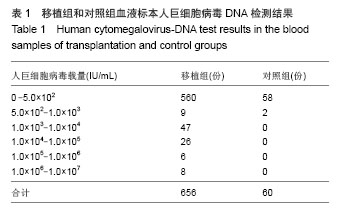
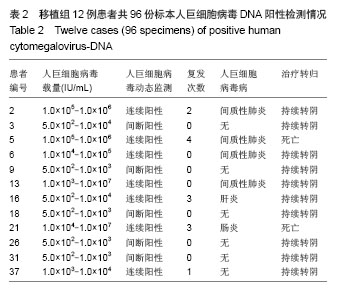
| [1]Griffiths PD, Emery VC, Milne RC. Cytomegalovirus[A]// Douglas D,Richmand, Whitley RJ, ed. Clinical virology[M]. 3rd ed. ASM Press,2009: 475-506.
[2]周文强,石炳毅,蔡明,等.器官移植受者术后巨细胞病毒感染的监测[J].中国组织工程研究与临床康复,2008,12(18):3479-3481.
[3]Ichihara H, Nakamae H, Hirose A,et al. Immunoglobulin prophylaxis against cytomegalovirus infection in patients at high risk of infection following allogeneic hematopoietic cell transplantation.Transplant Proc. 2011;43(10):3927-3932.
[4]Cantoni N, Hirsch HH, Khanna N,et al.Evidence for a bidirectional relationship between cytomegalovirus replication and acute graft-versus-host disease.Biol Blood Marrow Transplant. 2010;16(9):1309-1314.
[5]Borchers S, Luther S, Lips U,et al.Tetramer monitoring to assess risk factors for recurrent cytomegalovirus reactivation and reconstitution of antiviral immunity post allogeneic hematopoietic stem cell transplantation.Transpl Infect Dis. 2011;13(3):222-236.
[6]赵晓甦,刘代红,许兰平,等.异基因造血干细胞移植后巨细胞病毒肺炎临床特点分析[J].北京大学学报(医学版),2009,41(5): 548-553.
[7]Torres HA, Aguilera E, Safdar A,et al. Fatal cytomegalovirus pneumonia in patients with haematological malignancies: an autopsy-based case-control study.Clin Microbiol Infect. 2008; 14(12):1160-1166.
[8]Leruez-Ville M, Ouachée M, Delarue R, et al.Monitoring cytomegalovirus infection in adult and pediatric bone marrow transplant recipients by a real-time PCR assay performed with blood plasma.J Clin Microbiol. 2003;41(5):2040-2046.
[9]张之南,沈悌.血液病诊断及疗效标准[M].3版.北京:科学出版社, 2007.
[10]Muñoz-Cobo B, Solano C, Costa E,et al. Dynamics of cytomegalovirus (CMV) plasma DNAemia in initial and recurrent episodes of active CMV infection in the allogeneic stem cell transplantation setting: implications for designing preemptive antiviral therapy strategies.Biol Blood Marrow Transplant. 2011;17(11):1602-1611.
[11]Kekik C, Besisik SK, Seyhun Y,et al. Relationship between HLA tissue type, CMV infection, and acute graft-vs-host disease after allogeneic hematopoietic stem cell transplantation: single-center experience.Transplant Proc. 2009;41(9):3859-3862.
[12]Jang JE, Hyun SY, Kim YD,et al. Risk factors for progression from cytomegalovirus viremia to cytomegalovirus disease after allogeneic hematopoietic stem cell transplantation. Biol Blood Marrow Transplant. 2012;18(6):881-886.
[13]van der Bij W, Speich R. Management of cytomegalovirus infection and disease after solid-organ transplantation.Clin Infect Dis. 2001;33 Suppl 1:S32-37.
[14]陈育红,赵晓生,刘开彦,等.造血干细胞移植后患者血浆巨细胞病毒DNA拷贝数与巨细胞病毒病的关系[J].中华医学杂志,2009, 89(22): 1540-1543.
[15]Eid AJ, Razonable RR.New developments in the management of cytomegalovirus infection after solid organ transplantation.Drugs. 2010;70(8):965-981.
[16]Ariza-Heredia EJ, Nesher L, Chemaly RF.Cytomegalovirus diseases after hematopoietic stem cell transplantation: a mini-review.Cancer Lett. 2014 ;342(1):1-8.
[17]金欣,陈建魁,于农,等.血液系统恶性肿瘤患者巨细胞病毒和EB病毒定量检测的临床价值[J].国际检验医学杂志, 2011,32(7): 793-794.
[18]谢卫民,张曦,彭贵华,等.造血干细胞移植受者免疫抑制治疗与巨细胞病毒感染的关系[J].中南大学学报(医学版), 2010,35(11): 1162-1166.
[19]刘开彦.造血干细胞移植患者巨细胞病毒感染的诊治进展[J].中国实用内科杂志,2007,27(20):1597-1598.
[20]Ruiz-Camps I, Len O, de la Cámara R, et al.Valganciclovir as pre-emptive therapy for cytomegalovirus infection in allogeneic haematopoietic stem cell transplant recipients. Antivir Ther. 2011;16(7):951-957.
