Chinese Journal of Tissue Engineering Research ›› 2024, Vol. 28 ›› Issue (2): 272-279.doi: 10.12307/2023.475
Previous Articles Next Articles
MiRNA-122 contributes to the effect of exercise on non-alcoholic fatty liver
Guo Xiangying, Peng Zifu, He Yimin, Fang Hongbo, Jiang Ning
- Tianjin Key Laboratory of Sports Physiology and Sports Medicine, Tianjin University of Sport, Tianjin 301617, China
-
Received:2022-07-05Accepted:2022-08-08Online:2024-01-18Published:2023-06-30 -
Contact:Jiang Ning, PhD, Associate chief physician, Tianjin Key Laboratory of Sports Physiology and Sports Medicine, Tianjin University of Sport, Tianjin 301617, China -
About author:Guo Xiangying, Master candidate, Tianjin Key Laboratory of Sports Physiology and Sports Medicine, Tianjin University of Sport, Tianjin 301617, China -
Supported by:the National Natural Science Foundation of China, No. 31370021 (to JN)
CLC Number:
Cite this article
Guo Xiangying, Peng Zifu, He Yimin, Fang Hongbo, Jiang Ning. MiRNA-122 contributes to the effect of exercise on non-alcoholic fatty liver[J]. Chinese Journal of Tissue Engineering Research, 2024, 28(2): 272-279.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
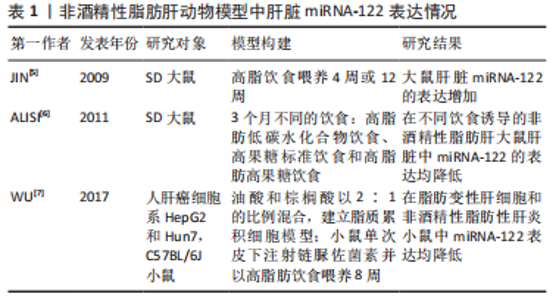
2.1 miRNA-122与NAFLD相关致病因素 2.1.1 miRNA-122在NAFLD中表达失调 miRNA-122是成人肝脏中含量最丰富的miRNA,分别占成年小鼠和人类整个肝脏miRNA的70%和52%,在其他细胞类型中的表达可以忽略不计。因此,它被认为是一种肝细胞特异性miRNA[3]。NAFLD是以弥漫性肝细胞大泡性脂肪变为主要临床特征的病理综合征,肝细胞中脂质的过度积累是NAFLD肝脂肪变性的第一个可识别阶段。这些聚集的脂质以微泡或大泡的形式可能引起肝细胞损伤,导致NAFLD发展到晚期,即以炎症和肝纤维化为特征的非酒精性脂肪性肝炎。非酒精性脂肪性肝炎患者进一步恶化,可发展为肝硬化和/或肝癌。通过使用肝细胞特异性Dicer敲除小鼠研究miRNA在肝脏中的广泛功能,SEKINE等[4]发现肝细胞特异性Dicer1敲除小鼠肝细胞中的miRNA加工被阻断,导致肝细胞中所有miRNA丢失;并且这些小鼠逐渐出现了肝脂肪变性、炎症和肝癌,这与miRNA-122敲除小鼠的研究结果相似。这表明miRNA-122在维持肝脏正常功能上发挥重要作用。然而,在NAFLD相关的研究中,miRNA-122的表达是失调的。 (1)肝脏miRNA-122表达与NAFLD:在饮食诱导的NAFLD动物模型中,JIN等[5]研究表明,在高脂饮食诱导的单纯性脂肪肝和脂肪性肝炎大鼠肝脏中,miRNA-122表达均增加。但ALISI等[6]研究表明,在不同饮食诱导的NAFLD大鼠肝脏中miRNA-122的表达显著下调。在游离脂肪酸诱导的脂肪变性肝细胞和链脲佐菌素和高脂饮食诱导的非酒精性脂肪性肝炎小鼠中miRNA-122表达也显著下调[7],这可能是因为模型构建的差异导致研究结果不同,见表1。CHEUNG等[8]研究表明,在非酒精性脂肪性肝炎患者肝脏中miRNA-122的表达显著降低。但也有研究报道,在单纯性脂肪变性和脂肪性肝炎患者肝脏中,miRNA-122相对表达量均升高[9]。MIYAAKI等[10]指出,轻度脂肪变性患者的肝脏miRNA-122水平明显低于重度脂肪变性,轻度纤维化患者的肝脏miRNA-122水平显著高于重度纤维化患者,这表明miRNA-122的表达水平可能随着疾病的发展进程发生改变。同样,PIROLA等[11]对单纯性脂肪变性患者和非酒精性脂肪性肝炎患者进行研究发现,与对照组相比,单纯性脂肪变性患者肝脏miRNA-122表达升高,与单纯性脂肪变性患者相比,非酒精性脂肪性肝炎患者肝脏miRNA-122表达下调,相关研究见表2。这进一步证明,miRNA-122可能在脂肪肝疾病的不同阶段表现出不同的表达水平,在NAFLD疾病早期,肝脏可能通过上调miRNA-122的表达来防御肝脏脂肪变性,而在非酒精性脂肪性肝炎中,miRNA-122的下调可能与NAFLD的进一步恶化有关。"

(2)循环miRNA-122水平在NAFLD 中增加:血清miRNA-122水平可能代表NAFLD疾病诊断新的非侵入性生物标志物。CERMELLI等[12]研究表明,与健康对照组相比,单纯性脂肪变性患者的血清miRNA-122水平较高;与单纯性脂肪变性患者相比,非酒精性脂肪性肝炎患者的血清miRNA-122水平进一步增加。他们还观察到,miRNA-122与丙氨酸氨基转移酶和天冬氨酸氨基转移酶呈强正相关,与总胆固醇水平和低密度脂蛋白之间也呈正相关。YAMADA等[13]研究也表明,在诊断为NAFLD的患者中,血清miRNA-122水平较高,血清miRNA-122水平与肝脏脂肪变性的严重程度相关。同样,陈轶等[14]报道,单纯性脂肪肝和非酒精性脂肪性肝炎患者血清中miRNA-122的表达均明显升高。SALVOZA等[15]研究表明,在NAFLD患者中miRNA-122的血清水平显著升高,miRNA-122与低密度脂蛋白-胆固醇和三酰甘油水平呈正相关,与小叶炎症和肝细胞气球形成之间存在正相关[16], 并且ROC曲线分析结果表明,血清miRNA-122的预测价值高于丙氨酸氨基转移酶。从单纯性脂肪变性到非酒精性脂肪性肝炎,血清miRNA-122水平与组织病理学特征之间呈正相关[17-18]。这些结果提示miRNA-122可能作为诊断NAFLD和评估组织学病变严重性的一种新的生物学标记物[19],同时有望成为一个潜在的治疗靶点。此外,BRANDT等[20]研究也表明,循环miRNA-122高表达与肥胖儿童NAFLD发病率之间存在明显的相关性,肥胖儿童的miRNA-122水平也可能是儿科NAFLD诊断的潜在生物标志物。上述多项研究均表明,NAFLD患者血清miRNA-122表达水平明显升高,且miRNA-122水平随着NAFLD患者脂肪肝的严重程度增加而显著升高[21],相关研究见表3。"

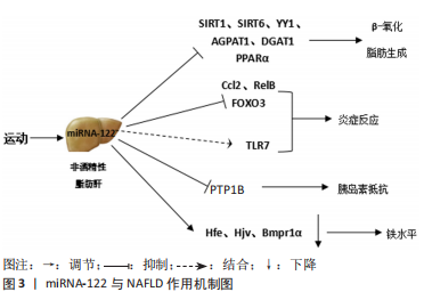
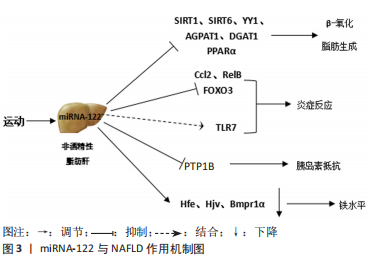
