中国组织工程研究 ›› 2022, Vol. 26 ›› Issue (4): 617-625.doi: 10.12307/2022.101
• 生物材料综述 biomaterial review • 上一篇 下一篇
基于透明质酸的复合水凝胶修复骨关节炎软骨损伤:应用与机制
张 通1,蔡金池1,袁志发1,赵海燕2,韩兴文2,王文己2
- 1兰州大学第一临床医学院,甘肃省兰州市 730000;2兰州大学第一医院骨科,甘肃省兰州市 730000
-
收稿日期:2020-08-07修回日期:2020-08-11接受日期:2020-09-19出版日期:2022-02-08发布日期:2021-12-06 -
通讯作者:王文己,主任医师,博士,博士生导师,兰州大学第一医院骨科,甘肃省兰州市 730000 -
作者简介:张通,男,1994年生,陕西省汉中市人,蒙古族,兰州大学第一临床医学院在读硕士,医师,主要从事骨科相关生物材料研究。 -
基金资助:兰州市人才创新创业项目(2019RC32),项目负责人:王文己
Hyaluronic acid-based composite hydrogel in cartilage injury caused by osteoarthritis: application and mechanism
Zhang Tong1, Cai Jinchi1, Yuan Zhifa1, Zhao Haiyan2, Han Xingwen2, Wang Wenji2
- 1The First Clinical Medical College of Lanzhou University, Lanzhou 730000, Gansu Province, China; 2Department of Orthopedics, First Hospital of Lanzhou University, Lanzhou 730000, Gansu Province, China
-
Received:2020-08-07Revised:2020-08-11Accepted:2020-09-19Online:2022-02-08Published:2021-12-06 -
Contact:Wang Wenji, Chief physician, MD, Doctoral supervisor, Department of Orthopedics, First Hospital of Lanzhou University, Lanzhou 730000, Gansu Province, China -
About author:Zhang Tong, Master candidate, Physician, The First Clinical Medical College of Lanzhou University, Lanzhou 730000, Gansu Province, China -
Supported by:Lanzhou Talent Innovation and Entrepreneurship Project, No. 2019RC32 (to WWJ)
摘要:

文题释义:
基于透明质酸的复合水凝胶:透明质酸是一种天然多糖,也是人体中常见的一种糖胺聚糖,因其生物相容性、可降解性等生物性能被制作为水凝胶广泛应用于组织工程中,尤其通过不同方法与生物因子、天然材料、合成材料、3D打印技术、多肽、机械刺激联合制成复合水凝胶,在软骨修复方面有着巨大的前景。
骨关节炎的软骨损伤:骨关节炎是一种退行性病变,软骨组织的慢性炎症通过免疫反应等机制破坏软骨的蛋白多糖及Ⅱ型胶原合成,软骨组织的自我修复因其缺乏血管而十分困难,最终导致软骨损伤。
背景:透明质酸是一种天然多糖,也是人体中常见的一种糖胺聚糖,在眼睛和关节中的浓度最高,因其生物相容性、可降解性和低免疫原性而被制成水凝胶广泛应用于组织工程中。
目的:介绍各类透明质酸复合水凝胶的特点以及在骨关节炎软骨损伤修复中的应用。
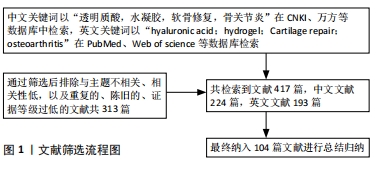
方法:以“透明质酸,水凝胶,软骨修复,骨关节炎”为中文关键词检索CNKI、万方等数据库,以“hyaluronic acid;hydrogel;Cartilage repair;osteoarthritis”为英文关键词检索PubMed、Web of science等数据库,检索1995年1月至2020年7月发表的文献,筛选后进一步分析总结。
结果与结论:透明质酸水凝胶优异的生物性能使其在骨关节炎软骨修复中得到了广泛研究,尤其是通过联合生物因子、天然材料、合成材料、3D打印技术、多肽、机械刺激等改善了透明质酸水凝胶的性能,推进了其在软骨组织工程中的应用。在未来随着化学、材料、物理、生物等多学科的综合发展,更加深入地了解透明质酸水凝胶降解的机制及探索骨关节炎的软骨损伤机制,有望设计出可以高效、无任何不良反应、更加适用软骨组织工程的透明质酸复合水凝胶。
https://orcid.org/0000-0002-9325-0230 (张通)
中国组织工程研究杂志出版内容重点:生物材料;骨生物材料; 口腔生物材料; 纳米材料; 缓释材料; 材料相容性;组织工程
中图分类号:
引用本文
张 通, 蔡金池, 袁志发, 赵海燕, 韩兴文, 王文己. 基于透明质酸的复合水凝胶修复骨关节炎软骨损伤:应用与机制[J]. 中国组织工程研究, 2022, 26(4): 617-625.
Zhang Tong, Cai Jinchi, Yuan Zhifa, Zhao Haiyan, Han Xingwen, Wang Wenji. Hyaluronic acid-based composite hydrogel in cartilage injury caused by osteoarthritis: application and mechanism[J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 617-625.
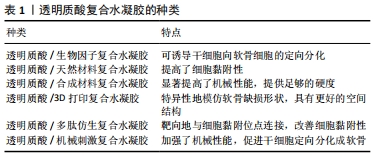
2.1 透明质酸水凝胶的特点 透明质酸是一种线性糖胺聚糖,由N-乙酰基-d-葡萄糖胺和d-葡萄糖醛酸的重复单元组成,单糖单元通过交替的β-1,3和β-1,4糖苷键连接在一起[11]。透明质酸是一种天然存在于关节的物质,是糖胺多糖的主要构成,有助于保持关节润滑所需的黏弹性,维持软骨对压缩力的结构抵抗力,还对软骨表面的润滑起着重要作用。当透明质酸在自由基或某些酶的作用下降解时,滑膜液失去润滑性能,导致关节磨损加剧。在骨关节炎中,炎性因子渗出和滑膜细胞功能异常可使透明质酸浓度降低33%-50%,润滑减少会对病变软骨产生进一步的压力,从而破坏软骨表面完整性,并导致进一步的疼痛[12-13]。根据链长透明质酸有两种形式:低分子质量和高分子质量,分别为≤2×106 Da和> 2×106 Da。高分子质量透明质酸能够在水中结合10-10 000倍质量的水,从而填充空间,起到减震器和润滑剂的作用。从生物学角度来看,高分子质量透明质酸具有抗血管生成和抗炎作用,并具有免疫抑制能力;然而,高分子质量透明质酸可分解为低分子质量透明质酸,低分子质量透明质酸在内源性信号通路被激活后可分泌炎性细胞因子[14-15]。由于透明质酸分子和蛋白质的静电相互作用,透明质酸在关节的润滑、减震和黏弹性行为中起着重要作用,而黏弹性与透明质酸的分子质量和浓度直接相关。在缓慢运动过程中,透明质酸的线性链沿流动方向缓慢排列,表现为黏性流体;当关节受到快速冲击(即跑或跳)时,透明质酸分子没有足够的时间重新排列并表现出弹性行为,因此能够减震。在骨关节炎中,由于透明质酸分子质量和浓度的降低,关节液的黏弹性及其保护软骨的能力显著降低,加速关节的退变[16-17]。
2.1.1 透明质酸水凝胶的优势 透明质酸由于原有透明质酸酶的作用易受到各种酶降解过程的影响,为了控制降解速率和提高其机械性能,人们采用了交联不同的化学物质来制作一种稳定的透明质酸水凝胶,在保持其生物性能的同时获得更稳定的物理力学性能[18]。透明质酸中羟基和羧基等官能团的存在为各种化学改性提供了锚定物,有利于调控机械性能,从而构造一种适合软骨修复的水凝胶[19]。透明质酸水凝胶的制备方式包含物理和化学交联两种:①在物理交联中,非共价键和超分子相互作用因其独特的性质而被广泛应用于透明质酸水凝胶的设计中。由于这些相互作用是可逆的,非共价组装可以获得性能可调的水凝胶,并且对光、pH值、温度等各种信号都有响应。与共价交联相比,物理交联导致形成机械和化学稳定性较差的水凝胶[20]。包合及络合反应是制备物理凝胶最常用的策略,是超分子相互作用和结构互补的结果,2个分子称为“宿主”和“客体”。环糊精是应用最广泛的宿主之一,具有疏水性空腔,将透明质酸官能化后,通过对其他疏水性客体的亲和力进行交联形成透明质酸水凝
胶[21]。②化学交联是制备具有优异化学稳定性、机械稳定性和热稳定性透明质酸水凝胶的一种通用方法,然而这种方法存在一些局限性,如使用金属催化剂、光引发剂及反应产率低等[22]。透明质酸水凝胶可以通过缩合反应、酶交联、二硫键交联、点击化学、聚合等方法得到,也可以通过二乙烯基砜、戊二醛、碳二亚胺、双环氧化物等直接交联,但直接交联需要苛刻的反应条件,会产生有毒的副产品,并且使用的交联剂具有细胞毒性。由于点击化学具有高特异性、高产率和温和的反应条件,目前最有前途的水凝胶制备策略就是点击化学反应[23]。透明质酸是一种存在于所有结缔组织中的糖胺聚糖,是细胞外基质的主要组成部分,而细胞外基质是一种亲水性基质,呈均匀的凝胶状。透明质酸通过交联形成水凝胶,保持其三维结构而不溶解,这使得透明质酸水凝胶在结构上与细胞外基质相似,具有非常高的含水量、良好的生物相容性和较低的免疫原性,构造类似天然细胞外基质的微观结构,从而提供了结构支持和保护,以及细胞在三维环境中相互作用的能力,支持软骨细胞的生长修复[24]。
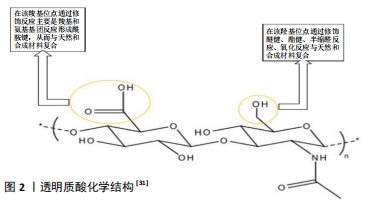
2.1.2 透明质酸水凝胶在软骨组织工程中的优势 天然聚合物在生物工程具有调节细胞反应和相互作用的特性,当植入体内时,天然聚合物表现出与体内生物分子非常相似的结构相容性,从而降低了免疫反应的可能性。在软骨组织工程中,透明质酸是软骨细胞外基质的主要组成,故拥有较其他聚合物更好的生物相容性、天然生物功能性、生物降解性、低免疫原性和多功能性,从而成为生物水凝胶设计中用来修复软骨最广泛的材料[25]。透明质酸与许多细胞表面受体互相作用,如CD44、透明质酸介导的运动性受体和细胞间黏附分子1,并参与组织流体力学、细胞增殖和迁移[26]。官能团(如硫醇、丙烯酸酯、胺等)可以通过温和的化学反应与透明质酸主链结合,并进一步用作交联位点,形成水凝胶,作为体外和体内细胞的支架[27]。当前应用在软骨组织工程中的透明质酸水凝胶主要是能够模拟细胞外基质的机械性能,并可被酶降解,足以承受体内周围组织的压力而不变形或塌陷。透明质酸水凝胶的机械性能可通过调节交联密度和透明质酸浓度进行调整,增加交联密度可以增强整个网络进而增加凝胶的硬度,随着透明质酸浓度的增长,凝胶的硬度加强[28],进而合成最适合软骨组织工程的透明质酸水凝胶。通过在组织工程中对软骨损伤修复的研究发现,透明质酸水凝胶较其他水凝胶具有以下优势:高水化微环境,可改善物质交换;模拟原始软骨细胞外基质的结构,以促进细胞与基质接触;与邻近组织的良好整合;间充质干细胞被其细胞表面受体CD44靶向引导聚集[28-29];良好的黏弹性可诱导间充质干细胞定向分化[30]。另一方面,由于细胞更喜欢黏附在中性或阳离子界面上,故透明质酸水凝胶因缺乏细胞黏附位点和阴离子性导致细胞黏附性较差,见图2[31]。因此,透明质酸水凝胶需要复合其他材料来提升机械性能和黏附性,推进其在软骨组织工程中的应用。
