中国组织工程研究 ›› 2026, Vol. 30 ›› Issue (35): 9281-9293.doi: 10.12307/2026.257
• 组织构建综述 tissue construction review • 上一篇 下一篇
非凋亡型调节性细胞死亡致缺血性脑卒中神经细胞损伤的机制
苏 旭1,张晓希1,杨娅青1,傅振燚2,刘佳鑫2
- 1昆明医科大学第六附属医院,云南省玉溪市 653100;2昆明理工大学基础医学院,云南省昆明市 650000
-
收稿日期:2025-08-08修回日期:2025-11-17出版日期:2026-12-18发布日期:2026-04-29 -
通讯作者:刘佳鑫,博士,讲师,昆明理工大学基础医学院,云南省昆明市 650000 -
作者简介:第一作者:苏旭,男,1981年生,云南省玉溪市人,汉族,副主任医师,主要从事中医内科临床及研究工作。 共同第一作者:张晓希,女,1990年生,云南省玉溪市人,汉族,硕士,主治医师,主要从事中医内科诊疗及研究工作。 -
基金资助:昆明理工大学联合专项项目(KUST-KH2023001Y) ,项目负责人:刘佳鑫;云南省中西医结合慢病防治重点实验室项目(YPKLS2024-004),项目负责人:刘佳鑫
Mechanism by which non-apoptotic regulated cell death induces neuronal injury in ischemic stroke
Su Xu1, Zhang Xiaoxi1, Yang Yaqing1, Fu Zhenyi2, Liu Jiaxin2
- 1The Sixth Affiliated Hospital of Kunming Medical University, Yuxi 653100, Yunnan Province, China; 2School of Basic Medicine, Kunming University of Science and Technology, Kunming 650000, Yunnan Province, China
-
Received:2025-08-08Revised:2025-11-17Online:2026-12-18Published:2026-04-29 -
Contact:Liu Jiaxin, MD, Lecturer, School of Basic Medicine, Kunming University of Science and Technology, Kunming 650000, Yunnan Province, China -
About author:Su Xu, Associate chief physician, The Sixth Affiliated Hospital of Kunming Medical University, Yuxi 653100, Yunnan Province, China Zhang Xiaoxi, MS, Attending physician, The Sixth Affiliated Hospital of Kunming Medical University, Yuxi 653100, Yunnan Province, China Su Xu and Zhang Xiaoxi contributed equally to this article. -
Supported by:Joint Special Project of Kunming University of Science and Technology, No. KUST-KH2023001Y (to LJX); Key Laboratory Project of Chronic Disease Prevention and Treatment of Integrated Traditional Chinese and Western Medicine in Yunnan Province, No. YPKLS2024-004 (to LJX)
摘要:
文题释义:
自噬:又称为Ⅱ型细胞死亡,是一种进化上保守的真核细胞过程,指细胞在自噬相关基因的调控下利用溶酶体降解自身受损细胞器和大分子物质的过程。脑缺血导致蛋白质错误折叠和聚集,并损害几乎所有类型的亚细胞器,包括线粒体、内质网、高尔基体、溶酶体等,不可修复的受损细胞器和不溶性蛋白质聚集体通常通过自噬去除。
铁死亡:是一种与铁依赖性活性氧和脂质过氧化物形成相关的新型调节性细胞死亡。细胞中铁代谢紊乱导致产生过多的铁离子,这些铁离子通过芬顿反应产生大量活性氧,活性氧靶向脂质膜内的多不饱和脂肪酸,产生脂质过氧化物,损害细胞膜的完整性,破坏线粒体活性,最终导致铁死亡。
背景:近年来,非凋亡型调节性细胞死亡参与缺血性脑卒中的发展已成为研究热点。
目的:总结现阶段自噬、铁死亡、铜死亡、双硫死亡、坏死性凋亡、细胞焦亡等非凋亡型调节性细胞死亡亚程序在缺血性脑卒中神经细胞损伤中发挥的作用及作用机制。
方法:通过中国知网和PubMed数据库检索有关非凋亡型调节性细胞死亡与缺血性脑卒中的文献,英文检索词为“ischemic stroke,regulated cell death,autophagy,ferroptosis, cuproptosis,disulfidptosis,necroptosis, pyroptosis,alkaliptosis,oxeiptosis,parthanatos,mitochondrial permeability transition-driven necrosis,neutrophil extracellular trap-related death,lysosome-dependent cell death,immunogenic cell death,anoikis”,中文检索词为“缺血性脑卒中,调节性细胞死亡,铁死亡,坏死性凋亡,自噬,焦亡,铜死亡,双硫死亡,碱死亡,氧死亡,聚ADP核糖聚合酶1依赖性细胞死亡,溶酶体依赖性细胞死亡,失巢凋亡,线粒体通透性转变驱动的坏死,免疫原性细胞死亡,中性粒细胞胞外陷阱相关死亡”。根据入选标准,最终纳入176篇文献进行分析总结。
结果与结论:非凋亡型调节性细胞死亡的调控机制主要包括自噬、铁死亡、失巢凋亡、铜死亡、双硫死亡、坏死性凋亡、焦亡、氧死亡、碱死亡、聚ADP核糖聚合酶1依赖性细胞死亡、线粒体通透性转换驱动的坏死、中性粒细胞胞外陷阱相关死亡、溶酶体依赖性细胞死亡和免疫原性细胞死亡。自噬对缺血性脑卒中神经细胞损伤起到双重调控作用,在缺血条件下自噬发挥神经细胞保护作用,而再灌注阶段的过度自噬可导致神经细胞死亡。铁死亡可通过铁过载、脂质过氧化加重缺血性脑卒中神经细胞损伤,铜死亡可通过调控蛋白铁氧还蛋白1调节谷胱甘肽诱导铁死亡,双硫死亡和铁死亡之间存在部分串扰,在葡萄糖匮乏的条件下,溶质载体家族7成员11的上调会消耗还原型烟酰胺腺嘌呤二核苷酸磷酸,导致二硫键化合物异常积累,促进神经细胞双硫死亡的发生。坏死性凋亡的重要参与者混合系激酶结构域样假激酶也与细胞焦亡相关蛋白NLRP3炎症小体的激活有关,在缺血性脑卒中坏死性凋亡发生期间进一步促进神经细胞焦亡。缺血性脑卒中的中性粒细胞胞外陷阱相关死亡主要是由瓜氨酸化、应激触发中性粒细胞胞外陷阱形成以及释放各种细胞毒性蛋白酶介导的炎症反应引起的。免疫原性细胞死亡等其他新兴亚型以多种特异性方式造成缺血性脑卒中的神经细胞损伤。
https://orcid.org/0009-0005-6815-2013 (苏旭)
中国组织工程研究杂志出版内容重点:干细胞;骨髓干细胞;造血干细胞;脂肪干细胞;肿瘤干细胞;胚胎干细胞;脐带脐血干细胞;干细胞诱导;干细胞分化;组织工程
中图分类号:
引用本文
苏 旭, 张晓希, 杨娅青, 傅振燚, 刘佳鑫. 非凋亡型调节性细胞死亡致缺血性脑卒中神经细胞损伤的机制[J]. 中国组织工程研究, 2026, 30(35): 9281-9293.
