[1] BUSCHAM TJ, EICHEL-VOGEL MA, STEYER AM, et al. Progressive axonopathy when oligodendrocytes lack the myelin protein CMTM5. Elife. 2022;11: e75523.
[2] YIN X, KIDD GJ, OHNO N, et al. Proteolipid protein-deficient myelin promotes axonal mitochondrial dysfunction via altered metabolic coupling. J Cell Biol. 2016;215(4):531-542.
[3] OSORIO-QUEREJETA I, SÁENZ-CUESTA M, MUÑOZ-CULLA M, et al. Models for Studying Myelination, Demyelination and Remyelination. Neuromolecular Med. 2017;19(2-3):181-192.
[4] 皮桐昊,刘瑞琦,贾佳佳,等.外泌体在药物递送系统中的研究进展[J].中国药剂学杂志,2023, 21(5):250-262.
[5] 居怡,SERAGAMANI HAMOOD ALI,施戈韬,等.外泌体作为药物递送载体的研究进展[J].药学进展,2023,47(11):804-816.
[6] KALAFATAKIS I, KARAGOGEOS D. Oligodendrocytes and Microglia: Key Players in Myelin Development, Damage and Repair. Biomolecules. 2021;11(7):1058.
[7] GARBERN JC, LEE RT. Heart regeneration: 20 years of progress and renewed optimism. Dev Cell. 2022;57(4):424-439.
[8] MITREČIĆ D, HRIBLJAN V, JAGEČIĆ D, et al. Regenerative Neurology and Regenerative Cardiology: Shared Hurdles and Achievements. Int J Mol Sci. 2022;23(2):855.
[9] DULAMEA AO. Role of Oligodendrocyte Dysfunction in Demyelination, Remyelination and Neurodegeneration in Multiple Sclerosis. Adv Exp Med Biol. 2017;958:91-127.
[10] 黄春满,李丽薇,黄勇彬,等.外泌体靶向修饰的研究进展[J].科学通报,2023,68(33):4532-4543.
[11] XIE F, HUANG Y, ZHAN Y, et al. Exosomes as drug delivery system in gastrointestinal cancer. Front Oncol. 2023;12:1101823.
[12] XIE S, SUN W, FU T, et al. Aptamer-Based Targeted Delivery of Functional Nucleic Acids. J Am Chem Soc. 2023;145(14):7677-7691.
[13] MILLÁN COTTO HA, PATHRIKAR TV, HAKIM B, et al. Cationic-motif-modified exosomes for mRNA delivery to retinal photoreceptors. J Mater Chem B. 2024;12(30):7384-7400.
[14] LI X, QU S, OUYANG Q, et al. A multifunctional composite nanoparticle with antibacterial activities, anti-inflammatory, and angiogenesis for diabetic wound healing. Int J Biol Macromol. 2024;260(Pt 2):129531.
[15] LI YJ, HE J, ZHANG QH, et al. Olig2-enriched exosomes: A novel therapeutic approach for cuprizone-induced demyelination. Neuroscience. 2024;555:41-51.
[16] JIA Z, LIU J, LI B, et al. Exosomes with FOXP3 from gene-modified dendritic cells ameliorate the development of EAE by regulating the balance of Th/Treg. Int J Med Sci. 2022;19(8): 1265-1274.
[17] TIMOFEEVA AM, PARAMONIK AP, SEDYKH SS, et al. Milk Exosomes: Next-Generation Agents for Delivery of Anticancer Drugs and Therapeutic Nucleic Acids. Int J Mol Sci. 2023;24(12):10194.
[18] 曾文,尹贝立,谭文松,等.体外诱导间充质干细胞向成纤维细胞分化的研究[J].华东理工大学学报(自然科学版),2024,50(3):383-390.
[19] 罗文彬,李若云,潘超凡,等.工程化外泌体修复组织损伤:应用潜力及优异的生物稳定性和靶向特异性[J].中国组织工程研究,2026, 30(1):204-217.
[20] SADEGHI S, TEHRANI FR, TAHMASEBI S, et al. Exosome engineering in cell therapy and drug delivery. Inflammopharmacology. 2023; 31(1):145-169.
[21] RAJPUT A, VARSHNEY A, BAJAJ R, et al. Exosomes as New Generation Vehicles for Drug Delivery: Biomedical Applications and Future Perspectives. Molecules. 2022;27(21):7289.
[22] 徐煌.神经干细胞来源囊泡联合姜黄素治疗缺血性脑卒中的安全性评估[D].大连:大连医科大学,2023.
[23] 高爽,任雪雷,韩翔宇,等.间充质干细胞来源的外泌体在心血管疾病中的研究现状[J].中国动脉硬化杂志,2022,30(11):1006-1012.
[24] 王建辉,吴东颖,郭小刚.干细胞外泌体在衰老和衰老相关疾病中的作用[J].中国生物化学与分子生物学报,2023,39(2):189-195.
[25] GURUNG S, PEROCHEAU D, TOURAMANIDOU L, et al. The exosome journey: from biogenesis to uptake and intracellular signalling. Cell Commun Signal. 2021;19(1):47.
