中国组织工程研究 ›› 2026, Vol. 30 ›› Issue (19): 5024-5032.doi: 10.12307/2026.220
• 干细胞综述 stem cell review • 上一篇 下一篇
间充质干细胞来源细胞外囊泡干预细胞调控网络治疗肺纤维化的机制与临床策略
丁 炎1,聂宏光1,孙 宇2
- 中国医科大学基础医学院,1干细胞与再生医学研究室,2病理生理学教研室,辽宁省沈阳市 110122
-
收稿日期:2025-08-18接受日期:2025-10-14出版日期:2026-07-08发布日期:2026-02-24 -
通讯作者:孙宇,高级实验师,中国医科大学基础医学院病理生理学教研室,辽宁省沈阳市 110122 -
作者简介:丁炎,女,1987年生,辽宁省沈阳市人,汉族,博士,高级实验师,主要从事干细胞与肺损伤研究。 -
基金资助:国家自然科学基金项目(82170093),项目负责人:聂宏光;辽宁省科学技术计划项目(2023JH2/20200072),项目负责人:丁炎;辽宁省自然科学基金计划项目(2023-BS-103),项目负责人:孙宇
Mechanisms and clinical strategies of mesenchymal stem cell-derived extracellular vesicles intervening in cell regulatory networks to treat pulmonary fibrosis
Ding Yan1, Nie Hongguang1, Sun Yu2
- 1Laboratory of Stem Cell and Regenerative Medicine Research, 2Department of Pathophysiology, College of Basic Medical Science, China Medical University, Shenyang 110122, Liaoning Province, China
-
Received:2025-08-18Accepted:2025-10-14Online:2026-07-08Published:2026-02-24 -
Contact:Sun Yu, Senior experimentalist, Department of Pathophysiology, College of Basic Medical Science, China Medical University, Shenyang 110122, Liaoning Province, China -
About author:Ding Yan, MD, Senior experimentalist, Laboratory of Stem Cell and Regenerative Medicine Research, College of Basic Medical Science, China Medical University, Shenyang 110122, Liaoning Province, China -
Supported by:National Natural Science Foundation of China, No. 82170093 (to NHG); Liaoning Province Science and Technology Plan Project, No. 2023JH2/20200072 (to DY); Liaoning Provincial Natural Science Foundation Project, No. 2023-BS-103 (to SY)
摘要:
文题释义:
间充质干细胞来源细胞外囊泡:是由间充质干细胞分泌的纳米级膜包裹的囊泡,直径通常在30-1 000 nm之间。这些囊泡是细胞间通讯的重要媒介,携带多种生物活性分子,包括蛋白质、脂质、核酸和代谢产物。间充质干细胞来源细胞外囊泡保留了母细胞的许多治疗特性,具有更低的免疫原性和更高的安全性。它们通过旁分泌机制发挥作用,能够穿越生物屏障(如血脑屏障),在组织修复和免疫调节中表现出显著的效果。肺纤维化:以肺泡结构进行性破坏、细胞外基质大量沉积和肺瘢痕形成为特征的间质性肺部疾病,伴随不可逆的肺功能下降及进行性呼吸衰竭。
摘要
背景:肺纤维化是一种以细胞外基质异常沉积为特征的慢性进行性肺疾病,目前缺乏有效的治疗手段。间充质干细胞来源细胞外囊泡凭借脂质包膜结构可穿越体内屏障,将多种抗纤维化、免疫调节因子(如生长因子、免疫调节细胞因子、趋化因子)、脂质和核酸(mRNA和miRNA)等生物活性物质直接递送至肺内靶细胞。
目的:系统综述间充质干细胞来源细胞外囊泡治疗肺纤维化的核心机制,总结和阐述间充质干细胞来源细胞外囊泡将携带的生物活性物质直接递送至肺内不同靶细胞,展现出调控肺纤维化微环境的独特优势,为将来利用间充质干细胞来源细胞外囊泡治疗肺纤维化疾病提供理论基础。
方法:应用计算机检索中国知网、PubMed数据库、clinicaltrials.gov数据库和中国临床试验注册中心,英文检索关键词为“Mesenchymal stem cells,Extracellular vesicles,Pulmonary fibrosis,Alveolar epithelium,Microvascular endothelium,Macrophages,Neutrophils”,中文检索关键词为“间充质干细胞,细胞外囊泡,肺纤维化,上皮细胞,血管内皮细胞,巨噬细胞,中性粒细胞”,最终纳入56篇文献进行总结。
结果与结论:在肺泡上皮细胞中,通过调控蛋白激酶B/糖原合成酶激酶3β、转化生长因子β/Smad等信号通路抑制上皮间质转化进程,以及特定miRNA(如miR-466f-3p、let-7等)阻断促纤维化通路网络。而在成纤维细胞和内皮细胞中,分别通过miR-21-5p和miR-218/miR-214-3p干预成纤维细胞的活化和内皮间质转化,同时从宏观上重编程单核细胞、调控巨噬细胞极化、抑制树突细胞成熟及平衡Th17/Treg反应,重塑免疫微环境。此外,基于工程化修饰(靶向肽修饰、药物共载)的间充质干细胞来源细胞外囊泡还可增强病变区域多细胞亚型的精准靶向性,而囊泡异质性、规模化制备标准及体内动态示踪技术仍是临床转化的关键瓶颈。未来研究需结合单细胞测序与空间多组学技术,深入解析间充质干细胞来源细胞外囊泡干预细胞调控网络的深入机制,制定以细胞为靶点的新型肺纤维化临床治疗策略。
中图分类号:
引用本文
丁 炎, 聂宏光, 孙 宇. 间充质干细胞来源细胞外囊泡干预细胞调控网络治疗肺纤维化的机制与临床策略[J]. 中国组织工程研究, 2026, 30(19): 5024-5032.
Ding Yan, Nie Hongguang, Sun Yu. Mechanisms and clinical strategies of mesenchymal stem cell-derived extracellular vesicles intervening in cell regulatory networks to treat pulmonary fibrosis[J]. Chinese Journal of Tissue Engineering Research, 2026, 30(19): 5024-5032.