2.2 透明质酸水凝胶复合不同材料在软骨修复中的应用 为了改善透明质酸水凝胶的性质,弥补应用在软骨修复时的不足,国内外学者主要通过将透明质酸水凝胶复合生物因子、天然材料、合成材料、3D打印技术、多肽、机械刺激提升了水凝胶的物理性能,促进了种子细胞的软骨分化和增殖,见图3。
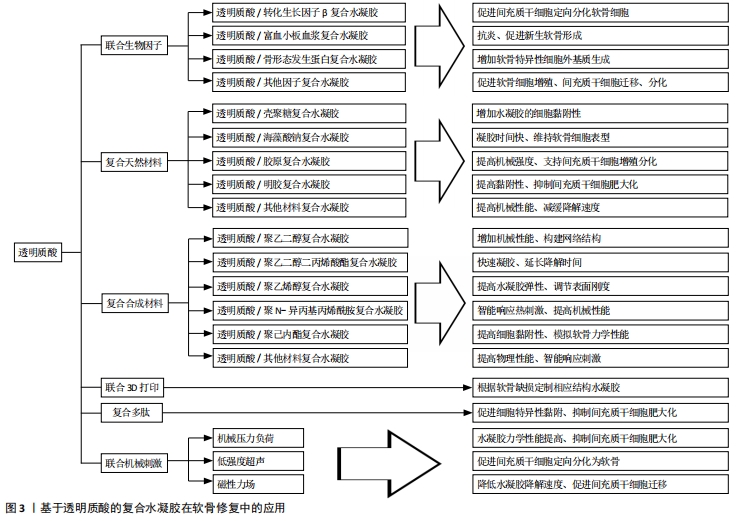
2.2.1 透明质酸/生物因子复合水凝胶在软骨修复中的应用 研究发现,许多生物调节因子可以诱导间充质干细胞的软骨形成和分化,这些生物调节因子包括转化生长因子β、骨形态发生蛋白、成纤维细胞生长因子、胰岛素样生长因子以及多种信号通路(hedgehog、notch、Wnt/β-catenin、血管生成和缺氧信号通路)[32]。
透明质酸/转化生长因子β复合水凝胶:转化生长因子β已被证明对软骨细胞表型的诱导和保持至关重要。DENG等[33]发现在透明质酸/聚内消旋聚乳酸复合水凝胶中加入转化生长因子β3,联合骨髓间充质干细胞培养可以增加糖胺聚糖沉积和软骨基因表达。虽然骨髓间充质干细胞能够在体外转化生长因子β诱导下形成软骨样组织,但移植到不受控制的体内环境中通常会导致细胞外基质的广泛钙化,导致更多的软骨下小梁骨形成[34]。BIAN等[35]采用一种由带相反电荷聚电解质制备的自组装双层纳米膜来减少转化生长因子β3从海藻酸钠微球中的初始释放,与一次性直接包埋转化生长因子β3相比,在透明质酸水凝胶包裹的海藻酸钠微球中单次给药可以促进间充质干细胞的软骨生成,并产生更好的透明软骨。地塞米松是一种合成的糖皮质激素,已知具有促软骨生成作用和抗炎作用。KIM等[36]研究发现,糖皮质激素联合透明质酸可增强糖皮质激素受体活性,以抑制p38 MAPK信号通路的激活。地塞米松、转化生长因子β3联合间充质干细胞培养可促进软骨形成,并抑制基质分解代谢。
透明质酸/富血小板血浆复合水凝胶:富血小板血浆易于提取,含多种生长因子、蛋白质和细胞因子,通过刺激细胞增殖和诱导软骨缺损部位的软骨生成来启动软骨修复,并且具有抗炎作用[37]。CHEN等[38]利用小鼠骨关节炎模型发现,富血小板血浆联合透明质酸水凝胶可抑制小鼠炎症相关的趋化因子和细胞因子,最终抑制免疫细胞的募集和激活。YAN等[39]用富血小板血浆联合透明质酸水凝胶治疗小猪骨关节炎模型,6个月后软骨表面光滑,与邻近软骨结合良好,力学性能得到改善,软骨细胞呈柱状分布,糖胺聚糖和Ⅱ型胶原含量提高,无肥大软骨形成。TURAJANE等[40]用自体外周血干细胞联合富血小板血浆、人粒细胞集落刺激因子和透明质酸治疗膝关节骨关节炎患者,治疗12个月后,治疗组在减轻疼痛和日常活动能力方面优于单纯透明质酸组。LIU等[37]通过在富血小板血浆中添加改性的透明质酸开发出一种光交联富血小板血浆/透明质酸复合水凝胶,可原位形成,实现生长因子的可控释放,提高水凝胶的软骨黏附性和整合性,在兔全层软骨缺损模型中能促成完整的透明软骨再生。
透明质酸/骨形态发生蛋白复合水凝胶:骨形态发生蛋白2是一种功能蛋白,可促进间充质干细胞定向分化成为软骨细胞,并刺激关节软骨细胞合成软骨基质,但骨形态发生蛋白2在软骨组织中的高扩散性和对酶降解的敏感性使其作用有限[41]。JHA等[42]将骨形态发生蛋白2用透明质酸凝胶粒子包裹并加入到透明质酸水凝胶中,形成的三维微环境不仅维持了软骨细胞表型,而且增加了软骨特异性细胞外基质的产生;骨形态发生蛋白2以可控的方式从水凝胶中释放,可长时间作用于间充质干细胞。XU等[41]合成了肝素修饰的透明质酸水凝胶粒子,微观颗粒呈球形,并包含适合生长因子封装的纳米孔;共价固定化肝素保留了其与骨形态发生蛋白2特异结合的能力,其释放动力学可以通过调节颗粒组成进行调节;与纯透明质酸颗粒相比,该粒子具有更高的骨形态发生蛋白2负载能力,可加强诱导小鼠间充质干细胞的软骨分化,增加软骨形成标志物的mRNA水平和软骨特异性细胞外基质成分。但AULIN等[43]认为关节中高浓度的骨形态发生蛋白2可能使软骨细胞向更退化的表型转移,导致肥大和异位骨形成而不是维持软骨细胞表型;还发现使用透明质酸水凝胶与骨形态发生蛋白2联合治疗不能改善兔诱导性软骨缺损的愈合,反而可诱导异位骨形成,严重影响关节功能。
透明质酸/其他生物因子复合水凝胶:血小板裂解物含有白蛋白、纤维蛋白原及大量生长因子,并且较富血小板血浆的免疫原性低。在一些研究中发现,血小板裂解物能促进细胞增殖、迁移和软骨分化[44]。JOOYBAR等[45]将血小板裂解物加入透明质酸酪胺水凝胶中,发现人骨髓间充质干细胞附着在基质中并扩散开来,逐渐沉积Ⅱ型胶原和蛋白多糖;新的细胞外基质的沉积与凝胶降解同时进行,最后形成坚硬致密的基质。这些发现证明了可注射透明质酸/血小板裂解物复合水凝胶作为软骨再生细胞输送系统的潜力。
在关节软骨中胰岛素样生长因子1是一种重要的合成代谢生长因子,促进软骨基质的生成和软骨细胞的增殖。YOON等[46]发现,在体外和体内透明质酸的加入可以增强胰岛素样生长因子与海藻酸钠水凝胶包埋软骨细胞的相互作用,但透明质酸在高质量浓度(5 g/L)下可以阻断胰岛素样生长因子1,干扰软骨细胞胰岛素样生长因子1信号转导,从而控制其向软骨细胞的传递以及内源性胰岛素样生长因子1的表达,抑制软骨细胞增殖。
成纤维细胞生长因子在软骨发生以及发育和修复过程中发挥各种作用,成纤维细胞生长因子在单层培养中增加了间充质干细胞的生长速度和寿命,提高了软骨细胞的迁移速度。KAYAKABE等[47]在间充质干细胞扩张期间单独使用成纤维细胞生长因子2可以促进增殖,延缓衰老,并为随后更好的软骨细胞分化做好准备。用透明质酸水凝胶移植成纤维细胞生长因子培养的间充质干细胞,能显著提高关节软骨的修复能力,增加Ⅱ型胶原表达。
2.2.2 透明质酸水凝胶复合天然材料在软骨损伤修复中的应用 软骨组织工程中常用制备水凝胶的天然材料包括琼脂糖、海藻酸钠、壳聚糖、胶原蛋白、纤维蛋白和透明质酸等。通过特定的表面受体,这些生物材料可以与细胞相互作用,促进细胞迁移、细胞外分子的产生和增殖[48]。不同的材料都有着各自的优点,将不同的天然材料通过物理或化学方式与透明质酸进行复合形成新的水凝胶,弥补了透明质酸水凝胶的不足,改善了其在软骨修复中的应用前景。
透明质酸/壳聚糖复合水凝胶:壳聚糖是一种线性多糖,由β-1,4糖苷键连接的D-氨基葡萄糖和N-乙酰-D-氨基葡萄糖残基衍生而来,在节肢动物骨骼中天然存在,其结构类似于天然存在的糖胺聚糖,在人体内被酶生物降解。REMYA等[49]以壳聚糖和透明质酸为原料制备了一种水凝胶,体外研究表明该复合水凝胶模拟了关节软骨的细胞外基质环境,促进软骨细胞的黏附和增殖,能有效支持软骨细胞的成软骨活性,并促进软骨细胞的基质生成,Ⅱ型胶原表达上调,去分化标志物即Ⅰ型胶原基因表达明显下降。壳聚糖的阳离子性质允许保留活性软骨细胞分泌的阴离子糖胺聚糖、蛋白多糖和其他带负电荷的物质,从而进一步增加细胞外基质浓度。LIU等[50]以壳聚糖和透明质酸为原料制备复合水凝胶,发现其在体外能促进骨髓间充质干细胞的增殖和向软骨分化,而随着细胞外基质颗粒在水凝胶中浓度的增长,水凝胶的抗压强度提高、降解速率加快,并且促进细胞表达更多的软骨细胞标记物(如SOX9、蛋白聚糖和Ⅱ型胶原)。
PARK等[51]利用光交联制备了一种由甲基丙烯酸乙二醇壳聚糖和透明质酸组成的可注射水凝胶,在交联前将透明质酸溶解在光交联壳聚糖溶液中制备复合水凝胶,壳聚糖网络捕获高分子指令的透明质酸形成半互穿网络;随后将软骨细胞包裹在水凝胶中,21 d后发现大多数细胞具有软骨细胞的圆形特征以及周围的软骨陷窝,并且观察到细胞增殖和糖胺聚糖的含量增加;在37 ℃溶菌酶溶液中培养42 d后复合水凝胶的剩余重量为50%,明显延缓了水凝胶的降解。RIEGER等[52]将透明质酸/壳聚糖复合水凝胶注射入兔子骨关节炎模型中,发现软骨下骨微结构参数(骨小梁厚度和骨小梁体积分数)明显改变,软骨下骨板和小梁骨密度降低,可显著保护软骨和延缓骨关节炎的发展。
透明质酸/海藻酸钠复合水凝胶:海藻酸钠是一种著名的天然多糖,由α-L-古鲁糖酸酯和β-D-甘露聚糖残基组成,因为它可以很容易地与二价阳离子形成水凝胶,所以可作为组织工程支架输送细胞和生物活性分子[53]。PARK等[54]以乙二胺为连接剂将透明质酸接枝到海藻酸钠链上,合成了不同含量的透明质酸/海藻酸钠复合水凝胶,该复合水凝胶的机械刚度随着透明质酸含量的增加而增长;然而当透明质酸与海藻酸盐的比例达到2∶1时,大量的透明质酸会阻碍离子交联,导致凝胶硬度下降;联合软骨细胞培养发现,该复合水凝胶成功地调节软骨细胞分化和维持软骨细胞表型,观察到硫酸化糖胺聚糖的大量分泌和软骨形成标志基因的表达。MAHAPATRA等[55]以海藻酸钠和透明质酸为基础、加入Ⅰ型胶原制备了透明质酸/海藻酸钠/Ⅰ型胶原复合水凝胶,进一步加强了分子间的化学相互作用,改善了凝胶的热稳定性和动态力学性能。将小鼠软骨细胞在复合水凝胶中培养表现出更强的增殖能力,软骨细胞表型(包括SOX9、Ⅱ型胶原和蛋白聚糖)的mRNA水平显著上调,糖胺聚糖的分泌更多,但其在体内的功能尚需更深入的研究。
REPPEL等[56]将沃顿胶体间充质干细胞和骨髓间充质干细胞分别植入透明质酸/海藻酸钠复合水凝胶,4周后观察到沃顿胶体间充质干细胞能够适应三维环境,表达特定的软骨相关基因和基质蛋白,比骨髓间充质干细胞表现出更强的Ⅱ型胶原合成能力,并且降低成骨转录因子Runx2和细胞肥大化标志物Ⅹ型胶原的表达。CHEn等[57]制备了透明质酸/海藻酸钠互穿网络复合水凝胶,在植入大鼠骨关节炎模型后发现其能在8周内修复软骨损伤,随后将其结合黄连素,通过激活Wnt/β-catenin通路可显著加强骨髓间充质干细胞的成骨分化,修复软骨下骨损伤。
透明质酸/胶原复合水凝胶:胶原蛋白作为细胞外基质的重要组成部分是一种天然的生物材料,普遍存在于皮肤、骨骼、软骨、血管、牙齿、肌腱等组织中,被广泛应用于生物和医学领域。Ⅰ型胶原是一种黏附分子,能使许多类型的细胞通过整合素识别基质,如α1β1、α2β1、α10β1和α11β1[58],可支持间充质干细胞的黏附、生长、扩散和向软骨分化,为构建工程化软骨结构提供了条件[59]。大量研究发现,Ⅱ型胶原支持间充质干细胞的增殖和软骨形成[60]。
CHEN等[61]将Ⅰ型胶原与透明质酸共混交联制成注射型复合水凝胶,可减轻Ⅰ型胶原的体外收缩,克服透明质酸细胞黏附性小的弱点。胶原与透明质酸为7∶3的比例制备的水凝胶具有最佳的注射效果,注射力为3.5 N时凝胶化时间仅为10 s,频率为10 Hz时凝胶的储能模量可达11 kPa。而该水凝胶包埋软骨细胞时表现出优异的增殖潜能、表型维持和生物相容性;另外,细胞同时产生的谷胱甘肽可使水凝胶逐渐分解,为细胞增殖提供充足的空间。