2.1.2 miRNA-122与NAFLD脂质代谢 NAFLD脂肪变性是由于脂肪组织中脂肪酸的输送增加,伴随着脂质降解和新生脂质合成之间的不平衡,导致肝脏中脂质的过度积累而引发的。在肝脏中,26%的三酰甘油来自脂肪从头合成[22]。在NAFLD患者中,脂肪生成途径增加,脂肪生成率是健康个体的3倍[23]。此外,在患有NAFLD的患者和动物模型肝脏中,参与脂肪酸和胆固醇合成的固醇调节元件结合蛋白1/2 (sterol regulatory element binding protein 1/2,SREBP1/2)的表达/活性持续增加[24],参与脂肪酸β-氧化的主转录因子过氧化物酶体增殖物激活受体α(peroxisome proliferators-activated receptors α,PPARα)的表达/激活显著减少。这些研究表明,在NAFLD中脂质代谢发生了紊乱,脂质代谢紊乱导致肝脏中大量脂质沉积,从而导致肝细胞脂肪变性。因此,改善脂质代谢对改善NAFLD具有重要意义。 刘丽雅[25]研究表明,与对照组相比,高脂饮食诱导的NAFLD大鼠肝脏中miRNA-122表达明显降低,脂肪酸合酶蛋白和mRNA表达显著升高,肝组织有明显的脂肪性炎性改变,这提示miRNA-122可能在肝脏脂类代谢过程中发挥重要的调控作用,并参与NAFLD的病理生理学过程。NAFLD的致病因素包括肝细胞内脂肪酸的过度积累,这可归因于肝内三酰甘油从头合成升高和β-氧化还原下降。 miRNA-122是脂质代谢的关键,肝脏miRNA-122的表达与脂肪生成基因的表达呈负相关,miRNA-122可能通过抑制脂肪生成基因的表达在肝脏中发挥抗脂肪生成的作用。在使用HEG2细胞的体外研究中,miRNA-122的沉默导致3-羟基-3-甲基戊二酰辅酶A还原酶(3-hydroxy-3- methylglutaryl CoA reductase,HMGCR)和脂肪酸合成酶(fatty acid synthase,FAS)、SREBP-1c的过表达,而miRNA-122的表达可导致这些基因的表达水平显著降低[8]。同样,MIYAAKI等[10]发现,miRNA-122敲除导致脂质代谢基因(如FAS、HMGCR、SREBP)上调。miRNA-122 肝脏特异性敲除小鼠脂蛋白合成增强但分泌减少,肝脏三酰甘油含量增加[26]。WU等[7]研究发现,在游离脂肪酸诱导前用miRNA-122模拟物转染肝细胞,可通过降低阴阳1 mRNA的稳定性,上调法尼类X受体和小异二聚体伴侣信号传导抑制体外脂滴形成和三酰甘油积聚。miRNA-122还可通过靶向三酰甘油生物合成途径中的1-酰基-sn-甘油-3-磷酸酰基转移酶-β-1(1-acyl-sn-glycerol-3-phosphate acyltransferase-beta-1,AGPAT1)和二酰基甘油-酰基转移酶-1(diacylglycerol-acyltranferase-1,DGAT1)来减少三酰甘油的积累[27]。此外,在高脂饮食喂养的小鼠中,miRNA-122拮抗剂通过减少β-氧化而恶化脂肪肝,在小鼠肝脏中,随着miRNA-122水平的降低,脂肪滴和总三酰甘油含量增加,β-氧化和能量消耗减少[28]。在游离脂肪酸诱导剂处理的小鼠中抑制miRNA-122可增加食物摄入和三酰甘油合成途径的基因表达,增加肝脏和肌肉组织中的三酰甘油水平,降低β-羟基丁酸酯的表达,抑制β-氧化。这些结果表明,miRNA-122的表达对NAFLD具有保护作用,miRNA-122可能通过调节三酰甘油代谢和胆固醇生物合成途径中的脂质生成基因及脂肪β-氧化,在脂质稳态中发挥重要作用。 然而,也有研究表明,miRNA-122的表达促进NAFLD脂代谢紊乱。正常小鼠中miRNA-122抑制导致肝脏脂肪酸β-氧化增加,miRNA-122靶点沉默信息调节因子2相关酶6(sirtuin6,SIRT6)参与脂肪合成相关基因表达和脂肪酸β-氧化[29]。高脂饮食喂养小鼠肝脏和棕榈酸干预HepG2细胞中,miRNA-122的表达增加,FAS、乙酰辅酶A羧化酶(acetyl CoA carboxylase,ACC)的mRNA及蛋白表达增加,肉毒碱棕榈酰转移酶1A(carnitine palmitoyl transferase 1A,CPT1A)的mRNA及蛋白表达减少[30]。ESAU等[31]在正常小鼠中抑制miRNA-122表达可降低血浆总胆固醇水平,增加肝脏脂肪酸氧化,降低肝脏脂肪酸和总胆固醇合成速率;同样,在饮食诱导的肥胖小鼠模型中抑制miRNA-122后,肝脏和血浆总胆固醇、三酰甘油水平降低,肝脏脂肪酸氧化增加,肝脏脂肪变性显著改善,同时伴有几种脂质合成相关基因的减少。在游离脂肪酸处理的HepG2或Huh-7细胞中敲除miRNA-122后,可以有效抑制过度脂质沉积和三酰甘油分泌[32]。SENDI等[33]报道,miRNA-122抑制可通过下调几种在胆固醇生物合成中起作用的基因来降低血清胆固醇水平,在miRNA-122抑制后,胆固醇生物合成相关基因HMGCR表达降低,胆固醇合成胆汁酸的限速酶CYP7A1表达增加。这些研究表明,抑制miRNA-122可以降低循环中的三酰甘油和总胆固醇水平,并且miRNA-122对脂质生物合成的影响是通过调节脂质生物合成相关基因表达介导的。此外,LONG等[32]报道,抑制miRNA-122可通过上调沉默信息调节因子1和激活AMPK通路抑制脂肪生成,进而改善NAFLD脂肪变性和脂肪生成,保护肝细胞免受NAFLD等脂质代谢紊乱的损害。 综上所述, miRNA-122参与NAFLD脂质代谢过程,其作用机制可能是通过调控FAS、ACC、SREBP-1c、HMGCR等脂质生物合成相关基因表达和脂肪酸β-氧化间接参与脂质代谢过程。 2.1.3 miRNA-122与NAFLD炎症反应 肝细胞内脂质的积累可触发慢性炎症反应,炎症反应是NAFLD发生和发展的关键因素[34]。研究表明,促炎基因的重要核因子κB(nuclear factorκB,NF-κB)及其产物的过度表达可能参与NAFLD肝脏炎症的发生和发展。在NAFLD动物模型和患者的肝脏中,促炎基因的重要核因子κB显著升高[35]。HAJIGHASEM等[36]研究表明,NAFLD可能与促炎症递质如白细胞介素6、肿瘤坏死因子α和白细胞介素1β的大量释放和上调有关。在NAFLD大鼠肝脏中,炎症递质肿瘤坏死因子α和肿瘤坏死因子β含量增加,肝小叶有轻度至中度的炎性细胞浸润[37]。这表明,减轻NAFLD炎症因子表达,改善机体炎症反应可能有助于减缓NAFLD的进程。 研究表明,miRNA-122的基因缺失严重影响肝脏脂质代谢,导致肝脏微结构退化和炎症反应,并且随着小鼠年龄的增长,这些病理改变发展为非酒精性脂肪性肝炎和肝纤维化[2]。在肝脏类器官中的研究也表明,miRNA-122的抑制导致炎症、坏死、脂肪变性和纤维化,并且miRNA-122抑制剂感染后, 白细胞介素6、肿瘤坏死因子α表达显著增加[33]。此外,CLARKE等[38]研究结果表明,肝细胞中miRNA-122的降低与NAFLD相关。由此可知,miRNA-122对肝脏炎症反应也是至关重要的。HSU等[2]研究表明, miRNA-122的缺失导致小鼠肝脂肪变性和肝脏炎症反应的发展与促炎细胞因子白细胞介素6、肿瘤坏死因子α的过度分泌有关,并且miRNA-122可通过抑制其靶点Ccl2的表达增强肝脏的抗炎作用。miRNA-122 敲除小鼠肝脏中促炎性趋化因子Ccl2、Ccl4、Ccl20、Cxcl2、Cxcl10和RelB的表达增加[39]。而miRNA-122 敲除小鼠中miRNA-122水平的恢复可通过抑制趋化因子Ccl2逆转肝脏炎症反应,该靶点显示可在肝内招募CD11bhiGr1+炎症细胞和促纤维化Krüppel样因子6(Krüppel like factor 6,KLF6)[40]。这些结果表明,miRNA-122可能通过靶向炎症相关通路的蛋白(如促炎性趋化因子CCL2、RelB)间接调控炎症因子的表达,进而参与NAFLD炎症反应。然而,miRNA-122是否还有其他参与炎症反应的靶向因子?这还需要进一步研究。除了通过靶向mRNA上的碱基互补配对位点进行翻译抑制或直接对mRNA进行降解,miRNAs还可以直接作为某些RNA受体的生理配体。