[1] 赵国峰,宋克东,刘天庆,等.低氧条件下微囊化成骨细胞调控造血干/祖细胞扩增的研究[J].高校化学工程学报,2009, 23(4):623-631.
[2] LlESVELD JL. Trouble in the niche? Send in astatin. Blood. 2016;128(25):2877-2878.
[3] 张灵莉,刘晓倩,黄晓芹.成骨细胞对造血的调控及传统药物在该调控中的作用研究进展[J].中药与临床,2018,9(3):64-66+63.
[4] LIU H, HE J, BAGHERI-YARMAND R, et al. Osteocyte CIITA aggravates osteolytic bone lesions in myeloma. Nat Commun. 2022; 13(1):3684.
[5] BOSCH-FORTEA M, MARCIANO D, GAUTROT JE. Biomimetic artificial bone marrow niches for the scale up of hematopoietic stem and progenitor cells. Biomaterials. 2026;325:123612.
[6] BAUER M, VAXEVANIS C, JAEKEL N, et al. Association of the composition of the bone marrow tumor microenvironment in BCR: ABL1-negative myeloproliferative neoplasms with IFN-γ signaling and driver mutations. Leukemia. 2025;39(10):
2391-2405.
[7] DU L, FREITAS-CORTEZ MA, ZHANG J, et al. Periarteriolar niches become inflamed in aging bone marrow, remodeling the stromal microenvironment and depleting lymphoid progenitors. Proc Natl Acad Sci U S A. 2025; 122(11):e2412317122.
[8] TIKHONOVA AN, DOLGALEV I, HU H, et al. The bone marrow microenvironment at single-cell resolution. Nature. 2019;569(7755): 222-228.
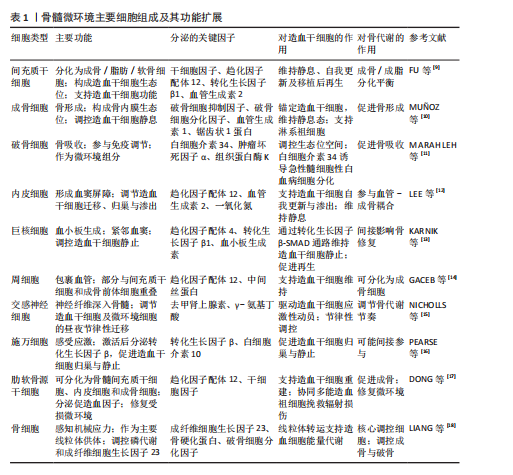
[9] FU J, WANG Y, JIANG Y, et al. Systemic therapy of MSCs in bone regeneration: a systematic review and meta-analysis. Stem Cell Res Ther. 2021;12(1):377.
[10] MUÑOZ J, AKHAVAN NS, MULLINS AP, et al. Macrophage Polarization and Osteoporosis: A Review. Nutrients. 2020;12(10):2999.
[11] MARAHLEH A, KITAURA H, OHORI F, et al. The osteocyte and its osteoclastogenic potential. Front Endocrinol (Lausanne). 2023; 14:1121727.
[12] LEE C, KIM MJ, KUMAR A, et al. Vascular endothelial growth factor signaling in health and disease: from molecular mechanisms to therapeutic perspectives. Signal Transduct Target Ther. 2025;10(1):170.
[13] KARNIK SJ, NAZZAL MK, KACENA MA, et al. Megakaryocyte Secreted Factors Regulate Bone Marrow Niche Cells During Skeletal Homeostasis, Aging, and Disease. Calcif Tissue Int. 2023;113(1):83-95.
[14] GACEB A, BARBARIGA M, ÖZEN I, et al. The pericyte secretome: Potential impact on regeneration. Biochimie. 2018;155:16-25.
[15] NICHOLLS AJ, WEN SW, HALL P, et al. Activation of the sympathetic nervous system modulates neutrophil function. J Leukoc Biol. 2018;103(2):295-309.
