中国组织工程研究 ›› 2022, Vol. 26 ›› Issue (34): 5553-5561.doi: 10.12307/2022.468
• 生物材料综述 biomaterial review • 上一篇 下一篇
组织工程技术修复半月板损伤的替代策略
杜朝政,智佳佳,王 越,王新军,袁银鹏,王宇泽
- 山西医科大学第二医院骨科,山西省太原市 030001
-
收稿日期:2020-11-14接受日期:2020-12-25出版日期:2022-12-08发布日期:2022-04-16 -
通讯作者:王宇泽,博士,副主任医师,山西医科大学第二医院骨科,山西省太原市 030001 -
作者简介:杜朝政,男,1996年生,安徽省阜阳市人,汉族,山西医科大学第二医院在读硕士,医师,主要从事关节软骨修复研究。
Alternative strategies for tissue engineering repair of meniscus injury
Du Chaozheng, Zhi Jiajia, Wang Yue, Wang Xinjun, Yuan Yinpeng, Wang Yuze
- Department of Orthopedics, Second Hospital of Shanxi Medical University, Taiyuan 030001, Shanxi Province, China
-
Received:2020-11-14Accepted:2020-12-25Online:2022-12-08Published:2022-04-16 -
Contact:Wang Yuze, MD, Associate chief physician, Department of Orthopedics, Second Hospital of Shanxi Medical University, Taiyuan 030001, Shanxi Province, China -
About author:Du Chaozheng, Master candidate, Physician, Department of Orthopedics, Second Hospital of Shanxi Medical University, Taiyuan 030001, Shanxi Province, China
摘要:
文题释义:
组织各向异性:半月板组织由内到外可分为白-白区、红-白区和红-红区3个区域,白-白区主要由纤维软骨细胞、放射状分布的Ⅱ型胶原蛋白和较高浓度的蛋白聚糖组成,红-红区主要由成纤维细胞样细胞、沿半月板长轴环状分布的Ⅰ型胶原蛋白和较低浓度的蛋白聚糖组成,红-白区结构组成介于两区之间。因此,半月板组织内侧具有较高的压缩模量,外侧具有较高的拉伸模量。
支架:在半月板组织工程中,支架主要发挥塑造形态、模仿半月板组织各向异性、为干细胞提供生长环境、生物因子载体和加强支架力学特性的功能。通过进行组织工程支架形态的设计以达到契合不同形状的半月板损伤,同时向损伤处持续递送干细胞和生物因子促进组织愈合。
背景:目前的手术方法不能提供半月板再生和替换的长期解决方案,但组织工程技术可以提供替代的治疗策略。
目的:就半月板损伤修复的临床策略及组织工程研究现状作一综述。
方法:应用计算机检索PubMed数据库、MEDLINE数据库、Google学术数据库、CNKI中国期刊全文数据库、万方医学网数据库收录的文献,英文引文检索词为“meniscus injury;tissue engineering;meta-analysis;allografts;meniscus repair;scaffold;polymers;hydrogels;polyurethane; intestinal submucosa;polycaprolactone;polylactic;self-assembly;bone marrow MSC;meniscus derived stem cells;synovium derived MSCs;adipose derived MSC;co-culture;growth factor;transforming growth factor-beta;basic fibroblast growth factor;Insulin growth factor”,中文引文检索词为“半月板;间充质干细胞;支架;生长因子;组织工程;压缩模量;拉伸模量;水凝胶;静电纺丝”,检索期限为2005-01-01/2020-10-01。
结果与结论:半月板组织工程作为半月板治疗研究的热点方向,虽然在各个方面都取得了丰富的研究成果,但均缺乏大量长期的临床试验来验证结论的准确性。生物分子作为组织工程不可或缺的工具,目前仍然需要更多的人或动物实验来确定不同生物分子准确的作用信号通路、应用的最佳浓度和组合方式及生物分子的递送方式。尽管已发现很多种具有半月板表型分化潜力的干细胞,但对于各种干细胞在组织工程中的作用仍待深入研究,最具优势或最适合半月板组织工程的干细胞种类依然颇具争议。与此同时,半月板在细胞组成、细胞外基质成分及生物力学特性上与关节软骨和椎间盘等组织相似,并且关节软骨和椎间盘的修复再生策略同样备受关注,相似领域的研究进展可以相互借鉴、相互促进。
https://orcid.org/0000-0003-0750-5506(杜朝政)
中国组织工程研究杂志出版内容重点:生物材料;骨生物材料; 口腔生物材料; 纳米材料; 缓释材料; 材料相容性;组织工程
中图分类号:
引用本文
杜朝政, 智佳佳, 王 越, 王新军, 袁银鹏, 王宇泽. 组织工程技术修复半月板损伤的替代策略[J]. 中国组织工程研究, 2022, 26(34): 5553-5561.
Du Chaozheng, Zhi Jiajia, Wang Yue, Wang Xinjun, Yuan Yinpeng, Wang Yuze. Alternative strategies for tissue engineering repair of meniscus injury[J]. Chinese Journal of Tissue Engineering Research, 2022, 26(34): 5553-5561.
2.1.1 天然高分子材料 天然高分子材料主要有胶原蛋白、纤维蛋白、弹性蛋白、丝素蛋白、纤维素、海藻酸盐、明胶、琼脂糖、壳聚糖等,具有良好的生物相容性、可降解性,无毒。胶原蛋白作为天然半月板细胞外基质的主要成分具有优秀的生物相容性,已经成为半月板组织工程的首选,但由于极低的刚度和快速降解作用使其在临床中的应用受到限制[10]。海藻酸盐同样是半月板组织工程的热门材料,因为其支持半月板纤维软骨细胞表型的分化和蛋白聚糖的产生,可用来改善支架的黏弹性。一项系统评估4种生物医学级和2种非生物医学级海藻酸盐能力的研究显示,在3D水凝胶微环境中生物医学级高甘露糖醛酸含量的海藻酸盐在增强人类半月板纤维软骨细胞蛋白聚糖生产方面具有广阔的前景[11]。琼脂糖和明胶也具备作为半月板组织工程材料的潜力。BAHCECIOGLU等[12]通过3D打印半月板形聚己内酯支架,然后分别在内部和外部区域用琼脂糖和明胶甲基丙烯酸酯水凝胶浸渍来构建模仿天然组织的人造半月板,在培养载有猪纤维软骨细胞的构建体8周后,检测到琼脂糖的存在使糖胺聚糖的合成增加约4倍,而明胶甲基丙烯酸酯增加胶原蛋白的合成约50倍。丝素蛋白材料在实验中一般不单独应用,其与聚己内酯的复合材料支架已被证实拥有优秀的促进细胞黏附、增殖、分化及细胞外基质产生的性能,同时展示了卓越的生物力学功能[13-14]。目前许多天然高分子材料已被应用于临床前和动物实验中,这些材料更具体的生物、力学和化学方面的特性有待于更多的实验来发掘与验证。
2.1.2 脱细胞细胞外基质材料 天然半月板脱细胞细胞外基质支架已显示出与天然半月板相似的生物力学特性,并且具有低免疫原性、潜在的再生潜力和区域特定的生物活性[15],但其与自然半月板相比的生物力学性能(压缩和拉伸刚度)较差[16-18]。小肠黏膜下层是一种自然存在的脱细胞细胞外基质,已被公认为是组织工程的理想支架并广泛用于修复各种组织和器官[19]。最近的一项研究在大鼠模型中比较了3种真皮分离物(人、胎牛和交联猪)和两种小肠分离物(猪和交联猪)的生物活性,犬半月板细胞、滑膜细胞、肌腱成纤维细胞和骨髓祖细胞在5种支架中共培养,猪小肠分离物表现出最大促进这些细胞保留、浸润和存活的能力[20]。虽然小肠黏膜下层显示出显著的生物活性,并被认为可以诱导一些组织再生,但以此为基础构建的支架力学性质强度不足[21]。脱细胞细胞外基质材料在生物力学特性上的缺点是制约其应用的主要原因。
2.1.3 合成聚合物材料 考虑到天然聚合物的成本、可用性、加工、纯化和免疫反应,合成聚合物受到许多组织工程研究人员的青睐。目前常用的可降解合成聚合物有:聚己内酯、聚乙醇酸、聚乳酸及其共聚物、聚碳酸三甲乙烯和可降解聚氨酯等[13,22-24]。聚合物作为支架应该具有生物相容性和生物降解性,而且在植入人体一段时间后应该被原生组织所取代,其还应该允许原生细胞在聚合物组织或支架中附着、迁移和增殖。根据聚合物表面的不同,存在于介质中的溶解蛋白可能以不同的速率附着,这取决于存在于介质中不同蛋白质的亲和力及聚合物表面的生物吸引力[25]。聚己内酯的力学性能优异且与生物组织制品相匹配,但降解率极低及缺乏生物活性使其很少单独作为半月板支架材料。聚己内酯与天然材料所组成的复合材料,既能克服天然材料的力学特性不足缺陷,又能克服合成聚合物降解率极低及缺乏生物活性的缺点。研究人员合成了一种由再生丝素蛋白制成的新型高度互连的可缝合多孔支架,该支架在横向平面用3D打印聚己内酯网格增强,以增强缝合线的固定能力,结果显示用聚己内酯网眼增强丝素蛋白支架可使缝线保留强度提高至400%[26]。临床应用显示,聚氨酯支架在术后2年内的随访中取得了良好临床效果[27],聚氨酯支架似乎具有促进血管向内生长的潜力。KOCH 等[22]的研究发现,用装载或未装载间充质干细胞的聚氨酯支架治疗兔外侧无血管和血管区域大的约7 mm宽的半月板损伤,在两治疗组的整个修复组织中均检测到密集的血管形成。
2.1.4 复合聚合物材料 复合支架是由2种或2种以上化学成分和物理性质不同的聚合物合成的支架。使用复合聚合物的主要优点是可以在组合中获得期望的最佳性能,而这是在使用单一聚合物时无法实现的。两种不同聚合物形成的复合材料可以增加生物和机械性能,并最小化单一聚合物在复合材料中的局限性[6]。以合成聚合物聚己内酯为例,单纯的聚己内酯材料降解率低及生物活性差,但其具有良好的力学特性和可塑性;聚己内酯-丝素蛋白共混物具有优良的性能,聚己内酯-丝素蛋白支架较聚己内酯支架可更好地促进植入细胞的黏附、增殖、分化及细胞外基质的生成,同时极大地平衡生物力学性能和降解速率;聚己内酯-丝素蛋白支架较丝素蛋白支架显著增强了缝合线固定能力及压缩模量[13-14, 26];聚己内酯-半月板脱细胞细胞外基质电纺丝材料同样也显示出较单一材料更为优越的性质[28-29]。