JIANG等[62]制备了一种胶原/透明质酸/硫酸软骨素复合水凝胶,在胶原/透明质酸水凝胶中加入硫酸软骨素能通过改变胶原纤维的生成速率、纤维直径和网络构成来提高胶原/透明质酸复合水凝胶的压缩模量和黏弹性,提高机械强度,体外实验证实其可作为同种异体软骨细胞的载体生成异位软骨,促进软骨修复。GUO等[63]通过胶原自组装和交联聚合制备了具有互穿网络结构的胶原/透明质酸/硫酸软骨素复合水凝胶,体外培养兔关节软骨细胞结果表明该水凝胶具有更好的细胞相容性,可上调软骨特异性基因表达,促进软骨细胞合成糖胺聚糖和Ⅱ型胶原。
YANG等[64]研究发现与透明质酸/胶原复合水凝胶相比,加入淫羊藿苷可促进间充质干细胞在条件培养基中的向软骨分化,支持软骨细胞增殖,维持软骨细胞表型,促进蛋白多糖和胶原基质的分泌,并且抑制胶原蛋白和蛋白多糖的降解,包裹在复合水凝胶中的细胞趋向于聚集成更大的团簇,成软骨基因表达水平显著上调,糖胺聚糖和Ⅱ型胶原的基质合成明显增强。
透明质酸/明胶复合水凝胶:明胶由一系列精氨酸-甘氨酸-天冬氨酸序列组成,有利于提高细胞黏附力和基质金属蛋白酶的能力,有杰出的生物降解性、生物相容性和细胞亲和力[60]。
LIN等[65]以明胶和透明质酸为原料通过光交联制成甲基丙烯酸明胶/甲基丙烯酸透明质酸复合水凝胶,当明胶与透明质酸比例为9 ∶1时能使骨髓间充质干细胞肥大程度降到最低,糖胺聚糖产量升高;此外,当将其植入到兔骨软骨缺损时,12周后发现透明软骨和骨的生成,这表明其在修复骨软骨缺损方面具有潜在的临床应用价值。JIN等[66]在透明质酸/明胶复合水凝胶中加入了绿茶提取物,由于绿茶提取物是一种多酚化合物可以清除氧自由基,防止炎症引起的氧化损伤,也可抑制白细胞介素1刺激的软骨细胞中的金属蛋白酶、环氧化酶2和核因子kB[67],将该复合水凝胶注射到动物骨关节炎模型3周后没有发现任何磨损和细胞外基质丢失,表明这也是一种在软骨修复中具有广泛前景的方法。
除了与种子细胞联用,水凝胶承载药物注射入关节腔也是较好的软骨修复策略。GARCíA-FERNáNDEZ等[68]设计了一个可注射和可生物降解的药物传递系统,将氧化葡聚糖和明胶通过席夫碱反应形成水凝胶,再与透明质酸混合后形成明胶/氧化葡聚糖/透明质酸半互穿网络复合水凝胶,再将水凝胶装载2种不同的抗炎药:广泛用于抑制骨关节炎症和软骨损伤的非类固醇抗炎药——萘普生,和通过关节内注射用于治疗骨关节炎的抗炎激素——地塞米松,两组分别注入新西兰兔膝关节骨性关节炎模型后,负载地塞米松的水凝胶组恢复更好,软骨的保存和细胞外基质中Ⅱ型胶原的表达更为明显。
透明质酸水凝胶复合其他天然材料:结冷胶是一种天然、无毒、生物相容性和可生物降解的物质,Ca2+、Mg2+可以诱导结冷胶水凝胶的形成。KIM等[69]研究了一种注入CaCl2溶液使其物理共混的结冷胶/透明质酸复合水凝胶,当结冷胶与透明质酸比例为1 ∶ 0.75时其物理力学性能表现出良好的适用性,包裹软骨细胞的存活率和增殖率明显升高,软骨特异性蛋白和基因表达增强;兔子体内实验显示,当软骨缺陷部位植入复合水凝胶后可以有效再生。
菊粉是一种具有生物相容性、水溶性的天然多糖,是由D-果糖经β糖苷键连接而成的线性直链多糖,末端常带有一个葡萄糖残基,并被美国贝克曼库尔特有限公司批准应用在生物医学领域[70]。PALUMBO等[71]将二乙烯基砜功能化菊粉通过迈克尔加成反应交联了两种透明质酸衍生物(分别含有侧链乙二胺和十八烷基侧基),并分别制成复合水凝胶,物理力学测定发现复合水凝胶提高了机械性能,降低了对透明质酸酶水解的敏感性,联合牛软骨细胞培养时细胞活性显著增加。
卡拉胶是结构上与琼脂有关的杂多糖,由红藻类植物中提取而成[72]。当关节软骨细胞被包裹在透明质酸和纤维蛋白组成的水凝胶中时,观察到支架在几天内急剧收缩。因此,为了提高水凝胶的质量,PEREIRA等[73]将卡拉胶引入透明质酸/纤维蛋白凝胶中,发现收缩效应消失,凝胶呈现出良好的弹性和更好的刚度;包裹的人关节软骨细胞也保持着活力和增殖状态,维持着典型软骨形成标志基因的表达,并促进了软骨细胞外基质沉积。由此可采取关节镜下注射的方式将体外扩增的关节软骨细胞输送到关节软骨损伤部位,从而用微创技术修复关节软骨损伤。
2.2.3 透明质酸水凝胶复合合成材料在软骨修复中的应用 合成材料也被广泛研究并应用于软骨组织工程中,包括聚乙二醇、聚N-异丙基丙烯酰胺、聚己内酯、聚乳酸及其衍生物左旋聚乳酸、聚乳酸-羟基乙酸共聚物、右旋聚乳酸、聚氨酯、聚乙二醇二丙烯酸酯和聚乙烯醇。这些聚合物相对容易生产,表现出较强的机械性能,在与透明质酸水凝胶复合后显著提高了复合水凝胶的物理学性能,弥补了机械强度的不足,并且维持了优秀的生物相容性和可降解性[48]。
透明质酸/聚乙二醇复合水凝胶:聚乙二醇以其柔软性、生物相容性、亲水性和溶解能力而著称,在大多数有机溶剂和无机溶剂中也观察到聚乙二醇的高溶解度,并在空
气-水界面形成单层膜,因此其在溶解和渗透中起着重要作用[74]。WANG等[75]通过光交联反应和热诱导点击化学反应制备了具有均匀网络结构和良好力学性能的透明质酸/聚乙二醇复合水凝胶,光交联30 s内迅速凝胶化,成功地将软骨细胞原位包裹,并显示出良好的细胞相容性,其可注射性有利于保持被包裹细胞的活性,便于临床操作。
软骨细胞外基质的相对浓度从浅层到深层逐渐变化,Ⅱ型胶原浓度从浅层到深层逐渐降低,而Ⅹ型胶原和蛋白多糖浓度增加,从而导致力学性能的变化[76]。天然和人工合成生物材料的特殊组合可以将骨髓间充质干细胞诱导成不同表型的软骨细胞,类似于关节软骨浅层、过渡区和深层的细胞。NGUYEN等[77]设计了一种3层结构的复合水凝胶:聚乙二醇水凝胶与硫酸软骨素、基质金属蛋白酶敏感肽结合在顶层,硫酸软骨素和聚乙二醇水凝胶并入中间层,在底部加入透明质酸/聚乙二醇复合水凝胶。将该复合水凝胶与间充质干细胞共培养后,这些不同的生物材料成分可以刺激干细胞定向地分化成不同表型的软骨细胞,从而形成力学性能具有空间变化性的天然关节软骨。
透明质酸/聚乙二醇二丙烯酸酯复合水凝胶:聚乙二醇二丙烯酸酯是一种生物惰性材料,在体内可以存在较长时间,特别适用于组织工程和药物传递。PLACE等[78]用透明质酸水凝胶装载了高剂量的转化生长因子β,再将聚乙二醇二丙烯酸酯与透明质酸水凝胶相交联,体外实验显示该复合水凝胶缓慢释放转化生长因子β且降低了局部爆发释放引起的不良反应,促进软骨细胞增殖和基质合成。LI等[79]从关节腔冲洗液中分离出间充质干细胞,再以聚乙二醇二丙烯酸酯与透明质酸为原料制备的复合水凝胶进行包裹,能显著修复大鼠软骨缺损,观察到透明软骨形成;该复合水凝胶能在生理条件下自发快速地实现凝胶化,便于干细胞原位包封,支持其存活、自我更新和分化。
透明质酸/聚乙烯醇复合水凝胶:聚乙烯醇是聚醋酸乙烯酯的水解形式,具有良好的生物相容性、高弹性模量和易于调节的物理性能,能一定程度上弥补透明质酸机械性能较差的不足[80]。
KIM等[81]用大孔聚乙烯醇海绵作为基础结构,以提供弹性增强和三维互联的孔隙空间,再通过在聚乙烯醇海绵孔内植入透明质酸来模拟软骨组织的生理结构和功能特性,在大鼠模型中观察到软骨特异性糖胺聚糖和Ⅱ型胶原在支架内的堆积。OH等[82]用液氮逐步冻融法交联聚乙烯醇与透明质酸产生刚度梯度为20-200 kPa的复合水凝胶,体外培养时观察到人类骨髓间充质干细胞在不同的刚度范围被诱导分化成特定的细胞类型(0-20 kPa为神经细胞,21-40 kPa为肌细胞,41-80 kPa为软骨细胞,81-190 kPa为成骨细胞),在软骨修复工程上具有巨大的前景。
透明质酸/聚N-异丙基丙烯酰胺复合水凝胶:聚N-异丙基丙烯酰胺是一种标准的热敏聚合物,在水溶液中的最低临界溶液温度在32 ℃左右,常用来制备热敏水凝胶[83]。ATOUFI等[84]合成了聚N-异丙基丙烯酰胺/透明质酸新型热敏水凝胶,内部含有以壳聚糖-丙烯酸包覆的聚乳酸-羟基乙酸共聚物纳米粒子,具有药物释放速度快、单体分离率低、多孔结构连通、机械强度高、生物黏附性好、生物活性高等特点,间充质干细胞被包裹在水凝胶中发现细胞活性正常并且糖胺聚糖合成增多,提示可用于软骨修复。WANG等[85]采取物理和化学交联法分别制备了两种透明质酸/聚N-异丙基丙烯酰胺复合水凝胶,体外实验结果表明两种复合水凝胶均能显著提高细胞存活率、软骨形成标志基因(聚蛋白多糖和Ⅱ型胶原)的表达和硫化物糖胺聚糖的形成,增加了水凝胶的亲水性、膨胀率,降低了收缩率,并且联合兔子脂肪干细胞注射在兔滑膜腔后发现大量透明软骨形成。
透明质酸/聚己内酯复合水凝胶:聚己内酯具有良好的热塑性、生物相容性和可降解性,通过表面侵蚀和整体降解进行缓慢降解,已被批准用作生物可吸收植入物和缝合材
料[86]。MINTZ等[87]以透明质酸水凝胶与聚己内酯制成的复合支架,在拉伸和压缩方面表现出与人类关节软骨相似的力学性能,牛软骨细胞在复合支架中表现出圆形的形态,并且Ⅱ型胶原和聚蛋白多糖的蛋白表达水平正常,因此可作为软骨细胞的适合载体。LEE等[88]将透明质酸水凝胶融入到聚己内酯支架中形成软/硬两相支架,发现该支架增加了软骨细胞的附着数量,为其提供更好的微环境来产生更多的糖胺聚糖、Ⅱ型胶原。
透明质酸水凝胶复合其他合成材料:OWIDA等[89]采用了一种三明治式的制造方法,表面和中间区域由2层透明质酸水凝胶组成,包裹着定向或随机分布的聚乳酸纳米纤维网;深层为透明质酸水凝胶,具有多个垂直通道。通过这种方式可以使软骨细胞在中间区域随机聚集,深层区域柱状聚集,并表达更多的Ⅱ型胶原、糖胺聚糖,减少Ⅰ型胶原产生。
有研究发现,一些新的热敏聚合物通过与透明质酸复合也有着不错的效应,有望在组织工程中推广。聚N-乙烯基己内酰胺是一种热敏聚合物,能够在34.8-36.8 ℃改变其分子构象,形成水凝胶。LYNCH等[90]将聚N-乙烯基己内酰胺与透明质酸制成复合水凝胶,发现可在缺氧条件下提高软骨细胞活性和细胞外基质合成。Pluronic F-127是一种由聚氧化乙烯和聚丙烯氧化物组成的共聚物,遇热凝胶。SEOL等[91]将Pluronic F-127和透明质酸制成复合热敏水凝胶,联合骨髓间充质干细胞注入大鼠软骨损伤部位可形成透明软骨样基质,有利于软骨修复。
2.2.4 透明质酸水凝胶与3D打印技术结合 现有的生物支架由于以下缺点未能获得满意的软骨再生:外部三维形状和内部孔隙结构的控制不精确;降解速率与软骨再生不匹配;机械强度较低,无法实现软骨再生[92]。为了解决这些不足,目前研究者将3D打印技术引进并结合开发了新的生物支架。3D生物打印是一种新兴的再生结构制造技术,它允许细胞和生物材料精确的分层定位,因此可用于制造符合组织结构的三维模型[93]。软骨组织工程的3D生物打印最常用的生物材料是水凝胶,因为它们能保证细胞和生物因子的均匀包裹,并支持相关类型细胞的存活,如间充质干细胞和软骨细胞。
ANTICH等[94]通过将海藻酸盐与透明质酸物理交联制成复合水凝胶,再用聚乳酸打印出用软件设计好的3D框架,最后将含有软骨细胞的复合水凝胶注入聚乳酸结构的孔隙中,4周后发现透明软骨特异性基因(如SOX9、COL2A1、ACAN)的表达加强,而纤维化标记基因COL1A1和肥大标记基因COL10A1的表达降低。
XIA等[95]将明胶和透明质酸混合后引入甲基丙烯酸酐和光引发剂,使其具有光固化作用,并采用3D打印技术制成复合水凝胶,最后利用冷冻干燥技术进一步提高机械性能,延长降解时间。将该复合水凝胶与软骨细胞结合,在体外及山羊模型中成功再生出具有典型软骨陷窝结构和软骨特异性细胞外基质的透明软骨。
