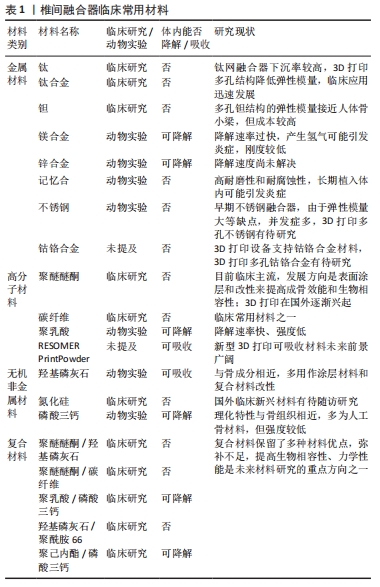
[1] KOH K, YAMADA K, ENOMOTO T, et al. Sex-Specific Impact of Pain Severity, Insomnia, and Psychosocial Factors on Disability due to Spinal Degenerative Disease. Pain Res Manag. 2020;2020:1-6.
[2] 汤泽瀚.新型3D打印颈椎椎间融合器的研制及动物实验初步研究[D].上海:中国人民解放军海军军医大学,2019.
[3] RAO PJ, PELLETIER MH, WALSH WR, et al. Spine interbody implants: material selection and modification, functionalization and bioactivation of surfaces to improve osseointegration. Orthop Surg. 2014;6:81-89.
[4] 黎庆初,闫慧博.前路椎体间融合术治疗腰椎退变性疾病[J].中国矫形外科杂志,2020,28(1):47-52.
[5] OLIVARES-NAVARRETE R, HYZY SL, SLOSAR PJ, et al. Implant materials generate different peri-implant inflammatory factors: poly-ether-ether- ketone promotes fibrosis and microtextured titanium promotes osteogenic factors. Spine (Phila Pa 1976). 2015;40:399-404.
[6] GUYER RD, ABITBOL JJ,O HNMEISS DD, et al.Evaluating osseointegration into a deeply porous titanium scaffold: a biomechanical comparison with PEEK and allograft. Spine (Phila Pa 1976). 2016;41:E1146-1150.
[7] 赵勃然,郑修军,马金荣.椎间融合器及其材料的研究与进展[J].中国组织工程研究,2017,21(2):315-321.
[8] 姜威,毛克亚.颈椎椎间融合器设计的历史沿革与研究进展[J].解放军医学院学报,2018,39(6):538-541.
[9] VAN BAEL S, CHAI YC, TRUSCELLO S, et al. The effect of pore geometry on the in vitro biological behavior of human periosteum-derived cells seeded on selective laser-melted Ti6Al4V bone scaffolds. Acta Biomater. 2012;8(7):2824-2834.
[10] PONADER S, VAIRAKTARIS E, HEINL P, et al. Effects of topographical surface modifications of electron beam melted Ti-6Al-4V titanium on human fetal osteoblasts. J Biomed Mater Res A. 2010;84A(4):1111-1119.
[11] 杨旭,赵晓峰,齐德泰,等.3D打印ACT钛金骨小梁椎间融合器行颈椎前路减压融合后颈椎的矢状位平衡变化[J].中国组织工程研究,2020,24(36):5741-5748.
[12] 刘正蓬,王雅辉,张义龙,等.3D打印椎间融合器置入治疗脊髓型颈椎病:颈椎曲度及椎间高度恢复的半年随访[J].中国组织工程研究,2021,25(6):849-853.
[13] 王林楠,杨曦,宋跃明.3D打印技术在椎间融合临床应用的研究进展[J].华西医学,2018,33(9):1061-1067.
[14] 贺瑞,张文志,尚希福,等.钽金属椎间融合器在腰椎间盘突出症减压术后复发融合术中的应用[J]. 颈腰痛杂志,2013(5):387-390.
[15] IV-JOHN, BEUTLER W, PEPPELMAN W, et al. Clinical Outcomes with Porous Tantalum in Lumbar Interbody Fusion. Spine J. 2010;10(9-supp-S):S147-S148.
[16] FERNÁNDEZ-FAIREN M, ALVARADO E, TORRES A. Eleven-year follow-up of two cohorts of patients comparing stand-alone porous tantalum cage versus autologous bone graft and plating in anterior cervical fusions. World Neurosurg. 2019;122: e156-e167.
