中国组织工程研究 ›› 2026, Vol. 30 ›› Issue (1): 153-162.doi: 10.12307/2025.572
• 干细胞综述 stem cell review • 上一篇 下一篇
牙髓干细胞及衍生产物在牙髓再生中的应用与进展
徐海超,罗丽花,潘乙怀
- 温州医科大学口腔医学院附属口腔医院,浙江省温州市 325000
-
收稿日期:2024-11-18接受日期:2025-01-17出版日期:2026-01-08发布日期:2025-07-02 -
通讯作者:潘乙怀,博士,教授,博士研究生导师,温州医科大学口腔医学院附属口腔医院,浙江省温州市 325000; 共同通讯作者:罗丽花,博士,副研究员,硕士研究生导师,温州医科大学口腔医学院附属口腔医院,浙江省温州市 325000 -
作者简介:徐海超,男,2002年生,浙江省台州市人,汉族,温州医科大学口腔专业在读学士,主要从事牙髓干细胞及其临床转化研究。 -
基金资助:浙江省基础公益研究计划项目(LGF21H140007),项目负责人:罗丽花
Application and progress of dental pulp stem cells and their derivatives in dental pulp regeneration
Xu Haichao, Luo Lihua, Pan Yihuai
- School and Hospital of Stomatology, Wenzhou Medical University, Wenzhou 325000, Zhejiang Province, China
-
Received:2024-11-18Accepted:2025-01-17Online:2026-01-08Published:2025-07-02 -
Contact:Pan Yihuai, PhD, Professor, Doctoral supervisor, School and Hospital of Stomatology, Wenzhou Medical University, Wenzhou 325000, Zhejiang Province, China; Co-corresponding author: Luo Lihua, PhD, Associate researcher, Master's supervisor, School and Hospital of Stomatology, Wenzhou Medical University, Wenzhou 325000, Zhejiang Province, China -
About author:Xu Haichao, School and Hospital of Stomatology, Wenzhou Medical University, Wenzhou 325000, Zhejiang Province, China -
Supported by:Zhejiang Provincial Basic Public Welfare Research Project, No. LGF21H140007 (to LLH)
摘要:
文题释义:
https://orcid.org/0009-0003-1693-3868 (徐海超)
牙髓再生:是指在移除炎症或坏死的牙髓组织后,利用特定的治疗方法包括干细胞、生物活性因子及生物支架材料等,促使新的牙髓组织生长,从而恢复牙齿的正常生理功能和维持原有的生命活力。牙髓再生是牙髓治疗和再生牙科学的一个重要分支,尤其适用于年轻患者的受损或发育不全的牙齿。
摘要
背景:牙髓干细胞是一类源自牙髓组织的牙源性间充质干细胞,具有良好的自我更新和多向分化潜力。近年来,牙髓干细胞及其衍生产物包括细胞外囊泡、条件培养液以及脱细胞基质等,在牙髓组织损伤修复再生中的应用研究取得了显著进展,显示出广阔的临床应用前景。
目的:系统综述牙髓干细胞及其衍生产物在牙髓组织工程中的研究成果和应用进展。
方法:检索PubMed数据库、中国生物医学文献数据库、中国知网,以“牙髓干细胞,细胞外囊泡,外泌体,凋亡小体,条件培养液,脱细胞基质,再生”为中文检索词,以“dental pulp stem cells,extracellular vesicles,exosomes,apoptotic bodies,conditioned medium,decellularized matrix,regeneration”为英文检索词进行检索,检索时限为2005年1月至2023年6月。根据文题和摘要对初检文献进行筛选,排除重复文献和与主题不相关的文献,最终纳入103篇与牙髓再生高度相关的文献进行综述分析。
结果与结论:牙髓干细胞及其衍生产物富含多种生物活性因子,能够有效促进成牙本质、成血管和成神经分化,在牙髓-牙本质复合体的形成过程中展现出巨大的应用潜力。然而,牙髓干细胞及其衍生产物在临床应用转化中仍面临挑战,未来的研究应着重于优化制备流程、明确作用机制和完善安全性评价,为推动临床牙髓损伤修复提供新的治疗策略。
中图分类号:
引用本文
徐海超, 罗丽花, 潘乙怀. 牙髓干细胞及衍生产物在牙髓再生中的应用与进展[J]. 中国组织工程研究, 2026, 30(1): 153-162.
Xu Haichao, Luo Lihua, Pan Yihuai. Application and progress of dental pulp stem cells and their derivatives in dental pulp regeneration[J]. Chinese Journal of Tissue Engineering Research, 2026, 30(1): 153-162.
境[21];通过调节培养环境中的pH值、氧气体积分数及电磁场等物理因素[22-23],可精确调控牙髓干细胞的增殖和分化;此外,基因转染技术通过调控特定转录因子的表达[24-26],促进牙髓干细胞向目标细胞类型分化,进一步提升牙髓干细胞在牙髓再生中的效果。
(1)牙髓干细胞活性:为了提高牙髓再生的效果,增强和维持牙髓干细胞的活性包括增殖、黏附、迁移、分化和免疫调节能力等方面,是实现有效修复的关键。研究者们提出了多种工程化干细胞方案来提高牙髓干细胞的活性,为后续血运重建以及形成牙髓-牙本质样结构奠定了坚实的基础[27-28]。YU等[18]发现大麻二酚通过下调促炎细胞因子如肿瘤坏死因子α和白细胞介素1β的表达,有效恢复炎症环境中牙髓干细胞的增殖与分化活性,进一步增强成牙分化潜力。AYADILORD等[29]将植物体姜黄素纳米颗粒作用于牙髓干细胞,发现通过调控多个miRNA(如miR-21、miR-23、miR-155等)和CD200的表达,抑制磷脂酰肌醇3激酶/蛋白激酶B/核因子κB信号通路,从而调节干细胞的免疫活性并降低促炎细胞因子的表达。WANG等[22]利用等离子体纳米材料的电磁场效应和光热效应的协同作用,靶向增加牙髓干细胞线粒体内羟脯氨酸的含量,从而促进牙髓干细胞向成牙本质细胞的定向分化。ZHANG等[23]构建了一种磁性纳米颗粒交联的超分子聚合物纳米纤维,并通过施加人工磁场成功实现对牙髓干细胞极性的调控,促进牙髓干细胞定向极化及延伸等细胞行为,这一机制策略为牙髓再生提供了新的研究方向。JEONG等[30]利用牙髓干细胞构建了一种牙髓牙本质样类器官,这些类器官能够对生物刺激器展现出适当的反应,具有干细胞和分化细胞(如成牙本质细胞)的特征,呈现了在牙髓再生领域作为新型研究工具的巨大潜力。MEZA等[31]首次将自体牙髓干细胞与白细胞富血小板纤维蛋白基质联合应用于不可逆牙髓炎患者的恒牙再生手术,结果表明,白细胞富血小板纤维蛋白基质不仅为牙髓干细胞提供了理想的黏附与迁移环境,还通过释放生长因子促进了细胞的增殖与分化,最终成功再生出具有血管结构的均匀致密牙髓样组织。LIU等[32]通过在牙本质切片表面修饰多巴胺-透明质酸涂层来增强牙髓干细胞与根管壁之间的黏附力,研究表明涂层处理后的牙本质组在细胞数量、细胞伸长率和黏附力上明显优于未处理的牙本质组,进而促进了牙髓干细胞在根管内的增殖和迁移。TERRANOVA等[33]将鞣酸修饰的聚己内酯微粒引入电纺聚乳酸纳米纤维支架中,得到的3D锥体结构支架具备优良的结构可控性,易于插入根管,同时具有高度多孔性,能够有效促进牙髓干细胞的浸润与定植,增强增殖、分化和迁移活性,为牙髓组织再生与修复提供有力支持。
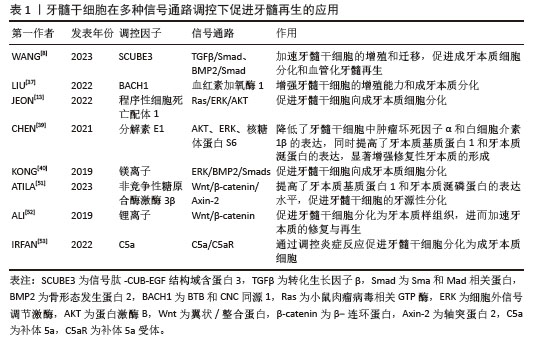
(2)牙髓干细胞向成牙本质细胞分化:牙髓-牙本质复合体的重建是牙髓再生的关键因素之一,大量研究表明,牙髓干细胞能够在多种信号通路的调控下向成牙本质细胞分化[34-36],从而促进牙髓-牙本质复合体的再生。LIU等[37]研究发现,过表达BTB和CNC同源1能够影响血红素加氧酶1信号通路从而促进牙髓干细胞向成牙本质细胞的分化。JEON和LIU等[13,38]研究发现,通过阻断程序性细胞死亡配体1信号,可以激活Ras-细胞外信号调节激酶(extracellular signal-regulated kinase,ERK)和蛋白激酶B信号通路,进一步促进牙髓干细胞的成牙本质分化。CHEN等[39]研究表明,分解素E1通过调节蛋白激酶B、ERK以及核糖体蛋白S6的磷酸化水平,显著促进牙髓干细胞中牙本质磷蛋白(dentin sialoprotein,DSP)的表达。另一项研究证明了镁离子(Mg2+)通过激活ERK/骨形态发生蛋白2/Smads信号通路促进牙髓干细胞向成牙本质细胞分化[40]。
WANG等[24]通过反转录病毒转染技术使牙髓干细胞过表达分泌型Frizzled相关蛋白2,并将其移植到上下颌牙拔除的兔模型中,实验组拔牙槽被大量高密度的牙本质样结构填充,并且表达了牙本质特异性蛋白,如牙本质涎磷蛋白(dentin sialophosphoprotein,DSPP)和牙本质基质蛋白1(dentin matrix protein 1,DMP1)等。XUAN等[41]使用自体乳牙牙髓干细胞治疗因创伤导致的年轻恒牙牙髓坏死,成功再生出含有正常成牙本质细胞层的牙髓组织,经过24个月的随访,发现再生牙髓仍然保持活性。另外,牙髓干细胞与生物材料的联合应用已被广泛探索,被认为是牙髓再生的有效策略之一。HAN等[42]采用双网络羧乙基几丁质水凝胶携载牙髓干细胞,在体外促进成牙本质分化,并在体内实验中展现出与天然牙髓组织生成相匹配的支架降解速率。JUN等[43]将氧化铈纳米颗粒引入到三氧化二矿物骨料并作用于牙髓干细胞,发现细胞内活性氧水平下降约70%,而牙髓干细胞成牙本质分化能力得到明显提升。其他支架材料如光交联丝素蛋白甲基丙烯酸酯复合水凝胶、富含氯化钾的卡拉胶/壳聚糖/明胶复合支架等为牙髓干细胞提供了优异的仿生微环境,进一步提升了牙髓干细胞的成牙本质分化能力[7,44-53]。相关文献见表1。