Su Xu, Zhang Xiaoxi, Yang Yaqing, Fu Zhenyi, Liu Jiaxin. Mechanism by which non-apoptotic regulated cell death induces neuronal injury in ischemic stroke[J]. Chinese Journal of Tissue Engineering Research, 2026, 30(35): 9281-9293.
2.1.1 自噬依赖性细胞死亡对缺血性脑卒中神经细胞损伤的作用及机制 自噬作为一种适应性机制,在维持细胞稳态和生物体存活方面发挥了重要作用,它通过自噬体-溶酶体降解过程去除长寿蛋白或受损的细胞器、蛋白质聚集体和多余的细胞成分来减轻细胞应激[1](图5)。在脑缺血后再灌注阶段,自噬可去除受损的线粒体,从而减轻线粒体损伤诱导的神经元凋亡和缺血损伤[14]。自噬的主要分子调节因子包括哺乳动物雷帕霉素靶蛋白复合体1、腺苷酸活化蛋白激酶、肿瘤抑制蛋白p53和内质网应激。腺苷酸活化蛋白激酶是丝氨酸/苏氨酸激酶组的成员,脑缺血后,磷酸化的腺苷酸活化蛋白激酶通过直接和间接途径激活Unc-51类激酶复合物来启动自噬途径[15]。在大鼠脑缺血预处理前5 min,注射单剂量化合物C可抑制大脑中动脉闭塞大鼠的自噬、增加梗死面积,表明腺苷酸活化蛋白激酶依赖性自噬有助于缺血缺氧引起的大脑神经细胞保护[16]。哺乳动物雷帕霉素靶蛋白是一种丝氨酸/苏氨酸激酶,是磷脂酰肌醇3-激酶相关激酶激酶家族的成员[17],哺乳动物雷帕霉素靶蛋白通过Unc-51类激酶来抑制自噬。哺乳动物雷帕霉素靶蛋白复合体1位于溶酶体的细胞质表面,与负责自噬降解的细胞器完全相同。在缺氧条件下,用雷帕霉素(哺乳动物雷帕霉素靶蛋白抑制剂)处理新生大鼠的神经元细胞可增加自噬标志物苄氯素1和微管相关蛋白1轻链3Ⅱ的表达、减少神经元死亡[17]。在大脑中动脉闭塞的啮齿动物中,施用雷帕霉素可以减少梗死体积、减少神经元和内皮细胞损伤、提高血脑屏障的通透性、改善神经功能[18]。在缺血性脑卒中鼠模
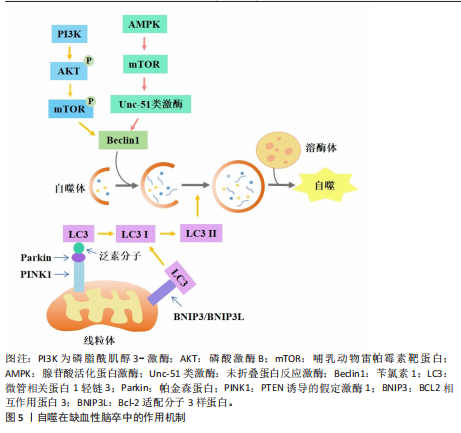
型中,用6-Gingerol(从生姜根茎中分离的化合物)给药处理可抑制蛋白激酶B/哺乳动物雷帕霉素靶蛋白/信号转导和转录激活物通路的激活,改善小胶质细胞自噬、减少炎症反应的发生[19]。表达成纤维细胞生长因子21的基因工程鼠伤寒沙门氏菌,通过成纤维细胞生长因子受体1/腺苷酸活化蛋白激酶/哺乳动物雷帕霉素靶蛋白通路促进缺血性脑卒中的神经功能恢复[20]。碱性成纤维细胞生长因子敲低通过抑制Caveolin-1的水平来抑制哺乳动物雷帕霉素靶蛋白的信号传导,在少年大鼠动脉缺血性脑损伤后加剧认知损害[21]。目前关于自噬在缺血性脑卒中的研究逐渐趋向于中草药及与生物工程相结合的药物开发研究,常与铁死亡等相关机制联合研究,深入探讨对缺血性脑卒中的双重作用机制。
在缺血条件下,自噬通过去除功能失调的细胞器和蛋白质来减轻细胞损伤,并保持细胞完整性;相反,在再灌注过程中,自噬可能会加剧损伤并导致细胞死亡[22]。磷酸肌醇-3激酶/蛋白激酶B信号通路与自噬调节密切相关。中医方剂通窍活血汤可以激活磷酸肌醇-3激酶/蛋白激酶B/哺乳动物雷帕霉素靶蛋白信号通路,减少脑缺血/再灌注损伤后脑微血管内皮细胞自噬,减少内皮细胞死亡[23]。有研究发现,N-乙酰转移酶10调节缺血损伤后星形细胞自噬,N-乙酰转移酶10直接催化组织金属蛋白酶抑制因子1 mRNA的 N4-乙酰基胞苷乙酰化,导致组织金属蛋白酶抑制因子1上调和组织金属蛋白酶抑制因子1依赖性自噬激活增加[24]。再灌注前24 h,自噬抑制剂 3-甲基腺嘌呤处理可引起较高的神经元死亡率;再灌注后48,72 h, 3-甲基腺嘌呤处理可显著保护神经元,原因可能为:瞬时糖氧剥夺诱导适应性自噬,消除受损细胞器,拯救神经元,而长时间糖氧剥夺引发过度自噬,导致神经元死亡[25]。毛蕊异黄酮调控信号转导及转录激活蛋白3/叉头框蛋白O3A信号通路抑制自噬,进而减轻脑缺血/再灌注损伤,通过下调微管相关蛋白1轻链3Ⅱ/微管相关蛋白1轻链3Ⅰ比值、上调SQSTM1表达来缓解脑缺血/再灌注诱导的过度自噬[26]。缺血性脑卒中后肉瘤融合蛋白聚集诱导过度自噬,促进脑星形胶质细胞活化,从而加重神经功能损伤[27]。上述证据表明,靶向自噬途径的药物具有治疗缺血性脑卒中的潜力,未来可能通过关注关键自噬调节因子及其相关途径,在增强自噬有益作用的同时最大限度地减少其有害影响。