WANG等[41]报道,受损小鼠肝细胞释放的循环miRNA-122在巨噬细胞中可结合Toll样受体7并激活炎症反应,导致巨噬细胞M1极化和炎性细胞因子的分泌,如白细胞介素6、肿瘤坏死因子α。 2.1.4 miRNA-122与NAFLD 其他致病因素 铁代谢受损与NAFLD炎症有关。研究表明,肝脏miRNA-122的表达对于预防血浆和肝脏缺铁、低铁利用率引起的造血损伤以及脾脏髓外红细胞生成至关重要[42]。CASTOLDI等[42]报道miRNA-122参与维持系统性铁稳态,有效且特异性地抑制miRNA-122可导致野生型小鼠血浆和肝脏铁水平降低,造血受损,髓外红细胞生成增加。此外,在肝组织和小鼠原代肝细胞中,miRNA-122缺失直接增加了控制全身铁水平的hepcidin转录激活剂的mRNA表达,如血色素沉着症基因(hemochromatosis,Hfe)、铁调素调节蛋白(hemojuvelin,Hjv)、骨形态发生蛋白受体1A型(bone morphogenetic protein receptor type 1A,Bmpr1α)[42]。此外,NAFLD的进展还与胰岛素抵抗有关。体内和体外研究表明,在高脂饮食诱导的胰岛素抵抗小鼠中,肝脏miRNA-122水平降低,miRNA-122可以在肝脏中调控蛋白酪氨酸磷酸酶1B(protein tyrosine phosphatase 1B,PTP1B)相关的胰岛素抵抗[43],见图3。"

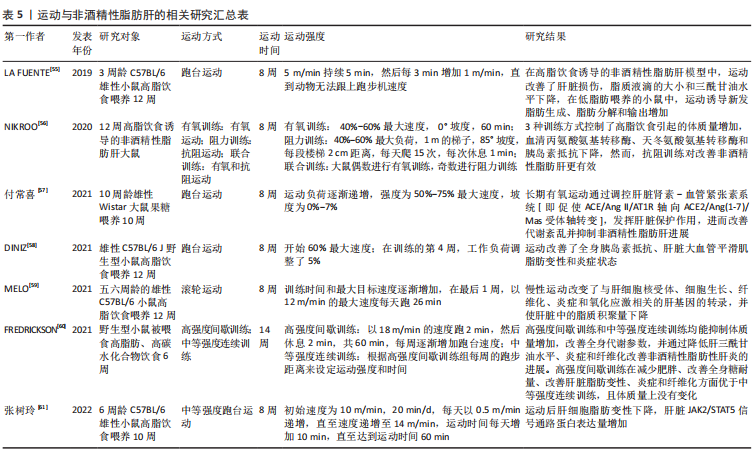
2.2 运动对NAFLD及miRNA-122表达的影响 2.2.1 运动对NAFLD的影响 NAFLD被认为是一种复杂的疾病,是许多环境和遗传因素相互作用的结果。生活方式的改变,特别是营养过剩以及缺乏运动,是引起NAFLD的主要环境因素。脂肪酸积累、炎症和胰岛素抵抗是NAFLD发生和发展的主要病理事件[44]。近年来,随着肥胖、2型糖尿病和代谢综合征流行的增加,NAFLD的发病率迅速上升。根据现有证据,除运动和饮食干预外,尚未开发出治疗NAFLD的特定药物[45]。通过定期锻炼,早期NAFLD可以得到很好的控制,经常性运动被认为是当前克服与脂肪肝相关的肝脏病理学最有效的策略[46]。 HASHIDA等[47]对有氧运动和抗阻运动进行系统评价发现,对于有氧运动,中位有效方案为4.8代谢当量,40 min/次,每周3次,持续12周;对于抗阻运动,中位有效方案为3.5 代谢当量,45 min/次,3次/周,持续12周;两种运动方式的持续时间、频率或运动量之间没有显著差异,这表明中等强度的有氧或抗阻运动可改善肝脏脂肪变性。KATSAGONI等[48]的一项荟萃分析也表明,有氧运动和抗阻运动均对肝脏参数有改善作用,且两种运动方式间没有显差异,但与中低运动量(120 min≤每周运动时长< 180 min)的中等强度运动训练相比,高强度间歇训练可能在减少肝脏脂肪变性方面产生更大的益处,而中高运动量(每周运动时长≥180 min)的中等强度持续训练比高强度间歇训练更为有益。 MEDRANO等[49]的荟萃分析表明,运动干预显著降低儿童和青少年的肝脏脂肪含量和 NAFLD 患病率,有氧和抗阻运动在高强度或中等至高强度运动下,运动时间≥60 min /次、每周3次的最低频率可显著降低肝脏脂肪含量。这表明,每周至少进行120 min的中等强度运动才可对NAFLD产生积极的改善作用。ZHOU等[50]分析了4种运动对NAFLD患者血脂和肝酶的干预效果,结果表明,有氧和抗阻运动结合的干预效果优于高强度间歇训练,高强度间歇训练优于抗阻运动,抗阻运动优于有氧运动。 BABU等[51]对316例NAFLD患者进行荟萃分析发现,运动对缓解NAFLD有积极影响,但不会显著减轻体质量;单独有氧运动显著降低肝内脂质、丙氨酸氨基转移酶和天冬氨酸氨基转移酶;阻力训练显著降低总胆固醇和三酰甘油;有氧和抗阻运动结合显著降低了肝内脂质。XIONG等[52]报道,有氧运动可以显著降低 NAFLD 患者的体质量指数、丙氨酸氨基转移酶、天冬氨酸氨基转移酶、低密度脂蛋白、三酰甘油、胆固醇水平,增加 NAFLD 患者的高密度脂蛋白水平;抗阻运动可以显著降低 NAFLD 患者的天冬氨酸氨基转移酶、胆固醇水平;高强度间歇训练可显著降低 NAFLD 患者的丙氨酸氨基转移酶。同样,另一项样本量更大的荟萃分析表明,有氧运动和阻力运动能显著改善肝脏酶、血脂和血糖参数,尤其是内脏脂肪组织[53]。高强度间歇训练在肝脏脂肪方面的改善与中等强度持续训练相当[54]。这表明,不同的运动方式对NAFLD均有改善作用,但对NAFLD相关参数的干预效果不同,相关研究见表4。"

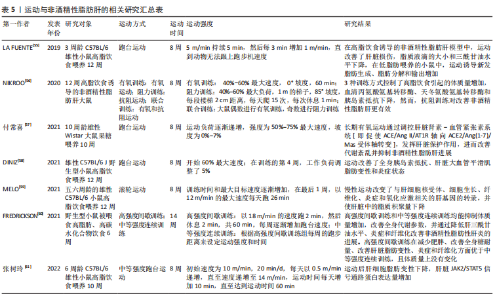
因此,NAFLD患者也可根据自身的疾病情况选择运动方式,如胆固醇或天冬氨酸氨基转移酶水平较高的患者可以进行有氧或抗阻运动,丙氨酸氨基转移酶水平高的患者采用有氧或高强度间歇训练。 虽然运动训练能有效改善NAFLD,但运动产生有益作用的潜在机制尚不清楚。一些针对NAFLD患者和动物模型的研究表明,运动可能通过降低肝细胞脂质含量,提高胰岛素敏感性,减轻肝脏炎症反应和氧化应激水平来防治该疾病的发生发展,相关研究见表5。在高脂饮食诱导的NAFLD小鼠中,8周跑台运动改善了肝脂滴大小,降低了三酰甘油水平,并减少了肝脏的损伤[55]。在高脂饮食诱导的NAFLD大鼠中,8周有氧、抗阻和联合训练均可控制体质量,改善胰岛素抵抗,降低血清丙氨酸氨基转移酶、门冬氨酸氨基转移酶水平,且抗阻训练似乎对改善NAFLD更有效[56]。在果糖喂养的Wistar大鼠中,8周中等强度跑台运动可通过调控肝脏肾素-血管紧张素系统发挥肝脏保护作用,进而改善糖脂代谢紊乱、肝脏损伤和炎症反应,并抑制NAFLD进展[57]。同样,DINIZ等[58]研究报道,8周中等强度跑台运动略微改善了肥胖小鼠的大泡性脂肪变性及全身胰岛素抵抗,降低了肿瘤坏死因子α、白细胞介素10、单核细胞趋化因子1和白细胞介素6的表达,并减轻了肝脏炎症反应。MELO等[59]研究表明,8周跑台运动使脂滴缩小和脂质累积标记物减少,改善了高脂饮食诱导的肝纤维化,减少了肝脏炎症和氧化应激。这表明有氧运动和抗阻运动均可改善NAFLD,抗阻运动在改善NAFLD病理上更显著。中等强度的有氧运动通过调节糖脂代谢紊乱、减轻NAFLD炎症反应和胰岛素抵抗来改善NAFLD。此外,FREDRICKSON等[60]认为,高强度运动在改善非酒精性脂肪性肝炎疾病进展方面比中等强度运动更有效,包括显著改善肝脏炎症和脂质生物合成。张树玲等[61]实验表明,运动对NAFLD肝内脂质沉积及肝脏脂肪变性程度的改善作用可能与JAK2/STAT5信号通路有关。"

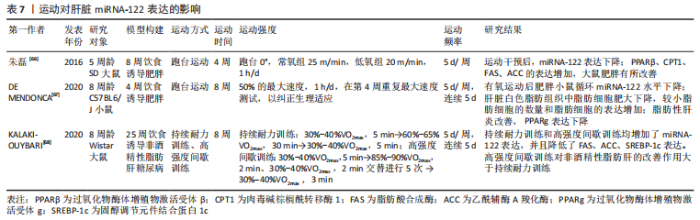
综上所述,运动是减轻肝脂肪变性、降低肝脏炎症反应和胰岛素抵抗、防治NAFLD的可行策略。不同的运动方式和运动强度均可改善NAFLD病理进程。然而,运动改善NAFLD的分子机制尚未完全了解,还需要更多的研究来探讨其中的作用机制。 2.2.2 运动对循环miRNA-122水平的影响 miRNA-122在维持肝脏正常功能上发挥重要作用。研究表明,适量的体育运动似乎能够调节miRNA-122的表达。BRANDSTETTER[62]对15名男性进行铁人三项等力竭运动,随后测定赛前、赛后以及赛后1 d和7 d运动员的全血microRNA表达谱,结果发现,成熟miRNA-122水平在比赛后直接下调了2倍以上,甚至在比赛结束后1,7 d仍低于赛前水平。CUI等[63]对18名健康的年轻男性进行短跑间歇骑车训练后,其循环miRNA-122水平下降,并且miRNA-122水平与峰值功率比相关。然而,在马拉松小鼠模型的研究中,3周自主跑轮运动后,经长期选定的马拉松小鼠循环miRNA-122水平高于未经选择的对照组[64]。另一项研究也表明,9 d的跑步机训练增加了小鼠循环miRNA-122水平,并通过靶向内皮细胞中AGPAT1表达增强脂肪酸的利用,促进血管生成[65]。这些结果表明,短期、顺时的运动降低了循环miRNA-122水平,而长期的运动训练增加了循环miRNA-122水平,相关研究见表6。"