(3)牙髓干细胞向血管内皮细胞分化:在牙髓重建过程中,新生血管为周围组织提供必要的营养支持[54],有效促进牙髓干细胞向血管内皮细胞分化,并分泌相关血管生成活性因子。为了克服牙髓干细胞在单细胞培养模型中增殖缓慢和微环境模拟不足等问题,研究者们通过在单细胞培养模型中引入其他类型的细胞[55]、各种生长因子和活性材料建立细胞共培养体系,显著提高了牙髓干细胞的血管生成能力[56-58]。吕继忠[58]将小鼠成纤维细胞来源的诱导多能干细胞诱导分化为血管内皮样细胞后,与牙髓干细胞共培养,发现共培养体系中牙髓干细胞的血管内皮生长因子mRNA表达水平显著上升。KATATA等[4]以及ITOH等[59]的研究表明,使用内皮分化培养基对牙髓干细胞进行预处理,显著增强了牙髓干细胞的血管生成能力。BINDAL等[60]研究发现,人血小板裂解液可以显著提升牙髓干细胞中血管生成相关基因的表达。LIANG等[61]在比格犬和裸鼠体内模型中的研究表明,骨形态发生蛋白7能够诱导牙髓干细胞迁移,并促进新生血管向内生长,同时促进丰富的牙髓样组织形成。WU等[62]通过铜离子(Cu2+)激活晚期牙髓干细胞中染色盒蛋白同源物7 (chromobox protein homolog 7,CBX7)的表达,并将干细胞注射到裸鼠皮下移植模型中,发现CD31表达上调,提示有血管生成。NAKASHIMA等[63]在犬牙髓切除的临床前模型中,通过自体牙髓干细胞与粒细胞集落刺激因子联合移植到根管中,成功促进了血管和神经丰富的牙髓样组织形成,且治疗后的犬临床体征恢复正常。在验证干细胞疗法用于牙髓再生的临床前安全性和有效性后,NAKASHIMA等[64]首次将自体牙髓干细胞应用于人体多根磨牙的牙髓再生治疗,治疗后4周,患牙电活力测试呈阳性反应,患者未见全身毒性;24周时,核磁共振成像结果显示再生牙髓与对照牙髓几乎无差异,并形成血管-神经支配的牙本质样矿化组织;48周时,锥形束计算机断层扫描未见根尖周射线可透性,表明治疗后的牙髓组织稳定无渗漏,证明该疗法在人体牙髓再生中的临床安全性与疗效性。同时,研究者们构建封装牙髓干细胞与血管内皮生长因子的可注射海藻酸钠/锂皂石水凝胶微球,不但可以注射到常规细胞片层或刚性支架难以覆盖的区域,同时能够持续释放血管内皮生长因子长达28 d[65]。ATILA等[51,66]在水凝胶核/壳结构上分别负载了褪黑素和非竞争性糖原合酶激酶3β,成功实现了双重可控释放,其中非竞争性糖原合酶激酶3β作为首要释放成分,用于刺激血管生成,褪黑素起到延迟释放的作用,有效调控牙髓干细胞的早期增殖速率。此外,LI等[26]提出了一种新型非病毒基因纳米载体,通过携载血管内皮生长因子基因显著提升了转染后的牙髓干细胞血管生成潜力,有效促进了牙根血运重建与牙髓组织再生。
2.2 牙髓干细胞来源活性因子在牙髓组织工程中的应用
2.2.1 细胞外囊泡
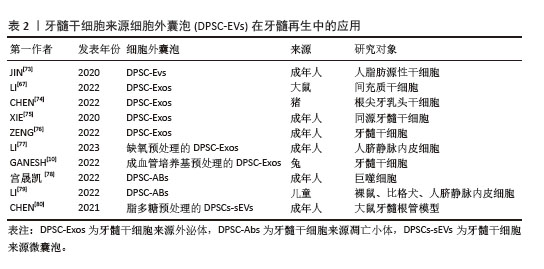
(1)细胞外囊泡的定义及特征:牙髓干细胞来源细胞外囊泡是由牙髓干细胞分泌的一类被细胞膜包裹的纳米级囊泡,具有高度稳定性和生物相容性。根据国际细胞外囊泡学会的建议,通常将细胞外囊泡根据大小和形成机制分为3类:小的内体衍生的外泌体(直径30-150 nm)、大的质膜衍生的微囊泡(直径100-1 000 nm)和凋亡过程中产生的凋亡小体(直径50-1 000 nm)。由于牙髓干细胞基因工程的可行性和便利性,可作为细胞外囊泡的合适来源[67]。通过收集牙髓干细胞上清液,离心即可获得富含蛋白质、脂质和核酸如信使RNA(messenger RNA,mRNA)、微小RNA(microRNA,miRNA)、长链非编码RNA(long non-coding RNA,lncRNA)等活性因子的牙髓干细胞来源细胞外囊泡[68-70],牙髓干细胞来源细胞外囊泡可以停留在原始细胞附近或进入生物体液[71],通过细胞膜融合或内吞作用进入靶细胞,发挥信号通路传递作用。因较低的免疫排斥风险和较高的基因工程改造可行性[67,72],牙髓干细胞来源细胞外囊泡被认为是牙髓组织工程中一种极具潜力的无细胞治疗手段[73-80],相关文献见表2。
(2)细胞外囊泡在牙髓再生方面的应用
促进成牙本质分化:在牙髓组织修复过程中,牙髓干细胞来源细胞外囊泡内部的活性因子能够通过多种机制有效调控干细胞向成牙本质细胞的分化。研究表明,牙髓干细胞来源细胞外囊泡通过激活多条信号通路如ERK、C-Jun氨基末端激酶(C-Jun N-terminal kinase,JNK)和丝裂原活化蛋白激酶(mitogen-activated protein kinase,MAPK)等[73,81],促进人脂肪来源干细胞的成骨分化,效果与美国食品和药物管理局批准的骨形态发生蛋白2相当。此外,SWANSON等[82]实验表明,牙髓干细胞来源外泌体能够通过内吞作用刺激人源性间充质干细胞激活DSPP的转录,促进其向成牙本质细胞分化。CHEN等[74]研究发现,牙髓干细胞来源外泌体通过细胞归巢机制,成功招募了根尖牙乳头干细胞,并在体内形成牙髓样结缔组织。值得注意的是,牙髓干细胞来源细胞外囊泡中的miRNA在促进成牙本质分化过程中也发挥了重要作用。XIE等[75]研究发现,在牙髓干细胞来源外泌体被同型牙髓干细胞吞噬后,上调的环状溶血磷脂酸受体1与miR-31结合,消除了对牙髓干细胞成骨分化的抑制作用。LI等[67]研究表明,牙髓干细胞来源外泌体携载的长链非编码RNA锚定蛋白重复结构域26能够通过调控miR-150/Toll样受体4信号通路促进成骨细胞分化。
促进成血管和成神经分化:牙髓干细胞来源细胞外囊泡在牙髓再生中的另一个关键作用在于其强大的促进血管和神经再生能力。牙髓神经的再生依赖于健康血管提供的营养支持,牙髓干细胞来源细胞外囊泡通常包含可溶性血管生成相关蛋白(如血管内皮生长因子和成纤维细胞生长因子)及相关miRNA[83-85],在血管再生方面发挥着尤为重要的作用。ZHANG等[9]通过将含有牙髓干细胞来源细胞外囊泡的纤维蛋白凝胶与牙髓干细胞共培养,发现牙髓干细胞来源细胞外囊泡中的miRNA促进了血管内皮生长因子的分泌,并且伴随多种胶原蛋白的沉积,后者为新生血管的成熟和稳定提供了支持。GANESH等[10]发现,血管分化条件下的牙髓干细胞来源外泌体在促进血管生成方面有显著优势。蓝彬园等[86]研究发现,脂多糖预处理的牙髓干细胞分泌的外泌体能够在根管内形成更多新生血管密度的牙髓样组织。LI等[87]进一步表明,脂多糖预处理的牙髓干细胞分泌的外泌体还能够增强施万细胞的增殖、迁移及成牙本质分化的能力。CHEN等[80]将脂多糖预处理的牙髓干细胞产生的微囊泡植入到大鼠牙髓根管模型中,发现血管内皮生长因子、神经成束蛋白和牙本质涎磷蛋白等标志物表达显著上升,显示出脂多糖预处理的牙髓干细胞分泌的微囊泡在血管、神经和牙本质再生中的潜力。此外,不同年龄供者来源的牙髓干细胞衍生细胞外囊泡在血管和神经再生能力方面存在差异[88],年轻组的牙髓干细胞来源外泌体相较于年老组在促进血管内皮生长因子和肝细胞生长因子分泌上具有显著优势,显示出更高的无细胞治疗潜力。MAS-BARGUES等[89]研究发现,衰老牙髓干细胞来源细胞外囊泡能够诱导年轻牙髓干细胞中抗氧化基因过表达,进而增强抗氧化能力,并促进年轻牙髓干细胞的增殖、迁移与存活,从而调节年轻牙髓干细胞的微环境。
2.2.2 条件培养液
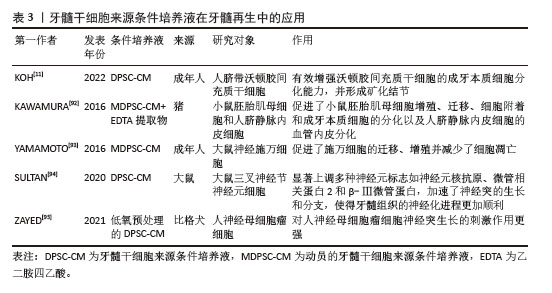
(1)条件培养液的定义及特征:牙髓干细胞来源条件培养液是通过收集在体外培养牙髓干细胞的基液,经过过滤、离心和浓缩等处理步骤,从而获得高生物活性的条件培养液。牙髓干细胞来源条件培养液富含细胞因子、生长因子、细胞外基质的分解产物和代谢产物等,能够通过旁分泌机制调节周围细胞的行为。由于含有丰富的生物活性成分和低免疫排斥风险,牙髓干细胞来源条件培养液被认为在组织修复领域具有重要的应用潜力[90]。与牙髓干细胞来源细胞外囊泡相比,牙髓干细胞来源条件培养液含有更多的分解产物和代谢因子,能够在微环境中提供更有利的修复条件,促进成牙本质分化,诱导新生血管形成并减少内皮细胞凋亡,在牙髓再生领域相关文献如表3所示,受到了广泛关注[91-95]。
(2) 条件培养液在牙髓再生方面的应用
促进成牙本质分化:牙髓干细胞来源条件培养液在促进成牙本质分化过程中扮演着关键角色。研究表明,牙髓干细胞来源条件培养液中高度表达的神经营养因子3和骨形态发生蛋白4能够诱导成牙本质细胞的分化,加速牙髓牙本质复合体的形成[96]。KOH等[11]通过将牙髓干细胞来源条件培养液与沃顿胶间充质干细胞共培养,成功诱导了沃顿胶间充质干细胞向成牙本质细胞分化。SARRA等[97]使用牙髓干细胞来源条件培养液联合三氧化二矿骨料在大鼠牙髓炎症模型上进行盖髓治疗,发现联合组形成了结构更有序的牙本质桥。KAWAMURA等[92]将动员的牙髓干细胞来源条件培养液与乙二胺四乙酸可溶性成分联合进行牙齿异位重建,观察到有良好的牙髓样组织形成。
促进成血管和成神经分化:在牙髓再生过程中,血管再生通常优先于轴突再生[93],施万细胞和轴突会随着血管生成进一步生长延伸。牙髓干细胞来源条件培养液中的血管内皮生长因子、脑源性神经营养因子和胶质源性神经营养因子等在血管生成和神经再生过程中发挥着重要作用[98],它们通过促进内皮细胞的迁移和血管管腔的形成,加速血管再生,为牙髓再生提供充足的营养和氧气供应。LAMBRICHTS等[99]发现牙髓干细胞来源条件培养液能够促进内皮细胞迁移,展现了促进血管生成的作用。牙髓干细胞来源条件培养液还能够通过旁分泌机制提高神经细胞的存活率,促进受损牙髓神经的再生,尤其是对损伤后的三叉神经节神经元细胞具有显著的神经营养作用,加速了神经突的生长,使得牙髓组织的神经化进程更加顺利[94]。YAMAMOTO等[93,100-102]重点研究了粒细胞集落刺激因子梯度动员牙髓干细胞,发现动员牙髓干细胞不仅能促进施万细胞的抗凋亡、增殖和迁移作用,还能够分泌神经源/血管生成因子(如血管内皮生长因子、脑源性神经营养因子、胶质源性神经营养因子等)促进周围神经再生,且已在临床牙髓炎患者中验证了安全性和可行性。ZAYED等[95]对比研究了低氧预处理的牙髓干细胞来源条件培养液与动员牙髓干细胞来源条件培养液,发现低氧预处理的牙髓干细胞来源条件培养液对人神经母细胞瘤细胞的神经突生长具有更强的刺激作用。