参与缺血性脑卒中调节的自噬还有依赖/非依赖PTEN诱导的假定激酶1/帕金森蛋白线粒体自噬。线粒体自噬通过选择性地消除和降解受损与多余的线粒体,从而阻止线粒体脱氧核糖核酸突变的积累,促进细胞代谢的重编程,从而在细胞稳态中发挥关键作用。PTEN诱导的假定激酶1/帕金森蛋白参与的自噬同样具有双向调节功能[28]。帕金森蛋白感知线粒体跨膜潜在损失,并在受损的线粒体中积累[29]。大脑中动脉闭塞诱导大鼠缺血/再灌注模型可导致线粒体功能严重受损、线粒体膜电位和腺嘌呤核苷三磷酸水平显著降低与线粒体受损聚集,而PTEN诱导的假定激酶1/帕金森蛋白的激活可促进受损的线粒体清除。有研究表明,电针通过脑缺血再灌注中PTEN诱导的假定激酶1/帕金森蛋白介导的线粒体自噬清除减少受损线粒体的积累,进而改善神经元损伤;酸性后处理可通过促进帕金森蛋白线粒体易位,增加帕金森蛋白介导的线粒体自噬[30]。增强的帕金森蛋白Parkin依赖性线粒体自噬进一步将再灌注窗口延长至缺血后至少6 h[29]。β-细辛醚通过靶向PTEN诱导的假定激酶1/帕金森蛋白依赖性线粒体自噬减轻脑缺血/再灌注损伤[31]。有趣的是,在糖氧剥夺/复氧处理的人神经母细胞瘤细胞SH-SY5Y细胞模型中,YU等[32]发现抑制线粒体钙单向转运蛋白减弱了糖氧剥夺/复氧诱导的PTEN诱导的假定激酶1/帕金森蛋白依赖性线粒体自噬,保护神经元免受缺血/再灌注损伤。BCL2相互作用蛋白3和Bcl-2适配分子3样蛋白是位于线粒体外膜上的促凋亡性线粒体蛋白,它们与抗凋亡蛋白Bcl-2的竞争性结合会使Bcl-2-苄氯素1复合物解离并释放苄氯素1,从而激活线粒体自噬,减少细胞凋亡[33]。在正常情况下,BCL2相互作用蛋白3/Bcl-2适配分子3样蛋白在大多数器官中的表达较低,但在缺氧缺血条件下,它的转录水平受缺氧诱导因子1α直接调节并被激活[34]。在新生缺氧缺血动物模型中,BCL2相互作用蛋白3和Bcl-2适配分子3样蛋白以“延迟”方式高度表达,并且与脑卒中后延迟神经元丢失有关。当BCL2相互作用蛋白3沉默时,线粒体自噬水平不受Bcl-2适配分子3样蛋白急剧上调的影响,表明Bcl-2适配分子3样蛋白的主要功能是维持生理水平的线粒体自噬,而BCL2相互作用蛋白3触发大脑缺氧缺血中的过度线粒体自噬[35]。缺血再灌注时,体内外敲除帕金森蛋白基因后,Bcl-2适配分子3样蛋白仍能促进线粒体自噬,并且可检测到帕金森蛋白、FUN14结构域包含蛋白1、Bcl2、Bcl-2适配分子3样蛋白/ BCL2相互作用蛋白3等线粒体自噬相关蛋白[33]。使用特定分子靶向线粒体自噬相关通路可能为某些缺血性脑卒中患者提供治疗益处。
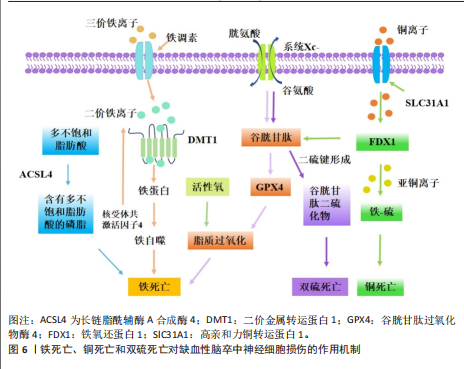
2.1.2 铁死亡、铜死亡和双硫死亡对缺血性脑卒中神经细胞损伤的作用及机制 铁死亡是一种新型的调节性细胞死亡,主要特征是细胞内铁代谢异常导致脂质过氧化,从而引发细胞膜的破裂和细胞死亡[36]。谷胱甘肽是一种来源于半胱氨酸、谷氨酸和甘氨酸的三肽[37],是合成谷胱甘肽过氧化酶4的底物,谷胱甘肽过氧化酶4可将细胞内毒性磷脂氢过氧化物转化为无毒醇[38]。谷胱甘肽的消耗会抑制谷胱甘肽过氧化酶4的表达,使脂质过氧化物积累,发生铁死亡[39]。补充谷胱甘肽可通过激活磷酸肌醇-3激酶/蛋白激酶B和Bcl-2等神经保护通路减少脑梗死面积和缺血性细胞死亡[40]。谷胱甘肽过氧化酶4过表达可抑制脑卒中模型中神经元的铁死亡[41]。有研究表明,通过抑制或调节脂质过氧化酶,如赖氨酰氧化酶、环氧合酶、长链酰基辅酶A合成酶4和TOLL样受体4,可能减轻缺血性细胞死亡并保持神经元功能[42]。脑缺血时,腺嘌呤核苷三磷酸的消耗导致膜去极化,引发谷氨酸的大量释放,随后谷氨酸受体的过度激活显著导致细胞内钙超载;脑卒中后,细胞内钙水平升高可激活胞质胞浆型磷脂酶A2α,启动膜磷脂的水解,从而引发缺血性脑损伤后长链酰基辅酶A合成酶4依赖性铁死亡[43]。凝血酶通过长链酰基辅酶A合成酶4和溶血卵磷脂酰基转移酶3增强含磷脂酰乙醇胺的多不饱和脂肪酸合成,而多不饱和脂肪酸是脂质过氧化和铁沉积的底物[44]。研究表明,TOLL样受体4可能通过激活核因子kB[45]、NLRP3炎症小体等多种炎症信号通路在脑卒中后脂质过氧化发展中发挥作用[46]。TOLL样受体4过表达诱导活性氧、腺嘌呤核苷三磷酸和铁离子的产生,从而促进糖氧剥夺/复氧诱导的海马神经元细胞HT22铁死亡相关蛋白表达[47]。在脑缺血再灌注期间,铁死亡主要通过铁超载、兴奋性毒性、抗氧化系统破坏和脂质过氧化物等机制加剧神经元损伤[48]。当脑缺血再灌注发生时,以谷氨酸为代表的兴奋性氨基酸的释放增加并在突触嵴中积累,细胞内谷氨酸的摄取减少,而细胞外谷氨酸的释放增加,导致系统Xc-的抑制[49]。胱氨酸由半胱氨酸谷氨酸逆向转运蛋白系统Xc-运输,它由细胞中的胱氨酸/谷氨酸逆向转运蛋白溶质载体家族7成员11组成,用于合成谷胱甘肽[50]。高浓度谷氨酸通过抑制系统Xc-来减少胱氨酸的摄取,引发细胞内谷胱甘肽耗竭、活性氧积累,造成脂质过氧化增加、线粒体损伤,最终导致铁死亡[43]。在大鼠大脑中动脉闭塞模型中,缺血性区域的系统Xc-活性显著增加,在脑卒中后第3-7天达到峰值;通过免疫组化检测证明神经元、星形胶质细胞和小胶质细胞中的系统Xc-水平上