[26] 范星宇,廖晓倩,王梓仪,等.NLRP3炎症小体介导的细胞焦亡在慢性心力衰竭中的机制及中医药研究进展[J].中国中药杂志,2024, 49(8):2106-2116.
[27] ZHANG S, SHANG D, SHI H, et al. Function of Astrocytes in Neuroprotection and Repair after Ischemic Stroke. Eur Neurol. 2021;84(6):426-434.
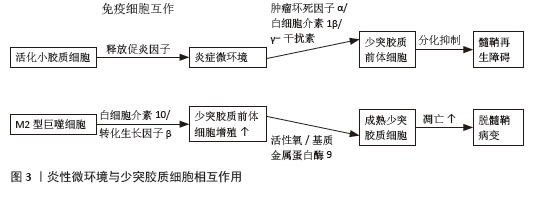
[28] 刘丽,李琦,杜欣珂,等.M2型小胶质细胞极化促进少突胶质祖细胞的分化:促进多发性硬化症髓鞘再生的有效途径[J].中国比较医学杂志,2022,32(2):126-132.
[29] KRYLOVA SV, FENG D. The Machinery of Exosomes: Biogenesis, Release, and Uptake. Int J Mol Sci. 2023;24(2):1337.
[30] 原平利,刘刚,马兰香,等.毛兰素通过抑制NLRP3炎症体介导的细胞焦亡减轻心肌缺血再灌注损伤[J].山西医科大学学报,2023, 54(9):1200-1207.
[31] ZHANG J, BULLER BA, ZHANG ZG, et al. Exosomes derived from bone marrow mesenchymal stromal cells promote remyelination and reduce neuroinflammation in the demyelinating central nervous system. Exp Neurol. 2022;347:113895.
[32] 傅馨莹,孙天松,朱丛旭,等.基于NLRP3/caspase-1/GSDMD通路研究黄精改善糖尿病大血管焦亡损伤的作用机制[J].中国中药杂志, 2023,48(24):6702-6710.
[33] LI C, QIN T, ZHAO J, et al. Bone Marrow Mesenchymal Stem Cell-Derived Exosome-Educated Macrophages Promote Functional Healing After Spinal Cord Injury. Front Cell Neurosci. 2021;15:725573.
[34] ZHANG C, PATHRIKAR TV, BABY HM, et al. Charge-Reversed Exosomes for Targeted Gene Delivery to Cartilage for Osteoarthritis Treatment. Small Methods. 2024;8(9):e2301443.
[35] GAO R, SONG SJ, TIAN MY, et al. Myelin debris phagocytosis in demyelinating disease. Glia. 2024;72(11):1934-1954.
[36] MARTIN NA, HYRLOV KH, ELKJAER ML, et al. Absence of miRNA-146a Differentially Alters Microglia Function and Proteome. Front Immunol. 2020;11:1110.
[37] SHENG X, ZHAO J, LI M, et al. Bone Marrow Mesenchymal Stem Cell-Derived Exosomes Accelerate Functional Recovery After Spinal Cord Injury by Promoting the Phagocytosis of Macrophages to Clean Myelin Debris. Front Cell Dev Biol. 2021;9:772205.
[38] SOCK E, WEGNER M. Using the lineage determinants Olig2 and Sox10 to explore transcriptional regulation of oligodendrocyte development. Dev Neurobiol. 2021;81(7):892-901.
[39] SCHMIDT AL, KREMP M, ARATAKE T, et al. The myelination-associated G protein-coupled receptor 37 is regulated by Zfp488, Nkx2.2, and Sox10 during oligodendrocyte differentiation. Glia. 2024;72(7):1304-1318.
[40] MECHA M, YANGUAS-CASÁS N, FELIÚ A, et al. Involvement of Wnt7a in the role of M2c microglia in neural stem cell oligodendrogenesis. J Neuroinflammation. 2020;17(1):88.
[41] FEDDER-SEMMES KN, APPEL B. The Akt-mTOR Pathway Drives Myelin Sheath Growth by Regulating Cap-Dependent Translation. J Neurosci. 2021;41(41):8532-8544.
[42] XIN H, LIU Z, BULLER B, et al. MiR-17-92 enriched exosomes derived from multipotent mesenchymal stromal cells enhance axon-myelin remodeling and motor electrophysiological recovery after stroke. J Cereb Blood Flow Metab. 2021;41(5):1131-1144.
[43] DAHL KD, ALMEIDA AR, HATHAWAY HA, et al. mTORC2 Loss in Oligodendrocyte Progenitor Cells Results in Regional Hypomyelination in the Central Nervous System. J Neurosci. 2023;43(4):540-558.
[44] QIN D, WANG C, LI D, et al. Exosomal miR-23a-3p derived from human umbilical cord mesenchymal stem cells promotes remyelination in central nervous system demyelinating diseases by targeting Tbr1/Wnt pathway. J Biol Chem. 2024;300(1):105487.