2.3 牙髓干细胞来源的支架材料在牙髓组织工程中的应用
2.3.1 脱细胞基质的定义及特征 牙髓干细胞来源脱细胞基质是一类通过物理、化学或酶处理去除牙髓干细胞成分,只保留细胞外基质的三维支架材料。与其他生物材料相比,牙髓干细胞来源脱细胞基质在高度重现原始牙髓组织的结构和微环境方面具有明显优势,且能更高效地利用原代牙髓资源。牙髓干细胞来源脱细胞基质中包含与牙体形成、血管生成和神经再生相关的特定蛋白质[12],这些成分可以促进牙髓组织的多向再生。同时,由于去除了免疫原性细胞成分,牙髓干细胞来源脱细胞基质具有较低的异种移植排斥风险。在牙髓组织工程中,牙髓干细胞来源脱细胞基质不仅为细胞提供了物理支架,还通过残留的生长因子和细胞黏附分子,改善细胞的增殖、迁移和分化,并通过仿生微环境促进成牙本质细胞分化以及血管和神经生成,为牙髓再生的研究提供了新的方向。
2.3.2 脱细胞基质在牙髓再生方面的应用 LI等[12]将牙髓干细胞接种到人牙髓来源脱细胞基质水凝胶上并进行培养,实验结果显示,人牙髓来源脱细胞基质水凝胶能够显著促进牙髓干细胞向成牙本质细胞的分化,经过14 d的诱导,牙髓干细胞表现出CD31和神经特异性β-Ⅲ微管蛋白表达上调,表明人牙髓来源脱细胞基质水凝胶能够促进牙髓干细胞向血管、神经分化。ZHENG等[103]提出了一种将牙髓干细胞来源脱细胞基质与水凝胶微球相结合(dpECM modified GelMA microspheres, dpECM@GM)的策略,并将含牙髓干细胞的dpECM@GM植入裸鼠皮下3个月后,发现dpECM@GM在局部微环境中提供了多种生长因子(如转化生长因子β和成纤维细胞生长因子)支持,有效促进了牙髓干细胞的成牙本质、成血管和成神经分化,最终实现了类牙髓组织和新牙本质的再生。
| [1] LIANG Q, LIANG C, LIU X, et al. Vascularized dental pulp regeneration using cell-laden microfiber aggregates. J Mater Chem B. 2022; 10(48):10097-10111. [2] SUI B, CHEN C, KOU X, et al. Pulp Stem Cell–Mediated Functional Pulp Regeneration. J Dent Res. 2019;98(1):27-35. [3] ROTHERMUND K, CALABRESE TC, SYED-PICARD FN. Differential Effects of Escherichia coli- Versus Porphyromonas gingivalis-derived Lipopolysaccharides on Dental Pulp Stem Cell Differentiation in Scaffold-free Engineered Tissues. J Endod. 2022;48(11):1378-1386.e2. [4] KATATA C, SASAKI JI, LI A, et al. Fabrication of Vascularized DPSC Constructs for Efficient Pulp Regeneration. J Dent Res. 2021;100(12): 1351-1358. [5] XIE Z, SHEN Z, ZHAN P, et al. Functional Dental Pulp Regeneration: Basic Research and Clinical Translation. Int J Mol Sci. 2021;22(16):8991. [6] LAI CF, SHEN J, BALIC A, et al. Nogo-A Regulates the Fate of Human Dental Pulp Stem Cells toward Osteogenic, Adipogenic, and Neurogenic Differentiation. Cells. 2022; 11(21):3415. [7] LOUKELIS K, MACHLA F, BAKOPOULOU A, et al. Kappa-Carrageenan/Chitosan/Gelatin Scaffolds Provide a Biomimetic Microenvironment for Dentin-Pulp Regeneration. Int J Mol Sci. 2023;24(7):6465. [8] WANG Z, CHEN C, ZHANG J, et al. Epithelium-derived SCUBE3 promotes polarized odontoblastic differentiation of dental mesenchymal stem cells and pulp regeneration. Stem Cell Res Ther. 2023;14(1):130. [9] ZHANG S, THIEBES AL, KREIMENDAHL F, et al. Extracellular Vesicles-Loaded Fibrin Gel Supports Rapid Neovascularization for Dental Pulp Regeneration. Int J Mol Sci. 2020;21(12): 4226. [10] GANESH V, SEOL D, GOMEZ-CONTRERAS PC, et al. Exosome-Based Cell Homing and Angiogenic Differentiation for Dental Pulp Regeneration. Int J Mol Sci. 2022;24(1):466. [11] KOH B, AB RAHMAN FH, MATLAN NA, et al. Potential role of dental pulp stem cells conditioned medium for odontoblastic differentiation. Biol Res. 2022;55(1):11. [12] LI J, RAO Z, ZHAO Y, et al. A Decellularized Matrix Hydrogel Derived from Human Dental Pulp Promotes Dental Pulp Stem Cell Proliferation, Migration, and Induced Multidirectional Differentiation In Vitro. J Endod. 2020;46(10):1438-1447.e5. [13] JEON SM, LIM JS, PARK SH, et al. Blockade of PD-L1/PD-1 signaling promotes osteo-/odontogenic differentiation through Ras activation. Int J Oral Sci. 2022;14(1):18. [14] KIM H, OH N, KWON M, et al. Exopolysaccharide of Enterococcus faecium L15 promotes the osteogenic differentiation of human dental pulp stem cells via p38 MAPK pathway. Stem Cell Res Ther. 2022;13(1):446. [15] ZHAI S, LIU C, VIMALRAJ S, et al. Glucagon-like peptide-1 receptor promotes osteoblast differentiation of dental pulp stem cells and bone formation in a zebrafish scale regeneration model. Peptides. 2023;163: 170974. [16] MANIMARAN K, SHARMA R, SANKARANARAYANAN S, et al. Regeneration of mandibular ameloblastoma defect with the help of autologous dental pulp stem cells and buccal pad of fat stromal vascular fraction. Ann Maxillofac Surg. 2016;6(1):97-100. [17] TAKAOKA S, UCHIDA F, ISHIKAWA H, et al. Transplanted neural lineage cells derived from dental pulp stem cells promote peripheral nerve regeneration. Hum Cell. 2022;35(2): 462-471. [18] YU L, ZENG L, ZHANG Z, et al. Cannabidiol Rescues TNF-α-Inhibited Proliferation, Migration, and Osteogenic/Odontogenic Differentiation of Dental Pulp Stem Cells. Biomolecules. 2023;13(1):118. [19] ALGHUTAIMEL H, YANG X, DRUMMOND B, et al. Investigating the vascularization capacity of a decellularized dental pulp matrix seeded with human dental pulp stem cells: in vitro and preliminary in vivo evaluations. Int Endod J. 2021;54(8):1300-1316. [20] PHAN TV, OO Y, AHMED K, et al. Salivary gland regeneration: from salivary gland stem cells to three-dimensional bioprinting. SLAS Technol. 