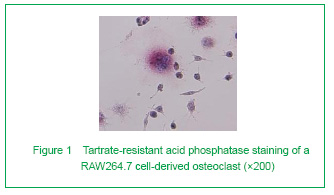
RAW264.7 cells treated with macrophage-colony stimulating factor and RANKL could be induced into mature osteoclasts
TRAP and resorption lacuna were differentiation markers for the osteoclasts.
To see if the cells were successfully induced into mature osteoclasts, TRAP staining and resorption assay were applied. We first examined TRAP staining. The result clearly confirmed our method. Osteoclasts were observed among the RAW264.7 cells. These big, multinucleated cells had irregular unclear boundaries and red-stained cytoplasm. On the contrary, the RAW264.7 cells were smaller and negatively stained (Figure 1).
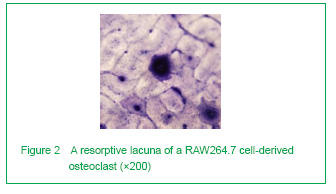
We next examined the formation of resorption lacunae. They were found big, with a round or irregular shape. Contrarily, the bone lacunae in the Haversian system were much smaller (Figure 2).
Physiological strain inhibits osteoclast apoptosis, and pathological strain has nearly no impact upon osteoclast apoptosis (Figure 3)
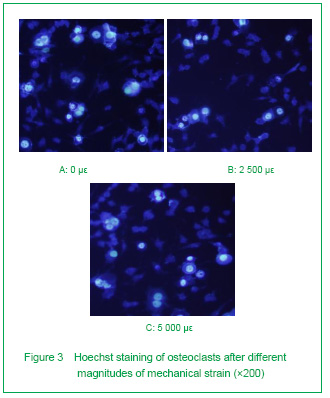
To see if mechanical strain modulated the apoptosis rate of osteoclasts, Hoechst staining was used. As shown in Figure 3, osteoclasts displaying characteristics of apoptosis as chromatin condensation and DNA fragmentation can be easily distinguished from normal cells by Hoechst staining. We found that osteoclasts underwent apoptosis spontaneously without any mechanical strain.
Then we calculated the apoptotic rate of each group. The apoptotic rate was (21.45±0.99)% in the control group, (15.34±1.50)% in the 2 500 με group and (21.95±1.06)% in the 5 000 με group.
Compared with the control group, the apoptosis rate of osteoclasts under 2 500 με decreased significantly (P < 0.05). Whereas, the apoptosis rate of osteoclasts under 5 000 με increased back to the unstrained ones (P > 0.05).
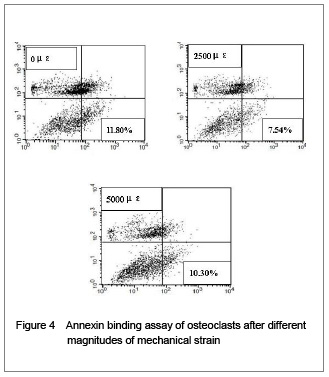
In addition, the Annexin-V/PI binding assay was applied to further confirm this effect. As shown in Figure 4, we obtained a same tendency as the Hoechst staining test showed. These results indicate that physiological strain inhibits osteoclast apoptosis, while higher strain magnitude beyond the physiological strain has nearly no impact upon osteoclast apoptosis, suggesting that mechanical strain plays an important role in the regulation of osteoclast apoptosis.
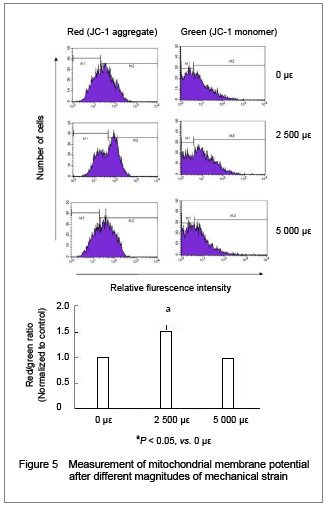
Mitochondria play an important role in regulating cell apoptosis. To study the role of the mitochondria in the response of mechanical strain to osteoclast apoptosis, we examined the effect of mechanical strain on the mitochondrial membrane potential. After JC-1 staining, the ratio of the mitochondrial aggregates (red) to cytoplasmic monomeric form (green) of JC-1 was observed and analyzed with flow cytometry. The increase of this ratio means that fewer cells undergo depolarization of the mitochondrial membrane.
As shown in Figure 5, the red/green ratio of the cells under physiological strain increased significantly (P < 0.05), while cells under pathologic strain had almost the same level of mitochondrial membrane potential, compared to the unstrained ones (P > 0.05).