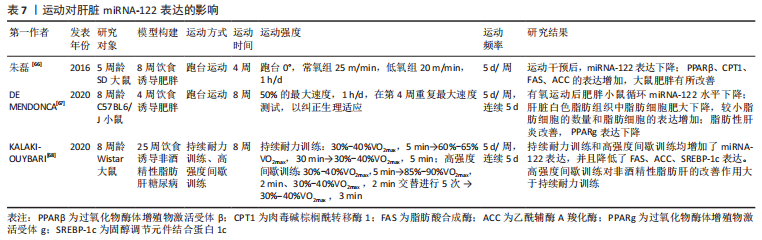
2.2.3 运动对肝脏miRNA-122表达的影响 运动似乎能够通过调节miRNA-122来防治NAFLD的发生和发展,相关研究表明,运动训练可作为诱导miRNA-122改善NAFLD的非药理学干预手段,见表7。朱磊[66]的研究表明,耐力训练和低氧耐力训练下调了肥胖大鼠肝脏 miRNA-122 表达,提高了脂代谢相关因子过氧化物酶体增殖物激活受体β(peroxisome proliferators-activated receptors β,PPARβ)、肉毒碱棕榈酰转移酶1(carnitine palmitoyl transferase 1,CPT1)、FAS、ACC 的表达,减轻了大鼠肥胖。然而,De Mendonca等[67]的研究表明,8周有氧训练降低高脂饮食诱导的肥胖小鼠血清miRNA-122水平,增加肝组织miRNA-122表达,降低了肝脏过氧化物酶体增殖物激活受体γ(peroxisome proliferators-activated receptors γ,PPARγ)和炎症标志物白细胞介素6、肿瘤坏死因子α的表达,减弱肥胖诱导的脂肪细胞肥大,改善肥胖小鼠的胰岛素抵抗和肝脂肪变性。KALAKI-JOUYBARI等[68]对高脂饮食诱导的糖尿病NAFLD大鼠的研究表明,高强度间歇训练和持续耐力训练均增加NAFLD糖尿病大鼠肝脏miRNA-122的表达,降低脂代谢相关因子FAS、ACC、SREBP-1的表达,减轻肝脏脂肪变性,这可能是因为研究模型所处的病理阶段不同,在肥胖的初期,miRNA-122可能作为一种代偿因子,在肝脏中表达增加,而运动抑制了肥胖的发生,进而恢复miRNA-122的表达。但随着肥胖的加重,miRNA-122不足以代偿,且表达减少,加重疾病的发展,这时运动可能通过上调miRNA-122的表达来改善肥胖和NAFLD。此外,也可能是选用的实验对象不同或运动干预方式和运动干预强度不同所致。"
| [1] FARZANEGI P, DANA A, EBRAHIMPOOR Z, et al. Mechanisms of beneficial effects of exercise training on non-alcoholic fatty liver disease (NAFLD): Roles of oxidative stress and inflammation. Eur J Sport Sci. 2019;19(7):994-1003. [2] HSU SH, WANG B, KOTA J, et al. Essential metabolic, anti-inflammatory, and anti-tumorigenic functions of miR-122 in liver. J Clin Invest. 2012;122(8):2871-2883. [3] GIRARD M, JACQUEMIN E, MUNNICH A, et al. miR-122, a paradigm for the role of microRNAs in the liver. J Hepatol. 2008;48(4):648-656. [4] SEKINE S, OGAWA R, ITO R, et al. Disruption of Dicer1 induces dysregulated fetal gene expression and promotes hepatocarcinogenesis. Gastroenterology. 2009;136(7): 2304-2315.e1-4. [5] JIN X, YE YF, CHEN SH, et al. MicroRNA expression pattern in different stages of nonalcoholic fatty liver disease. Dig Liver Dis. 2009;41(4): 289-297. [6] ALISI A, DA SACCO L, BRUSCALUPI G, et al. Mirnome analysis reveals novel molecular determinants in the pathogenesis of diet-induced nonalcoholic fatty liver disease. Lab Invest. 2011;91(2):283-293. [7] WU GY, RUI C, CHEN JQ, et al. MicroRNA-122 Inhibits Lipid Droplet Formation and Hepatic Triglyceride Accumulation via Yin Yang 1. Cell Physiol Biochem. 2017;44(4): 1651-1664. [8] CHEUNG O, PURI P, EICKEN C, et al. Nonalcoholic steatohepatitis is associated with altered hepatic MicroRNA expression. Hepatology. 2008;48(6):1810-1820. [9] 黄慧. MicroRNA122在非酒精性脂肪肝病的表达及临床意义[D].南充:川北医学院,2013. [10] MIYAAKI H, ICHIKAWA T, KAMO Y, et al. Significance of serum and hepatic microRNA-122 levels in patients with non-alcoholic fatty liver disease. Liver Int. 2014; 34(7):e302-e307. [11] PIROLA CJ, FERNÁNDEZ GIANOTTI T, CASTAÑO GO, et al. Circulating microRNA signature in non-alcoholic fatty liver disease: from serum non-coding RNAs to liver histology and disease pathogenesis. Gut. 2015;64(5):800-812. [12] CERMELLI S, RUGGIERI A, MARRERO JA, et al. Circulating microRNAs in patients with chronic hepatitis C and non-alcoholic fatty liver disease. PLoS one. 2011;6(8):e23937. [13] YAMADA H, SUZUKI K, ICHINO N, et al. Associations between circulating microRNAs (miR-21, miR-34a, miR-122 and miR-451) and non-alcoholic fatty liver. Clin Chim Acta. 2013;424:99-103. [14] 陈轶,陈益耀,蔡蔓妮,等.miR-122在非酒精性脂肪肝病患者血清中的表达及其临床意义[J].