2023;28(3):199-209. [21] TIEN N, LEE JJ, LEE AK, et al. Additive Manufacturing of Caffeic Acid-Inspired Mineral Trioxide Aggregate/Poly-ε-Caprolactone Scaffold for Regulating Vascular Induction and Osteogenic Regeneration of Dental Pulp Stem Cells. Cells. 2021;10(11):2911. [22] WANG J, QU X, XU C, et al. Thermoplasmonic Regulation of the Mitochondrial Metabolic State for Promoting Directed Differentiation of Dental Pulp Stem Cells. Anal Chem. 2022; 94(27):9564-9571. [23] ZHANG B, YU Q, LIU Y. Polarization of Stem Cells Directed by Magnetic Field-Manipulated Supramolecular Polymeric Nanofibers. ACS Appl Mater Interfaces. 2021;13(8):9580-9588. [24] WANG C, WANG Y, WANG H, et al. SFRP2 enhances dental pulp stem cell-mediated dentin regeneration in rabbit jaw. Oral Dis. 2021;27(7):1738-1746. [25] MACHLA F, SOKOLOVA V, PLATANIA V, et al. Tissue engineering at the dentin-pulp interface using human treated dentin scaffolds conditioned with DMP1 or BMP2 plasmid DNA-carrying calcium phosphate nanoparticles. Acta Biomater. 2023;159:156-172. [26] LI Q, HU Z, LIANG Y, et al. Multifunctional peptide-conjugated nanocarriers for pulp regeneration in a full-length human tooth root. Acta Biomater. 2021;127:252-265. [27] ZHANG W, ZHENG Y, LIU H, et al. A non-invasive monitoring of USPIO labeled silk fibroin/hydroxyapatite scaffold loaded DPSCs for dental pulp regeneration. Mater Sci Eng C Mater Biol Appl. 2019; 103:109736. [28] QIU Y, SAITO T. Novel Bioactive Adhesive Monomer CMET Promotes Odontogenic Differentiation and Dentin Regeneration. Int J Mol Sci. 2021;22(23):12728. [29] AYADILORD M, NASSERI S, EMADIAN RAZAVI F, et al. Immunomodulatory effects of phytosomal curcumin on related-micro RNAs, CD200 expression and inflammatory pathways in dental pulp stem cells. Cell Biochem Funct. 2021;39(7):886-895. [30] JEONG SY, LEE S, CHOI WH, et al. Fabrication of Dentin-Pulp-Like Organoids Using Dental-Pulp Stem Cells. Cells. 2020;9(3):642. [31] MEZA G, URREJOLA D, SAINT JEAN N, et al. Personalized Cell Therapy for Pulpitis Using Autologous Dental Pulp Stem Cells and Leukocyte Platelet-rich Fibrin: A Case Report. J Endod. 2019;45(2):144-149. [32] LIU Y, QIU Y, NI S, et al. Mussel-Inspired Biocoating for Improving the Adhesion of Dental Pulp Stem Cells in Dental Pulp Regeneration. Macromol Rapid Commun. 2020;41(24):e2000102. [33] TERRANOVA L, LOUVRIER A, HÉBRAUD A, et al. Highly Structured 3D Electrospun Conical Scaffold: A Tool for Dental Pulp Regeneration. ACS Biomater Sci Eng. 2021; 7(12):5775-5787. [34] LIU Z, LI S, XU S, et al. Hsa_Circ_0005044 Promotes Osteo/Odontogenic Differentiation of Dental Pulp Stem Cell Via Modulating miR-296-3p/FOSL1. DNA Cell Biol. 2023;42(1): 14-26. [35] MU R, CHEN B, BI B, et al. LIM Mineralization Protein-1 Enhances the Committed Differentiation of Dental Pulp Stem Cells through the ERK1/2 and p38 MAPK Pathways and BMP Signaling. Int J Med Sci. 2022;19(8): 1307-1319. [36] 陈冬梅,徐丽丽,周建伟.姜黄素通过Wnt信号通路调节人牙髓干细胞的成牙本质分化[J].中国组织工程研究,2019,23(25): 4018-4024. [37] LIU C, YU J, LIU B, et al. BACH1 regulates the proliferation and odontoblastic differentiation of human dental pulp stem cells. BMC Oral Health. 2022;22(1):536. [38] LIU Y, JING H, KOU X, et al. PD-1 is required to maintain stem cell properties in human dental pulp stem cells. Cell Death Differ. 2018; 25(7):1350-1360. [39] CHEN J, XU H, XIA K, et al. Resolvin E1 accelerates pulp repair by regulating inflammation and stimulating dentin regeneration in dental pulp stem cells. Stem Cell Res Ther. 2021;12(1):75. [40] KONG Y, HU X, ZHONG Y, et al. Magnesium-enriched microenvironment promotes odontogenic differentiation in human dental pulp stem cells by activating ERK/BMP2/Smads signaling. Stem Cell Res Ther. 2019;10(1):378. [41] XUAN K, LI B, GUO H, et al. Deciduous autologous tooth stem cells regenerate dental pulp after implantation into injured teeth. Sci Transl Med. 2018;10(455):eaaf3227. [42] HAN B, CAO C, WANG A, et al. Injectable Double-Network Hydrogel-Based Three-Dimensional Cell Culture Systems for Regenerating Dental Pulp. ACS Appl Mater Interfaces. 2023;15(6):7821-7832. [43] JUN SK, YOON JY, MAHAPATRA C, et al. Ceria-incorporated MTA for accelerating odontoblastic differentiation via ROS downregulation. Dent Mater. 