2.1.5 无支架自组装 尽管支架具有优势,但是支架的使用也会导致降解相关毒性、应力屏蔽、细胞表型的改变和组织重构的阻碍[30],鉴于这些阻碍人们提出了无支架半月板技术。无支架方法分为自组织方法和自组装方法。自组织的再生需要外力干预(开放系统),在自组装过程中细胞在不借助外力的条件下再生组织(封闭系统)[31],常应用高密度球团培养法获得自组装组织,这需要高密度的细胞接种和特定生长因子的应用。自组装过程的主要优点是易于控制实验条件和通过种植特定的非黏连霉菌来改变初生组织的大小和厚度,这种方法最主要的局限是需要使用较高的细胞密度(1×1010-10×1010 L-1),这种高密度细胞的应用可以防止细胞附着,并通过减少自由能量来增强细胞间的通讯。钙黏蛋白跨膜蛋白和间隙连接增强了细胞间的相互作用,它们主要通过触发β-连环蛋白进一步激活了Wnt信号通路,从而进行细胞调节。连接蛋白是细胞连接形成过程中分泌的间隙连接蛋白,它在细胞聚集的组织形成中也发挥着重要作用[6]。通过使用生物化学和生物力学刺激,使用无支架方法获得的工程组织已获得与天然组织相似的功能特性。原生半月板组织的压缩和拉伸模量分别为0.1-0.15 MPa和10-30 MPa,无支架工程半月板的压缩和拉伸模量分别约为0.12 MPa和
5 MPa[32-33]。因此,无支架方法有可能规避与支架相关的挑战,产生具有生物力学功能的植入物。 各种半月板组织工程支架的特点见表1。

2.2 细胞来源 干细胞作为组织工程细胞的来源已得到广泛的研究和认可,因为它可以分化成软骨、骨和结缔组织等多种细胞,并且可以通过分泌免疫调节分子和旁分泌途径促进组织愈合[37]。干细胞具有广泛多样性,主要可分为胚胎干细胞、诱导多能干细胞与成体干细胞三类。成体干细胞包括造血干细胞、神经干细胞和间充质干细胞,其中间充质干细胞是目前临床上的主要研究目标。间充质干细胞主要具有以下几个优势:①来源广泛,可以从骨髓、脂肪、滑膜、肌肉和胎盘等组织中分离得到。②制备相对简单。③具备干细胞分化的多能性。④免疫原性低,安全性高。⑤不涉及社会伦理和法律方面的争议[38]。虽然间充质干细胞可以从各种组织中获得并具有共同的特征,但越来越多的报告描述了关于不同组织来源的间充质干细胞变异特性。一项研究通过收集人滑膜、半月板、关节内韧带、肌肉、脂肪组织和骨髓来分析集落形成细胞,发现所有这些细胞均表现出间充质干细胞特有的多能性和表面标记,但相对于关节外组织间充质干细胞的基因谱,关节内组织间充质干细胞和软骨细胞的基因谱表达更接近;在特定于关节内组织间充质干细胞的3个特征基因中选择富含脯氨酸-精氨酸的富含末端亮氨酸的重复蛋白进行分析,证实了软骨细胞和关节内组织间充质干细胞高表达富含脯氨酸-精氨酸的富含末端亮氨酸重复蛋白,滑膜间充质干细胞稳定表达富含脯氨酸-精氨酸的富含末端亮氨酸重复蛋白,而骨髓间充质干细胞在体外软骨形成过程中增加富含脯氨酸-精氨酸的富含末端亮氨酸重复蛋白表达,得到了关节内组织间充质干细胞的基因表达谱非常相似,而富含脯氨酸-精氨酸的富含末端亮氨酸重复蛋白高表达似乎是关节内组织间充质干细胞的特征[39]。该研究提示,关节内或者关节周围的间充质干细胞更适合作为软骨和半月板损伤修复再生的组织工程细胞来源。
2.2.1 骨髓来源间充质干细胞 目前骨髓治疗半月板损伤主要通过3种方式:①从骨髓中提取细胞,随后在实验室培养并最终在损伤部位移植。②骨髓抽吸浓缩直接植入损伤部位。③利用生长因子系统动员干细胞和其他骨髓前体[40]。虽然这3种应用方式均可显著促进半月板的再生,但治疗效应是由间充质细胞直接作用还是由某些刺激因子的分泌介导尚不清楚。缺乏对潜在机制的认识,是在临床实践中应用这些基于干细胞治疗策略的监管负担之一[41]。骨髓来源间充质干细胞具有优异的软骨分化倾向、Ⅱ型胶原蛋白沉积及蛋白基质合成能力,这些特性都十分有利于半月板的修复再生,然而骨髓来源间充质干细胞具有明显的肥大表型分化倾向,该倾向是其在半月板组织工程应用的一个难题。骨髓来源间充质干细胞作为临床上最先用于再生医学的细胞来源,已被用于临床前半月板修复研究。SHIMOMURA等[15]将骨髓来源间充质干细胞接种到去细胞半月板细胞外基质支架中,用含重组人转化生长因子β3的软骨形成培养基培养,发现内侧去细胞半月板细胞外基质促进了骨髓来源间充质干细胞的纤维软骨分化,而外侧去细胞半月板细胞外基质促进了骨髓来源间充质干细胞的成纤维细胞表型分化。KOCH等[22]建立了7 mm宽横跨兔半月板的血管和非血管区域的半月板病变动物模型,用加载和未加载骨髓来源间充质干细胞的聚氨酯支架进行治疗,术后6-12周两组都获得了良好的愈合和半月板样修复组织。虽然各个关于骨髓来源间充质干细胞应用于人体和动物实验的报道均得出其有利于半月板的修复与再生,然而高表达的肥大分化却是一个无法回避的障碍,肥大分化与钙化往往意味着愈合不良与失败,目前主要通过添加生长因子及与半月板纤维软骨细胞共培养的方式来抑制肥大分化的表达[42];同时,骨髓来源间充质干细胞的应用还受限于细胞来源的获取,骨髓间充质干细胞的分离是一个痛苦的侵入性过程,并且产率低[43]。
2.2.2 滑膜来源间充质干细胞 滑膜来源间充质干细胞可能是成年期关节修复的主要驱动因素[44-45]。新的证据表明,关节软骨和滑膜组织从相同基因配置的前体中发育而来,并且滑膜来源间充质干细胞有稳定的软骨分化潜力[46],与此同时滑膜是丰富的间充质干细胞来源[47],这些都说明了滑膜来源间充质干细胞具有优秀的软骨及半月板损伤修复潜力。滑膜来源间充质干细胞在滑膜组织中的定位尚未达成统一意见,滑膜组织在组织学上可分为表面、间质和血管周围区域3个区域,有研究显示,在3个区域中血管周区域的滑膜间充质干细胞具有最高的增殖和软骨形成潜能[48],该研究为高质量滑膜来源间充质干细胞的获取提供了参考。相对于其他组织来源的间充质干细胞,滑膜来源间充质干细胞似乎具有更加优秀的增殖分化及细胞外基质产生的潜力。SASAKI等[49]收集了6条犬的滑膜、髌骨下脂肪垫、腹股沟脂肪和骨髓组织,检查了4种组织来源的间充质干细胞的基因表位表达、增殖能力和三系分化潜能,结果显示:4种组织来源的贴壁细胞CD90和CD44均呈阳性,CD45和CD11b呈阴性,滑膜来源间充质干细胞的CD90阳性率高于脂肪来源和骨髓来源间充质干细胞;滑膜来源和髌骨下脂肪垫来源间充质干细胞显示出显著的增殖能力,并且4个种群都经历了三系分化,在软骨形成过程中滑膜来源间充质干细胞衍生的软骨颗粒湿质量大于其他3种来源。BAMI等[50]分离和描述兔骨髓、脂肪、滑膜、肌腱膜、韧带和肌腱来源间充质干细胞的特征,发现滑膜来源间充质干细胞在软骨生长、成骨、肌生成和肌腱生成方面表现出优势。基因的匹配对于滑膜来源间充质干细胞的治疗效果似乎存在显著的影响。OKUNO等[51]比较了同基因和同种异体滑膜间充质干细胞移植对大鼠半月板再生的影响,切除F344大鼠双膝内侧半月板前半部,在F344大鼠膝关节内注射来自F344(同基因移植)、Lewis(小错配移植)和ACI(大错配移植)的滑膜来源间充质干细胞,最终得到滑膜间充质干细胞同基因和小错配移植对半月板再生的促进作用优于大错配移植的结果。2013年,Katagiri等[52]在大鼠模型中观察到滑膜来源间充质干细胞聚集体的移植比关节内注射滑膜来源间充质干细胞悬浮液更有效地促进半月板的修复再生。虽然目前的许多动物实验已经证明了滑膜来源间充质干细胞的治疗半月板损伤的潜力,但仍然缺乏大型动物模型以及中长期实验来验证其长期临床效果。
2.2.3 脂肪来源间充质干细胞 脂肪组织包括嵌入细胞外基质的成熟脂肪细胞和血管周围的结缔组织,后者被称为基质血管部分。通过吸脂获得的基质血管部分除了血管周围干细胞外,还含有一群异质单核细胞,如脂肪前细胞、成纤维细胞、血管平滑肌细胞、内皮细胞、单核细胞、巨噬细胞和淋巴细胞。近年来这种未分化的基质血管部分已成为再生医学研究人员关注的对象,因为它代表着丰富的干细胞来源[53]。马、兔、猪和大鼠等动物实验已证实,脂肪来源间充质干细胞具有与骨髓来源间充质干细胞相似的成软骨分化功能,可以替代骨髓来源间充质干细胞作为软骨再生细胞来源[54-57]。由于脂肪组织基质血管部分简单的干细胞提取程序、微小的侵袭性和重复性、相对短的分离时间和高产量、大量的细胞产量和最小的不适,使其为现代医学许多领域非常有吸引力的干细胞来源。与来自皮下脂肪组织的间充质干细胞相比,来自膝盖髌下脂肪垫的间充质干细胞具有更强的成软骨潜能[58]。髌下脂肪垫在膝关节中通过稳定髌骨和减少摩擦来防止髌骨损伤,同时也具有修复膝关节损伤的效果,主要是因为髌下脂肪垫-脂肪来源间充质干细胞既可以分化为软骨,也可以分泌血小板来源生长因子、血管内皮生长因子和成纤维细胞生长因子,并且可在转化生长因子β作用情况下发挥抗凋亡作用[59]。一项研究对比了不同组织来源间充质干细胞的半月板组织产生能力,包括源自红-红区和白-白区域的半月板细胞、人类骨髓来源、滑膜来源及髌下脂肪垫来源的间充质干细胞,将所有细胞接种到对齐的电纺I型胶原蛋白支架上,并随机地包封在3组分水凝胶中产生单层或多层构建体,在具有特定生长因子的培养基中培养2周后,所有细胞类型经过转化生长因子β1和转化生长因子β3的处理均会增加Ⅰ型胶原蛋白、软骨寡聚基质蛋白、腱生蛋白C和Scleraxis基因的表达及Ⅰ型胶原蛋白的沉积;然而与其他研究的细胞类型相比,髌下脂肪垫-间充质干细胞产生的半月板样组织具有更高的半月板基因表达、更好的机械性能和细胞分布[34]。由于脂肪来源间充质干细胞作为半月板组织工程修复细胞来源的时间尚短,因此目前文献研究缺乏大型动物数据及中长期随访数据,因此需要更长时间及更多的实验数据来了解脂肪来源间充质干细胞作为组织工程细胞的潜力。