肺纤维化临床治疗体系呈现三级架构:基础方案采用糖皮质激素联合免疫抑制剂延缓肺功能衰退,辅以抗纤维化药物改善生存质量;终末期患者依赖肺移植,但受限于供体稀缺和术后并发症,5年生存率不足50%;氧疗等支持性措施仅具有症状缓解作用。现有疗法均无法逆转纤维化进程,患者中位生存期仍不足3年,核心瓶颈在于药物靶点集中于转化生长因子β/Smad等下游通路,未能干预上皮细胞异常凋亡/成纤维细胞活化等始动环节[9]。值得注意的是,COVID19病毒感染重症患者肺间质纤维化发生率高达12%-17%,其进展速度较肺纤维化快两三倍,且对激素治疗耐药率提升40%[10-11],这揭示病毒残留蛋白可能通过重塑免疫微环境加速纤维化,提示靶向病毒相关分子或异常免疫记忆的创新疗法亟待突破。
2.2 间充质干细胞来源细胞外囊泡的作用机制 间充质干细胞凭借多组织来源(图3,脐带、骨髓、脂肪等)及多重生物学特性(抗炎、免疫调节、促修复),已成为再生医学治疗肺纤维化的核心策略[12]。间充质干细胞的治疗机制通过三轴联动实现:①直接分化为肺泡上皮细胞修复损伤;②分泌肝细胞生长因子、人肿瘤坏死因子诱导蛋白6等因子抑制转化生长因子β/Smad通路,阻断成纤维细胞活化;③调控Th17/Treg免疫平衡重塑微环境。静脉注射因便捷性成为主要给药途径,较肺移植创伤显著降低,但临床应用受制于两大瓶颈:肺部截留效应(静脉注射后,间充质干细胞因肺毛细血管网被肺微血管机械截留)及微环境胁迫(缺氧条件下72 h存活率骤降至12%-15%)[13]。这促使研究转向外泌体等无细胞疗法,细胞外囊泡不仅规避了细胞存活难题,更具备独特优势:①低免疫原性与穿透性强,可递送药物/蛋白/RNA至靶点;②组分可通过预处理精准调控,实现疾病特异性治疗;③冻存稳定性更优,便于临床转化。细胞外囊泡疗法正成为突破现有治疗困局的新焦点。
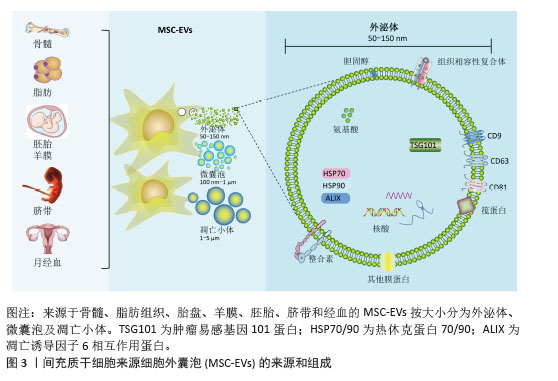
根据国际细胞外囊泡协会2024年发布的最新版细胞外囊泡研究指南(MISEV2023),细胞外囊泡是指从细胞中释放的颗粒,由脂质双层界定,并且不能自行复制(即不包含功能性细胞核)[14]。这些细胞外囊泡在细胞间信息传递、免疫应答调节及肿瘤进展等多个生物学过程中扮演着关键角色。细胞外囊泡作为新型无细胞治疗载体,根据生物发生、释放途径、大小、内容物和功能等大致分为3类(图3):外泌体(50-150 nm)、微囊泡(100-1 000 nm)及凋亡小体(> 1 000 nm)[15]。其中间充质干细胞来源细胞外囊泡展现核心治疗优势:①携带mRNA/miRNA、细胞因子及血管生成因子等活性组分,直接抑制成纤维细胞活化并促进上皮修复[16];②规避活细胞治疗的增殖失控和免疫排斥风险,治疗剂量耐受性提升3-5倍;③具有天然低免疫原性(CD47表达)和长效循环特性(半衰期> 24 h),靶向效率较干细胞提升60%以上。相较于传统干细胞疗法,细胞外囊泡治疗实现了三大技术跨越:生物安全性标准化(制剂活性成分可控)、治疗靶向精准化(表面修饰CD44/肝细胞生长因子受体)以及生产工艺规模化[整合振荡剪切微流控系统单批次产能(1.5×1013)±(0.3×1013)颗粒,符合ISO14644-1 Class 5标准]。这为开发可逆转肺纤维化的下一代生物制剂奠定了转化基础。
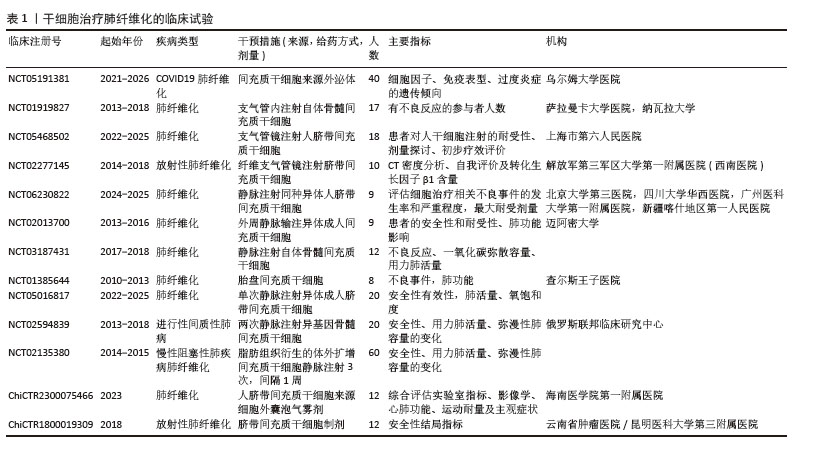
在中国知网、PubMed数据库筛选出近10年内涉及间充质干细胞来源细胞外囊泡治疗肺纤维化的56篇研究,所涉及的具体细节按照间充质干细胞种类,造模方法、细胞移植途径、剂量、时间等具体细节见附表。

2.2.1 抑制上皮-间充质转化/抗凋亡坏死进而维持肺泡上皮稳态 肺纤维化的病理重构始于肺泡上皮细胞的级联功能失调,核心表现为:上皮-间充质转化进程中,上皮细胞通过表型转化(E-cadherin下调> 80%)获得间充质特性,分泌转化生长因子β/肿瘤坏死因子α等因子直接导致细胞外基质异常沉积(基质金属蛋白酶9水平升高5-8倍);在遗传易感个体中,反复微损伤导致再上皮化障碍(修复周期延长2.3倍),形成Ⅰ/Ⅲ型胶原异常交联[占比达(78.0±5.2)%]的恶性修复模式;血小板衍生生长因子受体β/成纤维细胞生长因子受体1信号轴异常激活(磷酸化水平增加3.5倍),驱动α-平滑肌肌动蛋白(表达量升高4.2倍)等间充质标志物表达,加速肺泡空间纤维化重塑[17]。针对上皮-间充质转化进程的靶向调控、再生微环境重建及异常信号通路阻断,已成为干预肺纤维化病理进展的三大核心策略。
研究表明,不同间充质干细胞来源细胞外囊泡可通过多通路协同抑制上皮-间充质转化进程,从而发挥抗肺纤维化作用。①通过传递miR-466f-3p活性分子靶向调控蛋白激酶B/糖原合成酶激酶3β信号轴[18],抑制SNAIL和Twist蛋白活性,逆转上皮-间充质转化相关标志物表达(E-cadherin表达提升2.1倍,α-平滑肌肌动蛋白/N-cadherin降低60%);let-7i-5p靶向c-Met拮抗转化生长因子β驱动的细胞外基质沉积及成纤维细胞迁移活化(迁移率降低70%)[19-20];另一研究显示,递送的miR-23a-3p和miR-182-5p通过沉默Ikbkb基因并破坏核因子κB抑制蛋白激酶β蛋白稳定性,抑制核因子κB和Hedgehog通路的过度激活,协同逆转上皮-间充质转化进程[21];②通过抑制转化生长因子β1/Smad2/3信号通路及其下游上皮-间充质转化进程,显著降低促纤维化递质表达,且在早期干预效果更为显著[22-24];③调控Toll样受体4/髓样分化因子88/核因子κB、NOD1/核因子κB/NOD样受体家族Pyrin域蛋白3(NOD-like receptor family pyrin domain containing 3,NLRP3)炎症信号级联反应,抑制蛋白激酶B/核因子κB通路,减轻肺泡上皮-间充质转化[25-27];④阻断Wnt/β-catenin通路过度激活,通过糖原合成酶激酶3β/β-catenin轴抑制二氧化硅诱导的上皮-间充质转化进展[8]。这些研究共同揭示间充质干细胞来源细胞外囊泡可通过多miRNA组合策略,同时干预SNAIL、β-catenin等关键上皮-间充质转化调控因子,靶向转化生长因子β、Wnt等多条促纤维化通路网络,在辐射、博来霉素等肺纤维化模型中有效维持肺泡上皮稳态,抑制胶原沉积和病理重构。
间充质干细胞来源细胞外囊泡通过多维度调控肺泡上皮细胞的命运(包括凋亡、增殖、炎症等),从而在肺纤维化中发挥关键保护作用,作用机制包括:①抑制凋亡与坏死:间充质干细胞来源细胞外囊泡通过递送miR-21和热休克蛋白70激活磷脂酰肌醇3激酶/蛋白激酶B抗凋亡通路,显著减少博来霉素诱导的肺泡上皮细胞凋亡[28];而脂肪间充质干细胞来源细胞外囊泡和人脐带间充质干细胞来源微粒则分别通过抑制PM2.5诱导的细胞坏死及miR-100/哺乳动物雷帕霉素靶蛋白依赖的自噬增强[29-31],降低氧化应激和线粒体损伤,如线粒体DNA损伤、NLRP3炎性小体激活,进一步阻止上皮细胞程序性死亡;②促进增殖与修复:骨髓间充质干细胞通过旁分泌肝细胞生长因子、角质细胞生长因子和骨形态发生蛋白7等因子,直接刺激肺泡上皮细胞增殖,加速损伤修复并抑制促纤维化因子释放[32-33];③调控炎症与代谢:外泌体携带的miRNA(如let-7)通过下调肺泡上皮细胞线粒体活性氧水平、脂氧合酶1表达和线粒体DNA损伤,抑制炎症相关信号(如NLRP3)的激活[34],同时阻断Wnt/β-catenin通路的过度活化,减少促炎因子(白细胞介素6、肿瘤坏死因子α)分泌及成纤维细胞-肌成纤维细胞转分化[8],从而缓解炎症微环境对上皮细胞稳态的破坏[35]。此外,间充质干细胞通过激活核因子E2相关因子2通路,显著减轻电离辐射诱导的肺上皮细胞铁死亡,进而减轻放射性肺损伤[36]。综上,间充质干细胞来源细胞外囊泡通过靶向调控上皮细胞命运的核心信号网络(磷脂酰肌醇3激酶/蛋白激酶B、Wnt/β-catenin、自噬-凋亡平衡等),协同改善上皮损伤、炎症反应及纤维化进程。
近年来,基于外泌体的协同增效策略在肺纤维化治疗中展现出突破性潜力。研究显示,间充质干细胞来源外泌体既可作为药物递送载体,也可通过工程化改造实现靶向治疗。CAI团队[37]将酪氨酸激酶抑制剂尼达尼布与脂肪间充质干细胞来源外泌体结合形成的复合系统,通过双重抑制活性氧和转化生长因子β/Smad信号通路,显著缓解博莱霉素诱导的肺纤维化,其疗效超越单药治疗,证实了外泌体与抗纤维化药物的协同增效作用。同时,miR-486-5p修饰的间充质干细胞来源外泌体通过抑制Smad2和激活蛋白激酶B发挥抗纤维化作用,在体外抑制了上皮细胞的铁死亡和纤维化,在体内减轻了血管紧张素转化酶2人源化小鼠肺纤维化症状[38]。另一项研究中,CD38膜抗原修饰的工程化间充质干细胞来源外泌体能精准靶向衰老CD38高表达的Ⅱ型肺泡上皮细胞,通过递送抗纤维化miRNA恢复细胞内NAD+代谢水平,逆转上皮-间充质转化进程,在老年小鼠模型中实现衰老细胞表型重塑和肺纤维化改善[39]。这些研究共同揭示了外泌体治疗的多维度优势,既能增强传统药物的靶向递送效率,又能通过工程化策略实现细胞代谢重编程与纤维化信号通路的双重干预,为开发肺纤维化联合治疗新模式提供了重要理论依据。
2.2.2 抑制成纤维细胞增殖分化及降低胶原生成 成纤维细胞是肺组织中合成胶原蛋白、弹性蛋白等细胞外基质的核心细胞,通过动态调控细胞外基质的生成与降解,维持肺组织的结构完整性和机械稳定性[4],这一功能在正常组织修复中至关重要,但在病理状态下可能失衡。在肺损伤早期,转化生长因子β、白细胞介素4/白细胞介素13等促纤维化因子可激活成纤维细胞,诱导其分化为高表达α-平滑肌肌动蛋白的肌成纤维细胞[40-41],这种转化伴随胶原蛋白分泌量显著增加,导致细胞外基质异常沉积,成为肺纤维化的直接驱动因素。肺纤维化中的肌成纤维细胞不仅来源于局部成纤维细胞激活,还可通过上皮-间充质转化、周细胞分化、循环骨髓干细胞迁移等多种途径,形成异质性细胞群。单细胞测序研究证实,肺纤维化进展期多种间充质细胞(如Pdgfrb+间充质细胞、Cthrc1+成纤维细胞)均通过上调细胞外基质相关基因,扩大纤维化效应[42]。持续活化的肌成纤维细胞通过收缩特性改变肺组织结构,形成不可逆的瘢痕组织,最终导致肺泡结构破坏和肺功能衰竭。这一过程与转化生长因子β/Smad、Wnt/β-catenin等信号通路的异常激活密切相关[5,7],提示成纤维细胞的分子调控网络是抗纤维化治疗的关键靶点。