武警医学,2015,26(1):11-12,15. [15] SALVOZA NC, KLINZING DC, GOPEZ-CERVANTES J, et al. Association of Circulating Serum miR-34a and miR-122 with Dyslipidemia among Patients with Non-Alcoholic Fatty Liver Disease. PLoS One. 2016;11(4):e0153497. [16] AUGUET T, ARAGONÈS G, BERLANGA A, et al. miR33a/miR33b* and miR122 as Possible Contributors to Hepatic Lipid Metabolism in Obese Women with Nonalcoholic Fatty Liver Disease. Int J Mol Sci. 2016;17(10):1620. [17] JAMPOKA K, MUANGPAISARN P, KHONGNOMNAN K, et al. Serum miR-29a and miR-122 as Potential Biomarkers for Non-Alcoholic Fatty Liver Disease (NAFLD). Microrna. 2018;7(3):215-222. [18] DAN Y, ZHANG T, LOU G, et al. Plasma miR-17, miR-20a, miR-20b and miR-122 as potential biomarkers for diagnosis of NAFLD in type 2 diabetes mellitus patients. Life Sci. 2018;208:201-207. [19] LIU CH, AMPUERO J, GIL-GÓMEZ A, et al. miRNAs in patients with non-alcoholic fatty liver disease: A systematic review and meta-analysis. J Hepatol. 2018;69(6):1335-1348. [20] BRANDT S, ROOS J, INZAGHI E, et al. Circulating levels of miR-122 and nonalcoholic fatty liver disease in pre-pubertal obese children. Pediatr Obes. 2018;13(3):175-182. [21] 张广玉,钤培国,孙晓娜,等.消脂护肝汤对非酒精性脂肪性肝炎患者肝脏的影响[J].中医学报,2019,34(8):1735-1739. [22] DONNELLY KL, SMITH CI, SCHWARZENBERG SJ, et al. Sources of fatty acids stored in liver and secreted via lipoproteins in patients with nonalcoholic fatty liver disease. J Clin Invest. 2005;115(5):1343-1351. [23] LAMBERT JE, RAMOS-ROMAN M, BROWNING JD, et al. Increased De Novo Lipogenesis Is a Distinct Characteristic of Individuals With Nonalcoholic Fatty Liver Disease. Gastroenterology. 2014;146(3):726-735. [24] JU UI, JEONG DW, SEO J, et al. Neddylation of sterol regulatory element-binding protein 1c is a potential therapeutic target for nonalcoholic fatty liver treatment. Cell Death Dis. 2020;11(4):283. [25] 刘丽雅.复方茵陈脂肝颗粒调控miR-122及其下游通路治疗非酒精性脂肪肝病的作用机制研究[D].福州:福建中医药大学,2014. [26] SU Q, KUMAR V, SUD N, et al. MicroRNAs in the pathogenesis and treatment of progressive liver injury in NAFLD and liver fibrosis. Adv Drug Deliv Rev. 2018;129:54-63. [27] CHAI C, RIVKIN M, BERKOVITS L, et al. Metabolic Circuit Involving Free Fatty Acids, microRNA 122, and Triglyceride Synthesis in Liver and Muscle Tissues. Gastroenterology. 2017;153(5):1404-1415. [28] CHAI C, COX B, YAISH D, et al. Agonist of RORA Attenuates Nonalcoholic Fatty Liver Progression in Mice via Up-regulation of MicroRNA 122. Gastroenterology. 2020;159(3):999-1014.e9. [29] ELHANATI S, BEN-HAMO R, KANFI Y, et al. Reciprocal Regulation between SIRT6 and miR-122 Controls Liver Metabolism and Predicts Hepatocarcinoma Prognosis. Cell Rep. 2016;14(2):234-242. [30] 杨立英.水飞蓟宾通过调控miR-122的表达改善非酒精性脂肪肝的体内外研究[D].石家庄:河北医科大学,2019. [31] ESAU C, DAVIS S, MURRAY SF, et al. miR-122 regulation of lipid metabolism revealed by in vivo antisense targeting. Cell Metab. 2006;3(2):87-98. [32] LONG JK, DAI W, ZHENG YW, et al. miR-122 promotes hepatic lipogenesis via inhibiting the LKB1/AMPK pathway by targeting Sirt1 in non-alcoholic fatty liver disease. Mol Med. 2019;25(1):26. [33] SENDI H, MEAD I, WAN M, et al. miR-122 inhibition in a human liver organoid model leads to liver inflammation, necrosis, steatofibrosis and dysregulated insulin signaling. PLoS One. 2018;13(7):e0200847. [34] SCHUSTER S, CABRERA D, ARRESE M, et al. Triggering and resolution of inflammation in NASH. Nat Rev Gastroenterol Hepatol. 2018;15(6):349-364. [35] TAN DY, SHI HY, LI CP, et al. Effect of nuclear factor-κB and angiotensin Ⅱ receptor type 1 on the pathogenesis of rat non-alcoholic fatty liver disease. World J Gastroenterol. 