MORRISON等[96]根据软骨缺损形状用计算机辅助设计三维模型后,用聚己内酯打印出模型,将接种有脂肪间充质干细胞和软骨细胞的透明质酸/胶原水凝胶注入支架中进行体外培养和大鼠模型实验,发现了Ⅱ型胶原和软骨的生成,为患者特异性的软骨修复提供了可能,同时提高了细胞利用率,避免了外源性生长因子的需要。
2.2.5 透明质酸/多肽仿生复合水凝胶 根据胶原结构特征设计的具有(Gly-Xaa-Yaa)n重复序列的胶原模拟肽,具有抑制骨髓间充质干细胞在软骨分化过程中肥大表型的潜力。胶原模拟肽拥有较天然胶原蛋白更好的溶解度和低免疫原性,可以模拟胶原蛋白的结构、生物活性,并且可以整合某些生物活性位点[97]。REN等[98]使用马来酰亚胺改性透明质酸作为主链,使用基质金属蛋白酶敏感肽作为交联剂,将一种专门设计的胶原模拟肽即(GPO)8-CG-RGDS与透明质酸结合形成复合水凝胶,它不仅能结合RGD序列(具有促细胞黏附作用的序列),还可与具有基质金属蛋白酶降解位点的肽序列结合,诱导骨髓间充质干细胞向软骨分化,并抑制分化过程中的肥大表型,增加COL2A1、ACAN和SOX9等软骨特异性基因表达。
由具有良好生物相容性的天然小分子氨基酸组成的芳香族多肽超分子,可被体内酶特异识别和降解。WANG等[99]将合成多肽BPAA-AFF-OH与透明质酸通过化学自交联和物理自组装,有效构建了共价/非共价杂化网络,体内外研究结果表明,该复合水凝胶能促进软骨细胞增殖和基质的分泌,避免软骨细胞肥大化,促进透明软骨的形成。该仿生水凝胶具有良好的生物相容性和可注射性,操作简单,支架材料安全性高,在软骨组织再生中具有潜在的应用前景。
2.2.6 透明质酸水凝胶联合机械刺激在软骨修复中的应用 除了通过复合其他材料,适当的机械刺激也可以凭借改善孔隙率[100]、增加交联密度等来增强透明质酸水凝胶的物理性能,推进在软骨组织工程中的应用。BIAN等[101]将人骨髓间充质干细胞接种在透明质酸水凝胶中,在细胞培养的环境下给予动态压缩负荷,即在无侧限压缩下通过不透水的加载板进行加载,每天4 h,每周5 d。培养70 d后,动态压缩负荷增加了透明质酸水凝胶的力学性能、糖胺聚糖和胶原含量,显著降低了人骨髓间充质干细胞肥大标志物的表达。LIN
等[102]用透明质酸水凝胶封装间充质干细胞并在生物反应器中用动态压缩负荷(与BIAN等的负荷方案一致)进行处理14 d,再将其植入大鼠软骨缺损模型,发现动态压缩载荷可以显著增加软骨形成的潜能和细胞存活的能力,肥大标志物Ⅹ型胶原和基质金属蛋白酶13的基因表达水平下调,水凝胶的杨氏模量和最大载荷分别提高了207.1%和172.8%。
CHOI等[103]用纤维蛋白/透明质酸水凝胶包裹兔骨髓间充质干细胞,在低强度超声刺激下进行软骨分化4周,结果发现有效促进了骨髓间充质干细胞的软骨向分化,生成大量的硫化糖胺聚糖和胶原,无侧限抗压实验发现该复合水凝胶的机械强度明显升高。
ZHANG等[104]制备了Ⅱ型胶原/透明质酸/聚乙二醇复合水凝胶,并将磁性纳米粒子加入到复合水凝胶中。当大量磁性纳米粒子加入到支架中时,复合水凝胶内部微环境由许多微小磁场组成一个加强磁场,磁场效应可能会影响细胞膜上的离子通道,并在磁场下引发细胞骨架结构的变化。研究显示在分组培养的骨髓间充质干细胞中,磁凝胶组的细胞黏附密度最大,细胞增殖和迁移更加活跃,并且降低了水凝胶的降解速度,提高了整体性。由此可见,不同形式的机械刺激增加了水凝胶在软骨修复中的适用性,有望在临床中进一步开展。

| [1] MAUDENS P, JORDAN O, ALLÉMANN E. Recent advances in intra-articular drug delivery systems for osteoarthritis therapy. Drug Discov Today. 2018;23(10):1761-1775. [2] GOLDRING SR, GOLDRING MB. Changes in the osteochondral unit during osteoarthritis: structure, function and cartilage-bone crosstalk. Nat Rev Rheumatol. 2016;12(11):632-644. [3] 廖建钊,章晓云,张璇.骨性关节炎发生发展中的分子信号通路[J].中国组织工程研究,2020,24(21):3394-3400. [4] CHEN S, FU P, WU H, et al. Meniscus, articular cartilage and nucleus pulposus: a comparative review of cartilage-like tissues in anatomy, development and function. Cell Tissue Res. 2017;370(1):53-70. [5] ZHANG Y, LIU S, GUO W, et al. Coculture of hWJMSCs and pACs in Oriented Scaffold Enhances Hyaline Cartilage Regeneration In Vitro. Stem Cells Int. 2019; 2019:5130152. [6] RAI V, DILISIO MF, DIETZ NE, et al. Recent strategies in cartilage repair: A systemic review of the scaffold development and tissue engineering. J Biomed Mater Res A. 2017;105(8):2343-2354. [7] KAHN TL, SOHEILI A, SCHWARZKOPF R. Outcomes of total knee arthroplasty in relation to preoperative patient-reported and radiographic measures: data from the osteoarthritis initiative. Geriatr Orthop Surg Rehabil. 2013;4(4):117-126. [8] HUANG K, LI Q, LI Y, et al. Cartilage Tissue Regeneration: The Roles of Cells, Stimulating Factors and Scaffolds. Curr Stem Cell Res Ther. 2018;13(7):547-567. [9] REY-RICO A, MADRY H, CUCCHIARINI M. Hydrogel-Based Controlled Delivery Systems for Articular Cartilage Repair. Biomed Res Int. 2016; 2016:1215263. [10] PRÈ ED, CONTI G, SBARBATI A. Hyaluronic Acid (HA) Scaffolds and Multipotent Stromal Cells (MSCs) in Regenerative Medicine. Stem Cell Rev Rep. 2016;12(6): 664-681. [11] VASI AM, POPA MI, BUTNARU M, et al. Chemical functionalization of hyaluronic acid for drug delivery applications. Mater Sci Eng C Mater Biol Appl. 2014;38:177-185. [12] BROCKMEIER SF, SHAFFER BS. Viscosupplementation therapy for osteoarthritis. Sports Med Arthrosc Rev. 2006;14(3):155-162. [13] ZHANG B, THAYAPARAN A, HORNER N, et al. Outcomes of hyaluronic acid injections for glenohumeral osteoarthritis: a systematic review and meta-analysis. J Shoulder Elbow Surg. 2019;28(3):596-606. [14] STERN R, ASARI AA, SUGAHARA KN. Hyaluronan fragments: an information-rich system. Eur J Cell Biol. 2006;85(8):699-715. [15] BOLLYKY PL, BOGDANI M, BOLLYKY JB, et al. The role of hyaluronan and the extracellular matrix in islet inflammation and immune regulation. Curr Diab Rep. 2012;12(5):471-480. [16] HENROTIN Y, RAMAN R, RICHETTE P, et al. Consensus statement on viscosupplementation with hyaluronic acid for the management of osteoarthritis. Semin Arthritis Rheum. 2015;45(2):140-149. [17] CONROZIER T, MATHIEU P, VIGNON E, et al. Differences in the osteoarthritic synovial fluid composition and rheology between patients with or without flare: a pilot study. Clin Exp Rheumatol. 2012; 30(5):729-734. [18] LARSEN NE, DURSEMA HD, POLLAK CT, et al. Clearance kinetics of a hylan-based viscosupplement after intra-articular and intravenous administration in animal models. J Biomed Mater Res B Appl Biomater. 2012;100(2):457-462. [19] KENNE L, GOHIL S, NILSSON EM, et al. Modification and cross-linking parameters in hyaluronic acid hydrogels--definitions and analytical methods. Carbohydr Polym. 2013;91(1):410-418. [20] ZHENG Z, HU J, WANG H, et al. Dynamic Softening or Stiffening a Supramolecular Hydrogel by Ultraviolet or Near-Infrared Light. ACS Appl Mater Interfaces. 