间充质干细胞来源细胞外囊泡通过递送特异性miRNA,靶向调控成纤维细胞的纤维化进程。其中,细胞外囊泡携带的miR-21-5p和miR-23-3p通过抑制转化生长因子β和转化生长因子β受体2的表达进而抑制成纤维细胞的活化[43];let-7i-5p通过抑制转化生长因子β/Smad信号通路,阻断成纤维细胞的活化与分化,并诱导成纤维细胞凋亡及衰老[19,44];miR-29b-3p通过下调卷曲蛋白6表达,抑制成纤维细胞的增殖、迁移、侵袭及分化[45];miR-186靶向SOX4/DKK1信号轴,诱导细胞周期阻滞并促进凋亡,从而抑制成纤维细胞增殖和侵袭[46];miR-630则通过Thy-1-整合素通路实现精准递送,靶向抑制促纤维化基因表达[47-48]。
miR-486-5p通过靶向成纤维细胞生长因子9来调控成纤维细胞的分化[49]。此外,干扰素γ预处理通过重塑免疫微环境增强间充质干细胞来源细胞外囊泡的抗肺纤维化疗效,且作用呈剂量依赖性,低浓度触发促炎反应,高浓度激活抗纤维化通路,显著提升对系统性硬化症小鼠肺纤维化的治疗效果[50]。间充质干细胞来源细胞外囊泡通过递送miR-21-5p、let-7i-5p等关键miRNA[51-52],显著降低α-平滑肌肌动蛋白表达、胶原蛋白沉积等。如果联合干扰素γ预处理可增强细胞外囊泡的抗纤维化疗效,有效抑制成纤维细胞异常活化及细胞外基质过度沉积,最终在临床中发挥多机制协同治疗肺纤维化的治疗效应。
2.2.3 保护毛细血管内皮细胞以促进血管重塑 内皮-间充质转化是内皮细胞通过表型转化获得间充质特性的生物学过程。转化生长因子β、骨形态发生蛋白2及甲基化CpG结合蛋白2等信号通路通过激活Snail、Twist和Slug等转录因子驱动内皮-间充质转化进程。在此过程中,内皮标志物(如E-cadherin)表达下调,间质标志物(α-平滑肌肌动蛋白、成纤维细胞特异性蛋白1)显著上调,细胞间连接丧失,迁移和侵袭能力增强。这些转化细胞可迁移至周围组织,进而加速肺组织细胞外基质异常沉积和纤维瘢痕形成,最终推动肺纤维化病理进展。
甲基化CpG结合蛋白2通过双重机制驱动肺纤维化进展,一方面直接激活肌成纤维细胞中α-平滑肌肌动蛋白的表达,另一方面抑制骨形态发生蛋白2信号通路,打破转化生长因子β与骨形态发生蛋白2的平衡。在博来霉素诱导的肺纤维化模型中,转化生长因子β持续激活与骨形态发生蛋白2信号抑制协同加剧细胞外基质沉积,加速纤维化进程。ZHAO团队[53]研究发现,人脐带间充质干细胞来源外泌体通过递送miR-218,逆转转化生长因子β/甲基化CpG结合蛋白2介导的骨形态发生蛋白2抑制,从而恢复内皮细胞功能并阻断内皮-间充质转化。敲低miR-218可削弱外泌体的抗纤维化效应,而过表达miR-218则显著提升骨形态发生蛋白2水平,抑制胶原沉积并减轻肺纤维化。人胎盘间充质干细胞来源外泌体通过递送miR-214-3p,靶向下调ATM/P53/P21信号通路,抑制辐射诱导的DNA损伤及衰老相关分泌表型,从而缓解肺血管损伤、炎症反应及纤维化进展[54]。
2.2.4 抑制单核-巨噬细胞转化和调控巨噬细胞极化 研究显示单核细胞计数增加与肺纤维化患者不良预后呈正相关[55]。在纤维化早期,循环单核细胞会浸润至受损肺组织,并通过成熟分化为纤维化炎性肺泡巨噬细胞参与病理进程[6]。这些巨噬细胞通过分泌趋化因子,介导炎症细胞募集并激活肌成纤维细胞增殖,从而启动纤维化级联反应。巨噬细胞具有表型可塑性,在不同的环境和细胞因子的刺激下可分化为促进炎症的M1型和抗炎的M2型两种表型,呈现功能极化。M1型在急性炎症期占主导,分泌肿瘤坏死因子α等促纤维化炎症因子,调控肺纤维化炎症反应;抗炎M2型受慢性刺激后由M1型转化而来,通过分泌白细胞介素4、白细胞介素10等抗炎因子,抑制白细胞介素1β、肿瘤坏死因子α等促炎递质,同时表现出促纤维化特性。M2型巨噬细胞的过度极化会导致双重效应:一方面通过抑制炎症反应促进组织修复,另一方面异常激活的增殖反应和胶原沉积加速纤维化进程。多种调节因子(包括细胞因子、趋化因子、递质及免疫调节细胞)通过干预巨噬细胞极化平衡,形成复杂调控网络,最终影响肺纤维化的发展方向及严重程度。这种动态平衡的打破可能是决定肺纤维化病理转归的关键环节。
近年来,间充质干细胞来源外泌体在肺纤维化治疗中的免疫调控机制逐渐明确。首先,间充质干细胞来源外泌体通过干预单核细胞表型及功能发挥关键作用。研究表明,人间充质干细胞来源外泌体靶向趋化因子配体2信号级联,将促炎的“经典”单核细胞重编程为抗炎的“非经典”单核细胞,抑制促炎及促纤维化因子(如肿瘤坏死因子α、白细胞介素1β、白细胞介素6、转化生长因子β)的释放,并促进免疫抑制性白细胞介素10的合成[28,56];同时,外泌体携带的Let-7i-5p能够进一步抑制肺浸润单核细胞中白细胞介素6和肿瘤坏死因子α的产生,从而阻断炎症驱动的组织损伤和纤维化进程[19,44]。其次,间充质干细胞来源外泌体通过调节巨噬细胞数量及极化状态缓解纤维化。例如,脂肪间充质干细胞来源外泌体直接减少肺泡间隔和肉芽肿内的巨噬细胞数量[57];多能干细胞来源外泌体通过miR-302a-3p/TET1轴抑制M2型巨噬细胞分化[58];人脐带间充质干细胞来源细胞外囊泡通过升高miR-486-5p水平,上调抗纤维化基因(白细胞介素10、基质金属蛋白酶13和肝细胞生长因子)表达,下调骨桥蛋白表达,推动巨噬细胞向M2表型极化[59];而人胎盘间充质干细胞来源外泌体通过抑制肺泡巨噬细胞中核因子κB信号通路,降低炎症因子(肿瘤坏死因子α、白细胞介素1β)表达及细胞凋亡[60];人脐带间充质干细胞来源细胞外囊泡调节细胞外调节蛋白激酶1/2信号通路来抑制趋化因子配体2的表达,从而有效抑制了单核细胞和巨噬细胞的迁移,进而缓解肺纤维化病情[61]。此外,间充质干细胞来源外泌体靶向炎症信号通路调控成纤维细胞活化,如脐带间充质干细胞来源外泌体通过circPWWP2A/miR-223-3p/NLRP3轴双重抑制NLRP3炎症小体活性(减少白细胞介素1β、白细胞介素18等促炎因子释放)并阻断成纤维细胞活化,最终减轻二氧化硅暴露诱导的肺部炎症及纤维化[62]。综上,间充质干细胞来源外泌体通过多途径协同调控免疫细胞(单核细胞、巨噬细胞)表型及炎症相关通路(趋化因子配体2、核因子κB、NLRP3),平衡促炎/抗炎反应,抑制成纤维细胞活化,为肺纤维化的治疗提供了新的策略。未来研究需进一步明确不同来源外泌体的作用异同及临床转化潜力。
2.2.5 诱导树突状细胞向耐受表型分化 间充质干细胞来源细胞外囊泡的免疫调控作用不仅限于肺泡巨噬细胞,还可通过重塑肺浸润性树突状细胞的表型与功能发挥关键作用。作为连接先天免疫与适应性免疫的核心抗原呈递细胞,树突状细胞广泛分布于肺泡间隔等肺部外周组织,通常以未成熟状态驻留并专职捕获抗原。值得注意的是,间充质干细胞来源外泌体通过双向调节细胞因子表达,即下调促炎因子嗜酸性粒细胞趋化因子(Eotaxin)及白细胞介素25,同时上调免疫抑制因子白细胞介素10与转化生长因子β,显著抑制树突状细胞的成熟进程。实验研究表明,经间充质干细胞来源外泌体干预后,小鼠肺内未成熟树突状细胞的共刺激分子(CD40、CD80/CD86)表达水平显著降低,导致抗原呈递能力受限,进而削弱CD4+Th2细胞的活化强度,最终减轻Th2细胞主导的肺部炎症级联反应[63]。
2.2.6 减少中性粒细胞浸润 肺纤维化患者支气管肺泡灌洗液中中性粒细胞比例升高可作为早期病死率的独立预测因子[64]。在疾病初期,中性粒细胞被CXC趋化因子配体1、CXC趋化因子配体2/3、白细胞介素8/ CXC趋化因子配体8等趋化因子募集至肺部,穿透血管屏障并浸润肺实质,通过释放弹性蛋白酶等促炎递质加剧局部炎症,同时激活转化生长因子β1信号,驱动成纤维细胞增殖、肌成纤维细胞分化及胶原沉积,形成“炎症-纤维化”恶性循环,最终导致肺组织结构破坏和功能丧失。研究发现,间充质干细胞可通过外泌体及条件培养基调控中性粒细胞功能:在急性炎症期,间充质干细胞条件培养基通过抑制中性粒细胞趋化标志物(MIP-2/Ly-6G)及炎症信号通路(核因子κB)阻断炎症向纤维化转化[65];在慢性纤维化期,人羊膜上皮细胞来源外泌体通过下调中性粒细胞髓过氧化物酶活性,减轻氧化应激及胶原沉积[66]。
人脐带间充质干细胞来源细胞外囊泡可以通过降低肺泡灌洗液中淋巴细胞、多形核细胞的数量,增加CD206阳性巨噬细胞数量,而降低CD80和CD68阳性巨噬细胞数量,发挥抗炎和抗纤维化的作用[67]。尽管间充质干细胞调控中性粒细胞的具体机制尚未完全阐明,但中性粒细胞作为介导炎症反应、直接促纤维化及微环境失衡的核心效应细胞,仍是肺纤维化治疗的重要靶点。
2.2.7 矫正Th17/Treg失衡 T细胞亚群的功能失衡及其相互作用在肺纤维化的炎症驱动和纤维化进程中发挥核心作用[68]。其中Th17细胞通过分泌白细胞介素17激活中性粒细胞释放蛋白酶(如弹性蛋白酶)和活性氧,放大炎症反应并加剧肺组织破坏[69],同时Th17细胞分泌的白细胞介素17和白细胞介素22可直接刺激成纤维细胞活化、分化为肌成纤维细胞,促进细胞外基质沉积与瘢痕形成;此外,Th17细胞通过抑制Treg的免疫抑制功能打破免疫稳态,加速纤维化进程。Th1/Th2失衡通过干扰素γ、肿瘤坏死因子α、白细胞介素4、白细胞介素5等因子驱动巨噬细胞活化和成纤维细胞增殖,加速纤维化进展。Th17细胞与中性粒细胞的正反馈环路协同Th9细胞增强成纤维细胞活化,最终形成“炎症-纤维化”恶性循环。骨髓间充质干细胞来源外泌体通过吲哚胺2,3-双加氧酶依赖性机制,下调Th17细胞分化关键基因(RORγt、白细胞介素17A)表达并增强Treg转录因子FoxP3活性,促进Treg扩增,进而抑制肺部炎症与纤维化进程[70-71]。羊水间充质干细胞来源外泌体在动物模型中通过靶向Th17细胞显著抑制促炎因子(白细胞介素17、肿瘤坏死因子α、白细胞介素1β)及促纤维化因子表达,同时上调人外周血单核细胞中抗炎因子白细胞介素10分泌,通过重塑Th17/Treg免疫平衡阻断肺部炎症-纤维化级联反应,凸显它对肺纤维化的治疗潜力[72]。