[21]Boeckh M, Boivin G.Quantitation of cytomegalovirus: methodologic aspects and clinical applications.Clin Microbiol Rev. 1998;11(3):533-554.
[22]SY Y, YP C. The development of laboratory diagnosis of human cytomegalovirus infection. Foreign Medical Sciences Section of Pediatric. 2005; 32: 284-287.
[23]Bhatia J, Shah BV, Mehta AP,et al. Comparing serology, antigenemia assay and polymerase chain reaction for the diagnosis of cytomegalovirus infection in renal transplant patients.J Assoc Physicians India. 2004;52:297-300.
[24]Griscelli F, Barrois M, Chauvin S,et al.Quantification of human cytomegalovirus DNA in bone marrow transplant recipients by real-time PCR.J Clin Microbiol. 2001;39(12):4362-4369.
[25]翟文静,魏嘉琳,赵明峰,等.巨细胞病毒定量PCR与pp65抗原测定监测异基因造血干细胞移植巨细胞病毒感染的比较[J].中国实验血液学杂志,2009,17(6):1522-1526.
[26]Boeckh M, Gooley TA, Myerson D,et al.Cytomegalovirus pp65 antigenemia-guided early treatment with ganciclovir versus ganciclovir at engraftment after allogeneic marrow transplantation: a randomized double-blind study.Blood. 1996;88(10):4063-4071.
[27]Gerna G, Baldanti F, Middeldorp J, et al. Use of CMV transcripts for monitoring of CMV infections in transplant recipients.Int J Antimicrob Agents. 2000;16(4):455-460.
[28]Einsele H, Ehninger G, Hebart H,et al.Polymerase chain reaction monitoring reduces the incidence of cytomegalovirus disease and the duration and side effects of antiviral therapy after bone marrow transplantation.Blood. 1995;86(7): 2815-2820.
[29]Lu SC, Chin LT, Wu FM,et al. Seroprevalence of CMV antibodies in a blood donor population and premature neonates in the south-central Taiwan.Kaohsiung J Med Sci. 1999;15(10):603-610.
[30]张晓艳,李建勇,吴汉新,等.造血干细胞移植后对人巨细胞病毒感染病毒载量的检测[J].中华传染病杂志,2006,24(6):401-405.
[31]邱志祥,王茫佶,王莉红,等.异基因造血干细胞移植后血巨细胞病毒定量监测及临床意义[J].中华内科杂志,2012,51(5):371-375.
[32]陈欢,刘开彦,许兰平,等.异基因造血干细胞移植后实时定量聚合酶链反应在巨细胞病毒感染诊断和治疗中的应用[J].中华血液学杂志,2009,30(2): 77-81.
[33]Winston DJ, Ho WG, Bartoni K,et al.Ganciclovir prophylaxis of cytomegalovirus infection and disease in allogeneic bone marrow transplant recipients. Results of a placebo-controlled, double-blind trial.Ann Intern Med. 1993;118(3):179-184.
[34]Crumpacker CS.Ganciclovir. N Engl J Med. 1996;335(10): 721-729.
[35]李春元,陈本川.更昔洛韦的临床应用进展[J].国外医药:合成药生化药制剂分册,1998,19(3):172.
[36]胡增建,蒋华良,杨玉社.抗病毒药物研究新进展[J].中国药理学通报,1996,12(5):385.
[37]Tokimasa S, Hara J, Osugi Y, et al. Ganciclovir is effective for prophylaxis and treatment of human herpesvirus-6 in allogeneic stem cell transplantation.Bone Marrow Transplant. 2002;29(7):595-598.
[38]刘鑫荣.更昔洛韦对巨细胞病毒感染的治疗应用[J].国外医药:抗生素分册,1996,17(1):66.
[39]Henkin CC, Griener JC, Ten Eick AP. Stability of valganciclovir in extemporaneously compounded liquid formulations. Am J Health Syst Pharm. 2003;60(7):687-690.
[40]Emery VC, Hassan-Walker AF. Focus on new drugs in development against human cytomegalovirus. Drugs. 2002; 62(13):1853-1858.
[41]Weiskittel P.Valganciclovir hydrochloride (Valcyte): a new antiviral agent.Nephrol Nurs J. 2003;30(1):93-95.
[42]Bozkaya H, Yurdaydin C, Bozdayi AM,et al. Oral ganciclovir for treatment of lamivudine-resistant hepatitis B virus infection: a pilot study.Clin Infect Dis. 2002;35(8):960-965.
[43]Griffiths P, Whitley R, Snydman DR,et al.Contemporary management of cytomegalovirus infection in transplant recipients: guidelines from an IHMF workshop, 2007.Herpes. 2008;15(1):4-12.
[44]Winston DJ, Yeager AM, Chandrasekar PH,et al. Randomized comparison of oral valacyclovir and intravenous ganciclovir for prevention of cytomegalovirus disease after allogeneic bone marrow transplantation.Clin Infect Dis. 2003;36(6): 749-758.
[45]Meyers JD, Reed EC, Shepp DH,et al.Acyclovir for prevention of cytomegalovirus infection and disease after allogeneic marrow transplantation.N Engl J Med. 1988;318(2):70-75. |