2017;9(29):24511-24517. [21] CHEN G, JIANG M. Cyclodextrin-based inclusion complexation bridging supramolecular chemistry and macromolecular self-assembly. Chem Soc Rev. 2011;40(5):2254-2266. [22] APPEL EA, DEL BARRIO J, LOH XJ, et al. Supramolecular polymeric hydrogels. Chem Soc Rev. 2012;41(18):6195-6214. [23] JIANG Y, CHEN J, DENG C, et al. Click hydrogels, microgels and nanogels: emerging platforms for drug delivery and tissue engineering. Biomaterials. 2014;35(18):4969-4985. [24] HAMLET SM, VAQUETTE C, SHAH A, et al. 3-Dimensional functionalized polycaprolactone-hyaluronic acid hydrogel constructs for bone tissue engineering. J Clin Periodontol. 2017;44(4):428-437. [25] MOHAMMADINEJAD R, KUMAR A, RANJBAR-MOHAMMADI M, et al. Recent Advances in Natural Gum-Based Biomaterials for Tissue Engineering and Regenerative Medicine: A Review. Polymers (Basel). 2020;12(1):176. [26] ISHIDA O, TANAKA Y, MORIMOTO I, et al. Chondrocytes are regulated by cellular adhesion through CD44 and hyaluronic acid pathway. J Bone Miner Res. 1997;12(10):1657-1663. [27] OH EJ, PARK K, KIM KS, et al. Target specific and long-acting delivery of protein, peptide, and nucleotide therapeutics using hyaluronic acid derivatives. J Control Release. 2010;141(1):2-12. [28] BORZACCHIELLO A, RUSSO L, MALLE BM, et al. Hyaluronic Acid Based Hydrogels for Regenerative Medicine Applications. Biomed Res Int. 2015;2015:871218. [29] SUN Y, LI X, ZHANG L, et al. Cell Permeable NBD Peptide-Modified Liposomes by Hyaluronic Acid Coating for the Synergistic Targeted Therapy of Metastatic Inflammatory Breast Cancer. Mol Pharm. 2019; 16(3):1140-1155. [30] LEI Y, GOJGINI S, LAM J, et al. The spreading, migration and proliferation of mouse mesenchymal stem cells cultured inside hyaluronic acid hydrogels. Biomaterials. 2011;32(1):39-47. [31] LAM J, TRUONG NF, SEGURA T. Design of cell-matrix interactions in hyaluronic acid hydrogel scaffolds. Acta Biomater. 2014;10(4):1571-1580. [32] GREEN JD, TOLLEMAR V, DOUGHERTY M, et al. Multifaceted signaling regulators of chondrogenesis: Implications in cartilage regeneration and tissue engineering. Genes Dis. 2015;2(4):307-327. [33] DENG Y, SUN AX, OVERHOLT KJ, et al. Enhancing chondrogenesis and mechanical strength retention in physiologically relevant hydrogels with incorporation of hyaluronic acid and direct loading of TGF-β. Acta Biomater. 2019;83:167-176. [34] SCOTTI C, TONNARELLI B, PAPADIMITROPOULOS A, et al. Recapitulation of endochondral bone formation using human adult mesenchymal stem cells as a paradigm for developmental engineering. Proc Natl Acad Sci U S A. 2010;107(16):7251-7256. [35] BIAN L, ZHAI DY, TOUS E, et al. Enhanced MSC chondrogenesis following delivery of TGF-β3 from alginate microspheres within hyaluronic acid hydrogels in vitro and in vivo. Biomaterials. 2011;32(27):6425-6434. [36] KIM M, GARRITY ST, STEINBERG DR, et al. Role of dexamethasone in the long-term functional maturation of MSC-laden hyaluronic acid hydrogels for cartilage tissue engineering. J Orthop Res. 2018;36(6): 1717-1727. [37] LIU X, YANG Y, NIU X, et al. An in situ photocrosslinkable platelet rich plasma - Complexed hydrogel glue with growth factor controlled release ability to promote cartilage defect repair. Acta Biomater. 2017;62:179-187. [38] CHEN WH, LO WC, HSU WC, et al. Synergistic anabolic actions of hyaluronic acid and platelet-rich plasma on cartilage regeneration in osteoarthritis therapy. Biomaterials. 2014;35(36):9599-9607. [39] YAN W, XU X, XU Q, et al. Platelet-rich plasma combined with injectable hyaluronic acid hydrogel for porcine cartilage regeneration: a 6-month follow-up. Regen Biomater. 2020;7(1):77-90. [40] TURAJANE T, CHAVEEWANAKORN U, FONGSARUN W, et al. Avoidance of Total Knee Arthroplasty in Early Osteoarthritis of the Knee with Intra-Articular Implantation of Autologous Activated Peripheral Blood Stem Cells versus Hyaluronic Acid: A Randomized Controlled Trial with Differential Effects of Growth Factor Addition. Stem Cells Int. 2017; 2017:8925132. [41] XU X, JHA AK, DUNCAN RL, et al. Heparin-decorated, hyaluronic acid-based hydrogel particles for the controlled release of bone morphogenetic protein 2. Acta Biomater. 2011;7(8):3050-3059. [42] JHA AK, MALIK MS, FARACH-CARSON MC, et al. Hierarchically structured, hyaluronic acid-based hydrogel matrices via the covalent integration of microgels into macroscopic networks. Soft Matter. 2010; 6(20):5045-5055. [43] AULIN C, JENSEN-WAERN M, EKMAN S, et al. Cartilage repair of experimentally 11 induced osteochondral defects in New Zealand White rabbits. Lab Anim. 2013;47(1):58-65. [44] MASOUDI E, RIBAS J, KAUSHIK G, et al. Platelet-Rich Blood Derivatives for Stem Cell-Based Tissue Engineering and Regeneration. Curr Stem Cell Rep. 2016;2(1):33-42. [45] JOOYBAR E, ABDEKHODAIE MJ, ALVI M, et al. An injectable platelet lysate-hyaluronic acid hydrogel supports cellular activities and induces chondrogenesis of encapsulated mesenchymal stem cells. Acta Biomater. 