2.3 基于间充质干细胞来源细胞外囊泡多靶点协同作用的肺纤维化治疗策略 间充质干细胞来源细胞外囊泡通过携带生物活性分子构建了一个多靶点协同调控网络,在肺纤维化治疗中展现出独特优势。研究表明,let-7i-5p作为关键效应分子,在肺纤维化微环境中呈现多细胞靶向特性:能够同时作用于肺泡上皮细胞和成纤维细胞,通过抑制转化生长因子β/Smad3通路,显著降低上皮-间充质转化进程和胶原沉积,并通过调控单核细胞抑制炎症的产生。不同组织来源间充质干细胞衍生的细胞外囊泡具有特异性miRNA表达谱,骨髓间充质干细胞来源细胞外囊泡富含的miR-29b-3p通过卷曲蛋白6/DKK1抑制肌成纤维细胞转化;脐带间充质干细胞来源细胞外囊泡高表达miR-218,逆转转化生长因子β/甲基化CpG结合蛋白2介导的骨形态发生蛋白2抑制,从而恢复内皮细胞功能促进血管重塑;脂肪间充质干细胞来源细胞外囊泡则依赖分泌miR-21、miR-29,抑制甚至逆转成纤维细胞向肌成纤维细胞转化。在免疫调控方面则通过三重机制发挥协同作用,诱导巨噬细胞向M2型极化,促进耐受性树突状细胞分化以降低抗原提呈,并矫正Th17/Treg失衡来抑制过度炎症。基于上述机制,临床转化可设计“工程化细胞外囊泡鸡尾酒疗法”,将组织特异性miRNA与C-X-C趋化因子受体4靶向修饰结合,实现组织特异性miRNA递送系统病灶定位与时序释放。联合干扰素γ预处理增强疗效,同时配伍尼达尼布等抗纤维化药物形成多模式治疗方案,为肺纤维化患者的治疗带来新希望。
2.4 临床应用现状 间充质干细胞来源细胞外囊泡作为具有多靶点调控特性的生物疗法,在组织纤维化治疗领域展现出独特优势。临床前研究表明,间充质干细胞来源细胞外囊泡通过多重机制在肺、肝、肾等多器官纤维化模型中表现出显著的抗纤维化和促组织修复作用[73]。当前全球范围内共有11项基于间充质干细胞治疗肺纤维化的临床试验注册于clinicaltrials.gov,但均未公布最终研究数据。其中,针对COVID19病毒感染相关肺纤维化的临床研究采用符合CGMP标准的骨髓间充质干细胞来源外泌体,通过体外实验系统评估免疫调节机制(表1)。研究采用多组学技术(包括流式细胞术、单细胞测序等)分析外泌体(108-1010颗粒/mL)对免疫细胞表型、炎症因子网络及基因表达谱的影响,重点考察对T细胞亚群、中性粒细胞活化等关键免疫指标的调控作用。值得关注的是中国临床试验注册中心注册号为ChiCTR2300075466的Ⅰ期临床试验(表1),该研究采用随机单盲设计,探索雾化人脐带间充质干细胞来源外泌体对高分辨率CT确诊肺纤维化患者的安全性和初步疗效。研究方案设计严谨,实验组每日2次雾化吸入含2 mL 外泌体的混合制剂,通过标准化给药时间和严格的对照设置,重点评估呼吸道不良反应等安全性指标,同时监测肺功能、影像学及炎症因子等次要终点,患者获益显著[59]。2020年,SENGUPTA团队[74]开展的里程碑式研究首次评估了同种异体骨髓间充质干细胞来源细胞外囊泡(商品名ExoFlo)在重症COVID19病毒感染患者中的临床应用价值。这项开创性临床试验证实,单次静脉输注ExoFlo表现出良好的安全性特征,在所有观察时间点均未报告治疗相关不良事件;临床疗效数据显示:患者总体生存率达到83%,氧合指数显著改善(PaO2/FiO2提升> 50%);同时观察到显著的免疫调节效应,包括中性粒细胞计数降低32%,淋巴细胞亚群(CD3+、CD4+及CD8+)数量平均增加45%,有效缓解了COVID19病毒感染特征性的淋巴细胞减少症。机制研究表明,间充质干细胞来源细胞外囊泡通过三重作用机制发挥治疗效果:①抑制过度炎症反应(细胞因子风暴);②改善组织缺氧状态;③重建免疫稳态平衡。然而,现有临床证据仍存在明显局限性。目前关于间充质干细胞来源细胞外囊泡治疗肺纤维化的公开发表数据极为有限,长期安全性和治疗效果亟需通过设计严谨的多中心随机对照试验进一步验证。尽管如此,这些早期临床研究为间充质干细胞来源细胞外囊泡的转化医学研究提供了关键的剂量参考和安全性基准,为后续临床试验设计奠定了重要基础。
| [1] DING Y, HOU Y, LIU Y, et al. MiR-130a-3p Alleviates Inflammatory and Fibrotic Phases of Pulmonary Fibrosis Through Proinflammatory Factor TNF-α and Profibrogenic Receptor TGF-βRII. Front Pharmacol. 2022;13:863646. [2] SILVERSTEIN E, RICHMANN M, TYL D, et al. The Application of Mesenchymal Stem Cell Therapy in Treating Pulmonary Fibrosis: A Scoping Review. Cureus. 2024;16(11):e74611. [3] PARK KS, LÄSSER C, LÖTVALL J. Extracellular vesicles and the lung: from disease pathogenesis to biomarkers and treatments. Physiol Rev. 2025; 105(3):1733-1821. [4] LI M, LI J, WANG Y, et al. Umbilical cord-derived mesenchymal stem cells preferentially modulate macrophages to alleviate pulmonary fibrosis. Stem Cell Res Ther. 2024;15(1):475. [5] LIU Y, DING Y, HOU Y, et al. The miR-130a-3p/TGF-βRII Axis Participates in Inhibiting the Differentiation of Fibroblasts Induced by TGF-β1. Front Pharmacol. 2021;12:732540. [6] MISHARIN AV, MORALES-NEBREDA L, REYFMAN PA, et al. Monocyte-derived alveolar macrophages drive lung fibrosis and persist in the lung over the life span. J Exp Med. 2017; 214(8):2387-2404. [7] SHI J, LI F, LUO M, et al. Distinct Roles of Wnt/β-Catenin Signaling in the Pathogenesis of Chronic Obstructive Pulmonary Disease and Idiopathic Pulmonary Fibrosis. Mediators Inflamm. 2017;2017:3520581. [8] ZHANG E, GENG X, SHAN S, et al. Exosomes derived from bone marrow mesenchymal stem cells reverse epithelial-mesenchymal transition potentially via attenuating Wnt/β-catenin signaling to alleviate silica-induced pulmonary fibrosis. Toxicol Mech Methods. 2021;31(9):655-666. [9] 邓芝花,陈垚鑫,钱进先.间充质干细胞源性外泌体在特发性肺纤维化治疗中的研究进展[J].临床肺科杂志,2023,28(8):1267-1269. [10] CHUANG HM, HO LI, HARN HJ, et al. Recent Findings on Cell-Based Therapies for COVID19-Related Pulmonary Fibrosis. Cell Transplant. 2021; 30:963689721996217. [11] VISHNUPRIYA M, NAVEENKUMAR M, MANJIMA K, et al. Post-COVID pulmonary fibrosis: therapeutic efficacy using with mesenchymal stem cells - How the lung heals. Eur Rev Med Pharmacol Sci. 2021; 25(6):2748-2751. [12] LI X, YUE S, LUO Z. Mesenchymal stem cells in idiopathic pulmonary fibrosis. Oncotarget. 2017;8(60):102600-102616. [13] SHAN Y, ZHANG M, TAO E, et al. Pharmacokinetic characteristics of mesenchymal stem cells in translational challenges. Signal Transduct Target Ther. 2024;9(1):242. [14] WELSH JA, GOBERDHAN DCI, O’DRISCOLL L, et al. Minimal information for studies of extracellular vesicles (MISEV2023): From basic to advanced approaches. J Extracell Vesicles. 