2.2.4 软骨来源祖细胞 与以上不同组织来源的间充质干细胞相似,正常人类(非关节炎)关节软骨中也含有组织特异性的干细胞,这些细胞通常被称为软骨来源祖细胞[60]。通过纤维连接蛋白差异黏附实验可以有效地从健康关节软骨中获取软骨来源祖细胞,其已经被证实具有分化成骨、软骨和脂肪组织的能力,能够表达间充质干细胞标志性的表面标记CD44、CD49e、CD90、CD105、CD146和CD166[61-62]。WILLIAMS等[60]发现软骨来源祖细胞克隆群体中具有高端粒酶活性和维持端粒长度的能力,这是间充质干细胞的典型特征。软骨来源祖细胞在形态上表现为纤维软骨细胞,具有较高的集落形成效率、Notcht基因表达和软骨分化潜力[63-64];具有较低的肥大分化倾向,避免了成熟关节软骨细胞体外扩展培养的终末分化及去分化,提示软骨来源祖细胞是一种优秀的半月板修复再生细胞来源[64]。然而,限制软骨来源祖细胞在临床上应用的是其在软骨中较低的细胞含量,据报道软骨来源祖细胞占正常关节软骨细胞的(1.47±0.16)%。JAYASURIYA等[61]发现从健康人关节软骨中分离出的软骨来源祖细胞,与骨髓间充质干细胞一样对趋化因子基质细胞来源因子1有反应,它们可以成功迁移到半月板组织损伤区域,促进胶原蛋白桥接穿过内侧半月板撕裂;与骨髓来源间充质干细胞相比,软骨来源祖细胞在单层培养和半月板修复的移植器官培养模型中细胞肥大标志物表达降低。这项研究表明,来自健康人类软骨的软骨来源祖细胞可能是纤维软骨组织修复再生的重要工具,因为其可以抵抗细胞肥大,并在趋化因子信号传导的反应下动员起来。虽然该项研究取得了理想的结论,但仍需要更多的实验数据来支持和验证这一结论。
2.2.5 半月板来源干细胞 近年来,半月板中存在的一种具有多种分化潜力及优秀克隆能力的细胞越来越受人们的关注。1972年CAMERON等[65]在半月板的表面区域发现一种细胞,这些细胞呈扁平梭形形态,无细胞延伸,可能是具有治疗和再生能力的特异性祖细胞。GUI等[66]、GAMER等[67]分别在兔和小鼠的半月板中发现特异性祖细胞,兔半月板中的祖细胞表达出高水平的干细胞表面标记蛋白,包括SSEA-4、Nanog和核蛋白,并且具有高水平的Ⅱ型胶原蛋白表达;小鼠半月板中的祖细胞也表达出多种干细胞表面标记蛋白,包括CD44、Sca-1、CD90和CD73,并且具有较高水平的糖蛋白和I型胶原蛋白表达。有研究使用胶原酶消化人半月板组织且低密度播种细胞形成聚落,流式细胞术检测显示,该种细胞的表面标记蛋白CD44、CD105、CD29、CD90和CD166均为阳性,说明半月板来源干细胞具有类似于干细胞的特性,而且半月板来源干细胞表现出比骨髓来源和滑膜来源间充质干细胞更高的Ⅱ型胶原蛋白表达水平。由此可见,半月板来源干细胞确实在半月板组织工程的应用中具有巨大的潜力。
2.2.6 细胞共培养 在半月板组织工程的运用中,单一来源间充质干细胞往往无法达到天然半月板的各项生理、生化及生物力学指标,例如骨髓来源间充质干细胞在应用过程中总是表现出明显的肥大分化倾向。近些年的研究表明,两种不同的细胞共培养可达到加强半月板修复或改善支架性能的效果。MCCORRY等[68-70]在人和动物的细胞实验中发现,骨髓间充质干细胞与半月板纤维软骨细胞共培养的沉淀物中I型胶原蛋白、Ⅱ型胶原蛋白和聚集蛋白聚糖的mRNA表达上调,与骨髓来源间充质干细胞的单培养相比减少了间充质干细胞的肥大分化。SUN等[71-73]的研究发现,骨髓来源间充质干细胞与软骨细胞共培养可以促进组织中糖胺聚糖和胶原蛋白的合成,以及骨髓来源间充质干细胞向透明软骨细胞表型分化。不同的细胞比例得到的结果也不相同,为了获得最佳骨髓间充质干细胞与纤维软骨细胞种植比例,HAGMEIJER等[74]以细胞微球的形式在Ⅰ型胶原水凝胶中培养不同比例的人半月板细胞和骨髓间充质干细胞,共培养体系中观察到了缝隙连接的细胞通讯,并且半月板细胞/骨髓间充质干细胞为20∶80和10∶90时水凝胶中氨基多糖和胶原蛋白的含量最高,在颗粒培养中也发现了同样的统计趋势;并且与静态接种的种植复合体相比,注射种植复合体中的细胞明显增多、细胞分布更加均匀。这种共培养模式不仅对于骨髓来源间充质干细胞适用,对于滑膜来源间充质干细胞同样有促进效果。XIE等[75]设计了不同比率(3/1、1/1和1/3)滑膜来源间充质干细胞和半月板细胞的共培养系统,以滑膜来源间充质干细胞或半月板细胞的单一培养作为对照,发现1/3的滑膜间充质干细胞/半月板细胞共培养系统显示出最好的结果。HADIDI等[76]将自体关节软骨细胞与半月板、腱或韧带细胞共培养而成的自组装半月板形构建体,与转化生长因子β1在无血清培养基中培养4周,发现就力学性能和生化含量而言,使用肌腱细胞和韧带纤维细胞改造的构建体与使用半月板细胞改造的构建体等效或更高。对于共培养的研究才刚刚起步,相信以后会有更多的细胞组合应用到半月板组织工程中。半月板组织工程细胞来源见表2。

2.3 生物分子 生物分子是个体在生长发育过程中控制生长、增殖和细胞分化的重要分子[84],主要包括生长因子、受体、细胞因子、以碳水化合物为基础的生物分子、蛋白质和肽、脂类及其衍生物,它们在组织工程中发挥着加快组织整合、促进结构完整性、改善细胞增殖和增强组织的功能参数的作用。生物分子从支架上释放的效率和持续时间取决于生物分子的稳定性和可用性,影响生物分子可用性的因素有交联密度、聚合物的pH值和分子质量、降解速率及对生物分子的亲和力[85]。
在组织工程中,许多生长因子因其独特的功能而被应用,例如生物活性信号分子通过与细胞表面的特异性受体结合来实现细胞间的相互作用。常用的生物活性信号分子有血小板来源生长因子、骨形态发生蛋白、转化生长因子β、胰岛素样生长因子、血管内皮生长因子、表皮生长因子和成纤维细胞生长因子,其中成纤维细胞生长因子、转化生长因子β及胰岛素样生长因子最常用于临床试验,它们可增强细胞附着、增殖和分化的过程[86-87]。生长因子在创面愈合、细胞外基质分泌和组织再生中也发挥着重要作用。
2.3.1 成纤维细胞生长因子 成纤维细胞生长因子作为一种生物活性蛋白在组织工程的研究中主要表现以下几点生物功能:促进种植细胞增殖扩展;整合天然细胞、组织和支架;抑制骨形态发生蛋白的功能[88-90]。IONESCU等[88]在电纺丝聚丙内酯-己内酯支架中填充天然组织和细胞,培养在不同生长因子配方的培养基中4-8周,发现短期给予碱性成纤维细胞生长因子能提高幼稚和成熟支架组织的整合强度。CHIU等[90]在生物反应器中使用不同的培养基组成、不同数量的血清及有无生长因子培养软骨细胞,发现相对于未添加生长因子的生物反应器来说,添加生长因子(包括碱性成纤维细胞生长因子和转化生长因子β2)生物反应器中培养的构建物更厚、DNA和糖胺聚糖含量更大,也显示了最明显的蛋白多糖和Ⅱ型胶原染色。SONG等[80]在进行滑膜来源间充质干细胞与半月板软骨细胞共培养时发现,滑膜来源间充质干细胞可增强半月板软骨细胞的增殖和胶原合成,并且此作用可通过中和抗成纤维细胞生长因子1的抗体来阻断。
2.3.2 转化生长因子β 转化生长因子作为多功能细胞因子家族具有很多亚型(常用亚型转化生长因子β1、β3),并且作为受体激酶开启信息级联放大体系,激活多种下游基质和蛋白。转化生长因子同样也具有类似促进半月板再生的功能,但功能上特点上略有不同。转化生长因子的主要功能有:促进种植细胞的增殖扩展;促进间充质干细胞向半月板表型分化;促进胶原蛋白和糖胺聚糖的合成沉淀;整合天然细胞、组织和支架[86,90-92]。
2018年LIANG等[86]研究滑液来源间充质干细胞向半月板纤维软骨细胞表型分化的能力,发现没有补充生长因子(转化生长因子β3和胰岛素样生长因子1)的人半月板衍生细胞外基质不能使滑液来源间充质干细胞分化为半月板表型。精确地控制生长因子在组织缺损中持续发挥效用是组织工程应用目标之一,虽然转化生长因子β1在促进软骨形成中发挥关键作用,但治疗应用受限于其结构不稳定而易分解,导致软骨形成效率低下。CHOI等[93]开发了在可注射可见蓝光诱导的壳聚糖水凝胶体系中稳定转化生长因子β1释放的策略,成功调节了转化生长因子β1的递送。
2.3.3 胰岛素样生长因子 胰岛素样生长因子1是胰岛素相关肽的一种,在儿童生长发育和成年人组织代谢修复中起重要作用。PUETZER等[94]将牛半月板纤维软骨细胞接种到20 g/L的海藻酸盐中,以硫酸钙进行交联,然后在培养基中添加或不添加100 μg/L胰岛素样生长因子1的情况下构建组织工程半月板物,培养4周后进行了组织学、免疫组织学、生化和机械分析。结果显示,胰岛素样生长因子1治疗4周后与0周相比糖胺聚糖增加了26倍、胶原蛋白含量增加了10倍,与第2周相比平衡模量增加了3倍;经过胰岛素样生长因子1处理的半月板具有约60%的原生半月板组织糖胺聚糖含量,并且在培养2周后其压缩平衡模量与原生组织的特性相匹配;此外,胰岛素样生长因子1处理的半月板形成了一个与天然组织相似的表层结构,细胞拉长,胶原纤维平行于表面,存在I型和Ⅱ型胶原,并积累了润滑剂。同时,胰岛素样生长因子1在促进间充质干细胞向半月板表型分化也起着重要的作用[86]。半月板组织工程生物分子介绍见表3。

2.4 生物机械刺激 半月板在膝关节内不但承担压缩应力,同时承受环向拉伸应力和组织内的剪切力,因此机械应力刺激对半月板组织工程具有重要意义。在过去的研究中,对半月板工程生物力学功能的研究主要集中在复制固有的带状排列方式和基质层组织的生化成分。ZELLNER等[95]发现每天应用4 h的1 Hz正弦静水压力(0.55-5.03 MPa之间)刺激人纤维软骨细胞,会使内、外侧部分半月板纤维软骨细胞的Ⅱ型胶原蛋白存在很大差异,结果显示机械刺激可以用来帮助构建区域结构。PUETZER等[96]的研究发现,使用5%-10%压缩应变的生物反应器可在外区产生与原生组织排列相似的纤维性胶原基质。生物力学加载可以调节体外半月板的细胞分化和细胞外基质的产生,压缩应力加载可以促进糖胺聚糖生成及软骨细胞的形成,而拉伸应力加载可增多Ⅰ型胶原蛋白合成和提高支架抗拉强度[97]。WOO等[98]模拟微重力对原代人半月板细胞和脂肪间充质干细胞共培养的影响,生化、实时定量PCR和免疫荧光测定显示,模拟微重力显著增强了原半月板细胞和脂肪来源间充质干细胞共培养中软骨基质的形成,支架内区域的圆形细胞对压缩应变有响应,表明压缩载荷可诱导内源性和外源性干细胞/祖细胞的软骨形成,促进Ⅱ型胶原蛋白和糖胺聚糖的产生;支架外区域的成纤维细胞样细胞呈拉长状态,提示沿周向和放射状施加张力可能刺激干细胞/祖细胞分化为成纤维细胞,促进I型胶原蛋白的产生。因此适当的机械应力刺激有利于模拟自然半月板组织的各向异性和带性组织排列方式。