2019;83:233-244. [46] YOON DM, CURTISS S, REDDI AH, et al. Addition of hyaluronic acid to alginate embedded chondrocytes interferes with insulin-like growth factor-1 signaling in vitro and in vivo. Tissue Eng Part A. 2009; 15(11):3449-3459. [47] KAYAKABE M, TSUTSUMI S, WATANABE H, et al. Transplantation of autologous rabbit BM-derived mesenchymal stromal cells embedded in hyaluronic acid gel sponge into osteochondral defects of the knee. Cytotherapy. 2006;8(4):343-353. [48] JOHNSTONE B, ALINI M, CUCCHIARINI M, et al. Tissue engineering for articular cartilage repair--the state of the art. Eur Cell Mater. 2013; 25:248-267. [49] REMYA NS, NAIR PD. Engineering cartilage tissue interfaces using a natural glycosaminoglycan hydrogel matrix--an in vitro study. Mater Sci Eng C Mater Biol Appl. 2013;33(2):575-582. [50] LIU C, LIU D, WANG Y, et al. Glycol chitosan/oxidized hyaluronic acid hydrogels functionalized with cartilage extracellular matrix particles and incorporating BMSCs for cartilage repair. Artif Cells Nanomed Biotechnol. 2018;46(sup1):721-732. [51] PARK H, CHOI B, HU J, et al. Injectable chitosan hyaluronic acid hydrogels for cartilage tissue engineering. Acta Biomater. 2013;9(1): 4779-4786. [52] RIEGER R, BOULOCHER C, KADERLI S, et al. Chitosan in viscosupplementation: in vivo effect on rabbit subchondral bone. BMC Musculoskelet Disord. 2017;18(1):350. [53] LEE KY, MOONEY DJ. Alginate: properties and biomedical applications. Prog Polym Sci. 2012;37(1):106-126. [54] PARK H, LEE HJ, AN H, et al. Alginate hydrogels modified with low molecular weight hyaluronate for cartilage regeneration. Carbohydr Polym. 2017;162:100-107. [55] MAHAPATRA C, JIN GZ, KIM HW. Alginate-hyaluronic acid-collagen composite hydrogel favorable for the culture of chondrocytes and their phenotype maintenance. Tissue Eng Regen Med. 2016;13(5): 538-546. [56] REPPEL L, SCHIAVI J, CHARIF N, et al. Chondrogenic induction of mesenchymal stromal/stem cells from Wharton’s jelly embedded in alginate hydrogel and without added growth factor: an alternative stem cell source for cartilage tissue engineering. Stem Cell Res Ther. 2015;6:260. [57] CHEN P, XIA C, MO J, et al. Interpenetrating polymer network scaffold of sodium hyaluronate and sodium alginate combined with berberine for osteochondral defect regeneration. Mater Sci Eng C Mater Biol Appl. 2018;91:190-200. [58] YU HS, KIM JJ, KIM HW, et al. Impact of mechanical stretch on the cell behaviors of bone and surrounding tissues. J Tissue Eng. 2016;7:2041731415618342. [59] WANG SJ, JIANG D, ZHANG ZZ, et al. Biomimetic Nanosilica-Collagen Scaffolds for In Situ Bone Regeneration: Toward a Cell-Free, One-Step Surgery. Adv Mater. 2019;31(49):e1904341. [60] BAO W, LI M, YANG Y, et al. Advancements and Frontiers in the High Performance of Natural Hydrogels for Cartilage Tissue Engineering. Front Chem. 2020;8:53. [61] CHEN Y, SUI J, WANG Q, et al. Injectable self-crosslinking HA-SH/Col I blend hydrogels for in vitro construction of engineered cartilage. Carbohydr Polym. 2018;190:57-66. [62] JIANG X, LIU J, LIU Q, et al. Therapy for cartilage defects: functional ectopic cartilage constructed by cartilage-simulating collagen, chondroitin sulfate and hyaluronic acid (CCH) hybrid hydrogel with allogeneic chondrocytes. Biomater Sci. 2018;6(6):1616-1626. [63] GUO Y, YUAN T, XIAO Z, et al. Hydrogels of collagen/chondroitin sulfate/hyaluronan interpenetrating polymer network for cartilage tissue engineering. J Mater Sci Mater Med. 2012;23(9):2267-2279. [64] YANG J, LIU Y, HE L, et al. Icariin conjugated hyaluronic acid/collagen hydrogel for osteochondral interface restoration. Acta Biomater. 2018; 74:156-167. [65] LIN H, BECK AM, SHIMOMURA K, et al. Optimization of photocrosslinked gelatin/hyaluronic acid hybrid scaffold for the repair of cartilage defect. J Tissue Eng Regen Med. 2019;13(8):1418-1429. [66] JIN Y, KOH RH, KIM SH, et al. Injectable anti-inflammatory hyaluronic acid hydrogel for osteoarthritic cartilage repair. Mater Sci Eng C Mater Biol Appl. 2020;115:111096. [67] RASHEED Z, ANBAZHAGAN AN, AKHTAR N, et al. Green tea polyphenol epigallocatechin-3-gallate inhibits advanced glycation end product-induced expression of tumor necrosis factor-alpha and matrix metalloproteinase-13 in human chondrocytes. Arthritis Res Ther. 2009; 11(3):R71. [68] GARCÍA-FERNÁNDEZ L, OLMEDA-LOZANO M, BENITO-GARZÓN L, et al. Injectable hydrogel-based drug delivery system for cartilage regeneration. Mater Sci Eng C Mater Biol Appl. 2020;110:110702. [69] KIM WK, CHOI JH, SHIN ME, et al. Evaluation of cartilage regeneration of chondrocyte encapsulated gellan gum-based hyaluronic acid blended hydrogel. Int J Biol Macromol. 