2019;35(9): 1291-1299. [44] WANG L, ZHANG Y, XIA Y, et al. Photocross-linked silk fibroin/hyaluronic acid hydrogel loaded with hDPSC for pulp regeneration. Int J Biol Macromol. 2022;215:155-168. [45] XIA K, CHEN Z, CHEN J, et al. RGD- and VEGF-Mimetic Peptide Epitope-Functionalized Self-Assembling Peptide Hydrogels Promote Dentin-Pulp Complex Regeneration. Int J Nanomedicine. 2020; 15:6631-6647. [46] KHAYAT A, MONTEIRO N, SMITH EE, et al. GelMA-Encapsulated hDPSCs and HUVECs for Dental Pulp Regeneration. J Dent Res. 2017;96(2):192-199. [47] ZHANG Q, YANG T, ZHANG R, et al. Platelet lysate functionalized gelatin methacrylate microspheres for improving angiogenesis in endodontic regeneration. Acta Biomater. 2021;136:441-455. [48] PARK H, COLLIGNON AM, LEPRY WC, et al. Acellular dense collagen-S53P4 bioactive glass hybrid gel scaffolds form more bone than stem cell delivered constructs. Mater Sci Eng C Mater Biol Appl. 2021;120:111743. [49] BAKHTIAR H, ASHOORI A, RAJABI S, et al. Human amniotic membrane extracellular matrix scaffold for dental pulp regeneration in vitro and in vivo. Int Endod J. 2022;55(4): 374-390. [50] LUZURIAGA J, GARCÍA-GALLASTEGUI P, GARCÍA-URKIA N, et al. Osteogenic differentiation of human dental pulp stem cells in decellularised adipose tissue solid foams. Eur Cell Mater. 2022;43:112-129. [51] ATILA D, KESKIN D, LEE YL, et al. Injectable methacrylated gelatin/thiolated pectin hydrogels carrying melatonin/tideglusib-loaded core/shell PMMA/silk fibroin electrospun fibers for vital pulp regeneration. Colloids Surf B Biointerfaces. 2023;222:113078. [52] ALI M, OKAMOTO M, KOMICHI S, et al. Lithium-containing surface pre-reacted glass fillers enhance hDPSC functions and induce reparative dentin formation in a rat pulp capping model through activation of Wnt/β-catenin signaling. Acta Biomater. 2019;96: 594-604. [53] IRFAN M, KIM JH, MARZBAN H, et al. The role of complement C5a receptor in DPSC odontoblastic differentiation and in vivo reparative dentin formation. Int J Oral Sci. 2022;14(1):7. [54] 王舸,谢利,田卫东.牙髓再生中促进血管化策略的新进展[J].中国组织工程研究, 2022,26(30):4904-4911. [55] 张楚晗,张东敏,徐稳安.牙髓再生组织工程中的细胞共培养体系[J].中国组织工程研究,2023,27(15):2379-2384. [56] CATALDI A, AMOROSO R, DI GIACOMO V, et al. The Inhibition of the Inducible Nitric Oxide Synthase Enhances the DPSC Mineralization under LPS-Induced Inflammation. Int J Mol Sci. 2022;23(23):14560. [57] LIANG X, XIE L, ZHANG Q, et al. Gelatin methacryloyl-alginate core-shell microcapsules as efficient delivery platforms for prevascularized microtissues in endodontic regeneration. Acta Biomater. 2022;144: 242-257. [58] 吕继忠.成纤维细胞来源诱导性多能干细胞定向分化为血管内皮样细胞及与牙髓干细胞的共培养[J].中国组织工程研究,2018, 22(21):3371-3375. [59] ITOH Y, SASAKI JI, HASHIMOTO M, et al. Pulp Regeneration by 3-dimensional Dental Pulp Stem Cell Constructs. J Dent Res. 2018; 97(10):1137-1143. [60] BINDAL P, GNANASEGARAN N, BINDAL U, et al. Angiogenic effect of platelet-rich concentrates on dental pulp stem cells in inflamed microenvironment. Clin Oral Investig. 2019;23(10):3821-3831. [61] LIANG C, LIANG Q, XU X, et al. Bone morphogenetic protein 7 mediates stem cells migration and angiogenesis: therapeutic potential for endogenous pulp regeneration. Int J Oral Sci. 2022;14(1):38. [62] WU Y, LI B, YU D, et al. CBX7 Rejuvenates Late Passage Dental Pulp Stem Cells by Maintaining Stemness and Pro-angiogenic Ability. Tissue Eng Regen Med. 2023;20(3):473-488. [63] NAKASHIMA M, IOHARA K. Mobilized dental pulp stem cells for pulp regeneration: initiation of clinical trial. J Endod. 2014;40(4 Suppl): S26-32. [64] NAKASHIMA M, FUKUYAMA F, IOHARA K. Pulp Regenerative Cell Therapy for Mature Molars: A Report of 2 Cases. J Endod. 2022;48(10): 1334-1340.e1. [65] ZHANG R, XIE L, WU H, et al. Alginate/laponite hydrogel microspheres co-encapsulating dental pulp stem cells and VEGF for endodontic regeneration. Acta Biomater. 2020;113:305-316. [66] ATILA D, CHEN CY, LIN CP, et al. In vitro evaluation of injectable Tideglusib-loaded hyaluronic acid hydrogels incorporated with Rg1-loaded chitosan microspheres for vital pulp regeneration. Carbohydr Polym. 2022;278:118976. [67] LI L, GE J. Exosome‑derived lncRNA‑Ankrd26 promotes dental pulp restoration by regulating miR‑150‑TLR4 signaling. Mol Med Rep. 2022; 25(5):152. [68] GUO S, DEBBI L, ZOHAR B, et al. Stimulating Extracellular Vesicles Production from Engineered Tissues by Mechanical Forces. Nano Lett. 2021;21(6):2497-2504. [69] HUANG CC, NARAYANAN R, ALAPATI S, et al. Exosomes as biomimetic tools for stem cell differentiation: Applications in dental pulp tissue regeneration. Biomaterials. 