[45] HOU H, WANG Y, YANG L, et al. Exosomal miR-128-3p reversed fibrinogen-mediated inhibition of oligodendrocyte progenitor cell differentiation and remyelination after cerebral ischemia. CNS Neurosci Ther. 2023;29(5):1405-1422.
[46] WELSH JA, GOBERDHAN DCI, O’DRISCOLL L, et al. Minimal information for studies of extracellular vesicles (MISEV2023): From basic to advanced approaches. J Extracell Vesicles. 2024;13(2):e12404.
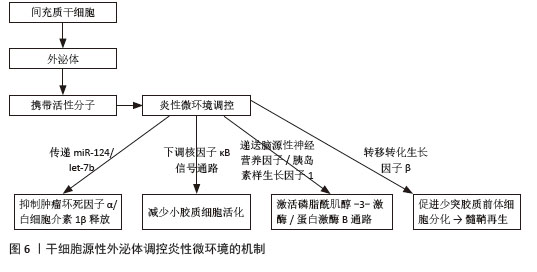
[47] 李佳林,张耀东,娄艳茹,等.间充质干细胞分泌组发挥作用的分子机制[J].中国组织工程研究,2025,29(7):1512-1522.
[48] 杨丽娜,杜欣珂,刘丽,等.基于“炎症-组织”稳态偶联实现多发性硬化症髓鞘修复的研究现状及展望[J].中国中药杂志,2023,48(1):5-12.
[49] ZHA Z, GAO YF, JI J, et al. Bu Shen Yi Sui Capsule Alleviates Neuroinflammation and Demyelination by Promoting Microglia toward M2 Polarization, Which Correlates with Changes in miR-124 and miR-155 in Experimental Autoimmune Encephalomyelitis. Oxid Med Cell Longev. 2021; 2021:5521503.
[50] LIU SS, ZHA Z, LI C, et al. The mechanism of exosomes of BMSCs modified with Bu Shen Yi Sui capsule in promoting remyelination via regulating miR-15b/Wnt signaling pathway-mediated differentiation of oligodendrocytes. J Ethnopharmacol. 2025;340:119283.
[51] JI J, SUN YQ, ZHA ZZ, et al. Bu Shen Yi Sui Capsules Promote Remyelination by Regulating MicroRNA-219 and MicroRNA-338 in Exosomes to Promote Oligodendrocyte Precursor Cell Differentiation. Evid Based Complement Alternat Med. 2022;2022:3341481.
[52] 刘念,董听明,王落芫,等.干细胞外泌体和生物材料辅助外泌体修复骨缺损[J].中国组织工程研究,2026,30(1):175-183.
[53] AUGUSTINE R, GEZEK M, NIKOLOPOULOS VK, et al. Stem Cells in Bone Tissue Engineering: Progress, Promises and Challenges. Stem Cell Rev Rep. 2024;20(7):1692-1731.
[54] 乔树雅,田田.干细胞外泌体用于缺血性疾病治疗的研究进展[J].药学进展,2023,47(11): 817-828.
[55] 孙庆,王海龙,米庆,等.外泌体在心力衰竭发生发展中的作用及临床价值[J].临床荟萃, 2024,39(3):259-263.
[56] 孙姝婷.干细胞外泌体的生物学机制与应用[J].张江科技评论,2024(4):108-110.
[57] LOZANO-VELASCO E, INÁCIO JM, SOUSA I, et al. miRNAs in Heart Development and Disease. Int J Mol Sci. 2024;25(3):1673.
[58] TIAN T, ZHANG HX, HE CP, et al. Surface functionalized exosomes as targeted drug delivery vehicles for cerebral ischemia therapy. Biomaterials. 2018;150:137-149.
[59] HE J, WANG Y, ZHAO ZH, et al. Exosome-specific loading Sox10 for the treatment of Cuprizone-induced demyelinating model. Biomed Pharmacother. 2024;171:116128.
[60] HUANG L, WU E, LIAO J, et al. Research Advances of Engineered Exosomes as Drug Delivery Carrier. ACS Omega. 2023;8(46):43374-43387.
[61] 李旻俊,彭泽琳,张良清.外泌体药物递送载体构建及治疗心血管疾病的研究进展[J].中华实用诊断与治疗杂志,2021,35(7):746-749.
[62] ROSZKOWSKI S. Therapeutic potential of mesenchymal stem cell-derived exosomes for regenerative medicine applications. Clin Exp Med. 2024;24(1):46.
[63] 王双敏,汪显耀,何志旭.工程化间充质干细胞来源外泌体在靶向递送抗肿瘤药物中的应用与问题[J].中国组织工程研究,2025,29(23): 4975-4983.
[64] SHAMS F, POURJABBAR B, HASHEMI N, et al. Current progress in engineered and nano-engineered mesenchymal stem cells for cancer: From mechanisms to therapy. Biomed Pharmacother. 2023;167:115505.
[65] GE L, WANG K, LIN H, et al. Engineered exosomes derived from miR-132-overexpresssing adipose stem cells promoted diabetic wound healing and skin reconstruction. Front Bioeng Biotechnol. 2023;11:1129538.
|