| [1] BAQUIE P, BRUKNER P. Injuries presenting to an Australian sports medicine centre: a 12-month study. Clin J Sport Med. 1997;7(1):28-31. [2] GEE SM, TENNENT DJ, CAMERON KL, et al. The Burden of Meniscus Injury in Young and Physically Active Populations. Clin Sports Med. 2020;39(1):13-27. [3] GUPTA R, KAPOOR A, SONI A, et al. Anterior cruciate ligament tear due to non-contact mode of injury associated with higher incidence of meniscal and chondral damage. J Clin Orthop Trauma. 2020;11(Suppl 3): S342-342S345. [4] POULSEN E, GONCALVES GH, BRICCA A, et al. Knee osteoarthritis risk is increased 4-6 fold after knee injury - a systematic review and meta-analysis. Br J Sports Med. 2019;53(23):1454-1463. [5] FAUCETT SC, GEISLER BP, CHAHLA J, et al. Meniscus Root Repair vs Meniscectomy or Nonoperative Management to Prevent Knee Osteoarthritis After Medial Meniscus Root Tears: Clinical and Economic Effectiveness. Am J Sports Med. 2019;47(3):762-769. [6] PILLAI MM, GOPINATHAN J, SELVAKUMAR R, et al. Human Knee Meniscus Regeneration Strategies: a Review on Recent Advances. Curr Osteoporos Rep. 2018;16(3):224-235. [7] MATSIKO A, GLEESON JP, O’BRIEN FJ. Scaffold mean pore size influences mesenchymal stem cell chondrogenic differentiation and matrix deposition. Tissue Eng Part A. 2015;21(3-4):486-497. [8] ZHANG ZZ, JIANG D, DING JX, et al. Role of scaffold mean pore size in meniscus regeneration. Acta Biomater. 2016;43:314-326. [9] BAKER BM, MAUCK RL. The effect of nanofiber alignment on the maturation of engineered meniscus constructs. Biomaterials. 2007; 28(11):1967-1977. [10] HEO J, KOH RH, SHIM W, et al. Riboflavin-induced photo-crosslinking of collagen hydrogel and its application in meniscus tissue engineering. Drug Deliv Transl Res. 2016;6(2):148-158. [11] REY-RICO A, KLICH A, CUCCHIARINI M, et al. Biomedical-grade, high mannuronic acid content (BioMVM) alginate enhances the proteoglycan production of primary human meniscal fibrochondrocytes in a 3-D microenvironment. Sci Rep. 2016;6:28170. [12] BAHCECIOGLU G, HASIRCI N, BILGEN B, et al. A 3D printed PCL/hydrogel construct with zone-specific biochemical composition mimicking that of the meniscus. Biofabrication. 2019;11(2):025002. [13] LI Z, WU N, CHENG J, et al. Biomechanically, structurally and functionally meticulously tailored polycaprolactone/silk fibroin scaffold for meniscus regeneration. Theranostics. 2020;10(11):5090-5106. [14] CENGIZ IF, MAIA FR, DA SILVA MORAIS A, et al. Entrapped in cage (EiC) scaffolds of 3D-printed polycaprolactone and porous silk fibroin for meniscus tissue engineering. Biofabrication. 2020;12(2):025028. [15] SHIMOMURA K, ROTHRAUFF BB, TUAN RS. Region-Specific Effect of the Decellularized Meniscus Extracellular Matrix on Mesenchymal Stem Cell-Based Meniscus Tissue Engineering. Am J Sports Med. 2017;45(3): 604-611. [16] CHEN Y, CHEN J, ZHANG Z, et al. Current advances in the development of natural meniscus scaffolds: innovative approaches to decellularization and recellularization. Cell Tissue Res. 2017;370(1):41-52. [17] ROTHRAUFF BB, SHIMOMURA K, GOTTARDI R, et al. Anatomical region-dependent enhancement of 3-dimensional chondrogenic differentiation of human mesenchymal stem cells by soluble meniscus extracellular matrix. Acta Biomater. 2017;49:140-151. [18] ROMANAZZO S, VEDICHERLA S, MORAN C, et al. Meniscus ECM-functionalised hydrogels containing infrapatellar fat pad-derived stem cells for bioprinting of regionally defined meniscal tissue. J Tissue Eng Regen Med. 2018;12(3):e1826-e1835. [19] GUO Q, LI CB, SHEN XZ, et al. A review of research on the application of small intestinal submucosa in repairing osteoarticular injury. Zhongguo Gu Shang. 2016;29(5):482-486. [20] COOK JL, FOX DB, KUROKI K, et al. In vitro and in vivo comparison of five biomaterials used for orthopedic soft tissue augmentation. Am J Vet Res. 2008;69(1):148-156. [21] MAKRIS EA, HADIDI P, ATHANASIOU KA. The knee meniscus: structure-function, pathophysiology, current repair techniques, and prospects for regeneration. Biomaterials. 2011;32(30):7411-7431. [22] KOCH M, ACHATZ FP, LANG S, et al. Tissue Engineering of Large Full-Size Meniscus Defects by a Polyurethane Scaffold: Accelerated Regeneration by Mesenchymal Stromal Cells. Stem Cells Int. 2018;2018:8207071. [23] BAEK J, CHEN X, SOVANI S, et al. Meniscus tissue engineering using a novel combination of electrospun scaffolds and human meniscus cells embedded within an extracellular matrix hydrogel. J Orthop Res. 2015;33(4):572-583. [24] FUKUSHIMA K. Poly(trimethylene carbonate)-based polymers engineered for biodegradable functional biomaterials. Biomater Sci. 2016;4(1):9-24. [25] SUTTON D, NASONGKLA N, BLANCO E, et al. Functionalized micellar systems for cancer targeted drug delivery. Pharm Res. 2007;24(6): 1029-1046. [26] CENGIZ IF, PEREIRA H, ESPREGUEIRA-MENDES J, et al. Suturable regenerated silk fibroin scaffold reinforced with 3D-printed polycaprolactone mesh: biomechanical performance and subcutaneous implantation. J Mater Sci Mater Med. 2019;30(6):63. [27] SCHÜTTLER KF, PÖTTGEN S, GETGOOD A, et al. Erratum to: Improvement in outcomes after implantation of a novel polyurethane meniscal scaffold for the treatment of medial meniscus deficiency. Knee Surg Sports Traumatol Arthrosc. 