2016;111:103-115. [70] KONG F, WU CT, GENG P, et al. Dental Pulp Stem Cell-Derived Extracellular Vesicles Mitigate Haematopoietic Damage after Radiation. Stem Cell Rev Rep. 2021;17(2): 318-331. [71] MA S, JIANG Y, QIAN Y, et al. The Emerging Biological Functions of Exosomes from Dental Tissue-Derived Mesenchymal Stem Cells. Cell Reprogram. 2023;25(2):53-64. [72] CHEN Y, KOSHY R, GUIRADO E, et al. STIM1 a calcium sensor promotes the assembly of an ECM that contains Extracellular vesicles and factors that modulate mineralization. Acta Biomater. 2021;120:224-239. [73] JIN Q, LI P, YUAN K, et al. Extracellular vesicles derived from human dental pulp stem cells promote osteogenesis of adipose-derived stem cells via the MAPK pathway. J Tissue Eng. 2020;11:2041731420975569. [74] CHEN Y, MA Y, YANG X, et al. The Application of Pulp Tissue Derived-Exosomes in Pulp Regeneration: A Novel Cell-Homing Approach. Int J Nanomedicine. 2022;17:465-476. [75] XIE L, GUAN Z, ZHANG M, et al. Exosomal circLPAR1 Promoted Osteogenic Differentiation of Homotypic Dental Pulp Stem Cells by Competitively Binding to hsa-miR-31. Biomed Res Int. 2020;2020:6319395. [76] ZENG J, HE K, MAI R, et al. Exosomes from human umbilical cord mesenchymal stem cells and human dental pulp stem cells ameliorate lipopolysaccharide-induced inflammation in human dental pulp stem cells. Arch Oral Biol. 2022;138:105411. [77] LI B, LIANG A, ZHOU Y, et al. Hypoxia preconditioned DPSC-derived exosomes regulate angiogenesis via transferring LOXL2. Exp Cell Res. 2023;425(2):113543. [78] 宫晟凯,杨晓姗,窦庚,等.牙髓干细胞来源凋亡小体调节巨噬细胞极化及炎症反应[J].口腔疾病防治,2022,30(1):12-19. [79] LI Z, WU M, LIU S, et al. Apoptotic vesicles activate autophagy in recipient cells to induce angiogenesis and dental pulp regeneration. Mol Ther. 2022;30(10):3193-3208. [80] CHEN WJ, XIE J, LIN X, et al. The Role of Small Extracellular Vesicles Derived from Lipopolysaccharide-preconditioned Human Dental Pulp Stem Cells in Dental Pulp Regeneration. J Endod. 2021;47(6):961-969. [81] LEE AE, CHOI JG, SHI SH, et al. DPSC-Derived Extracellular Vesicles Promote Rat Jawbone Regeneration. J Dent Res. 2023;102(3): 313-321. [82] SWANSON WB, ZHANG Z, XIU K, et al. Scaffolds with controlled release of pro-mineralization exosomes to promote craniofacial bone healing without cell transplantation. Acta Biomater. 2020;118:215-232. [83] LI B, XIAN X, LIN X, et al. Hypoxia Alters the Proteome Profile and Enhances the Angiogenic Potential of Dental Pulp Stem Cell-Derived Exosomes. Biomolecules. 2022; 12(4):575. [84] 陈婷,李心竹,徐稳安.外泌体和细胞因子促进牙髓血管生成的作用与调控机制[J].中国组织工程研究,2020,24(14):2263-2270. [85] ZHOU H, LI X, WU RX, et al. Periodontitis-compromised dental pulp stem cells secrete extracellular vesicles carrying miRNA-378a promote local angiogenesis by targeting Sufu to activate the Hedgehog/Gli1 signalling. Cell Prolif. 2021;54(5):e13026. [86] 蓝彬园,林熹,陈文瑨,等.脂多糖刺激人牙髓干细胞分泌的外泌体联合基质细胞衍生因子-1对牙髓再生的影响[J].中华口腔医学杂志,2022,57(1):60-67. [87] LI J, JU Y, LIU S, et al. Exosomes derived from lipopolysaccharide-preconditioned human dental pulp stem cells regulate Schwann cell migration and differentiation. Connect Tissue Res. 2021;62(3):277-286. [88] BRUNELLO G, ZANOTTI F, TRENTINI M, et al. Exosomes Derived from Dental Pulp Stem Cells Show Different Angiogenic and Osteogenic Properties in Relation to the Age of the Donor. Pharmaceutics. 2022;14(5):908. [89] MAS-BARGUES C, SANZ-ROS J, ROMERO-GARCÍA N, et al. Small extracellular vesicles from senescent stem cells trigger adaptive mechanisms in young stem cells by increasing antioxidant enzyme expression. Redox Biol. 2023;62:102668. [90] TAKEUCHI H, TAKAHASHI H, TANAKA A. Effects of Human Dental Pulp Stem Cell-Derived Conditioned Medium on Atrophied Submandibular Gland after the Release from Ligation of the Main Excretory Duct in Mice. J Hard Tissue Biol. 2020;29(3):183-192. [91] KICHENBRAND C, VELOT E, MENU P, et al. Dental Pulp Stem Cell-Derived Conditioned Medium: An Attractive Alternative for Regenerative Therapy. Tissue Eng Part B Rev. 2019;25(1):78-88. [92] KAWAMURA R, HAYASHI Y, MURAKAMI H, et al. EDTA soluble chemical components and the conditioned medium from mobilized dental pulp stem cells contain an inductive microenvironment, promoting cell proliferation, migration, and odontoblastic differentiation. Stem Cell Res Ther. 2016; 7(1):77. [93] YAMAMOTO T, OSAKO Y, ITO M, et al. Trophic Effects of Dental Pulp Stem Cells on Schwann Cells in Peripheral Nerve Regeneration. Cell Transplant. 