[17] 李洋.激光增材制造(3D打印)制备生物医用多孔金属工艺及组织性能研究[D].苏州:苏州大学,2015.
[18] 赵维彪.镍钛形状记忆合金的材料学特征与医学应用[J].中国组织工程研究,2007,11(22):4376-4379.
[19] ASSAD M, CHERNYSHOV AV, JARZEM P, et al. Porous Titanium-Nickel for Intervertebral Fusion in a Sheep Model: Part 2. Surface Analysis and Nickel Release Assessment. J Biomed Mater Res B Appl Biomater. 2003;64(2):121-129.
[20] HONG X, WU XT, ZHUANG SY, et al. New Cage for Posterior Minimally Invasive Lumbar Interbody Fusion: a Study in Vitro and in Vivo. Orthop Surg. 2014;6(1):47-53.
[21] ELKAIAM L, HAKIMI O, YOSAFOVICH-DOITCH G, et al. In Vivo Evaluation of Mg–5%Zn–2%Nd Alloy as an Innovative Biodegradable Implant Material. Ann Biomed Eng. 2019;48(2367):1-13.
[22] 许灏铖,张帆,吕飞舟,等.金属镁及其合金植入材料在脊柱外科中的应用[J].国际骨科学杂志,2016,37(5):269-273.
[23] DAENTZER D, WILLBOLD E, KALLA K, et al. Bioabsorbable interbody magnesium-polymer cage: degradation kinetics, biomechanical stiffness, and histological findings from an ovine cervical spine fusion model. Spine (Phila Pa 1976). 2014;39(20):E1220-E1227.
[24] 刘辰.骨植入用新型可降解镁合金材料设计及生物医学功能研究[D].南京:南京理工大学,2016.
[25] KAFRI A, OVADIA S, YOSAFOVICH-DOITCH G, et al. The Effects of 4%Fe on the Performance of Pure Zinc as Biodegradable Implant Material. Ann Biomed Eng. 2019;47(6):1400-1408.
[26] SEAMAN S, KEREZOUDIS P, BYDON M, et al. Titanium vs. polyetheretherketone (PEEK) interbody fusion: meta-analysis and review of the literature. J Clin Neurosci. 2017;44:23-29.
[27] 韩淑芬,陈伟伟,于洁.3D打印高分子材料研究进展[J].工程塑料应用,2017,45(10):146-150.
[28] PHILIPP H, NEHA S, BRANDO O, et al. Patient-Specific Surgical Implants Made of 3D Printed PEEK: Material, Technology, and Scope of Surgical Application. Biomed Res Int. 2018;2018:4520636.
[29] AHN H, PATEL RR, HOYT AJ, et al. Biological evaluation and finite-element modeling of porous poly( para -phenylene) for orthopaedic implants. Acta Biomater. 2018:S1742706118301454.
[30] 钱伯章.赢创推出首个用于医用植入物的3D打印聚醚醚酮长丝[J].合成纤维,2020,49(6):59-59.
[31] WILLEMS K, LAUWERYNS P, VERLEYE G, et al. Randomized controlled trial of posterior lumbar interbody fusion with Ti-and CaP-nanocoated polyetheretherketone cages: comparative study of the 1-year radiological and clinical outcome. Int J Spine Surg. 2020;13(6):575-587.
[32] HASEGAWA T, USHIROZAKO H, SHIGETO E, et al. The Titanium-coated PEEK Cage Maintains Better Bone Fusion With the Endplate Than the PEEK Cage 6 Months After PLIF Surgery: A Multicenter, Prospective, Randomized Study. Spine (Phila Pa 1976). 2020;45(15):E892-E902.
[33] BRANTIGAN JW, STEFFEE AD. A carbon fiber implant to aid interbody lumbar fusion. Two-year clinical results in the first 26 patients. Spine (Phila Pa 1976). 1993;18(14):2106-2107.
[34] WANG YT, WU XT, CHEN H, et al. Endoscopy-assisted posterior lumbar interbody fusion in a single segment. J Clin Neurosci. 2013;21(2):287-292.
[35] 宋成哲,金红旭.碳纤维椎间融合器与钛网椎间融合器修复腰椎退行性变:生物相容性比较[J].中国组织工程研究,2015,19(12): 1909-1913.
[36] 王树根,孙学猛,刘瑞存,等.两种椎间融合器联合椎弓根螺钉治疗腰椎滑脱症的疗效比较[J].中国医药指南,2017,15(4):8-9.