调[51]。星楼成气汤通过溶质载体家族7成员11/谷胱甘肽过氧化酶4信号传导抑制铁死亡,防止脑缺血/再灌注损伤[52]。
触发铁死亡的另一种方式是通过铁代谢失衡引起芬顿反应促进脂质过氧化途径。研究已证明,铁代谢的失调和之后Fe3+在大脑中的积累可对延迟神经变性和缺血后的不良预后发挥作用。缺血性脑卒中后脑组织会出现异常铁沉积[53]。缺血性脑卒中后,由于紧密连接蛋白的破坏,血脑屏障受到损害,血液中的转铁蛋白在转铁蛋白受体1的作用下进入脑组织,促进Fe3+从血液中释放到脑中,使脑组织中铁离子异常蓄积[54-55],铁还原酶将进入细胞内的Fe3?还原为Fe2?,为芬顿反应提供所需的底物,催化活性氧生成,进一步导致脂质过氧化、线粒体功能障碍和铁死亡的发生[56]。HU等[57]研究表明,脑缺血再灌注损伤改变了转铁蛋白独特肽、总白蛋白独特肽的表达水平,导致脑不稳定铁池失衡。铁调素是一种调节激素,在脑缺血铁超载中起关键作用。LUO等[58]发现缺血性脑卒中模型中白细胞介素6表达增加,并通过Janus 激酶信号转导及转录激活蛋白3途径提高铁调素水平,诱导铁调节转运蛋白1降解,导致铁释放减少,铁沉积增加。以上研究表明,靶向调控谷胱甘肽过氧化酶4、TOLL样受体4和转铁蛋白受体1等铁死亡相关蛋白可抑制脂质过氧化、减少铁死亡发生,对缺血性脑卒中的治疗具有重要的潜力。
铜死亡是一种全新的细胞死亡形式,与已知的细胞凋亡、坏死、焦亡和铁死亡不同,细胞内铜积累是铜死亡的主要触发因素[59]。研究表明,血浆铜水平升高会增加脑卒中的易感性[60],饮用水中微量的铜会通过损害小鼠的内皮祖细胞而加重脑缺血性损伤[61]。在脑缺血/再灌注损伤中,缺血或缺氧使线粒体腺嘌呤核苷三磷酸产生减少,从而限制铜的运输,导致铜积累[62]。在脑缺血/再灌注损伤中,Cu2+直接与线粒体三羧酸循环中的含硫成分结合,导致铜诱导的铁硫蛋白还原,从而触发蛋白质毒性应激,诱导铜依赖性细胞死亡;用右美托咪定和铜螯合剂D-青霉胺预处理可阻断铁氧还蛋白1介导的铜死亡发挥的脑损伤作用,使大鼠脑梗死体积显著减少、铜离子和铜凋亡相关蛋白含量降低,维持线粒体功能、增加谷胱甘肽水平,铁氧还蛋白1是铜死亡的主要调节蛋白,铁氧还蛋白1过表达部分逆转了右美托咪定的上述治疗作用[63]。和假手术组相比,槲皮素处理的大脑中动脉闭塞大鼠同侧大脑皮质中必需微量元素铜水平显著降低[64]。研究表明,双硫仑可以通过抑制铁氧还蛋白1来调节铜稳态,保护大脑中动脉闭塞小鼠神经髓鞘并缓解线粒体氧化应激;另外,铜死亡会导致热休克蛋白70应激和炎症反应的增加,进一步促进脑组织损伤[65]。一些研究通过基因组学探讨铜死亡在缺血性脑卒中中发挥的作用机制。在基因表达综合数据库中分析男性和女性缺血性脑卒中患者和健康人相比的差异表达基因,结果显示铜死亡相关差异基因含NLR家族Pyrin域蛋白3上调和金属硫蛋白1X下调[66]。有研究比较了基于铜死亡相关基因表达缺血性脑卒中患者的免疫特性,单细胞分析鉴定出19种铜死亡相关基因在缺血性脑卒中小鼠脑组织中不同细胞类型的不同表达模式,19种基因大多数与浆细胞、初始CD4+ T细胞、活化的树突状细胞和中性粒细胞4种免疫细胞类型显著相关;发现高亲和力铜转运蛋白1、铜离子转运ATP酶Α肽和铜离子转运ATP酶B肽是参与缺血性脑卒中的枢纽基因,高亲和力铜转运蛋白1在维持细胞铜稳态中起着至关重要的作用,它介导铜摄取,而铜离子转运ATP酶Α/B肽介导铜的输出,表明脑卒中可能是由于铜稳态失衡导致的铜死亡引起的,该研究还发现这些铜死亡相关基因富集于核转录因子信号通路、氧化磷酸化信号通路和肿瘤坏死因子信号通路,提示它们可能与小胶质细胞功能密切相关[67]。
双硫死亡是一种新型的调节性细胞死亡形式,由肌动蛋白之间的二硫键异常积累引起,肌动蛋白是维持细胞形状和存活的关键细胞结构[68]。二硫键是由2个半胱氨酸残基氧化形成的共价键,在促进蛋白质折叠和增强蛋白质稳定性方面发挥作用。然而,细胞质二硫键的异常积累会破坏细胞氧化还原稳态,导致硫化物应激诱导细胞毒性,人体主要通过硫氧还蛋白系统和谷胱甘肽系统消除有毒的二硫化物[69]。有报道显示,与对照组相比,缺血性脑卒中患者中双硫死亡相关基因GTP酶激活蛋白1、钙调素结合紧张素1、肌动蛋白结合蛋白β、倒转蛋白形成蛋白2和溶质载体家族7成员11的表达显著上调,认为双硫死亡在缺血性脑卒中脑损伤中发挥重要作用[70]。另有学者对脑卒中患者进行了双硫死亡基因分析,发现双硫死亡相关差异表达基因与各种细胞死亡途径和免疫过程相互关联[71]。在脑卒中小鼠模型中,经共定位分析确认过氧化还原酶1为缺血性脑卒中的关键靶点,脑卒中和脑卒中后适应都表现过氧化还原酶1水平的下调,提示氧化还原酶1在脑卒中后发挥的保护作用[72]。最新研究表明,泛醌氧化还原酶亚基11和泛醌氧化还原酶亚基S1-泛醌氧化还原酶亚基A11蛋白复合物在缺血性脑卒中的体外/体内模型及临床血液样本中表达均显著下调,泛醌氧化还原酶亚基11参与线粒体中呼吸链蛋白复合物的形成,因此泛醌氧化还原酶亚基11可作为双硫死亡生物标志物,通过靶向线粒体治疗缺血性脑卒中引起的双硫死亡是可能的[73]。