2016;25(1):183-193. [94] SULTAN N, AMIN LE, ZAHER AR, et al. Neurotrophic effects of dental pulp stem cells on trigeminal neuronal cells. Sci Rep. 2020;10(1):19694. [95] ZAYED M, IOHARA K, WATANABE H, et al. Characterization of stable hypoxia-preconditioned dental pulp stem cells compared with mobilized dental pulp stem cells for application for pulp regenerative therapy. Stem Cell Res Ther. 2021;12(1):302. [96] JOO KH, SONG JS, KIM S, et al. Cytokine Expression of Stem Cells Originating from the Apical Complex and Coronal Pulp of Immature Teeth. J Endod. 2018;44(1): 87-92.e1. [97] SARRA G, MACHADO MEL, CABALLERO-FLORES HV, et al. Effect of human dental pulp stem cell conditioned medium in the dentin-pulp complex regeneration: A pilot in vivo study. Tissue Cell. 2021;72:101536. [98] KARIMI-HAGHIGHI S, CHAVOSHINEZHAD S, SAFARI A, et al. Preconditioning with secretome of neural crest-derived stem cells enhanced neurotrophic expression in mesenchymal stem cells. Neurosci Lett. 2022;773:136511. [99] LAMBRICHTS I, DRIESEN RB, DILLEN Y, et al. Dental Pulp Stem Cells: Their Potential in Reinnervation and Angiogenesis by Using Scaffolds. J Endod. 2017;43(9S):S12-S16. [100] ZIAUDDIN SM, NAKASHIMA M, WATANABE H, et al. Biological characteristics and pulp regeneration potential of stem cells from canine deciduous teeth compared with those of permanent teeth. Stem Cell Res Ther. 2022;13(1):439. [101] MURAKAMI M, HORIBE H, IOHARA K, et al. The use of granulocyte-colony stimulating factor induced mobilization for isolation of dental pulp stem cells with high regenerative potential. Biomaterials. 2013;34(36): 9036-9047. [102] NAKASHIMA M, IOHARA K, MURAKAMI M, et al. Pulp regeneration by transplantation of dental pulp stem cells in pulpitis: a pilot clinical study. Stem Cell Res Ther. 2017; 8(1):61. [103] ZHENG L, LIU Y, JIANG L, et al. Injectable decellularized dental pulp matrix-functionalized hydrogel microspheres for endodontic regeneration. Acta Biomater. 2023;156:37-48. |
| [1] | 刘 洋, 刘东辉, 徐 磊, 展 旭, 孙昊博, 康 凯. 刺激响应型可注射水凝胶在心肌梗死精准化治疗中的作用与趋势[J]. 中国组织工程研究, 2026, 30(8): 2072-2080. |
| [2] | 赖 渝, 陈跃平, 章晓云. 生物活性材料治疗骨感染的研究热点与前沿趋势[J]. 中国组织工程研究, 2026, 30(8): 2132-2144. |
| [3] | 王奇飒, 卢雨征, 韩秀峰, 赵文玲, 石海涛, 徐 哲. 3D打印甲基丙烯酰化透明质酸/脱细胞皮肤水凝胶支架的细胞相容性[J]. 中国组织工程研究, 2026, 30(8): 1912-1920. |
| [4] | 王菘芃, 刘玉三, 于焕英, 高晓丽, 徐英江, 张晓明, 刘 敏. 沸石基咪唑盐框架8纳米材料的活性氧双向调控:从肿瘤治疗、抗菌到细胞保护[J]. 中国组织工程研究, 2026, 30(8): 2033-2013. |
| [5] | 王明琦, 冯诗雅, 韩银河, 于朋鑫, 郭丽娜, 贾子萱, 王秀丽. 神经化肠黏膜组织工程模型的构建及体外评价[J]. 中国组织工程研究, 2026, 30(4): 892-900. |
| [6] | 杨 肖, 白月辉, 赵甜甜, 王东昊, 赵 琛, 袁 硕. 颞下颌关节骨关节炎软骨退变:机制及再生的挑战[J]. 中国组织工程研究, 2026, 30(4): 926-935. |
| [7] | 余诗宇, 俞苏桐, 徐 杨, 镇祥燕, 韩凤选. 组织工程治疗策略在口腔黏膜下纤维化中的研究与应用进展[J]. 中国组织工程研究, 2026, 30(4): 936-948. |
| [8] | 王正业, 刘万林, 赵振群. 血管内皮生长因子A 靶向调控血管化治疗激素性股骨头坏死的机制[J]. 中国组织工程研究, 2026, 30(3): 671-679. |
| [9] | 杨 虎, 郑 宇, 贾承明, 王 通, 张广飞, 纪垚垚. 免疫微环境调节骨再生[J]. 中国组织工程研究, 2026, 30(3): 701-710. |
| [10] | 王 卓, 孙盼盼, 程焕芝, 曹婷婷. 壳聚糖在口腔软硬组织修复与再生中的应用[J]. 中国组织工程研究, 2026, 30(2): 459-468. |
| [11] | 王 域, 范民杰, 郑朋飞. 多重刺激响应性水凝胶在骨损伤修复中的应用:特殊响应能力及多样性功能[J]. 中国组织工程研究, 2026, 30(2): 469-479. |
| [12] | 顾健美, 袁坤山, 周 强, 张海军, . 激光微孔化脱细胞支架在组织再生中的应用[J]. 中国组织工程研究, 2026, 30(2): 499-507. |
| [13] | 杨凤丽, 周 朝, 熊 伟, 周宇翔, 李登顺, 王 鑫, 李展振. 3D打印聚乳酸骨支架修复骨缺损[J]. 中国组织工程研究, 2026, 30(2): 507-515. |
| [14] | 姜 侃, 阿力木江·阿不都肉苏力, 沙拉依丁·艾尔西丁, 艾克拜尔江·艾赛提, 库提鲁克·守克尔, 艾克热木江·木合热木. 生物材料与骨再生:研究热点及有影响力的500篇文献分析[J]. 中国组织工程研究, 2026, 30(2): 528-536. |
| [15] | 闫启全, 杨立斌, 李梦君, 倪亚卓, 陈科颖, 许 博, 李耀扬, 马士卿, 李 睿, 李建文. 负载抗菌肽KR-12-a5猪小肠黏膜下层复合纳米羟基磷灰石生物支架的制备及抗菌性能[J]. 中国组织工程研究, 2026, 30(2): 384-394. |
近年来,随着细胞学、生物学以及工程学技术的发展,牙髓组织工程利用细胞、活性因子与生物材料相结合,在体外构建有活性的牙髓组织,替代坏死的组织,从而达到牙髓功能的理想重建,已成为国内外口腔医学研究的热点。其中,牙髓干细胞是一种来源于牙齿相关组织的成体间充质干细胞,具有取材简便、相对微创、无伦理学问题等优点,在体外可直接向成牙本质、成血管以及成神经等方向进行诱导分化[6],在动物体内可分化形成血管化的牙髓牙本质复合体等[2,4,7-8],是牙髓组织工程重要的种子细胞来源之一。同时,如图1显示,牙髓干细胞源性衍生产物如细胞外囊泡(extracellular vesicles,EVs)、条件培养液(conditioned medium,CM)以及脱细胞基质(decellularized extracellular matrix,dECM)等,含有大量细胞活性因子,具有促进细胞增殖、分化以及组织修复的潜能,在牙髓损伤修复和再生领域也广泛研究[9-12]。此综述将深入探讨牙髓干细胞及其衍生产物在牙髓组织工程中的应用与进展,通过对一系列研究成果的综合分析,有望为不可逆性牙髓炎提供新的治疗策略,为牙髓组织工程未来的前景带来创新性的发展。
中国组织工程研究杂志出版内容重点:干细胞;骨髓干细胞;造血干细胞;脂肪干细胞;肿瘤干细胞;胚胎干细胞;脐带脐血干细胞;干细胞诱导;干细胞分化;组织工程
1.1.1 检索人及检索时间 由第一作者在2023年7月进行检索。
1.1.2 检索文献时限 2005-01-01/2023-06-30。
1.1.3 检索数据库 PubMed数据库、中国知网、中国生物医学文献数据库。
1.1.4 检索词 以“牙髓干细胞,细胞外囊泡,外泌体,凋亡小体,条件培养液,脱细胞基质,再生”为中文检索词,以“dental pulp stem cells,extracellular vesicles,exosomes,apoptotic bodies,conditioned medium,decellularized matrix,regeneration”为英文检索词。
1.1.5 检索文献类型 研究原著、系统综述、临床试验。