[37] VAN DIJK M, SMIT TH, BURGER EH, et al. Bioabsorbable poly-L-lactic acid cages for lumbar interbody fusion: three-year follow-up radiographic, histologic, and histomorphometric analysis in goats. Spine (Phila Pa 1976). 2002;27(23): 2706-2714.
[38] VAN DIJK M, SMIT TH, SUGIHARA S, et al. The effect of cage stiffness on the rate of lumbar interbody fusion: an in vivo model using poly (L- lactic acid) and Titanium cages. Spine (Phila Pa 1976). 2002;27(2): 682-688.
[39] JIYA T, SMIT T, DEDDENS J, et al. Posterior lumbar interbody fusion using nonresorbable poly-ether-ether-ketone versus resorbable poly-L-lactide-co-D,L-lactide fusion devices: a prospective, randomized study to assess fusion and clinical outcome. Spine (Phila Pa 1976). 2009;34(3):233-237.
[40] REGNELL ANDERSSON S, HAKKARAINEN M, INKINEN S, et al. Customizing the Hydrolytic Degradation Rate of Stereocomplex PLA through Different PDLA Architectures. Biomacromolecules. 2012; 13(4):1212-1222.
[41] 王乐,刘少喻.可吸收聚合物材料制作椎间融合器的临床应用[J].中国组织工程研究,2013,17(42):7462-7468.
[42] 王哲.赢创开发出全球首款面向植入式医疗器械3D打印的自由流动生物可吸收粉末[J].现代化工,2020,40(2):12.
[43] 林巧霞,殷萌,魏延,等.钛及钛合金表面羟基磷灰石涂层结合强度及稳定性[J].化学进展,2020,32(4):54-64.
[44] YAN L, LENG Y, WENG LT. Characterization of chemical inhomogeneity in plasma-sprayed hydroxyapatite coatings. Biomaterials. 2003;24(15): 2585-2592.
[45] CIZEK J, KHOR KA, PROCHAZKA Z. Influence of spraying conditions on thermal and velocity properties of plasma sprayed hydroxyapatite. Mater Sci Eng C. 2007;27(2):340-344.
[46] SADOWSKA JM, WEI F, GUO J, et al. Effect of nano-structural properties of biomimetic hydroxyapatite on osteoimmunomodulation. Biomaterials. 2018;181:318-332.
[47] SUH P B, PUTTLITZ C, LEWIS C, et al. The effect of cervical interbody cage morphology, material composition, and substrate density on cage subsidence. J Am Acad Orthop Surg. 2017;25(2):160-168.
[48] KERSTEN RFMR, WU G, POURAN B, et al. Comparison of polyetheretherketone versus silicon nitride intervertebral spinal spacers in a caprine model. J Biomed Mater Res B Appl Biomater. 2019; 107(3):688-699.
[49] MCENTIRE BJ, MASLIN G, BAL BS. Two-year results of a double-blind multicenter randomized controlled non-inferiority trial of polyetheretherketone (PEEK) versus silicon nitride spinal fusion cages in patients with symptomatic degenerative lumbar disc disorders. J Spine Surg. 2020;6(3): 523.
[50] 马瑞.纳米生物陶瓷/聚醚醚酮复合物骨生物活性的研究[D].上海:上海交通大学,2015.
[51] TSOU HK, HSIEH PY, CHI MH, et al. Improved osteoblast compatibility of medical-grade polyetheretherketone using arc ionplated rutile/anatase titanium dioxide films for spinal implants. J Biomed Mater Res A. 2012;100A(10):2787-2792.
[52] TAN KH, CHUA CK, LEONG KF, et al. Fabrication and characterization of three-dimensional poly(ether- ether- ketone)/-hydroxyapatite biocomposite scaffolds using laser sintering. Proc Inst Mech Eng H. 2005;219(3):183-194.
[53] WANG Z, GAO D. Friction and wear properties of stainless steel sliding against polyetheretherketone and carbon-fiber-reinforced polyetheretherketone under natural seawater lubrication. Mater Des. 2014;53:881-887.
[54] 刘学勇,荆延峰,朱洪欣,等.聚醚醚酮/羟基磷灰石/碳纤维复合材料的生物力学分析[J].生物医学工程与临床,2013(3):202-207.
[55] LI J, ZUO Y, CHENG X, et al. Preparation and characterization of nano-hydroxyapatite/polyamide 66 composite GBR membrane with asymmetric porous structure. J Mater Sci Mater Med. 2009;20(5):1031-1038.