双硫死亡和铁死亡之间存在部分串扰,铁死亡会进一步诱导双硫死亡的发生(图6)。在葡萄糖匮乏的条件下,溶质载体家族7成员11水平上调会消耗还原型烟酰胺腺嘌呤二核苷酸磷酸,导致胱氨酸等二硫键化合物的异常积累,从而诱导二硫键应激和细胞快速死亡[74],因此,减少二硫键化合物形成和纠正巯基-二硫键失衡的策略可能是改善缺血性脑卒中结局的关键。溶质载体家族7成员11的高表达可驱动双硫死亡并诱发脑卒中,可能影响细胞对特定代谢应激的反应。在缺血性脑卒中过程中,小胶质细胞中双硫死亡相关基因的表达水平显著升高,表明双硫死亡可能在脑卒中的炎症反应中发挥重要作用[75]。核因子E2相关因子2 siRNA注射降低了大脑中动脉闭塞模型大鼠硫氧还蛋白的蛋白质和mRNA表达,硫氧还蛋白受核因子E2相关因子2调控,具有神经保护功能;在缺血条件下,核因子E2相关因子2可以调控双硫死亡相关蛋白溶质载体家族7成员11和Trx系统[69],因此,核因子E2相关因子2是否是脑缺血后双硫死亡的调控靶点值得进一步研究。
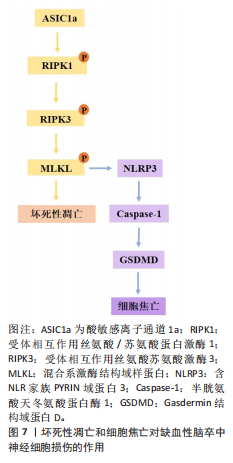
2.1.3 坏死性凋亡和细胞焦亡对缺血性脑卒中神经细胞损伤的作用机制 坏死性凋亡是调节性死亡的最典型形式,它由一系列激酶介导:受体相互作用丝氨酸/苏氨酸蛋白激酶1、受体相互作用丝氨酸苏氨酸激酶3和混合系激酶结构域样假激酶,在接受坏死性凋亡诱导的刺激后,受体相互作用丝氨酸/苏氨酸蛋白激酶1磷酸化并激活受体相互作用丝氨酸苏氨酸激酶3和混合系激酶结构域样假激酶,形成坏死体的复合物,坏死体引起质膜破坏和细胞裂解,诱导细胞发生坏死性凋亡。死亡受体肿瘤坏死因子α受体1、肿瘤坏死因子受体2、肿瘤坏死因子受体超家族成员6、肿瘤坏死因子相关凋亡诱导配体受体1和肿瘤坏死因子相关凋亡诱导配体受体2通过激酶的下游参与坏死性凋亡的启动[76]。缺血性脑卒中时,脑组织血流量减少导致腺嘌呤核苷三磷酸水平迅速下降,使离子泵失效和随后的膜电位耗散,这种破坏会引发不受控制的钙离子流入引起细胞肿胀,最终导致坏死性凋亡[77]。研究表明,受体相互作用丝氨酸/苏氨酸蛋白激酶1等激酶参与死亡受体激活后缺血性脑卒中的发病机制,用受体相互作用丝氨酸/苏氨酸蛋白激酶1特异性抑制剂坏死抑素1、GSK963和5-(3’,5’-二甲氧
基苯甲撑基)-2-硫代-咪唑-4-酮治疗可显著减少脑梗死体积和神经功能缺损评分,降低缺血诱导的神经炎症和活性氧的产生[78]。在大鼠和人的缺血性脑卒中脑组织中,受体相互作用丝氨酸/苏氨酸蛋白激酶1、混合系激酶结构域样假激酶和磷酸化混合系激酶结构域样假激酶的蛋白水平均显著升高,在体内大脑中动脉闭塞模型和体外糖氧剥夺/复氧细胞模型中,阻断上述信号通路可有效缓解缺血性脑卒中损伤[79]。通过与混合系激酶结构域样假激酶形成复合物,肿瘤坏死因子受体相关因子2保护小胶质细胞和神经元免受坏死性凋亡,敲除肿瘤坏死因子受体相关因子2会增加脑梗死体积,并通过坏死性凋亡机制促进细胞死亡[80]。在大脑中动脉闭塞小鼠模型中,环指蛋白216在神经元和小胶质细胞中被显著诱导,敲低环指蛋白216后可加速神经细胞死亡、增加小胶质细胞活性、促进炎性因子的释放,扩大脑梗死区域[81]。转化生长因子β激活激酶1在受体相互作用丝氨酸/苏氨酸蛋白激酶1的多个位点磷酸化中起重要作用,转化生长因子β激活激酶1在缺血性脑卒中中迅速激活;在从早期坏死性凋亡到细胞凋亡的过程中,小胶质细胞/浸润的巨噬细胞和神经元谱系中转化生长因子β激活激酶1的条件性敲除加剧了缺血性损伤后的神经元死亡和神经炎症[82]。酸敏感离子通道1a通过募集受体相互作用丝氨酸/苏氨酸蛋白激酶1到其C末端来介导酸性神经元坏死,而独立于其离子传导功能;酸敏感离子通道1a的N端与其CT相互作用形成自抑制,在静息条件下阻止受体相互作用丝氨酸/苏氨酸蛋白激酶1的募集/激活。在实验脑卒中模型中,酸敏感离子通道1a通过将受体相互作用丝氨酸/苏氨酸蛋白激酶1募集到酸敏感离子通道1a的C端并介导受体相互作用丝氨酸/苏氨酸蛋白激酶1激活,显著诱导并参与受体相互作用丝氨酸/苏氨酸蛋白激酶1的磷酸化[83]。ZHU等[84]的研究表明,受体相互作用丝氨酸/苏氨酸蛋白激酶1/受体相互作用丝氨酸苏氨酸激酶3/混合系激酶结构域样假激酶通路的激活,通过增强血管内皮生长因子D/血管内皮生长因子受体3信号传导促进星形胶质细胞增生和神经胶质细胞瘢痕形成,坏死抑素1可通过抑制上述信号通路减少星形胶质细胞增生和神经胶质细胞瘢痕形成,减轻脑损伤。小胶质细胞衍生的肿瘤坏死因子α及其受体肿瘤坏死因子α受体1介导内皮细胞坏死性凋亡,加重缺血性脑卒中后的血脑屏障破坏[85]。以上结果表明,参与缺血性脑卒中坏死性凋亡的细胞涉及神经元、星形胶质细胞、小胶质细胞和内皮细胞等多种脑细胞。