2016;24(9):3045. [28] GAO S, GUO W, CHEN M, et al. Fabrication and characterization of electrospun nanofibers composed of decellularized meniscus extracellular matrix and polycaprolactone for meniscus tissue engineering. J Mater Chem B. 2017;5(12):2273-2285. [29] GAO S, CHEN M, WANG P, et al. An electrospun fiber reinforced scaffold promotes total meniscus regeneration in rabbit meniscectomy model. Acta Biomater. 2018;73:127-140. [30] LEE JK, LINK JM, HU J, et al. The Self-Assembling Process and Applications in Tissue Engineering. Cold Spring Harb Perspect Med. 2017;7(11):a025668. [31] ATHANASIOU KA, ESWARAMOORTHY R, HADIDI P, et al. Self-organization and the self-assembling process in tissue engineering. Annu Rev Biomed Eng. 2013;15:115-136. [32] GUNJA NJ, HUEY DJ, JAMES RA, et al. Effects of agarose mould compliance and surface roughness on self-assembled meniscus-shaped constructs. J Tissue Eng Regen Med. 2009;3(7):521-530. [33] MAKRIS EA, MACBARB RF, PASCHOS NK, et al. Combined use of chondroitinase-ABC, TGF-β1, and collagen crosslinking agent lysyl oxidase to engineer functional neotissues for fibrocartilage repair. Biomaterials. 2014;35(25):6787-6796. [34] BAEK J, SOVANI S, CHOI W, et al. Meniscal Tissue Engineering Using Aligned Collagen Fibrous Scaffolds: Comparison of Different Human Cell Sources. Tissue Eng Part A. 2018;24(1-2):81-93. [35] KREMER A, RIBITSCH I, REBOREDO J, et al. Three-Dimensional Coculture of Meniscal Cells and Mesenchymal Stem Cells in Collagen Type I Hydrogel on a Small Intestinal Matrix-A Pilot Study Toward Equine Meniscus Tissue Engineering. Tissue Eng Part A .2017;23(9-10):390-402. [36] ACHATZ FP, KUJAT R, PFEIFER CG, et al. In Vitro Testing of Scaffolds for Mesenchymal Stem Cell-Based Meniscus Tissue Engineering-Introducing a New Biocompatibility Scoring System. Materials (Basel) 2016;9(4):1996-1944. [37] CAPLAN AI, DENNIS JE. Mesenchymal stem cells as trophic mediators. J Cell Biochem. 2006;98(5):1076-84. [38] UCCELLI A, MORETTA L, PISTOIA V. Mesenchymal stem cells in health and disease. Nat Rev Immunol. 2008;8(9):726-736. [39] SEGAWA Y, MUNETA T, MAKINO H, et al. Mesenchymal stem cells derived from synovium, meniscus, anterior cruciate ligament, and articular chondrocytes share similar gene expression profiles. J Orthop Res. 2009;27(4):435-441. [40] MARMOTTI A, DE GIROLAMO L, BONASIA DE, et al. Bone marrow derived stem cells in joint and bone diseases: a concise review. Int Orthop. 2014;38(9):1787-1801. [41] ANGELE P, KUJAT R, KOCH M, et al. Role of mesenchymal stem cells in meniscal repair. J Exp Orthop. 2014;1(1):12. [42] TWOMEY-KOZAK J, JAYASURIYA CT. Meniscus Repair and Regeneration: A Systematic Review from a Basic and Translational Science Perspective. Clin Sports Med. 2020;39(1):125-163. [43] WANG J, CHEN Z, SUN M, et al. Characterization and therapeutic applications of mesenchymal stem cells for regenerative medicine. Tissue Cell. 2020;64:101330. [44] MCGONAGLE D, BABOOLAL TG, JONES E. Native joint-resident mesenchymal stem cells for cartilage repair in osteoarthritis. Nat Rev Rheumatol. 2017;13(12):719-730. [45] TO K, ZHANG B, ROMAIN K, et al. Synovium-Derived Mesenchymal Stem Cell Transplantation in Cartilage Regeneration: A PRISMA Review of in vivo Studies. Front Bioeng Biotechnol. 2019;7:314. [46] CALDWELL KL, WANG J. Cell-based articular cartilage repair: the link between development and regeneration. Osteoarthritis Cartilage. 2015;23(3):351-362. [47] ZUPAN J, DROBNIČ M, STRAŽAR K. Synovium-Derived Mesenchymal Stem/Stromal Cells and their Promise for Cartilage Regeneration. Adv Exp Med Biol. 2020;1212:87-106. [48] MIZUNO M, KATANO H, MABUCHI Y, et al. Specific markers and properties of synovial mesenchymal stem cells in the surface, stromal, and perivascular regions. Stem Cell Res Ther. 2018;9(1):123. [49] SASAKI A, MIZUNO M, OZEKI N, et al. Canine mesenchymal stem cells from synovium have a higher chondrogenic potential than those from infrapatellar fat pad, adipose tissue, and bone marrow. PLoS One. 2018;13(8):e0202922. [50] BAMI M, SARLIKIOTIS T, MILONAKI M, et al. Superiority of synovial membrane mesenchymal stem cells in chondrogenesis, osteogenesis, myogenesis and tenogenesis in a rabbit model. Injury. 