[56] HU B, YANG X, HU Y, et al. The n-HA/PA66 cage versus the PEEK cage in anterior cervical fusion with single-level discectomy during 7 years of follow-up. World Neurosurg. 2019;123:e678-e684.
[57] YANG X, CHEN Q, LIU L, et al. Comparison of anterior cervical fusion by titanium mesh cage versus nano-hydroxyapatite/polyamide cage following single-level corpectomy. Int Orthop. 2013;37(12):2421-2427.
[58] NIE W, GAO Y, MCCOUL DJ, et al. Rapid mineralization of hierarchical poly (l-lactic acid)/poly (ε-caprolactone) nanofibrous scaffolds by electrodeposition for bone regeneration. Int J Nanomedicine. 2019;14: 3929.
[59] 王丽,王少俊,范仲勇,等.立构共聚聚乳酸/纳米β-磷酸三钙复合材料制备可吸收双层椎间融合器及其降解性能[J].复旦学报(自然科学版),2012,51(3):325-334.
[60] CAO L, CHEN Q, JIANG LB, et al. Bioabsorbable self-retaining PLA/nano-sized β-TCP cervical spine interbody fusion cage in goat models: An in vivo study. Int J Nanomedicine. 2017;12:7197-7205.
[61] 空军军医大学西京医院新型聚己内酯—磷酸三钙可降解颈椎椎间融合器首次应用临床[J].医学争鸣,2020,11(2):6.
[62] MILLER LE, BLOCK JE. Safety and effectiveness of bone allografts in anterior cervical discectomy and fusion surgery. Spine (Phila Pa 1976). 2011;36(24):2045-2050.
[63] YUE WM, BRODNER W, HIGHLAND TR. Long-term results after anterior cervical discectomy and fusion with allograft and plating: a 5-to 11-year radiologic and clinical follow-up study. Spine (Phila Pa 1976). 2005;30(19):2138-2144.
[64] KUSHIOKA J, KAITO T, MAKINO T, et al. Difference in the fusion rate and bone formation between artificial bone and iliac autograft inside an inter-body fusion cage - A comparison between porous hydroxyapatite/type 1 collagen composite and autologous iliac bone. J Orthop Sci. 2018:S0949265818300927.
[65] KIM KT, KIM KG, CHOI UY, et al. Safety and Tolerability of Stromal Vascular Fraction Combined with β-Tricalcium Phosphate in Posterior Lumbar Interbody Fusion: Phase I Clinical Trial. Cells. 2020;9(10):2250.
[66] 王建华.椎间融合器内填充材料及技术[J].美中国际创伤杂志, 2005,4(4):32-33.
[67] JUN SH, LEE EJ, JANG TS, et al. Bone morphogenic protein-2 (BMP-2) loaded hybrid coating on porous hydroxyapatite scaffolds for bone tissue engineering. J Mater Sci Mater Med. 2013;24(3):773-782.
[68] XIE XW, JIANG H, LI N, et al. Recombinant human bone morphogenetic protein-2 induces bone repair. Chin J Tissue Eng Res. 2013;5 Suppl 7:S27-29.
[69] SINGH K, NANDYALA SV, MARQUEZ-LARA A, et al. Epidemiological trends in the utilization of bone morphogenetic protein in spinal fusions from 2002 to 2011. Spine (Phila Pa 1976). 2014;39(6):491-496.
[70] ANNIS P, BRODKE DS, SPIKER WR, et al. The Fate of L5-S1 with Low dose BMP-2 and Pelvic Fixation, with or without Interbody Fusion, in Adult Deformity Surgery. Spine (Phila Pa 1976). 2015;40(11):634-639.
[71] WEISBROD LJ, ARNOLD PM, LEEVER JD. Radiographic and CT Evaluation of Recombinant Human Bone Morphogenetic Protein-2–assisted Cervical Spinal Interbody Fusion. Clin Spine Surg. 2019;32(2):71-79.
[72] 禹名卉,赵彦涛,衷鸿宾,等.骨形态发生蛋白2在腰椎融合术中应用的效果与并发症[J].中国骨与关节杂志,2017,6(3):228-231.
[73] TANNOURY CA, AN HS. Complications with the use of bone morphogenetic protein 2 (BMP-2) in spine surgery. Spine J. 2014;14(3):552-559. |