焦亡是于2001年发现的一种由炎症系统激活开始的细胞死亡[86],特征是细胞肿胀、膜破裂及炎症小体激活后的炎症递质释放[87]。导致细胞焦亡的分子级联反应始于模式识别受体检测缺血相关的危险信号,随后激活炎性小体复合物。Nod样受体家族是一种多蛋白复合物,它由含NLR家族Pyrin域蛋白3、凋亡相关斑点样蛋白和半胱氨酸蛋白酶1组成[88]。经典的焦亡途径是由含NLR家族Pyrin域蛋白3炎性小体作用激活半胱氨酸蛋白酶1触发的。脑缺血再灌注导致含NLR家族Pyrin域蛋白3激活,促使半胱氨酸蛋白酶1前体蛋白转化为半胱氨酸蛋白酶1,然后裂解Gasdermin结构域蛋白D在细胞膜上形成孔,释放炎症因子白细胞介素1β和白细胞介素18导致细胞死亡[89]。缺血性脑卒中后,炎症小体、Gasdermin结构域蛋白D、半胱天冬酶和炎症因子水平升高,通过介导细胞焦亡而加剧脑损伤[90]。在小鼠脑缺血再灌注模型中,促炎消退介质Maresin1通过抑制半胱氨酸蛋白酶3/Gasdermin结构域蛋白E介导的焦亡来减轻神经炎症[91]。星形胶质细胞衍生的外泌体miR-378a-5p通过调节含NLR家族Pyrin域蛋白3介导的细胞焦亡来减轻脑缺血性神经炎症[92]。电针可通过抑制小胶质细胞RAS同源基因家族成员A/含Pyrin域蛋白/Gasdermin结构域蛋白D信号通路改善脑缺血再灌注损伤[93]。高迁移率族蛋白B1是一种外细胞释放且独特的核蛋白,是损伤相关分子模式,它可以被含NLR家族Pyrin域蛋白3炎性小体检测。脑室内给药毛蕊异黄酮可抑制高迁移率族蛋白B1依赖性细胞焦亡,从而减轻大鼠脑缺血再灌注损伤[94]。
然而也有研究表明,Gasdermin结构域蛋白D在大脑中动脉闭塞和中药解毒活血汤治疗组大鼠脑梗死周围和对侧健康大脑半球中高度表达,暗示在脑缺血期间细胞焦亡可能不完全是促炎性损伤,Gasdermin结构域蛋白D也可能对脑缺血存在潜在的保护机制[95]。另有研究表明,贝拉卡萨(一种特异性半胱氨酸蛋白酶1抑制剂)通过抑制晚期糖基化终产物/丝裂原活化蛋白激酶通路的受体改善缺血诱导的焦亡激活,恢复血脑屏障的通透性[96]。皮树脂醇作为一种新型的过氧化物酶体增殖物激活受体γ共激活因子1α激活剂,可以通过促进过氧化物酶体增殖物激活受体α/谷草酰乙酸转氨酶1轴来减轻缺血性脑卒中脑微血管内皮细胞的焦亡[97],说明内皮细胞的焦亡可进一步促进缺血诱导的血脑屏障功能障碍。
坏死性凋亡的重要参与者混合系激酶结构域样假激酶,也与含NLR家族Pyrin域蛋白3炎症小体的激活有关,突出表现在免疫反应和细胞死亡机制研究的多个方面作用[98]。研究表明,混合系激酶结构域样假激酶在某些刺激下可直接激活含NLR家族Pyrin域蛋白3炎性体,导致成熟白细胞介素1β和白细胞介素18细胞因子释放,诱导细胞焦亡[99]。混合系激酶结构域样假激酶还可以通过促进受损线粒体释放线粒体脱氧核糖核酸间接激活含NLR家族Pyrin域蛋白3炎性体,这种线粒体脱氧核糖核酸的释放触发了含NLR家族Pyrin域蛋白3炎性体的组装和随后的焦亡诱导[100]。因此,靶向这些神经元焦亡的调控信号通路可能是治疗缺血性脑卒中的一种新的治疗策略(图7)。
2.1.4 中性粒细胞胞外陷阱相关死亡对缺血性脑卒中神经细胞损伤的作用机制 中性粒细胞胞外陷阱于1996年首次发现,是一种由中性粒细胞释放的细胞结构,用于捕获和杀死细菌、真菌和其他病原体,这些陷阱由脱氧核糖核酸纤维和蛋白质组成,形成一种网状结构。当活化的中性粒细胞将解聚的脱氧核糖核酸片段释放到细胞外间隙时,形成中性粒细胞胞外陷阱,这个过程称为中性粒细胞胞外陷阱相关死亡(图8)[101]。过量的中性粒细胞胞外陷阱可能导致血脑屏障崩溃、压倒性的神经炎症和神经元损伤。新生儿中性粒细胞胞外陷阱抑制肽预防中性粒细胞胞外陷阱形成,在不改变中性粒细胞向大脑运输的情况下,可减少健康小鼠以及患有合并症小鼠的缺血性脑卒中脑损伤[102]。中性粒细胞胞外陷阱释放各种细胞毒性蛋白酶,如弹性蛋白酶和髓过氧化物酶,这些蛋白激活干扰素基因刺激因子依赖性的Ⅰ型