[16] PEARSE DD, MARCILLO AE, OUDEGA M, et al. Transplantation of Schwann cells and olfactory ensheathing glia after spinal cord injury: does pretreatment with methylprednisolone and interleukin-10 enhance recovery? J Neurotrauma. 2004;21(9):1223-1239.
[17] DONG R, LING Z, FAN P, et al. A Novel Therapeutic Strategy for Bone Marrow Failure: Niche Rejuvenation Using Costal Cartilage-Derived Stem Cells. Adv Sci (Weinh). Published online August 27, 2025.
[18] LIANG W, WEI T, HU L, et al. An integrated multi-omics analysis reveals osteokines involved in global regulation. Cell Metab. 2024; 36(5):1144-1163.e7.
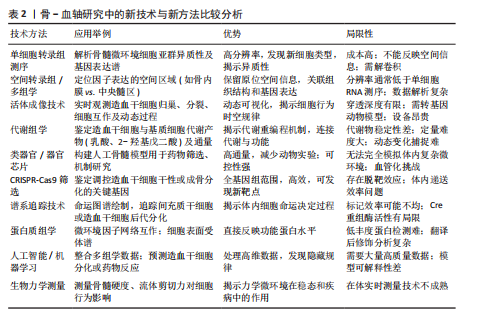
[19] FRÖBEL J, LANDSPERSKY T, PERCIN G, et al. The Hematopoietic Bone Marrow Niche Ecosystem. Front Cell Dev Biol. 2021;9:705410.
[20] KANDARAKOV O, BELYAVSKY A, SEMENOVA E. Bone Marrow Niches of Hematopoietic Stem and Progenitor Cells. Int J Mol Sci. 2022; 23(8):4462.
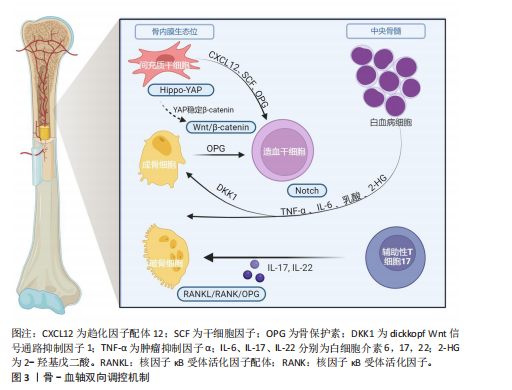
[21] LI D, XUE W, LI M, et al. VCAM-1+ macrophages guide the homing of HSPCs to a vascular niche. Nature. 2018;564(7734):119-124.
[22] GHAFOURI-FARD S, GLASSY MC, ABAK A, et al. The interaction between miRNAs/lncRNAs and Notch pathway in human disorders. Biomed Pharmacother. 2021;138:111496.
[23] BRUNS I, LUCAS D, PINHO S, et al. Megakaryocytes regulate hematopoietic stem cell quiescence through CXCL4 secretion. Nat Med. 2014;20(11):1315-1320.
[24] ZHAN H, KAUSHANSKY K. Megakaryocytes as the Regulator of the Hematopoietic Vascular Niche. Front Oncol. 2022;12:912060.
[25] TSIRKINIDIS P, TERPOS E, BOUTSIKAS G, et al. Bone metabolism markers and angiogenic cytokines as regulators of human hematopoietic stem cell mobilization. J Bone Miner Metab. 2018;36(4):399-409.
[26] BISWAS L, CHEN J, DE ANGELIS J, et al. Lymphatic vessels in bone support regeneration after injury. Cell. 2023;186(2):382-397.e24.