2024;13(2):e12404. [15] HARRELL CR, DJONOV V, VOLAREVIC A, et al. Molecular Mechanisms Responsible for the Therapeutic Potential of Mesenchymal Stem Cell-Derived Exosomes in the Treatment of Lung Fibrosis. Int J Mol Sci. 2024;25(8):4378. [16] KLETUKHINA S, MUTALLAPOVA G, TITOVA A, et al. Role of Mesenchymal Stem Cells and Extracellular Vesicles in Idiopathic Pulmonary Fibrosis. Int J Mol Sci. 2022;23(19):11212. [17] WANG Y, LI S, ZHAO J, et al. Snail-mediated partial epithelial mesenchymal transition augments the differentiation of local lung myofibroblast. Chemosphere. 2021;267:128870. [18] LI Y, SHEN Z, JIANG X, et al. Mouse mesenchymal stem cell-derived exosomal miR-466f-3p reverses EMT process through inhibiting AKT/GSK3β pathway via c-MET in radiation-induced lung injury. J Exp Clin Cancer Res. 2022;41(1):128. [19] XU C, HOU L, ZHAO J, et al. Exosomal let-7i-5p from three-dimensional cultured human umbilical cord mesenchymal stem cells inhibits fibroblast activation in silicosis through targeting TGFBR1. Ecotoxicol Environ Saf. 2022;233:113302. [20] ZHAO R, WANG L, WANG T, et al. Inhalation of MSC-EVs is a noninvasive strategy for ameliorating acute lung injury. J Control Release. 2022;345:214-230. [21] XIAO K, HE W, GUAN W, et al. Mesenchymal stem cells reverse EMT process through blocking the activation of NF-κB and Hedgehog pathways in LPS-induced acute lung injury. Cell Death Dis. 2020;11(10):863. [22] 杨静,胡华钟,张书勤,等.脐带间充质干细胞来源的外泌体通过抑制上皮间质转化缓解肺纤维化[J].南方医科大学学报,2020,40(7):988-994. [23] YANG S, LIU P, GAO T, et al. Every road leads to Rome: therapeutic effect and mechanism of the extracellular vesicles of human embryonic stem cell-derived immune and matrix regulatory cells administered to mouse models of pulmonary fibrosis through different routes. Stem Cell Res Ther. 2022;13(1):163. [24] GAO Y, LIU MF, LI Y, et al. Mesenchymal stem cells-extracellular vesicles alleviate pulmonary fibrosis by regulating immunomodulators. World J Stem Cells. 2024;16(6):670-689. [25] 刘树安,陈天恩,经慧,等.Toll样受体4在骨髓间充质干细胞外泌体调控上皮间质转化抑制急性呼吸窘迫综合征后肺纤维化中的作用机制[J].临床肺科杂志,2024,29(11):1633-1637. [26] CHEN W, PENG J, TANG X, et al. MSC-derived exosome ameliorates pulmonary fibrosis by modulating NOD 1/NLRP3-mediated epithelial-mesenchymal transition and inflammation. Heliyon. 2024;11(2):e41436. [27] WANG LL, OUYANG MY, YANG ZE, et al. Mesenchymal stem cells-derived exosomes alleviate radiation induced pulmonary fibrosis by inhibiting the protein kinase B/nuclear factor kappa B pathway. World J Stem Cells. 2025; 17(6):106488. [28] MANSOURI N, WILLIS GR, FERNANDEZ-GONZALEZ A, et al. Mesenchymal stromal cell exosomes prevent and revert experimental pulmonary fibrosis through modulation of monocyte phenotypes. JCI Insight. 2019;4(21): e128060. [29] GAO Y, SUN J, DONG C, et al. Extracellular Vesicles Derived from Adipose Mesenchymal Stem Cells Alleviate PM2.5-Induced Lung Injury and Pulmonary Fibrosis. Med Sci Monit. 2020;26:e922782. [30] CHEN WX, ZHOU J, ZHOU SS, et al. Microvesicles derived from human Wharton’s jelly mesenchymal stem cells enhance autophagy and ameliorate acute lung injury via delivery of miR-100. Stem Cell Res Ther. 2020;11(1):113. [31] KRISHNAN A, HARIKRISHNAN VS, SABAREESWARAN A, et al. Human Wharton’s Jelly Mesenchymal Stem Cells and their Extracellular Vesicles in the Management of Bleomycin-induced Lung Injury in Model Animals: A Comparative Preclinical Study Focused on Histomorphometric Analysis. Curr Stem Cell Res Ther. 2025. doi: 10.2174/011574888X366742250417065341. [32] LI X, WANG Y, AN G, et al. Bone marrow mesenchymal stem cells attenuate silica-induced pulmonary fibrosis via paracrine mechanisms. Toxicol Lett. 2017;270:96-107. [33] CHEN QH, ZHANG Y, GU X, et al. Microvesicles derived from mesenchymal stem cells inhibit acute respiratory distress syndrome-related pulmonary fibrosis in mouse partly through hepatocyte growth factor. World J Stem Cells. 2024;16(8): 811-823. [34] SUN L, ZHU M, FENG W, et al. Exosomal miRNA Let-7 from Menstrual Blood-Derived Endometrial Stem Cells Alleviates Pulmonary Fibrosis through Regulating Mitochondrial DNA Damage. Oxid Med Cell Longev. 2019;2019:4506303. [35] 黄坤,周勇,刘美芳,等.甘草酸二铵联合骨髓间充质干细胞治疗大鼠肺纤维化急性加重的实验研究[J]. 中国呼吸与危重监护杂志, 2020,19(1):64-69. [36] 谭鑫,张沛,杜乐辉,等.间充质干细胞通过抑制铁死亡减轻小鼠放射性肺损伤的作用机制研究[J].