干扰素β的产生,破坏内皮细胞及血脑屏障,阻碍脑卒中后的血管重建和修复[103]。缺血性脑卒中转录因子特异性蛋白1表达增加可导致冷诱导RNA结合蛋白表达升高并从神经元释放,随后与中性粒细胞相互作用并促进中性粒细胞胞外陷阱形成,导致脑内皮屏障破坏和脑水肿[104]。加载人参皂苷Rg1的囊泡可增强小胶质细胞对中性粒细胞的清除能力,减少中性粒细胞胞外陷阱的释放,改善缺血性脑卒中后的微环境,促进血管重塑和功能恢复[105]。靶向造血祖细胞激酶1缺失可抑制外周中性粒细胞过度激活和中性粒细胞胞外陷阱聚集,逐渐缓解缺血性脑卒中后的全身炎症及神经功能损害[106]。P-选择素/P-选择素糖蛋白配体1作为脑缺血性损伤的新机制,调节中性粒细胞胞外陷阱相关死亡[107]。P-选择素通过P-选择素糖蛋白配体1信号转导激活白细胞,促进肽酰基精氨酸脱亚氨酶4的表达,肽酰基精氨酸脱亚氨酶4可通过将带正电荷的精氨酸残基转换为中性瓜氨酸残基来修饰蛋白质,该过程称为瓜氨酸化,瓜氨酸化会触发中性粒细胞胞外陷阱。研究发现,大脑中动脉闭塞鼠脑组织肽酰基精氨酸脱亚氨酶4水平显著升高,肽酰基精氨酸脱亚氨酶4抑制剂BB-CL-酰胺鼻内给药可显著降低梗死体积,并在大脑中动脉闭塞后24 h改善神经系统和功能障碍,在除急性至亚急性期外具有显著的中性粒细胞胞外陷阱相关死亡抑制作用[108]。中性粒细胞胞外陷阱在血小板激活、组织因子依赖性凝血激活和内皮细胞扰动等血栓形成过程中发挥关键作用,
中性粒细胞胞外陷阱与不良结局和再灌注抗性有关,会导致血栓形成,并稳定这些血栓防止其降解[109]。血小板衍生的高迁移率族蛋白B1在血栓性脑卒中模型中激活中性粒细胞,并通过TOLL样受体4依赖性方式诱导中性粒细胞胞外陷阱相关死亡的发生[110]。脱氧核糖核酸酶Ⅰ通过降低血浆和皮质中性粒细胞胞外陷阱相关死亡水平来减少脑动脉闭塞后24 h的梗死体积,并改善神经功能结果[111]。靶向髓特异性整合素α 9通过限制缺血后/再灌注血栓炎症改善了预先存在合并症模型的短期和长期脑卒中结局[112]。脂氧素A4通过甲基转移酶样3的叉头框P2基因依赖性调节抑制中性粒细胞胞外陷阱形成,从而减轻缺血性脑卒中的脑损伤[113]。在缺血性脑卒中,中性粒细胞弹性酶靶向仿生纳米平台可通过抑制中性粒细胞胞外陷阱介导的黑色素瘤缺乏因子2炎性体激活来实现神经血管重构[114]。脑边界源的趋化因子配体2阳性中性粒细胞扩张可能会通过释放缺失性衰减网络来损害血管再生[115]。有研究表明,在佛波酯刺激的人中性粒细胞中,用苄丝肼处理可减少中性粒细胞胞外陷阱,防止与佛波酯刺激的人中性粒细胞共培养中人神经母细胞瘤细胞SH-SY5Y的死亡,同时苄丝肼能够减少脑卒中后中性粒细胞浸润到脑实质中,起到神经保护作用[116]。
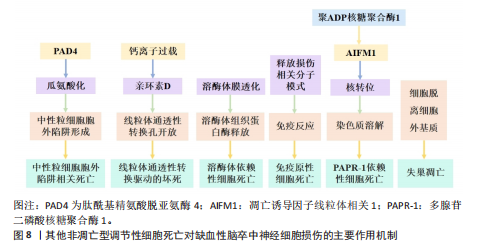
2.1.5 其他非凋亡型调节性细胞死亡对缺血性脑卒中神经细胞损伤的作用机制 除上述缺血性脑卒中常见的非凋亡型调节性细胞死亡亚型外,还有一些其他新兴的细胞死亡途径,包括溶酶体依赖性细胞死亡、免疫原性细胞死亡、聚ADP核糖聚合酶1依赖性细胞死亡、线粒体通透性转换驱动的坏死和失巢凋亡。2003年发现了线粒体通透性转换驱动的坏死,这是一种具有钙离子过载、亲环素D激活、线粒体通透性转换孔开放和线粒体膜去极化特征的调节性细胞死亡[117]。线粒体通透性转换孔是由细胞内钙离子浓度过高触发的在线粒体内膜上形成的孔[118]。在再灌注过程中,神经元线粒体中的钙超负荷和氧化应激导致线粒体通透性转换孔过度开放,发生氧化磷酸化解偶联、线粒体肿胀和线粒体外膜破裂,使神经元和小胶质细胞释放大量活性氧,促进神经细胞死亡[119]。亲环素D是线粒体基质中的肽基脯氨酸顺式反式异构酶,是唯一确认的线粒体通透性转换孔成分,根据它对钙离子的敏感性介导线粒体通透性转换孔开放[120]。亲环素D敲除后,大脑中动脉闭塞小鼠脑梗死面积显著减小、脑缺血/缺氧损伤恢复,早期细胞色素C从线粒体通透性转换孔向胞浆的渗漏可诱导神经细胞死亡[121]。
血小板中亲环素D缺失小鼠脑缺血再灌注损伤后的中性粒细胞和血小板-中性粒细胞聚集减少,脑血流明显增强,神经和运动功能改善,脑死面积减少,表明亲环素D是线粒体通透性转换孔介导缺血性脑卒中坏死的重要介质[122]。糖原合成酶激酶3β通过与亲环素D相互作用从细胞质转移到线粒体,诱导线粒体通透性转换孔开放,抑制糖原合成酶激酶3β水平可抑制神经损伤,减轻大脑中动脉闭塞小鼠模型中的线粒体功能障碍,减少脑梗死体积[78]。肿瘤抑制蛋白p53在线粒体基质中积累,并通过与亲环素D的物理相互作用触发线粒体通透性转换孔的开放,在缺血/再灌注期间会形成强大的肿瘤抑制蛋白p53-亲环素D复合物,降低肿瘤抑制蛋白p53水平可有效抑制缺血/再灌注造成的脑损伤[123]。淫羊藿苷通过抑制线粒体通透性转换孔开放来改善缺血性脑卒中期间的氧化应激损伤,淫羊藿苷在体内通过代谢产生淫羊藿素,淫羊藿素可通过抑制过氧化氢诱导的线粒体通透性转换孔过度开放来恢复线粒体稳态[124]。