2019;141:51-59. [70] LICCIARDI M, SCIALABBA C, SARDO C, et al. Amphiphilic inulin graft co-polymers as self-assembling micelles for doxorubicin delivery. J Mater Chem B. 2014;2(27):4262-4271. [71] PALUMBO FS, FIORICA C, DI STEFANO M, et al. In situ forming hydrogels of hyaluronic acid and inulin derivatives for cartilage regeneration. Carbohydr Polym. 2015;122:408-416. [72] BARTKOWIAK A, HUNKELER D. Carrageenan-oligochitosan microcapsules: optimization of the formation process(1). Colloids Surf B Biointerfaces. 2001;21(4):285-298. [73] PEREIRA RC, SCARANARI M, CASTAGNOLA P, et al. Novel injectable gel (system) as a vehicle for human articular chondrocytes in cartilage tissue regeneration. J Tissue Eng Regen Med. 2009;3(2):97-106. [74] D’SOUZA AA, SHEGOKAR R. Polyethylene glycol (PEG): a versatile polymer for pharmaceutical applications. Expert Opin Drug Deliv. 2016; 13(9):1257-1275. [75] WANG G, CAO X, DONG H, et al. A Hyaluronic Acid Based Injectable Hydrogel Formed via Photo-Crosslinking Reaction and Thermal-Induced Diels-Alder Reaction for Cartilage Tissue Engineering. Polymers (Basel). 2018;10(9):949. [76] SCHINAGL RM, GURSKIS D, CHEN AC, et al. Depth-dependent confined compression modulus of full-thickness bovine articular cartilage. J Orthop Res. 1997;15(4):499-506. [77] NGUYEN LH, KUDVA AK, SAXENA NS, et al. Engineering articular cartilage with spatially-varying matrix composition and mechanical properties from a single stem cell population using a multi-layered hydrogel. Biomaterials. 2011;32(29):6946-6952. [78] PLACE ES, NAIR R, CHIA HN, et al. Latent TGF-β hydrogels for cartilage tissue engineering. Adv Healthc Mater. 2012;1(4):480-484. [79] LI J, HUANG Y, SONG J, et al. Cartilage regeneration using arthroscopic flushing fluid-derived mesenchymal stem cells encapsulated in a one-step rapid cross-linked hydrogel. Acta Biomater. 2018;79:202-215. [80] BAKER MI, WALSH SP, SCHWARTZ Z, et al. A review of polyvinyl alcohol and its uses in cartilage and orthopedic applications. J Biomed Mater Res B Appl Biomater. 2012;100(5):1451-1457. [81] KIM HD, LEE Y, KIM Y, et al. Biomimetically Reinforced Polyvinyl Alcohol-Based Hybrid Scaffolds for Cartilage Tissue Engineering. Polymers (Basel). 2017;9(12):655. [82] OH SH, AN DB, KIM TH, et al. Wide-range stiffness gradient PVA/HA hydrogel to investigate stem cell differentiation behavior. Acta Biomater. 2016;35:23-31. [83] ASHRAFUL ALAM M, TAKAFUJI M, IHARA H. Thermosensitive hybrid hydrogels with silica nanoparticle-cross-linked polymer networks. J Colloid Interface Sci. 2013;405:109-117. [84] ATOUFI Z, KAMRAVA SK, DAVACHI SM, et al. Injectable PNIPAM/Hyaluronic acid hydrogels containing multipurpose modified particles for cartilage tissue engineering: Synthesis, characterization, drug release and cell culture study. Int J Biol Macromol. 2019;139:1168-1181. [85] WANG CZ, ESWARAMOORTHY R, LIN TH, et al. Enhancement of chondrogenesis of adipose-derived stem cells in HA-PNIPAAm-CL hydrogel for cartilage regeneration in rabbits. Sci Rep. 2018;8(1):10526. [86] OLIVEIRA JT, CRAWFORD A, MUNDY JM, et al. A cartilage tissue engineering approach combining starch-polycaprolactone fibre mesh scaffolds with bovine articular chondrocytes. J Mater Sci Mater Med. 2007;18(2):295-302. [87] MINTZ BR, COOPER JA JR. Hybrid hyaluronic acid hydrogel/poly(ɛ-caprolactone) scaffold provides mechanically favorable platform for cartilage tissue engineering studies. J Biomed Mater Res A. 2014; 102(9):2918-2926. [88] LEE MY, TSAI WW, CHEN HJ, et al. Laser sintered porous polycaprolacone scaffolds loaded with hyaluronic acid and gelatin-grafted thermoresponsive hydrogel for cartilage tissue engineering. Biomed Mater Eng. 2013;23(6):533-543. [89] OWIDA HA, YANG R, CEN L, et al. Induction of zonal-specific cellular morphology and matrix synthesis for biomimetic cartilage regeneration using hybrid scaffolds. J R Soc Interface. 2018;15(143):20180310. [90] LYNCH B, CRAWFORD K, BARUTI O, et al. The effect of hypoxia on thermosensitive poly(N-vinylcaprolactam) hydrogels with tunable mechanical integrity for cartilage tissue engineering. J Biomed Mater Res B Appl Biomater. 2017;105(7):1863-1873. [91] SEOL D, MAGNETTA MJ, RAMAKRISHNAN PS, et al. Biocompatibility and preclinical feasibility tests of a temperature-sensitive hydrogel for the purpose of surgical wound pain control and cartilage repair. J Biomed Mater Res B Appl Biomater. 2013;101(8):1508-1515. [92] LI J, CHEN M, FAN X, et al. Recent advances in bioprinting techniques: approaches, applications and future prospects. J Transl Med. 2016;14: 271. [93] Murphy SV, Atala A. 3D bioprinting of tissues and organs. Nat Biotechnol. 2014;32(8):773-785. [94] ANTICH C, DE VICENTE J, JIMÉNEZ G, et al. Bio-inspired hydrogel composed of hyaluronic acid and alginate as a potential bioink for 3D bioprinting of articular cartilage engineering constructs. Acta Biomater. 