1.1.6 检索策略 以PubMed数据库检索策略为例,见图2。
1.2 入选标准
纳入标准:①所有关于牙髓干细胞及其衍生产物用于牙髓组织再生修复的相关文献;②所有关于牙髓干细胞及其衍生产物促进其他组织再生修复但与牙髓组织再生机制有关的文献,包括成骨、成血管、成神经等。
排除标准:①重复性研究的文献;②与文章主题不相关的文献;③资料无法提取的部分文献。
1.3 数据的提取及文献证据综合提炼 研究内容由1人独立提取,信息记录主要关注牙髓干细胞及其衍生物在牙髓组织再生方面的相关文献。计算机初检得到1 903篇文献。为了确保文献能够充分反映牙髓干细胞及其衍生物在牙髓再生中的研究进展,不仅对文献的题目和摘要进行了初步筛选,还进一步审查了文献的研究设计、方法和结果等内容,排除了重复性研究以及与主题无关的文献,以确保最终纳入的文献能够更准确代表该领域的前沿进展。最终依据严格的纳入和排除标准,纳入103篇文献进行综述,见图3。
中国组织工程研究杂志出版内容重点:干细胞;骨髓干细胞;造血干细胞;脂肪干细胞;肿瘤干细胞;胚胎干细胞;脐带脐血干细胞;干细胞诱导;干细胞分化;组织工程
然而,与其他类型的组织工程研究相比,牙髓组织工程的研究范围和深度仍显不足。在种子细胞的应用方面,虽然有研究已证实牙髓干细胞具备较强的自我更新能力和多向分化潜力,能够有效促进牙髓损伤后的再生修复,但这类方法通常依赖于活细胞移植。研究者们尝试通过优化支架结构或引入生物活性因子等策略,以提高干细胞活性和稳定性,但这些问题仍未得到有效解决,牙髓再生修复的效果仍然受到牙髓干细胞本身特性的限制。未来的研究应聚焦于优化支架材料和细胞外基质的构建,以支持牙髓干细胞的稳定生长和定向分化。理想的支架材料应具备可注射性、可缓释性和生物可降解性,通过调整孔隙率、刚度等结构特性,结合相关生物活性因子来模拟牙髓微环境,从而提高干细胞的增殖与分化潜力。此外,基因编辑技术可通过精确调控干细胞的基因表达,增强血管化、神经再生等关键功能,进一步提升牙髓干细胞的再生能力。通过结合支架设计、生物活性因子和基因编辑等工程化干细胞策略,未来牙髓干细胞移植有望在牙髓组织工程中实现更精准、高效的临床应用。 在这一背景下,无细胞疗法作为牙髓组织工程领域的重要研究方向,逐渐引起了广泛关注。无细胞疗法是通过利用细胞外基质、细胞因子、外泌体等生物活性分子,促进组织的修复和再生,而不依赖于活细胞的移植。与传统细胞疗法相比,无细胞疗法避免了细胞移植可能引发的免疫排斥反应和伦理争议,展现出更大的临床应用潜力。然而,尽管牙髓干细胞衍生物在体外和动物模型中展现出显著的再生能力和无细胞疗法的应用潜力,在临床转化方面仍面临多重挑战。
首先,牙髓干细胞衍生物的免疫原性、代谢稳定性以及长期使用中的潜在风险仍未得到充分验证。尽管已有大量研究探讨了牙髓干细胞衍生物中生长因子、细胞因子和细胞外基质成分的潜在作用,但具体分子机制和作用途径仍未被完全揭示。例如,牙髓干细胞来源细胞外囊泡在成牙本质细胞分化中的调控作用与信号通路仍需深入研究。同时,牙髓干细胞来源条件培养液和脱细胞基质中的关键成分及与微环境的相互作用机制尚不明确,这可能导致发生不良反应。此外,无细胞疗法的治疗效果在很大程度上依赖于细胞外基质、细胞因子和外泌体等生物活性分子的有效递送与释放,而这一过程需要相应的机制研究和支持。未来的研究应结合高通量测序、单细胞组学、蛋白质组学等前沿技术手段,全面深入地解析牙髓干细胞衍生物中的活性因子成分,明确在组织再生修复过程中的具体作用机制。此外,基于对这些机制的深入理解,未来研究还应致力于开发更为高效的活性因子释放系统,比如纳米颗粒、微囊、微针阵列等智能药物递送系统,并探索通过脂多糖、低氧预处理等条件调控衍生物的释放效果,实现对活性因子的精准调控,不仅包括剂量和释放速率的控制,还应综合考虑释放的靶向性和持续性,从而为临床应用提供更加个性化和定向的治疗方案。
其次,批次差异性、技术难度以及临床验证的时间和成本是制约牙髓干细胞衍生物无细胞疗法广泛应用的一大障碍。目前,由于缺乏统一的标准化大规模的制备流程和严格的质量控制体系,牙髓干细胞衍生物的不同研究和生产批次之间在成分、纯度及生物活性方面可能存在显著差异,从而影响临床疗效的一致性与稳定性。此外,无细胞疗法的实施通常依赖于复杂的生产工艺和精细的质量控制技术,如高通量筛选、活性因子的提取与纯化等,这些环节对技术支持和设施的要求较高,且伴随较高的成本,限制了在大规模临床应用中的推广。同时,临床研究的验证通常需要长期随访,以评估不同患者群体的疗效与安全性。这个过程不仅消耗大量资金和资源,还可能面临伦理等挑战,进而延长临床转化的周期并增加整体成本。随着精准医学的兴起,未来的牙髓再生治疗应更加注重个性化与靶向性,通过结合患者的临床特征(如年龄、牙髓损伤程度、免疫状况等),设计出更加个性化的治疗方案,精准选择合适的牙髓干细胞衍生物,从而显著提高治疗效果并降低不良反应的风险,加速临床应用的步伐。
3.2 作者综述区别于他人他篇的特点 与现有综述文献相比,此综述在多个方面展现出显著的创新性。首先,此综述聚焦于牙髓干细胞及其衍生物在牙髓再生中的最新研究进展,全面回顾并总结了该领域的相关研究成果。研究认为,牙髓干细胞及其衍生物通过促进干细胞的增殖与分化,能够显著推动牙髓再生,在牙髓损伤修复领域具有巨大的临床应用潜力,有望为牙髓组织工程带来创新性突破。其次,在探讨牙髓干细胞及其衍生物促进牙髓再生的机制时,此综述深入分析了影响再生效果的多重因素,整理并总结了若干关键机制。研究认为,牙髓组织的成功再生依赖于微环境的精确调控,尤其是细胞间信号传导及细胞外基质的重塑。在此基础上,牙髓干细胞及其衍生物通过多种生物因子信号通路、支架材料优化等手段,有效构建出一个有利于干细胞迁移和增殖的再生微环境,进一步促进干细胞向成牙本质、成血管及成神经等细胞类型的分化,从而推动牙髓组织的修复与再生。相关总结不仅深化了对牙髓再生微环境调控作用的认识,也突出了此综述在机制研究方面的创新性贡献。最后,此综述还深入探讨了当前研究在临床转化过程中所面临的技术挑战,并针对这些瓶颈提出了具体的解决方案。尤其在牙髓干细胞衍生物无细胞疗法的临床应用中,文中强调了治疗机制、生产工艺、安全性评估以及个性化治疗等方面的优化需求,为突破临床转化中的技术瓶颈提供了理论依据。通过对此综述的深入探讨,期望为牙髓再生领域的持续发展提供新的视角,并为未来技术进步和临床应用的实现提供有价值的见解与发展方向。
3.3 综述的局限性 尽管此综述对牙髓干细胞及其衍生物在牙髓再生中的研究进展进行了全面总结,但仍存在一定的局限性。首先,关于牙髓干细胞来源的凋亡小体、微囊泡和脱细胞基质在牙髓再生中的研究尚显匮乏,部分体外实验和临床试验的相关文献较少,未能在综述中深入探讨这一方面。其次,检索策略存在一定的局限性,采用的筛选和排除标准较为简化,这导致最终纳入的文献数量相对有限,未能充分覆盖该领域所有相关的研究成果。尤其在文献筛选过程中,部分可能具有重要参考价值的文献未能被纳入,造成了一定的遗漏。由于缺乏大规模、高质量的临床和实验数据,对某些分子机制仍未能完全阐明。最后,由于不同研究在实验设计、样本量及研究方法上的差异,所引用的数据可能受到这些因素的影响,导致结果的普遍性存在一定的不确定性。尽管此综述存在这些局限性,但仍为牙髓组织工程领域的后续研究提供了有价值的参考和启示。
3.4 综述的重要意义 此综述在总结牙髓干细胞及其衍生物的研究进展方面具有重要意义。首先,它系统性地梳理了牙髓干细胞及其衍生物在牙髓再生中的最新研究成果,为该领域的研究者提供了较为全面的参考框架。其次,此综述不仅揭示了当前研究中的关键问题和挑战,还为未来的研究方向提供了指导,具有一定的前瞻性和实践意义。
3.5 课题专家组对未来的建议 牙髓自我修复能力有限,难以通过机体自身防御系统实现有效修复,一旦发生炎症反应,容易引发不可逆的牙髓坏死,因此牙髓再生一直是组织工程研究的关键领域。牙髓损伤常由龋病、机械性创伤、化学性损伤以及其他外部因素引起,损伤主要发生在牙髓腔和根尖区域,其中根尖区的牙髓损伤尤为显著。尽管牙髓组织工程可以借鉴其他组织工程领域的先进技术,但其在研究和临床应用中依然面临一系列独特的挑战。例如,由于根管的狭窄结构,牙髓的血液供应受到限制,且只能通过小于
1 mm的根尖孔进行血液交换,这限制了营养物质的有效扩散及再生所需环境的构建。此外,牙髓的再生过程受到复杂微环境的调控,涉及细胞信号传导、免疫反应以及细胞外基质的作用等多方面因素的相互影响。因此,未来的研究不仅应充分借鉴其他领域的共性发现,还应着力解决牙髓再生中的特定问题,以提高再生效果和治疗策略的可行性。
中国组织工程研究杂志出版内容重点:干细胞;骨髓干细胞;造血干细胞;脂肪干细胞;肿瘤干细胞;胚胎干细胞;脐带脐血干细胞;干细胞诱导;干细胞分化;组织工程
文题释义:
牙髓干细胞:是一种来源于成人正畸牙、埋伏牙以及第三磨牙等牙髓组织的牙源性间充质干细胞,具有易获取、无伦理问题等优点。同时,牙髓干细胞具有自我更新和多向分化潜能,短期内可大量增殖,并能够定向分化为多种不同类型的细胞,包括成牙本质细胞、血管内皮细胞和成神经细胞等,被广泛应用于牙髓组织工程再生领域。
牙髓再生:是指在移除炎症或坏死的牙髓组织后,利用特定的治疗方法包括干细胞、生物活性因子及生物支架材料等,促使新的牙髓组织生长,从而恢复牙齿的正常生理功能和维持原有的生命活力。牙髓再生是牙髓治疗和再生牙科学的一个重要分支,尤其适用于年轻患者的受损或发育不全的牙齿。
#br#
中国组织工程研究杂志出版内容重点:干细胞;骨髓干细胞;造血干细胞;脂肪干细胞;肿瘤干细胞;胚胎干细胞;脐带脐血干细胞;干细胞诱导;干细胞分化;组织工程
近年来,细胞学、生物学和工程学技术的进展推动了牙髓组织工程的快速发展,其中,牙髓干细胞作为一种具有显著多向分化潜力的间充质干细胞,能够在特定诱导条件下分化为成牙本质、成血管和成神经等细胞,为牙髓-牙本质复合体的重建提供关键细胞来源,已成为牙髓组织工程领域中的研究热点。此外,牙髓干细胞衍生的细胞外囊泡、条件培养液和脱细胞基质等产物在牙髓再生修复中的研究也展现出巨大的潜力。本文综述了这些关键要素在牙髓组织再生修复领域中的最新研究成果与应用进展,旨在为不可逆性牙髓炎等临床治疗提供新思路,并为牙髓组织工程未来的发展提供理论支持。
#br#
中国组织工程研究杂志出版内容重点:干细胞;骨髓干细胞;造血干细胞;脂肪干细胞;肿瘤干细胞;胚胎干细胞;脐带脐血干细胞;干细胞诱导;干细胞分化;组织工程
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||