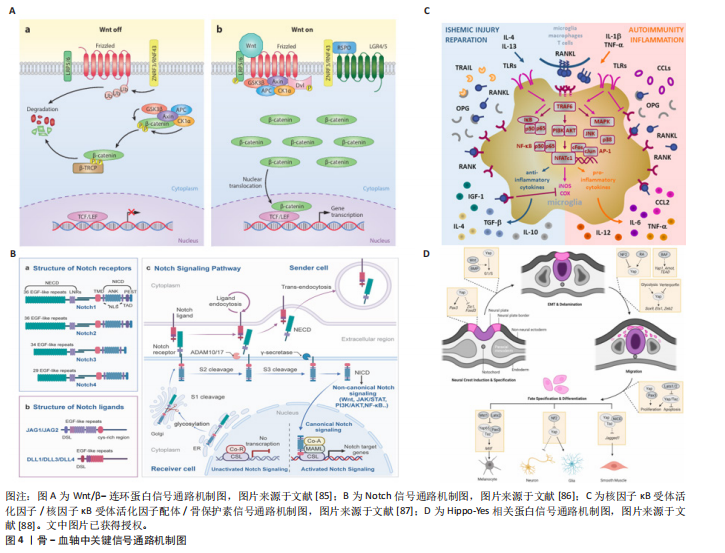
[27] RAMASAMY SK, KUSUMBE AP, WANG L, et al. Endothelial Notch activity promotes angiogenesis and osteogenesis in bone. Nature. 2014;507(7492):376-380.
[28] SUGIMURA R, HE XC, VENKATRAMAN A, et al. Noncanonical Wnt signaling maintains hematopoietic stem cells in the niche. Cell. 2012;150(2):351-365.
[29] ZHAO M, PERRY JM, MARSHALL H, et al. Megakaryocytes maintain homeostatic quiescence and promote post-injury regeneration of hematopoietic stem cells. Nat Med. 2014;20(11):1321-1326.
[30] ZHAO M, TAO F, VENKATRAMAN A, et al. N-Cadherin-Expressing Bone and Marrow Stromal Progenitor Cells Maintain Reserve Hematopoietic Stem Cells. Cell Rep. 2019; 26(3):652-669.e6.
[31] KAJIUME T, KAWAHARA Y, YUGE L, et al. Osteoblastic adherence regulates hematopoietic stem cell self-renewal and differentiation: a conceptional in vitro and in vivo study. Stem Cell Investig. 2021;8:21.
[32] LEE S, ZHAN H. Deciphering the differential impact of thrombopoietin/MPL signaling on hematopoietic stem/progenitor cell function in bone marrow and spleen. Stem Cell Reports. 2024;19(2):211-223.
[33] HIMBURG HA, TERMINI CM, SCHLUSSEL L, et al. Distinct Bone Marrow Sources of Pleiotrophin Control Hematopoietic Stem Cell Maintenance and Regeneration. Cell Stem Cell. 2018;23(3):370-381.e5.
[34] NIAZI V, GHAFOURI-FARD S, VERDI J, et al. Hypoxia preconditioned mesenchymal stem cell-derived exosomes induce ex vivo expansion of umbilical cord blood hematopoietic stem cells CD133+ by stimulation of Notch signaling pathway. Biotechnol Prog. 2022;38(1):e3222.
[35] GILCHRIST AE, HARLEY BAC. Connecting secretome to hematopoietic stem cell phenotype shifts in an engineered bone marrow niche. Integr Biol (Camb). 2020;12(7): 175-187.
[36] YU Z, YANG W, HE X, et al. Endothelial cell-derived angiopoietin-like protein 2 supports hematopoietic stem cell activities in bone marrow niches. Blood. 2022;139(10):1529-1540.
[37] LIM Y, BAEK KH, KIM HJ, et al. Changes in trabecular bone score and bone mineral density following allogeneic hematopoietic stem cell transplantation. Bone. 2019;124: 40-46.
[38] CANTONE AF, BURGALETTO C, DI BENEDETTO G, et al. Rebalancing Immune Interactions within the Brain-Spleen Axis Mitigates Neuroinflammation in an Aging Mouse Model of Alzheimer’s Disease. J Neuroimmune Pharmacol. 2025;20(1):15.