2020;106:114-123. [95] XIA H, ZHAO D, ZHU H, et al. Lyophilized Scaffolds Fabricated from 3D-Printed Photocurable Natural Hydrogel for Cartilage Regeneration. ACS Appl Mater Interfaces. 2018;10(37):31704-31715. [96] MORRISON RJ, NASSER HB, KASHLAN KN, et al. Co-culture of adipose-derived stem cells and chondrocytes on three-dimensionally printed bioscaffolds for craniofacial cartilage engineering. Laryngoscope. 2018; 128(7):E251-E257. [97] YU SM, LI Y, KIM D. Collagen Mimetic Peptides: Progress Towards Functional Applications. Soft Matter. 2011;7(18):7927-7938. [98] REN Y, ZHANG H, QIN W, et al. A collagen mimetic peptide-modified hyaluronic acid hydrogel system with enzymatically mediated degradation for mesenchymal stem cell differentiation. Mater Sci Eng C Mater Biol Appl. 2020;108:110276. [99] WANG Q, LI X, WANG P, et al. Bionic composite hydrogel with a hybrid covalent/noncovalent network promoting phenotypic maintenance of hyaline cartilage. J Mater Chem B. 2020;8(20):4402-4411. [100] 卢璐,吉鸿飞,郭各朴,等.超声增强藻酸钙凝胶支架材料孔隙率的研究[J].物理学报,2015,64(2):264-271. [101] BIAN L, ZHAI DY, ZHANG EC, et al. Dynamic compressive loading enhances cartilage matrix synthesis and distribution and suppresses hypertrophy in hMSC-laden hyaluronic acid hydrogels. Tissue Eng Part A. 2012;18(7-8):715-724. [102] LIN S, LEE WYW, FENG Q, et al. Synergistic effects on mesenchymal stem cell-based cartilage regeneration by chondrogenic preconditioning and mechanical stimulation. Stem Cell Res Ther. 2017;8(1):221. [103] CHOI JW, CHOI BH, PARK SH, et al. Mechanical stimulation by ultrasound enhances chondrogenic differentiation of mesenchymal stem cells in a fibrin-hyaluronic acid hydrogel. Artif Organs. 2013;37(7):648-655. [104] ZHANG N, LOCK J, SALLEE A, et al. Magnetic Nanocomposite Hydrogel for Potential Cartilage Tissue Engineering: Synthesis, Characterization, and Cytocompatibility with Bone Marrow Derived Mesenchymal Stem Cells. ACS Appl Mater Interfaces. 2015;7(37):20987-20998. |
| [1] | 谭新访, 郭艳幸, 秦晓飞, 张斌清, 赵东亮, 潘琨琨, 李瑜卓, 陈皓宇. 顺轴疲劳运动对兔髌股关节软骨损伤的影响[J]. 中国组织工程研究, 2022, 26(在线): 1-6. |
| [2] | 张吉超, 董跃福, 牟志芳, 张 震, 李冰言, 徐祥钧, 李佳意, 任 梦, 董万鹏. 骨关节炎患者在不同步态角度下膝关节内部生物力学变化的有限元分析[J]. 中国组织工程研究, 2022, 26(9): 1357-1361. |
| [3] | 姚晓玲, 彭建城, 许岳荣, 杨志东, 张顺聪. 可变角度零切迹前路椎间融合内固定系统治疗脊髓型颈椎病:30个月随访[J]. 中国组织工程研究, 2022, 26(9): 1377-1382. |
| [4] | 金 涛, 刘 林, 朱晓燕, 史宇悰, 牛建雄, 张同同, 吴树金, 杨青山. 骨关节炎与线粒体异常[J]. 中国组织工程研究, 2022, 26(9): 1452-1458. |
| [5] | 吴 聪, 贾全忠, 刘 伦. 成年关节软骨碎块化后转化生长因子β1表达与软骨细胞迁移的关系[J]. 中国组织工程研究, 2022, 26(8): 1167-1172. |
| [6] | 李志一, 贺鹏程, 边天月, 肖玉霞, 高 璐, 刘华胜. 铁死亡机制研究的文献计量学与可视化分析[J]. 中国组织工程研究, 2022, 26(8): 1202-1209. |
| [7] | 安维政, 何 萧, 任 帅, 刘建宇. 肌源干细胞在周围神经再生中的潜力[J]. 中国组织工程研究, 2022, 26(7): 1130-1136. |
| [8] | 张璟琳, 冷 敏, 朱博恒, 汪 虹. 干细胞源外泌体促进糖尿病创面愈合的机制及应用[J]. 中国组织工程研究, 2022, 26(7): 1113-1118. |
| [9] | 刘冬铖, 赵继军, 周子红, 吴沼锋, 俞颖豪, 陈宇浩, 冯德宏. 开放楔形胫骨高位截骨手术不同力线矫正参考方法的比较[J]. 中国组织工程研究, 2022, 26(6): 827-831. |
| [10] | 何俊君, 黄泽灵, 洪振强. 阳和汤对早期膝骨关节炎模型兔滑膜炎症的影响[J]. 中国组织工程研究, 2022, 26(5): 694-699. |
| [11] | 林旭晨, 祝海年, 王增顺, 祁腾民, 刘立民, 索南昂秀. 黄腐酚对骨关节炎模型小鼠炎性因子及关节软骨的作用[J]. 中国组织工程研究, 2022, 26(5): 676-681. |
| [12] | 许 磊, 韩晓强, 张锦涛, 孙海飚. 关节软骨细胞周围透明质酸产生、转化和功能特征[J]. 中国组织工程研究, 2022, 26(5): 768-773. |
| [13] | 沈 松, 徐 彬. 经皮椎体成形骨水泥呈弥散型分布可减少邻近椎体再骨折的发生率[J]. 中国组织工程研究, 2022, 26(4): 499-503. |
| [14] | 侯万星, 李洪伟, 郑 欣, 朱先任. 经皮椎体强化后骨水泥椎间盘渗漏与治疗前MRI发现的相关性[J]. 中国组织工程研究, 2022, 26(4): 504-509. |
| [15] | 黄传俊, 邹 煜, 周晓婷, 朱扬清, 钱 伟, 张 卫, 刘 星. 携带脐带间充质干细胞的RADA16-BDNF水凝胶支架定向移植促进脑出血大鼠神经功能恢复[J]. 中国组织工程研究, 2022, 26(4): 510-515. |
常用的关节软骨修复策略包括非类固醇抗炎药物治疗、微骨折、关节内注射玻璃酸钠、自体软骨细胞植入,但不能获得完整的正常透明软骨[6]。全膝关节置换是治疗终末期骨关节炎的有效方法,可以消除疼痛,帮助患者恢复正常活动,但具有感染、僵硬和假体寿命有限的缺点[7]。目前,由支架、种子细胞和有利的生长因子组成的组织工程已发展成为最有前途的软骨组织修复策略[8]。移植种子细胞后,由于细胞外基质支持不足和炎症造成大量细胞死亡,得不到预期的效果,所以最佳的生物支架应具有生物相容性和生物可吸收性,同时支持细胞生长和分化,提供适应的机械环境并允许细胞营养物质的运输。水凝胶作为交联亲水性聚合物是生物医学领域的一类重要生物材料,具有优异的生物相容性,可导致最小的炎症反应和组织损伤,以其形成3D网络的能力而备受关注,该网络可以被微调以修改其生物相容性和生物降解
性[9]。透明质酸遍布于结缔组织、上皮组织和神经组织,是细胞外基质的重要组成部分,有助于细胞增殖和迁移[10]。因此,透明质酸水凝胶因其生物相容性、生物降解性和低免疫原性而被普遍应用于组织工程领域,并且基于透明质酸的复合水凝胶在骨性关节炎的软骨修复中得到了广泛研究。通过查阅国内外文献,现就各类基于透明质酸的复合水凝胶在骨关节炎软骨损伤修复中的应用作一综述。 中国组织工程研究杂志出版内容重点:生物材料;骨生物材料; 口腔生物材料; 纳米材料; 缓释材料; 材料相容性;组织工程
1.2 选取与排除标准
纳入标准:阅读文献题目、摘要进行初步筛选,仔细阅读文献内容后选取与透明质酸复合水凝胶应用于软骨修复方面紧密相关的各类文献。
排除标准:文献与主题不相关、相关性低,以及重复的、陈旧的、证据等级过低的文献。
1.3 文献提取 共检索到文献417篇,中文文献224篇,英文文献193篇,通过筛选后排除与主题不相关、相关性低,以及重复的、陈旧的、证据等级过低的文献共313篇,纳入104篇文献进行综述,见图1。

文题释义:
基于透明质酸的复合水凝胶:透明质酸是一种天然多糖,也是人体中常见的一种糖胺聚糖,因其生物相容性、可降解性等生物性能被制作为水凝胶广泛应用于组织工程中,尤其通过不同方法与生物因子、天然材料、合成材料、3D打印技术、多肽、机械刺激联合制成复合水凝胶,在软骨修复方面有着巨大的前景。
骨关节炎的软骨损伤:骨关节炎是一种退行性病变,软骨组织的慢性炎症通过免疫反应等机制破坏软骨的蛋白多糖及Ⅱ型胶原合成,软骨组织的自我修复因其缺乏血管而十分困难,最终导致软骨损伤。
透明质酸水凝胶凭借着其优异的生物相容性、生物降解性和低免疫原性而被选择作为骨关节炎软骨损伤修复中的生物支架,但仍具有机械性能差、细胞黏附性低等不足。该文通过检索国内外数据库,归纳总结国内外学者通过将透明质酸水凝胶联合生物因子、天然材料、合成材料、3D打印技术、多肽、机械刺激等制成复合水凝胶,改善了透明质酸水凝胶的性能,进一步推进了在软骨组织工程中的应用。
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||