中国医学装备,2024,21(5):176-183. [37] CAI L, WANG J, YI X, et al. Nintedanib-loaded exosomes from adipose-derived stem cells inhibit pulmonary fibrosis induced by bleomycin. Pediatr Res. 2024;95(6):1543-1552. [38] ZHANG WY, WEN L, DU L, et al. S-RBD-modified and miR-486-5p-engineered exosomes derived from mesenchymal stem cells suppress ferroptosis and alleviate radiation-induced lung injury and long-term pulmonary fibrosis. J Nanobiotechnology. 2024;22(1):662. [39] LONG Y, YANG B, LEI Q, et al. Targeting Senescent Alveolar Epithelial Cells Using Engineered Mesenchymal Stem Cell-Derived Extracellular Vesicles To Treat Pulmonary Fibrosis. ACS Nano. 2024;18(9):7046-7063. [40] CHENG P, LI S, CHEN H. Macrophages in Lung Injury, Repair, and Fibrosis. Cells. 2021; 10(2):436. [41] DI X, LI Y, WEI J, et al. Targeting Fibrosis: From Molecular Mechanisms to Advanced Therapies. Adv Sci (Weinh). 2025;12(3):e2410416. [42] MAYR CH, SENGUPTA A, ASGHARPOUR S, et al. Sfrp1 inhibits lung fibroblast invasion during transition to injury-induced myofibroblasts. Eur Respir J. 2024;63(2):2301326. [43] SHI L, REN J, LI J, et al. Extracellular vesicles derived from umbilical cord mesenchymal stromal cells alleviate pulmonary fibrosis by means of transforming growth factor-β signaling inhibition. Stem Cell Res Ther. 2021;12(1):230. [44] XU C, ZHAO J, LI Q, et al. Exosomes derived from three-dimensional cultured human umbilical cord mesenchymal stem cells ameliorate pulmonary fibrosis in a mouse silicosis model. Stem Cell Res Ther. 2020;11(1):503. [45] WAN X, CHEN S, FANG Y, et al. Mesenchymal stem cell-derived extracellular vesicles suppress the fibroblast proliferation by downregulating FZD6 expression in fibroblasts via micrRNA-29b-3p in idiopathic pulmonary fibrosis. J Cell Physiol. 2020;235(11):8613-8625. [46] ZHOU J, LIN Y, KANG X, et al. microRNA-186 in extracellular vesicles from bone marrow mesenchymal stem cells alleviates idiopathic pulmonary fibrosis via interaction with SOX4 and DKK1. Stem Cell Res Ther. 2021;12(1):96. [47] SHENTU TP, WONG S, ESPINOZA C, et al. Extracellular vesicles isolated from human mesenchymal stem cells promote resolution of pulmonary fibrosis. FASEB J. 2016;30:160-162. [48] SHENTU TP, HUANG TS, CERNELC-KOHAN M, et al. Thy-1 dependent uptake of mesenchymal stem cell-derived extracellular vesicles blocks myofibroblastic differentiation. Sci Rep. 2017;7(1):18052. [49] SHI XF, TUO YJ, LIAO ZY, et al. Functions and mechanisms of UC-MSC-derived exosomal miR-486-5p in pulmonary fibrosis. Front Oncol. 2025; 15:1542008. [50] ROZIER P, MAUMUS M, MARIA ATJ, et al. Lung Fibrosis Is Improved by Extracellular Vesicles from IFNγ-Primed Mesenchymal Stromal Cells in Murine Systemic Sclerosis. Cells. 2021;10(10):2727. [51] BASALOVA N, SAGARADZE G, ARBATSKIY M, et al. Secretome of Mesenchymal Stromal Cells Prevents Myofibroblasts Differentiation by Transferring Fibrosis-Associated microRNAs within Extracellular Vesicles. Cells. 2020;9(5):1272. [52] CHAROENPHANNATHON JS, WONG PD, ROYCE SG, et al. Human bone marrow mesenchymal stem cell-derived extracellular vesicles induce inverse dose-dependent anti-fibrotic effects in human myofibroblast cultures and bleomycin-injured mice with pulmonary fibrosis. Biomed Pharmacother. 2025;190:118370. [53] ZHAO Y, DU L, SUN J, et al. Exosomal miR-218 derived from mesenchymal stem cells inhibits endothelial-to-mesenchymal transition by epigenetically modulating of BMP2 in pulmonary fibrosis. Cell Biol Toxicol. 2023;39(6):2919-2936. [54] LEI X, HE N, ZHU L, et al. Mesenchymal Stem Cell-Derived Extracellular Vesicles Attenuate Radiation-Induced Lung Injury via miRNA-214-3p. Antioxid Redox Signal. 2021;35(11):849-862. [55] SCOTT MKD, QUINN K, LI Q, et al. Increased monocyte count as a cellular biomarker for poor outcomes in fibrotic diseases: a retrospective, multicentre cohort study. Lancet Respir Med. 2019;7(6):497-508. [56] WILLIS GR, FERNANDEZ-GONZALEZ A, ANASTAS J, et al. Mesenchymal Stromal Cell Exosomes Ameliorate Experimental Bronchopulmonary Dysplasia and Restore Lung Function through Macrophage Immunomodulation. Am J Respir Crit Care Med. 2018;197(1):104-116. [57] BANDEIRA E, OLIVEIRA H, SILVA JD, et al. Therapeutic effects of adipose-tissue-derived mesenchymal stromal cells and their extracellular vesicles in experimental silicosis. Respir Res. 2018; 19(1):104. [58] ZHOU Y, GAO Y, ZHANG W, et al. Exosomes derived from induced pluripotent stem cells suppresses M2-type macrophages during pulmonary fibrosis via miR-302a-3p/TET1 axis. Int Immunopharmacol. 