2015;21(19):5877-5883. [36] HAJIGHASEM A, FARZANEGI P, MAZAHERI Z. Effects of combined therapy with resveratrol, continuous and interval exercises on apoptosis, oxidative stress, and inflammatory biomarkers in the liver of old rats with non-alcoholic fatty liver disease. Arch Physiol Biochem. 2019;125(2):142-149. [37] EL-DIN SH, SABRA AN, HAMMAM OA, et al. Pharmacological and antioxidant actions of garlic and.or onion in non-alcoholic fatty liver disease (NAFLD) in rats. J Egypt Soc Parasitol. 2014;44(2):295-308. [38] CLARKE JD, SHARAPOVA T, LAKE AD, et al. Circulating microRNA 122 in the methionine and choline-deficient mouse model of non-alcoholic steatohepatitis. J Appl Toxicol. 2014;34(6):726-732. [39] HSU KH, WEI CW, SU YR, et al. Upregulation of RelB in the miR-122 knockout mice contributes to increased levels of proinflammatory chemokines/cytokines in the liver and macrophages. Immunol Lett. 2020;226:22-30. [40] TSAI WC, HSU SD, HSU CS, et al. MicroRNA-122 plays a critical role in liver homeostasis and hepatocarcinogenesis. J Clin Invest. 2012;122(8):2884-2897. [41] WANG Y, LIANG H, JIN F, et al. Injured liver-released miRNA-122 elicits acute pulmonary inflammation via activating alveolar macrophage TLR7 signaling pathway. Proc Natl Acad Sci U S A. 2019;116(13):6162-6171. [42] CASTOLDI M, VUJIC SPASIC M, ALTAMURA S, et al. The liver-specific microRNA miR-122 controls systemic iron homeostasis in mice. J Clin Invest. 2011;121(4):1386-1396. [43] YANG YM, SEO SY, KIM TH, et al. Decrease of microRNA-122 causes hepatic insulin resistance by inducing protein tyrosine phosphatase 1B, which is reversed by licorice flavonoid. Hepatology. 2012;56(6):2209-2220. [44] BUZZETTI E, PINZANI M, TSOCHATZIS EA. The multiple-hit pathogenesis of non-alcoholic fatty liver disease (NAFLD). Metabolism. 2016;65(8):1038-1048. [45] CHALASANI N, YOUNOSSI Z, LAVINE JE, et al. The diagnosis and management of nonalcoholic fatty liver disease: Practice guidance from the American Association for the Study of Liver Diseases. Hepatology. 2018;67(1):328-357. [46] STEVANOVI J, BELEZA J, COXITO P, et al. Physical exercise and liver “fitness”: Role of mitochondrial function and epigenetics-related mechanisms in non-alcoholic fatty liver disease. Mol Metab. 2020;32:1-14. [47] HASHIDA R, KAWAGUCHI T, BEKKI M, et al. Aerobic vs. resistance exercise in non-alcoholic fatty liver disease: A systematic review. J Hepatol. 2017;66(1):142-152. [48] KATSAGONI CN, GEORGOULIS M, PAPATHEODORIDIS GV, et al. Effects of lifestyle interventions on clinical characteristics of patients with non-alcoholic fatty liver disease: A meta-analysis. Metabolism. 2017;68:119-132. [49] MEDRANO M, CADENAS-SANCHEZ C, ALVAREZ-BUENO C, et al. Evidence-Based Exercise Recommendations to Reduce Hepatic Fat Content in Youth- a Systematic Review and Meta-Analysis. Prog Cardiovasc Dis. 2018;61(2):222-231. [50] ZHOU BJ, HUANG G, WANG W, et al. Intervention effects of four exercise modalities on nonalcoholic fatty liver disease: a systematic review and Bayesian network meta-analysis. Eur Rev Med Pharmacol Sci. 2021;25(24):7687-7697. [51] BABU AF, CSADER S, LOK J, et al. Positive Effects of Exercise Intervention without Weight Loss and Dietary Changes in NAFLD-Related Clinical Parameters: A Systematic Review and Meta-Analysis. Nutrients. 