2020;51(12):2855-2865. [51] OKUNO M, MUNETA T, KOGA H, et al. Meniscus regeneration by syngeneic, minor mismatched, and major mismatched transplantation of synovial mesenchymal stem cells in a rat model. J Orthop Res. 2014; 32(7):928-936. [52] KATAGIRI H, MUNETA T, TSUJI K, et al. Transplantation of aggregates of synovial mesenchymal stem cells regenerates meniscus more effectively in a rat massive meniscal defect. Biochem Biophys Res Commun. 2013;435(4):603-609. [53] JAMES AW, ZARA JN, CORSELLI M, et al. An abundant perivascular source of stem cells for bone tissue engineering. Stem Cells Transl Med. 2012;1(9):673-84. [54] PAK J, LEE JH, LEE SH. Regenerative repair of damaged meniscus with autologous adipose tissue-derived stem cells. Biomed Res Int. 2014; 2014:436029. [55] GONZÁLEZ-FERNÁNDEZ ML, PÉREZ-CASTRILLO S, SÁNCHEZ-LÁZARO JA, et al. Assessment of regeneration in meniscal lesions by use of mesenchymal stem cells derived from equine bone marrow and adipose tissue. Am J Vet Res. 2016;77(7):779-788. [56] XIE X, WANG Y, ZHAO C, et al. Comparative evaluation of MSCs from bone marrow and adipose tissue seeded in PRP-derived scaffold for cartilage regeneration. Biomaterials. 2012;33(29):7008-7018. [57] TESSARO I, DI GIANCAMILLO A, BENASCIUTTI E, et al. Characterization of different in vitro culture conditions to induce a fibro-chondrogenic differentiation of swine adipose-derived stem cells. J Biol Regul Homeost Agents. 2018;32(6 Suppl. 1):97-103. [58] LOPA S, COLOMBINI A, STANCO D, et al. Donor-matched mesenchymal stem cells from knee infrapatellar and subcutaneous adipose tissue of osteoarthritic donors display differential chondrogenic and osteogenic commitment. Eur Cell Mater. 2014;27:298-311. [59] TAKATOKU K, SEKIYA H, HAYASHI M, et al. Influence of fat pad removal on patellar tendon length during growth. Knee Surg Sports Traumatol Arthrosc. 2005;13(8):706-713. [60] WILLIAMS R, KHAN IM, RICHARDSON K, et al. Identification and clonal characterisation of a progenitor cell sub-population in normal human articular cartilage. PLoS One. 2010;5(10):e13246. [61] JAYASURIYA CT, TWOMEY-KOZAK J, NEWBERRY J, et al. Human Cartilage-Derived Progenitors Resist Terminal Differentiation and Require CXCR4 Activation to Successfully Bridge Meniscus Tissue Tears. Stem Cells. 2019;37(1):102-114. [62] KHAN IM, BISHOP JC, GILBERT S, et al. Clonal chondroprogenitors maintain telomerase activity and Sox9 expression during extended monolayer culture and retain chondrogenic potential. Osteoarthritis Cartilage. 2009;17(4):518-528. [63] JAYASURIYA CT, CHEN Q. Potential benefits and limitations of utilizing chondroprogenitors in cell-based cartilage therapy. Connect Tissue Res. 2015;56(4):265-271. [64] MARCUS P, DE BARI C, DELL’ACCIO F, et al. Articular Chondroprogenitor Cells Maintain Chondrogenic Potential but Fail to Form a Functional Matrix When Implanted Into Muscles of SCID Mice. Cartilage. 2014; 5(4):231-240. [65] CAMERON HU, MACNAB I. The structure of the meniscus of the human knee joint. Clin Orthop Relat Res. 1972;89:215-219. [66] GUI J, ZHANG J, HUANG H. Isolation and characterization of meniscus derived stem cells from rabbit as a possible treatment for damaged meniscus. Curr Stem Cell Res Ther. 2015;10(4):353-363. [67] GAMER LW, SHI RR, GENDELMAN A, et al. Identification and characterization of adult mouse meniscus stem/progenitor cells. Connect Tissue Res. 2017;58(3-4):238-245. [68] MCCORRY MC, PUETZER JL, BONASSAR LJ. Characterization of mesenchymal stem cells and fibrochondrocytes in three-dimensional co-culture: analysis of cell shape, matrix production, and mechanical performance. Stem Cell Res Ther. 2016;7:39. [69] SALIKEN DJ, MULET-SIERRA A, JOMHA NM, et al. Decreased hypertrophic differentiation accompanies enhanced matrix formation in co-cultures of outer meniscus cells with bone marrow mesenchymal stromal cells. Arthritis Res Ther. 2012;14(3):R153. [70] MATTHIES NF, MULET-SIERRA A, JOMHA NM, et al. Matrix formation is enhanced in co-cultures of human meniscus cells with bone marrow stromal cells. J Tissue Eng Regen Med. 2013;7(12):965-973. [71] SUN H, HUANG Y, ZHANG L, et al. Co-culture of bone marrow stromal cells and chondrocytes in vivo for the repair of the goat condylar cartilage defects. Exp Ther Med. 2018;16(4):2969-2977. [72] PLEUMEEKERS MM, NIMESKERN L, KOEVOET J, et al. Trophic effects of adipose-tissue-derived and bone-marrow-derived mesenchymal stem cells enhance cartilage generation by chondrocytes in co-culture. PLoS One. 2018;13(2):e0190744. [73] KO CY, KU KL, YANG SR, et al. In vitro and in vivo co-culture of chondrocytes and bone marrow stem cells in photocrosslinked PCL-PEG-PCL hydrogels enhances cartilage formation. J Tissue Eng Regen Med. 2016;10(10):E485-485E496. [74] HAGMEIJER MH, VONK LA, FENU M, et al. Meniscus regeneration combining meniscus and mesenchymal stromal cells in a degradable meniscus implant: an in vitro study. Eur Cell Mater. 