2021;99:108075. [59] LI M, HUANG H, WEI X, et al. Clinical investigation on nebulized human umbilical cord MSC-derived extracellular vesicles for pulmonary fibrosis treatment. Signal Transduct Target Ther. 2025;10(1):179. [60] 铁华,曹佳伟,丁劭瑞,等.胎盘间充质干细胞外泌体通过调控NF-κB信号通路抑制矽肺肺纤维化的进展[J].宁夏医科大学学报,2024, 46(6):554-562. [61] LIANG X, LI Y, WU Y, et al. Human umbilical cord mesenchymal stem cell-derived microvesicles alleviate pulmonary fibrosis by inhibiting monocyte‒macrophage migration through ERK1/2 signaling-mediated suppression of CCL2 expression. Stem Cell Res Ther. 2025;16(1):145. [62] HOU L, ZHU Z, JIANG F, et al. Human umbilical cord mesenchymal stem cell-derived extracellular vesicles alleviated silica induced lung inflammation and fibrosis in mice via circPWWP2A/miR-223-3p/NLRP3 axis. Ecotoxicol Environ Saf. 2023;251: 114537. [63] CHO KS, KANG SA, KIM SD, et al. Dendritic cells and M2 macrophage play an important role in suppression of Th2-mediated inflammation by adipose stem cells-derived extracellular vesicles. Stem Cell Res. 2019;39:101500. [64] KINDER BW, BROWN KK, SCHWARZ MI, et al. Baseline BAL neutrophilia predicts early mortality in idiopathic pulmonary fibrosis. Chest. 2008;133(1):226-232. [65] SU VY, LIN CS, HUNG SC, et al. Mesenchymal Stem Cell-Conditioned Medium Induces Neutrophil Apoptosis Associated with Inhibition of the NF-κB Pathway in Endotoxin-Induced Acute Lung Injury. Int J Mol Sci. 2019;20(9):2208. [66] TAN JL, LAU SN, LEAW B, et al. Amnion Epithelial Cell-Derived Exosomes Restrict Lung Injury and Enhance Endogenous Lung Repair. Stem Cells Transl Med. 2018;7(2):180-196. [67] AMTIL-OUAHDI I, VERGARA F, RIO C, et al. EVs Biodistribution and Antifibrotic Impact in Aged Lung Fibrosis Model. Biofactors. 2025;51(3): e70021. [68] LEI L, ZHAO C, QIN F, et al. Th17 cells and IL-17 promote the skin and lung inflammation and fibrosis process in a bleomycin-induced murine model of systemic sclerosis. Clin Exp Rheumatol. 2016;34 Suppl 100(5):14-22. [69] KOUDSTAAL T, FUNKE-CHAMBOUR M, KREUTER M, et al. Pulmonary fibrosis: from pathogenesis to clinical decision-making. Trends Mol Med. 2023;29(12):1076-1087. [70] LAI P, CHEN X, GUO L, et al. A potent immunomodulatory role of exosomes derived from mesenchymal stromal cells in preventing cGVHD. J Hematol Oncol. 2018;11(1):135. [71] HARRELL CR, JOVICIC N, DJONOV V, et al. Mesenchymal Stem Cell-Derived Exosomes and Other Extracellular Vesicles as New Remedies in the Therapy of Inflammatory Diseases. Cells. 2019;8(12):1605. [72] HARRELL CR, MILORADOVIC D, SADIKOT R, et al. Molecular and Cellular Mechanisms Responsible for Beneficial Effects of Mesenchymal Stem Cell-Derived Product “Exo-d-MAPPS” in Attenuation of Chronic Airway Inflammation. Anal Cell Pathol (Amst). 2020;2020:3153891. [73] SANG L, GUO X, FAN H, et al. Mesenchymal Stem Cell-Derived Extracellular Vesicles as Idiopathic Pulmonary Fibrosis Microenvironment Targeted Delivery. Cells. 2022;11(15):2322. [74] SENGUPTA V, SENGUPTA S, LAZO A, et al. Exosomes Derived from Bone Marrow Mesenchymal Stem Cells as Treatment for Severe COVID-19. Stem Cells Dev. 2020;9(12):747-754. [75] LAI RC, ARSLAN F, LEE MM, et al. Exosome secreted by MSC reduces myocardial ischemia/reperfusion injury. Stem Cell Res. 2010;4(3): 214-222. [76] ZOU X, GU D, XING X, et al. Human mesenchymal stromal cell-derived extracellular vesicles alleviate renal ischemic reperfusion injury and enhance angiogenesis in rats. Am J Transl Res. 2016;8(10): 4289-4299. [77] WU H, YU Y, HUANG H, et al. Progressive Pulmonary Fibrosis Is Caused by Elevated Mechanical Tension on Alveolar Stem Cells. Cell. 2020;180(1):107-121.e17. [78] LIU Q, BI Y, SONG S, et al. Exosomal miR-17-5p from human embryonic stem cells prevents pulmonary fibrosis by targeting thrombospondin-2. Stem Cell Res Ther. 2023; 14(1):234. [79] BARI E, PERTEGHELLA S, DI SILVESTRE D, et al. Pilot Production of Mesenchymal Stem/Stromal Freeze-Dried Secretome for Cell-Free Regenerative Nanomedicine: A Validated GMP-Compliant Process. Cells. 2018;7(11):190. [80] DINH PC, PAUDEL D, BROCHU H, et al. Inhalation of lung spheroid cell secretome and exosomes promotes lung repair in pulmonary fibrosis. Nat Commun. 2020;11(1):1064. |