[39] HETTIARACHCHI SU, LI YH, ROY J, et al. Targeted inhibition of PI3 kinase/mTOR specifically in fibrotic lung fibroblasts suppresses pulmonary fibrosis in experimental models. Sci Transl Med. 2020;12(567):eaay3724.
[40] PÉPIN CM, ANDRÉ R, OCCELLI F, et al. Metastable water at several compression rates and its freezing kinetics into ice VII. Nat Commun. 2024;15(1):8239.
[41] 石琰,郭昊,郑凯娟,等.免疫细胞对骨髓间充质干细胞成骨分化作用及机制的研究进展[J].口腔颌面外科杂志,2024,34(2):150-153.
[42] XIONG Y, CHEN L, YAN C, et al. Retraction Note: M2 macrophagy-derived exosomal miRNA-5106 induces bone mesenchymal stem cells towards osteoblastic fate by targeting salt-inducible kinase 2 and 3. J Nanobiotechnology. 2024;22(1):238.
[43] LIU K, LUO X, LV ZY, et al. Macrophage-Derived Exosomes Promote Bone Mesenchymal Stem Cells Towards Osteoblastic Fate Through microRNA-21a-5p. Front Bioeng Biotechnol. 2022;9:801432.
[44] 徐枭喻,廖礼彬,张馨怡,等.剪切应力通过M2型巨噬细胞外泌体miR-5106促进骨髓间充质干细胞向成骨细胞分化[J].中国组织化学与细胞化学杂志,2024,33(3):209-216.
[45] NIKLASON LE, LAWSON JH. Bioengineered human blood vessels. Science. 2020; 370(6513):eaaw8682.
[46] RICHOZ N, TUONG ZK, LOUDON KW, et al. Distinct pathogenic roles for resident and monocyte-derived macrophages in lupus nephritis. JCI Insight. 2022;7(21):e159751.
[47] YUAN W, LIU M, YANG D, et al. Improvement in long-term survival with mesenchymal stem cell transplantation in systemic sclerosis patients: a propensity score-matched cohort study. Stem Cell Res Ther. 2025;16(1):128.
[48] WANG YM, LOVELESS M, MILLER E, et al. Phenotypes of adults with Fanconi anaemia. Br J Haematol. 2023;201(1):133-139.
[49] CHAROENNGAM N, RITTIPHAIROJ T, PONVILAWAN B. Fracture prevalence in thalassemia: a systematic review and meta-analysis. Arch Osteoporos. 2021;16(1):171.
[50] REAGAN MR, LIAW L, ROSEN CJ, et al. Dynamic interplay between bone and multiple myeloma: emerging roles of the osteoblast. Bone. 2015;75:161-169.
[51] KARAGIANNI A, RAVID K. Myeloproliferative disorders and their effects on bone homeostasis: the role of megakaryocytes. Blood. 2022;139(21):3127-3137.
[52] LALIOTI ME, ROMERO-MULERO MC, KARABACZ N, et al. Differentiation, ageing and leukaemia alter the metabolic profile of human bone marrow haematopoietic stem and progenitor cells. Nat Cell Biol. 2025; 27(8):1367-1380.
[53] WILLYANTO SE, ALIMSJAH YA, TANJAYA K, et al. Comprehensive analysis of the efficacy and safety of CAR T-cell therapy in patients with relapsed or refractory B-cell acute lymphoblastic leukaemia: a systematic review and meta-analysis. Ann Med. 2024; 56(1):2349796.
[54] RETTKOWSKI J, ROMERO-MULERO MC, SINGH I, et al. Modulation of bone marrow haematopoietic stem cell activity as a therapeutic strategy after myocardial infarction: a preclinical study. Nat Cell Biol. 2025;27(4):591-604.
[55] LI H, BAI L. Advances in mesenchymal stem cell and exosome-based therapies for aging and age-related diseases. Stem Cell Res Ther. 2025;16(1):401.