2019;38:51-62. [75] XIE X, ZHU J, HU X, et al. A co-culture system of rat synovial stem cells and meniscus cells promotes cell proliferation and differentiation as compared to mono-culture. Sci Rep. 2018;8(1):7693. [76] HADIDI P, PASCHOS NK, HUANG BJ, et al. Tendon and ligament as novel cell sources for engineering the knee meniscus. Osteoarthritis Cartilage. 2016;24(12):2126-2134. [77] ZELLNER J, PATTAPPA G, KOCH M, et al. Autologous mesenchymal stem cells or meniscal cells: what is the best cell source for regenerative meniscus treatment in an early osteoarthritis situation. Stem Cell Res Ther. 2017;8(1):225. [78] KOCH M, HAMMER S, FUELLERER J, et al. Bone Marrow Aspirate Concentrate for the Treatment of Avascular Meniscus Tears in a One-Step Procedure-Evaluation of an In Vivo Model. Int J Mol Sci 2019; 20(5):1120. [79] HORIE M, SEKIYA I, MUNETA T, et al. Intra-articular Injected synovial stem cells differentiate into meniscal cells directly and promote meniscal regeneration without mobilization to distant organs in rat massive meniscal defect. Stem Cells. 2009;27(4):878-887. [80] SONG X, XIE Y, LIU Y, et al. Beneficial effects of coculturing synovial derived mesenchymal stem cells with meniscus fibrochondrocytes are mediated by fibroblast growth factor 1: increased proliferation and collagen synthesis. Stem Cells Int. 2015;2015:926325. [81] NEWBERRY J, DESAI S, ADLER C, et al. SDF-1 preconditioned HPC scaffolds mobilize cartilage-derived progenitors and stimulate meniscal fibrocartilage repair in human explant tissue culture. Connect Tissue Res. 2020;61(3-4):338-348. [82] Huang H, Wang S, Gui J, et al. A study to identify and characterize the stem/progenitor cell in rabbit meniscus. Cytotechnology. 2016;68(5): 2083-2103. [83] DING Z, HUANG H. Mesenchymal stem cells in rabbit meniscus and bone marrow exhibit a similar feature but a heterogeneous multi-differentiation potential: superiority of meniscus as a cell source for meniscus repair. BMC Musculoskelet Disord. 2015;16:65. [84] CHAN BP, LEONG KW. Scaffolding in tissue engineering: general approaches and tissue-specific considerations. Eur Spine J. 2008;17 Suppl 4:467-479. [85] JIANG X, SUI X, LU Y, et al. In vitro and in vivo evaluation of a photosensitive polyimide thin-film microelectrode array suitable for epiretinal stimulation. J Neuroeng Rehabil. 2013; 10: 48. [86] LIANG Y, IDREES E, SZOJKA A, et al. Chondrogenic differentiation of synovial fluid mesenchymal stem cells on human meniscus-derived decellularized matrix requires exogenous growth factors. Acta Biomater. 2018;80:131-143. [87] MORAN CJ, BUSILACCHI A, LEE CA, et al. Biological augmentation and tissue engineering approaches in meniscus surgery. Arthroscopy. 2015;31(5):944-955. [88] IONESCU LC, LEE GC, HUANG KL, et al. Growth factor supplementation improves native and engineered meniscus repair in vitro. Acta Biomater. 2012;8(10):3687-3694. [89] MARSANO A, MILLWARD-SADLER SJ, SALTER DM, et al. Differential cartilaginous tissue formation by human synovial membrane, fat pad, meniscus cells and articular chondrocytes. Osteoarthritis Cartilage. 2007;15(1):48-58. [90] CHIU LL, TO WT, LEE JM, et al. Scaffold-free cartilage tissue engineering with a small population of human nasoseptal chondrocytes. Laryngoscope. 2017;127(3):E91-E99. [91] RIERA KM, ROTHFUSZ NE, WILUSZ RE, et al. Interleukin-1, tumor necrosis factor-alpha, and transforming growth factor-beta 1 and integrative meniscal repair: influences on meniscal cell proliferation and migration. Arthritis Res Ther. 2011;13(6):R187. [92] KOH RH, JIN Y, KANG BJ, et al. Chondrogenically primed tonsil-derived mesenchymal stem cells encapsulated in riboflavin-induced photocrosslinking collagen-hyaluronic acid hydrogel for meniscus tissue repairs. Acta Biomater. 