| [1] | 蒋星海, 宋玉林, 李德津, 邵建敏, 徐军志, 刘华凯, 吴应国, 沈岳辉, 冯思诚. 血管内皮生长因子165基因转染骨髓间充质干细胞构建血管化两亲性肽凝胶模块[J]. 中国组织工程研究, 2026, 30(8): 1903-1911. |
| [2] | 宋浦蓁, 马贺宾, 陈宏广, 章亚东. 骨髓间充质干细胞外泌体联合转化生长因子β1对巨噬细胞的作用[J]. 中国组织工程研究, 2026, 30(7): 1616-1623. |
| [3] | 周思瑞, 徐玉坤, 赵可伟. 白芷细胞外囊泡对抗黑色素的思路和方法[J]. 中国组织工程研究, 2026, 30(7): 1747-1754. |
| [4] | 黄嘉雯, 潘之怡, 薛文君, 廉源沛, 徐建达. 植物源性囊泡与恶性肿瘤治疗:跨物种交流并调节宿主细胞反应[J]. 中国组织工程研究, 2026, 30(7): 1828-1838. |
| [5] | 彭团辉, 宋洪明, 杨 玲, 丁小歌, 蒙鹏骏. 长期耐力运动对自然衰老小鼠kl/FGF23轴及钙磷代谢的影响[J]. 中国组织工程研究, 2026, 30(5): 1089-1095. |
| [6] | 刘可新, 郝凯敏, 庄文越, 李正祎. 自噬相关基因在肺纤维化模型中的表达:生物信息学分析及实验验证[J]. 中国组织工程研究, 2026, 30(5): 1129-1138. |
| [7] | 刘新月, 李春年, 李一卓, 徐世芳. 口腔牙槽骨缺损的再生修复[J]. 中国组织工程研究, 2026, 30(5): 1247-1259. |
| [8] | 郭嘉忱, 高 俊, 戴文昊, 廖华远, 蒋 优, 张 曦. 压应力微环境在骨折愈合过程中对细胞因子的影响[J]. 中国组织工程研究, 2026, 30(4): 908-916. |
| [9] | 杨 肖, 白月辉, 赵甜甜, 王东昊, 赵 琛, 袁 硕. 颞下颌关节骨关节炎软骨退变:机制及再生的挑战[J]. 中国组织工程研究, 2026, 30(4): 926-935. |
| [10] | 王正业, 刘万林, 赵振群. 血管内皮生长因子A 靶向调控血管化治疗激素性股骨头坏死的机制[J]. 中国组织工程研究, 2026, 30(3): 671-679. |
| [11] | 杨 虎, 郑 宇, 贾承明, 王 通, 张广飞, 纪垚垚. 免疫微环境调节骨再生[J]. 中国组织工程研究, 2026, 30(3): 701-710. |
| [12] | 刁有录, 高 佳, 潘国庆. 生物募集组织修复材料:调控细胞和因子的迁移及改善组织整合的优势[J]. 中国组织工程研究, 2026, 30(20): 5270-5281. |
| [13] | 白相宇, 霍 峰, 郝 妍, 王泽成, 郭晓宇. 负载血小板衍生生长因子BB的壳聚糖/还原氧化石墨烯支架修复牙槽骨缺损[J]. 中国组织工程研究, 2026, 30(2): 329-337. |
| [14] | 周世博, 俞 兴, 陈海龙, 熊 洋. 纳米晶胶原基骨联合补肾壮筋汤修复骨质疏松大鼠骨缺损[J]. 中国组织工程研究, 2026, 30(2): 354-361. |
| [15] | 钟焯岚, 彭芝娜, 田晓红, 韩翠菲, 张紫菡, 楚佳奇. 五加皮外泌体样纳米囊泡促进人骨髓间充质干细胞成骨分化[J]. 中国组织工程研究, 2026, 30(19): 4825-4835. |
中国组织工程研究杂志出版内容重点:干细胞;骨髓干细胞;造血干细胞;脂肪干细胞;肿瘤干细胞;胚胎干细胞;脐带脐血干细胞;干细胞诱导;干细胞分化;组织工程
1.1.1 检索人及检索时间 第一作者在2025年8月进行检索。
1.1.2 检索文献时限 2025年8月前发表的中英文文献,重点检索近10年发表的英文文献。
1.1.3 检索数据库 中国知网、PubMed数据库、clinicaltrials.gov数据库(https://clinicaltrials.gov/)、中国临床试验注册中心。
1.1.4 检索词 中文检索词:“间充质干细胞,细胞外囊泡,肺纤维化,上皮细胞,血管内皮细胞,巨噬细胞,中性粒细胞”;英文检索词:“Mesenchymal stem cells,Extracellular vesicles,Pulmonary fibrosis,Alveolar epithelium,Microvascular endothelium,Macrophages,Neutrophils”。
1.1.5 检索文献类型 研究性论文、综述。
1.1.6 手工检索情况 无。
1.1.7 检索策略 以中国知网和PubMed数据库检索策略为例,见图1。
1.1.8 检索文献量 初步检索到中文文献45篇,英文文献142篇。
1.2 入组标准
1.2.1 纳入标准 ①无学术不端、观点明确、论据充分、内容可靠、创新性强且与研究主题密切相关的文献;②间充质干细胞来源细胞外囊泡治疗肺纤维化密切相关的文献;③与肺纤维化病理进程相关的文献。
1.2.2 排除标准 内容重复相关性差、可靠性低、与研究主题不相符、无法完整获取或者重复陈旧性文献。
1.3 文献质量评估和数据的提取 初步检索相关文献总计187篇。通过阅读标题、摘要和全文后对保留的文献进行深入研究,最终纳入56篇文献进行归纳分析,其中来源于PubMed数据库50篇,来源于中国知网6篇。文献筛选流程图,见图2。
中国组织工程研究杂志出版内容重点:干细胞;骨髓干细胞;造血干细胞;脂肪干细胞;肿瘤干细胞;胚胎干细胞;脐带脐血干细胞;干细胞诱导;干细胞分化;组织工程
表2[28,39,59,75-78]。为规避干细胞疗法潜在的致瘤风险和安全性隐患,学界提出以干细胞来源细胞外囊泡替代治疗的新策略:此类无细胞疗法不仅可通过常规药物评估体系进行安全性验证,还能规避免疫排斥、感染风险及肿瘤生成等安全隐患,同时大幅降低细胞培养所需的时间和经济成本[27]。
间充质干细胞来源细胞外囊泡已被证实可通过递送蛋白质、miRNA等活性组分介导肺损伤修复,核心机制在于继承亲本细胞的免疫调节与再生潜能[38]。文章系统总结了间充质干细胞来源细胞外囊泡干预肺纤维化的细胞调控网络(图4):在肺泡上皮细胞中,通过多通路协同作用(如调控蛋白激酶B/糖原合成酶激酶3β、转化生长因子β/Smad、核因子κB等信号轴)抑制上皮-间充质转化进程,并靶向凋亡、增殖及炎症反应,从而在肺纤维化中维持肺泡上皮稳态、减少胶原沉积,核心机制是通过递送特定miRNA组合(如miR-466f-3p、let-7等)干预关键因子(SNAIL、β-catenin),同步阻断促纤维化通路网络(转化生长因子β/Wnt等),实现抗纤维化与组织修复;通过递送多种miRNA (如miR-21-5p、let-7i-5p、miR-29b-3p等)靶向抑制转化生长因子β/Smad、卷曲蛋白6、SOX4/DKK1等关键通路,阻断成纤维细胞的活化、增殖及迁移;通过递送miR-218靶向转化生长因子β/甲基化CpG结合蛋白2/骨形态发生蛋白2轴阻断内皮-间充质转化,递送miR-214-3p抑制ATM/P53/P21通路,协同减轻肺纤维化进程;通过重编程单核细胞(促炎→抗炎)、调控巨噬细胞极化(如miR-302a-3p/TET1抑制M2分化)及靶向炎症通路(如circPWWP2A/miR-223-3p/NLRP3轴),抑制促炎因子(肿瘤坏死因子α/白细胞介素1β等)释放并阻断成纤维细胞活化;抑制树突状细胞成熟、阻断中性粒细胞趋化/氧化应激、重塑Th17/Treg平衡协同阻断“炎症-纤维化”恶性循环,重塑肺部免疫微环境。这为间充质干细胞来源细胞外囊泡作为一种无细胞疗法治疗肺损伤提供了理论依据。值得注意的是,经靶向肽修饰或药物共载等工程化策略优化的间充质干细胞来源细胞外囊泡,可实现细胞调控网络的精准靶向,为肺纤维化治疗提供具备智能递送特性的新型解决方案。
中国组织工程研究杂志出版内容重点:干细胞;骨髓干细胞;造血干细胞;脂肪干细胞;肿瘤干细胞;胚胎干细胞;脐带脐血干细胞;干细胞诱导;干细胞分化;组织工程
文题释义:
间充质干细胞来源细胞外囊泡:是由间充质干细胞分泌的纳米级膜包裹的囊泡,直径通常在30-1 000 nm之间。这些囊泡是细胞间通讯的重要媒介,携带多种生物活性分子,包括蛋白质、脂质、核酸和代谢产物。间充质干细胞来源细胞外囊泡保留了母细胞的许多治疗特性,具有更低的免疫原性和更高的安全性。它们通过旁分泌机制发挥作用,能够穿越生物屏障(如血脑屏障),在组织修复和免疫调节中表现出显著的效果。
肺纤维化:以肺泡结构进行性破坏、细胞外基质大量沉积和肺瘢痕形成为特征的间质性肺部疾病,伴随不可逆的肺功能下降及进行性呼吸衰竭。
#br#
中国组织工程研究杂志出版内容重点:干细胞;骨髓干细胞;造血干细胞;脂肪干细胞;肿瘤干细胞;胚胎干细胞;脐带脐血干细胞;干细胞诱导;干细胞分化;组织工程
中国科研团队在间充质干细胞来源细胞外囊泡治疗肺纤维化领域已取得全球领先地位。清华大学、解放军总医院和海南医学院等机构构建了从基础研究到临床转化的完整创新链:在机制层面率先阐明miR-486-5p调控巨噬细胞极化的新通路;在技术层面开发出全球首个符合GMP标准的雾化细胞外囊泡制备系统;在临床层面完成了具有里程碑意义的人体试验。这些突破使中国在该领域的专利数量和国际论文引用量均居世界首位。特别值得注意的是,中国团队创新性地将雾化吸入这一传统给药方式与现代外泌体技术结合,克服了肺部递送效率低下的国际难题,为呼吸系统疾病治疗提供了新范式。
#br#
中国组织工程研究杂志出版内容重点:干细胞;骨髓干细胞;造血干细胞;脂肪干细胞;肿瘤干细胞;胚胎干细胞;脐带脐血干细胞;干细胞诱导;干细胞分化;组织工程
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||