2021;13(9):3135. [52] XIONG Y, PENG Q, CAO C, et al. Effect of Different Exercise Methods on Non-Alcoholic Fatty Liver Disease: A Meta-Analysis and Meta-Regression. Int J Environ Res Public Health. 2021;18(6):3242. [53] FU L, ZHANG W, AO Y, et al. Efficacy of aerobic and resistance exercises in improving visceral adipose in patients with non-alcoholic fatty liver: a meta-analysis of randomized controlled trials. Z Gastroenterol. 2022;60(11):1644-1658. [54] SABAG A, BARR L, ARMOUR M, et al. The Effect of High-intensity Interval Training vs Moderate-intensity Continuous Training on Liver Fat: A Systematic Review and Meta-Analysis. J Clin Endocrinol Metab. 2022;107(3):862-881. [55] LA FUENTE FP, QUEZADA L, SEPULVEDA C, et al. Exercise regulates lipid droplet dynamics in normal and fatty liver. Biochim Biophys Acta Mol Cell Biol Lipids. 2019; 1864(12):158519. [56] NIKROO H, HOSSEINI SRA, FATHI M, et al. The effect of aerobic, resistance, and combined training on PPAR-α, SIRT1 gene expression, and insulin resistance in high-fat diet-induced NAFLD male rats. Physiol Behav. 2020;227:113149. [57] 付常喜,陆阿明,孙一.有氧运动调控肝脏肾素-血管紧张素系统对大鼠非酒精性脂肪性肝病的作用与机制[J].山东体育学院学报,2021,37(4):75-85. [58] DINIZ TA, DE LIMA JUNIOR EA, TEIXEIRA AA, et al. Aerobic training improves NAFLD markers and insulin resistance through AMPK-PPAR-alpha signaling in obese mice. Life Sci. 2021;266:118868. [59] MELO L, BILICI M, HAGAR A, et al. The effect of endurance training on non-alcoholic fatty liver disease in mice. Physiol Rep. 2021;9(15):e14926. [60] FREDRICKSON G, BARROW F, DIETSCHE K, et al. Exercise of high intensity ameliorates hepatic inflammation and the progression of NASH. Mol Metab. 2021;53:101270. [61] 张树玲,李军汉,王佳倩,等.有氧运动干预非酒精性脂肪肝小鼠肝脏JAK2/STAT5信号通路的变化[J].中国组织工程研究,2022,26(17):2690-2695. [62] BRANDSTETTER S. Exhaustive exercise such as an Ironman triathlon alters MicroRNA expression pattern in whole blood. Wien: Universität Wien,2012. [63] CUI SF, LI W, NIU J, et al. Acute responses of circulating microRNAs to low-volume sprint interval cycling. Front Physiol. 2015;6:311. [64] OHDE D, BRENMOEHL J, WALZ C, et al. Comparative analysis of hepatic miRNA levels in male marathon mice reveals a link between obesity and endurance exercise capacities. J Comp Physiol B. 2016;186(8):1067-1078. [65] LOU J, WU J, FENG M, et al. Exercise promotes angiogenesis by enhancing endothelial cell fatty acid utilization via liver-derived extracellular vesicle miR-122-5p. J Sport Health Sci. 2022;11(4):495-508. [66] 朱磊.低氧训练诱导miR-27/PPARγ、miR-122/PPARβ调控肥胖大鼠肝脏脂代谢机理的研究[D].上海:上海体育学院,2016. [67] DE MENDONCA M, ROCHA KC, DE SOUSA E, et al. Aerobic exercise training regulates serum extracellular vesicle miRNAs linked to obesity to promote their beneficial effects in mice. Am J Physiol Endocrinol Metab. 2020;319(3):E579-E591. [68] KALAKI-JOUYBARI F, SHANAKI M, DELFAN M, et al. High-intensity interval training (HIIT) alleviated NAFLD feature via miR-122 induction in liver of high-fat high-fructose diet induced diabetic rats. Arch Physiol Biochem. 2020;126(3):242-249. |
| [1] | Wang Jingfeng, Wen Dengtai, Wang Shijie, Gao Yinghui. Atg-mediated autophagy, exercise and skeletal muscle aging [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(2): 295-301. |
| [2] | Long Yi, Yang Jiaming, Ye Hua, Zhong Yanbiao, Wang Maoyuan. Extracellular vesicles in sarcopenic obesity: roles and mechanisms [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(2): 315-320. |
