong. Proteomic analysis of cerebrospinal fluid exosomes derived from cerebral palsy children [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(6): 903-908. |
| [11] | Gao Ting, Ma Xiaohong, Li Xiaorong. Extraction and identification of exosomes from three different sources of ovarian granulosa cells [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(6): 860-865. |
| [12] | Yuan Bo, Xie Lide, Fu Xiumei. Schwann cell-derived exosomes promote the repair and regeneration of injured peripheral nerves [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(6): 935-940. |
| [13] | Xu Qijing, Yang Yichun, Lei Wei, Yang Ying, Yu Jiang, Xia Tingting, Zhang Meng, Zhang Tao, Zhang Qian. Advances and problems in cell-free treatment of diabetic skin chronic wounds [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(6): 962-969. |
| [14] | Chen Guanting, Zhang Linqi, Li Qingru. Meta-analysis of the value of exosomal miRNA for the diagnosis of chronic kidney disease [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(6): 970-976. |
| [15] | Xiong Zhilin, Sun Hong, Liu Miao, Zhuang Yong. Roles of ferroptosis in intervertebral disc degeneration and osteoarthritis [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(36): 5884-5890. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||