[56] KIM J, BIXEL MG. Intravital Multiphoton Imaging of the Bone and Bone Marrow Environment. Cytometry A. 2020;97(5):496-503.
[57] ALHARITHI YJ, PHILLIPS EA, WILSON TD, et al. Metabolomic and transcriptomic remodeling of bone marrow myeloid cells in response to maternal obesity. Am J Physiol Endocrinol Metab. 2025;328(2):E254-E271.
[58] FRENZ-WIESSNER S, FAIRLEY SD, BUSER M, et al. Generation of complex bone marrow organoids from human induced pluripotent stem cells. Nat Methods. 2024;21(5):868-881.
[59] AJORE R, MATTSSON J, PERTESI M, et al. Genome-wide CRISPR/Cas9 screen identifies regulators of BCMA expression on multiple myeloma cells. Blood Cancer J. 2024;14(1):21.
[60] WENG C, YU F, YANG D, et al. Deciphering cell states and genealogies of human haematopoiesis. Nature. 2024;627(8003): 389-398.
[61] KRAMER MH, ZHANG Q, SPRUNG R, et al. Proteomic and phosphoproteomic landscapes of acute myeloid leukemia. Blood. 2022;140(13):1533-1548.
[62] 肖宇俊,张燕,杨建民,等. 基于人工智能深度学习的骨髓细胞形态学检测的研究进展[J]. 中华生物医学工程杂志,2023, 29(4):457-461.
[63] JANSSEN E, VAN DEN DRIES K, VENTRE M, et al. Mechanobiology of myeloid cells. Curr Opin Cell Biol. 2024;86:102311.
[64] ZHANG D, DUQUE-JIMENEZ J, FACCHINETTI F, et al. Transferrin receptor targeting chimeras for membrane protein degradation. Nature. 2025;638(8051):787-795.
[65] HOFMAN MS, EMMETT L, SANDHU S, et al. Overall survival with [177Lu]Lu-PSMA-617 versus cabazitaxel in metastatic castration-resistant prostate cancer (TheraP): secondary outcomes of a randomised, open-label, phase 2 trial. Lancet Oncol. 2024;25(1):99-107.
[66] NA J, YANG Z, SHI Q, et al. Extracellular matrix stiffness as an energy metabolism regulator drives osteogenic differentiation in mesenchymal stem cells. Bioact Mater. 2024;35:549-563.
[67] LUO HH, REN WY, YE AH, et al. DDIT3 switches osteogenic potential of BMP9 to lipogenic by attenuating Wnt/β-catenin signaling via up-regulating DKK1 in mesenchymal stem cells. Aging (Albany NY). 2024;16(18):12543-12558.
[68] WANG J, XIE W, LI N, et al. Generation of a humanized mesonephros in pigs from induced pluripotent stem cells via embryo complementation. Cell Stem Cell. 2023;30(9): 1235-1245.e6.
[69] FAZIO A, DI MARTINO A, BRUNELLO M, et al. The involvement of signaling pathways in the pathogenesis of osteoarthritis: An update. J Orthop Translat. 2024;47:116-124.
[70] YAO XT, LI PP, LIU J, et al. Wnt/β-Catenin Promotes the Osteoblastic Potential of BMP9 Through Down-Regulating Cyp26b1 in Mesenchymal Stem Cells. Tissue Eng Regen Med. 2023;20(5):705-723.
[71] ZOLGHADR F, SADEGHIZADEH M, AMIRIZADEH N, et al. How benzene and its metabolites affect human marrow derived mesenchymal stem cells. Toxicol Lett. 2012; 214(2):145-153.
[72] GUO Z, XU Q, ZHANG K, et al. Sclerostin: A Potential Link between Osteoporosis and Alzheimer’s Disease. Neurodegener Dis. 2025;23:1-9.
[73] CHEN Y, LIU Z, ZOU J, et al. Low-dose tyrosine kinase inhibitors in patients with chronic myeloid leukemia: a retrospective study in China. Haematologica. 2022;107(8):1966-1970.
|