| [3] | Sun Yuan, Wang Qingbo, Pi Yihua, Lu Chunmin, Xu Chuanyi, Zhang Yan. Effects of early and late aerobic exercise on right heart failure induced by monocrotaline in rats with pulmonary hypertension [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(2): 177-185. |
| [4] | Liu Baofang, Xu Bin, Chen Lei. Pueraria decoction in the treatment of osteoarthritis: network pharmacology analysis and animal model validation [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(2): 193-199. |
| [5] | Yin Linwei, Huang Xiarong, Qu Mengjian, Yang Lu, Wang Jinling, Jia Feiyang, Liao Yang, Zhou Jun. Effects of treadmill exercise on osteoporosis and wnt/beta-catenin signal pathway in aged rats [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(2): 231-236. |
| [6] | Nong Fuxiang, Jiang Zhixiong, Li Yinghao, Xu Wencong, Shi Zhilan, Luo Hui, Zhang Qinglang, Zhong Shuang, Tang Meiwen. Bone cement augmented proximal femoral nail antirotation for type A3.3 intertrochanteric femoral fracturalysis [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(在线): 1-10. |
| [7] | Huang Linke, Wei Linhua, Jiang Jie, Liu Qian, Chen Weiwei. Effects of estrogen combined with treadmill exercise on bone mass and articular cartilage in ovariectomized mice [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(8): 1166-1171. |
| [8] | Ruan Ling, Wang Guanghua, Wu Rongping, Jin Zhan, Lyu Zhenqing, Zhang Nan, Li Shoubang. Correlation between exercise intensity and lipid metabolism disorder and oxidative stress in a high-diet rat model [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(8): 1149-1155. |
| [9] | Zhang Yan, He Ruibo, Wang Qingbo, Pi Yihua, Lu Chunmin, Xu Chuanyi, Ma Gang, Peng Peng. Effects of aerobic exercises with different load volumes on inflammatory response and insulin signaling pathway of skeletal muscle in obese rats [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(8): 1237-1244. |
| [10] | Wu Dongzhe, Gao Xiaolin, Li Chuangtao, Wang Hao. Constructing the prediction model of maximal oxygen uptake by back-propagation neural network based on the cardiorespiratory optimal point [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(8): 1224-1231. |
| [11] | Wang Ji, Zhang Min, Yang Zhongya, Zhang Long. A review of physical activity intervention in type 2 diabetes mellitus with sarcopenia [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(8): 1272-1277. |
| [12] | Liang Jiaqi, Liu Hengxu, Yang Jinxin, Yang Yi, Deng Xuhui, Tan Mingjian, Luo Jiong. Health benefit relationship between exercise and intestinal bacteria [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(8): 1292-1299. |
| [13] | Wang Jinling, Huang Xiarong, Qu Mengjian, Huang Fujin, Yin Lingwei, Zhong Peirui, Liu Jin, Sun Guanghua, Liao Yang, Zhou Jun. Effects of exercise training on bone mass and bone microstructure in aged osteoporotic rats [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(5): 676-682. |
| [14] | Lu Huixiu, Cao Haiyu, Lou Dan, Li Jianying, Liu Hongyuan, Sun Jing. Imiquimod combined with photodynamic therapy for hypertrophic scars: immune response and prognosis [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(5): 690-694. |
| [15] | Li Yujiao, Su Kunxia. High-intensity endurance exercise influences browning of white adipose tissue in a mouse model of high-fat diet induced obesity [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(5): 707-713. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||