2017;53:318-328. [93] CHOI B, KIM S, FAN J, et al. Covalently conjugated transforming growth factor-β1 in modular chitosan hydrogels for the effective treatment of articular cartilage defects. Biomater Sci. 2015;3(5):742-752. [94] PUETZER JL, BROWN BN, BALLYNS JJ, et al. The effect of IGF-I on anatomically shaped tissue-engineered menisci. Tissue Eng Part A. 2013;19(11-12):1443-1450. [95] ZELLNER J, MUELLER M, XIN Y, et al. Dynamic hydrostatic pressure enhances differentially the chondrogenesis of meniscal cells from the inner and outer zone. J Biomech. 2015;48(8):1479-1484. [96] PUETZER JL, BONASSAR LJ. Physiologically Distributed Loading Patterns Drive the Formation of Zonally Organized Collagen Structures in Tissue-Engineered Meniscus. Tissue Eng Part A. 2016; 22(13-14):907-916. [97] ZHANG ZZ, CHEN YR, WANG SJ, et al. Orchestrated biomechanical, structural, and biochemical stimuli for engineering anisotropic meniscus. Sci Transl Med. 2019;11(487):1946-6234. [98] WOO CH, KIM HK, JUNG GY, et al. Small extracellular vesicles from human adipose-derived stem cells attenuate cartilage degeneration. J Extracell Vesicles. 2020;9(1):1735249. |
| [1] | 姚晓玲, 彭建城, 许岳荣, 杨志东, 张顺聪. 可变角度零切迹前路椎间融合内固定系统治疗脊髓型颈椎病:30个月随访[J]. 中国组织工程研究, 2022, 26(9): 1377-1382. |
| [2] | 朱 婵, 韩栩珂, 姚承佼, 周 倩, 张 强, 陈 秋. 人体唾液成分与骨质疏松/骨量低下[J]. 中国组织工程研究, 2022, 26(9): 1439-1444. |
| [3] | 金 涛, 刘 林, 朱晓燕, 史宇悰, 牛建雄, 张同同, 吴树金, 杨青山. 骨关节炎与线粒体异常[J]. 中国组织工程研究, 2022, 26(9): 1452-1458. |
| [4] | 张立创, 徐 浩, 马迎辉, 熊梦婷, 韩海慧, 鲍嘉敏, 翟伟韬, 梁倩倩. 免疫调控淋巴回流功能治疗类风湿关节炎的机制及前景[J]. 中国组织工程研究, 2022, 26(9): 1459-1466. |
| [5] | 王 景, 熊 山, 曹 金, 冯林伟, 王 信. 白细胞介素3在骨代谢中的作用及机制[J]. 中国组织工程研究, 2022, 26(8): 1260-1265. |
| [6] | 肖 豪, 刘 静, 周 君. 脉冲电磁场治疗绝经后骨质疏松症的研究进展[J]. 中国组织工程研究, 2022, 26(8): 1266-1271. |
| [7] | 朱 婵, 韩栩珂, 姚承佼, 张 强, 刘 静, 邵 明. 针刺治疗帕金森病:动物实验显示的作用机制[J]. 中国组织工程研究, 2022, 26(8): 1272-1277. |
| [8] | 田 川, 朱向情, 杨再玲, 鄢东海, 李 晔, 王严影, 杨育坤, 何 洁, 吕冠柯, 蔡学敏, 舒丽萍, 何志旭, 潘兴华. 骨髓间充质干细胞调控猕猴卵巢的衰老[J]. 中国组织工程研究, 2022, 26(7): 985-991. |
| [9] | 胡 伟, 谢兴奇, 屠冠军. 骨髓间充质干细胞来源外泌体改善脊髓损伤后血脊髓屏障的完整性[J]. 中国组织工程研究, 2022, 26(7): 992-998. |
| [10] | 侯婧瑛, 郭天柱, 于萌蕾, 龙会宝, 吴 浩. 缺氧预处理通过激活MALAT1靶向抑制miR-195促进骨髓间充质干细胞的生存和血管形成[J]. 中国组织工程研究, 2022, 26(7): 1005-1011. |
| [11] | 周 颖, 张 幻, 廖 松, 胡凡琦, 易 静, 刘玉斌, 靳继德. 去铁胺联合干扰素γ预处理对人牙髓干细胞的免疫调节作用[J]. 中国组织工程研究, 2022, 26(7): 1012-1019. |
| [12] | 梁学振, 杨 曦, 李嘉程, 骆 帝, 许 波, 李 刚. 补肾活血胶囊介导Hedgehog信号通路调控大鼠骨髓间充质干细胞成骨成脂分化[J]. 中国组织工程研究, 2022, 26(7): 1020-1026. |
| [13] | 闻丹丹, 李 强, 沈才齐, 纪 哲, 金培生. 外用红色诺卡氏菌细胞壁骨架提高脂肪间充质干细胞活性修复糖尿病创面[J]. 中国组织工程研究, 2022, 26(7): 1038-1044. |
| [14] | 安维政, 何 萧, 任 帅, 刘建宇. 肌源干细胞在周围神经再生中的潜力[J]. 中国组织工程研究, 2022, 26(7): 1130-1136. |
| [15] | 范一鸣, 刘方煜, 张洪宇, 李 帅, 王岩松. 脊髓损伤后室管膜区内源性神经干细胞反应的系列问题[J]. 中国组织工程研究, 2022, 26(7): 1137-1142. |
半月板损伤作为最常见的运动损伤之一,已经随着各项体育运动的普及而逐渐引起人们的重视,在足球、篮球、网球、摔跤及长跑等运动中半月板损伤是最常见损伤之一[1-2]。半月板损伤常由于剧烈的剪切、挤压扭转和过伸运动导致,并且经常合并前交叉韧带撕裂与胫骨平台骨折[3],研究表明,半月板损伤和前交叉韧带合并半月板损伤肢体出现骨关节炎的概率是未受伤肢体的6倍[4]。近年来保留半月板结构和功能的半月板修复手术已经取代半月板部分或全部切除术,成为半月板损伤治疗的最佳选择。目前,临床常采用的半月板修复手术虽然可以解除疼痛且取得良好的短期临床效果,但对半月板组织的再生及阻止骨关节炎进展的效果不佳。2019年的一项报告显示,在过去的10年中半月板修复术、半月板切除术和非手术治疗导致骨关节炎的发生率分别为53.0%,99.3%和95.1%,全膝关节置换的发生率分别为33.5%,51.5%和45.5%[5],这些限制为生物组织工程研究提出了新的要求和挑战。在此基础上,生物组织工程在半月板再生修复及人工半月板两方面已经取得可观的进展,随着组织工程的研究和医学的发展,半月板损伤的成功治疗成为可能。
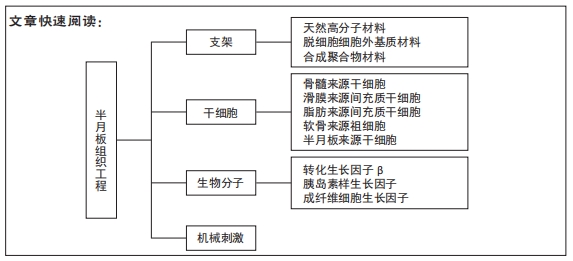
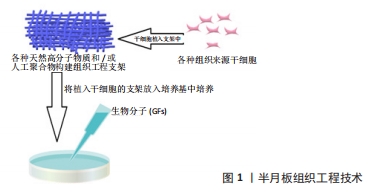
目前的手术方法不能提供半月板再生和替换的长期解决方案,但组织工程技术可以提供替代的治疗策略。半月板组织工程的目标是模仿和还原半月板组织复杂的各向异性结构及各个区域不同的细胞和生化组成,以达到同天然半月板相似的物理、生理功能。为了达到目标需要用到以下几种主要工具:支架、细胞、生物因子和生物机械刺激(图1),该文就以上4个方面目前的研究成果做一总结。
中国组织工程研究杂志出版内容重点:生物材料;骨生物材料; 口腔生物材料; 纳米材料; 缓释材料; 材料相容性;组织工程
1.2 纳入与排除标准
1.2.1 纳入标准 与半月板组织工程、支架、干细胞、生物因子、生物机械刺激相关文献。
1.2.2 排除标准 与该综述研究内容无关的文献、内容重复文献、综述类文献。
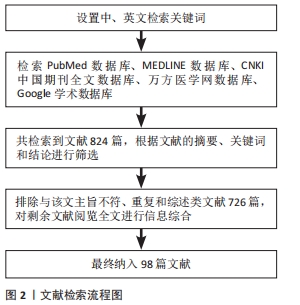
1.3 数据提取 共检索到824篇文献,其中中文文献32篇,英文文献792篇,排除与研究目的相关性差及内容陈旧、重复的文献726篇,纳入98篇符合标准的文献进行综述,见图2。
半月板组织工程作为半月板治疗研究的热点方向,虽然在各个方面都取得了丰富的研究成果,但各个成果均缺乏大量长期的临床试验来验证结论的准确性。生物分子作为组织工程不可或缺的工具,目前仍然需要更多的人或动物实验来确定不同生物分子准确的作用信号通路、应用的最佳浓度和组合方式及生物分子的递送方式。虽然已发现很多种具有半月板表型分化潜力的干细胞,但对于各种干细胞在组织工程中的作用仍待深入研究,最具优势或最适合半月板组织工程的干细胞种类依然颇具争议。与此同时,半月板在细胞组成、细胞外基质成分及生物力学特性上与关节软骨和椎间盘等组织相似,并且关节软骨和椎间盘的修复再生策略同样备受关注,相似领域的研究进展可以相互借鉴、相互促进。相信随着组织工程研究的迅速发展,针对个体半月板损伤的组织工程修复或者替代策略将会成为成熟并且可靠的治疗方案,这一天不会太遥远。
中国组织工程研究杂志出版内容重点:生物材料;骨生物材料; 口腔生物材料; 纳米材料; 缓释材料; 材料相容性;组织工程

文题释义:
组织各向异性:半月板组织由内到外可分为白-白区、红-白区和红-红区3个区域,白-白区主要由纤维软骨细胞、放射状分布的Ⅱ型胶原蛋白和较高浓度的蛋白聚糖组成,红-红区主要由成纤维细胞样细胞、沿半月板长轴环状分布的Ⅰ型胶原蛋白和较低浓度的蛋白聚糖组成,红-白区结构组成介于两区之间。因此,半月板组织内侧具有较高的压缩模量,外侧具有较高的拉伸模量。
支架:在半月板组织工程中,支架主要发挥塑造形态、模仿半月板组织各向异性、为干细胞提供生长环境、生物因子载体和加强支架力学特性的功能。通过进行组织工程支架形态的设计以达到契合不同形状的半月板损伤,同时向损伤处持续递送干细胞和生物因子促进组织愈合。
对于半月板损伤目前临床常用治疗方法有:保守治疗、半月板部分或全部切除术、半月板修复术和同种异体半月板移植术。但以上治疗方法均无法恢复半月板本身的愈合能力和天然的组织结构。半月板组织工程则有望解决半月板损伤和缺失的问题,该文总结近年来国内外半月板组织工程研究成果,分析归纳各种组织工程策略的优势和缺点,期待能够为以后的半月板组织工程研究提供参考和方向。
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||