溶酶体依赖性细胞死亡是一种由溶酶体膜损伤和溶酶体功能障碍引起的细胞死亡形式,通常由溶酶体膜透化引发,导致溶酶体组织蛋白酶释放触发细胞死亡[125]。从假手术组和永久性大脑中动脉阻塞(大脑中动脉闭塞)手术大鼠的大脑皮质中分离溶酶体和细胞质,发现大脑中动脉闭塞后24 h溶酶体区组织蛋白酶D和组织蛋白酶L水平降低,二者在胞质区的水平升高,大脑中动脉闭塞诱导溶酶体膜相关蛋白1水平显著降低,信号转导及转录激活蛋白3(酪氨酸激酶受体B的705位酪氨酸磷酸化)的增加促进大脑中动脉闭塞诱导的溶酶体膜透化[126]。暴露于糖氧剥夺/复氧条件后,原代SD大鼠皮质神经元或星形胶质细胞的溶酶体膜透化和组织蛋白酶B水平显著增加,坏死抑素1治疗可有效阻断大脑中动脉闭塞后6 h缺血皮质、糖氧剥夺诱导的神经元或星形胶质细胞活性组织蛋白酶B水平的增加,显著降低组织蛋白酶B水平;受体相互作用蛋白激酶1通过激活自噬-溶酶体途径促进缺血性脑卒中的神经元和星形胶质细胞死亡,坏死抑素1与组织蛋白酶B抑制剂联合治疗可进一步降低糖氧剥夺诱导的神经元或星形细胞死亡[127]。与野生型小鼠相比,组织蛋白酶B敲低小鼠海马区损伤面积显著减少,纹状体核心区存活神经元数量显著增加,大脑中动脉闭塞后小鼠神经功能显著改善[128]。
免疫原性细胞死亡是一种受监管的细胞死亡形式,它通过释放危险相关分子模式来激活免疫反应,包括钙网蛋白、腺嘌呤核苷三磷酸和高迁移率族蛋白B1[129]。免疫原性细胞死亡刺激导致原始抗原表位的产生和垂死细胞释放损伤相关分子模式,损伤相关分子模式可以与抗原呈递细胞(例如树突状细胞)结合,检测和吞噬死细胞抗原并将其递送到T细胞,以激发适应性免疫反应。免疫原性细胞死亡模式最初是在肿瘤治疗领域被发现和研究的,它在缺血性脑卒中中的研究还处于初步探索阶段。通过建立支持向量机模型,研究人员从差异表达的18个免疫原性细胞死亡相关基因预测缺血性脑卒中的发生,基因本体论结果显示,这些基因主要参与细胞因子产生的正调控、炎症反应的调节、白细胞介素1β产生的正调控、肿瘤坏死因子受体超家族结合和凋亡信号通
路[130],表明免疫原性细胞死亡可能在缺血性脑卒中预测中发挥作用。有研究确定了15个潜在的免疫原性细胞死亡调节因子和一个列线图模型,该模型能够准确预测缺血性脑卒中的患病率,这可能对缺血性脑卒中易感基因的临床筛查和病程监测具有重大意义,指导缺血性脑卒中患者诊断和个体化免疫疗法的发展[131]。免疫原性细胞死亡相关的枢纽基因丙酮酸脱氢酶激酶4、C-C基序趋化因子配体20和纤维蛋白,在老年女性缺血性脑卒中患者中具有良好的诊断价值,C-C基序趋化因子配体20和纤维蛋白与免疫细胞浸润程度显著相关,C-C基序趋化因子配体20与活化的CD4+ T细胞呈显著正相关;纤维蛋白与活化CD4+ T细胞和活化CD8+ T细胞等呈显著正相关,与巨噬细胞和中性粒细胞呈显著负相关,表明免疫原性细胞死亡相关基因可能对缺血性脑卒中个性化治疗提供新的诊断和治疗靶点[132]。
聚ADP核糖聚合酶1依赖性细胞死亡是脑缺血再灌注损伤中一种重要的程序性细胞死亡类型,它依赖于聚(ADP-核糖)聚合酶活化和聚(ADP-核糖)的形成。缺血性脑卒中后,聚ADP核糖聚合酶1表达的增加触发了凋亡诱导因子的核转位,随后通过与组蛋白变体H2AX的相互作用诱导染色质裂解和半胱天冬酶非依赖性细胞死亡,这种由聚ADP核糖聚合酶/凋亡诱导因子通路介导的不依赖半胱氨酸蛋白酶的细胞死亡,被命名为聚ADP核糖聚合酶1依赖性细胞死亡[133]。氧化应激和钙超负荷已被确定为导致聚ADP核糖聚合酶1依赖性细胞死亡的重要因素[134]。有研究表明,甲羟孕酮通过直接与胞外信号调节激酶结合并稳定活性磷酸化的细胞外调节蛋白激酶,从而抑制凋亡诱导因子易位,进而有效抑制依赖性细胞死亡,减少局灶性脑缺血小鼠模型的神经元死亡[135]。黄芪通过调节聚ADP核糖聚合酶1/Tax1结合蛋白1介导的线粒体呼吸链复合物I,拮抗脑缺血/再灌注损伤中的聚ADP核糖聚合酶1依赖性细胞死亡[135]。臭氧通过上调核因子E2相关因子2或激活过氧化物酶体增殖物激活受体γ减轻过氧化氢诱导人神经母细胞瘤细胞SH-SY5Y的聚ADP核糖聚合酶1依赖性细胞死亡,通过使用钙螯合剂和活性氧抑制剂发现,活性氧直接诱导聚ADP核糖聚合酶1依赖性细胞死亡并促进细胞内钙升高[136]。芹菜素、4’-O-甲基巴伐哈酮[137]、白藜芦醇通过抑制聚ADP核糖聚合酶1/凋亡诱导因子通路改善了大脑中动脉闭塞大鼠脑梗死体积和神经功能缺损[138],减轻了脑水肿,并减少了聚ADP核糖聚合酶1依赖性细胞死亡和细胞凋亡[139]。藏红花素通过抑制烟酰胺腺嘌呤二核苷酸磷酸氧化酶2和保留线粒体己糖激酶I来拮抗缺血性脑卒中的聚ADP核糖聚合酶1依赖性细胞死亡[140]。短暂性局灶性脑缺血期间的急性高血糖会增加组织酸中毒,神经细胞经N-甲基-N-硝基-N-亚硝基胍暴露后发生酸中毒,促进聚(ADP-核糖)聚合物的形成,凋亡诱导因子核易位,导致钙离子信号逐渐增加,用聚ADP核糖聚合酶抑制剂DR2313治疗可抑制与高血糖大脑中动脉闭塞相关的聚(ADP-核糖)形成增加[141]。
失巢凋亡是细胞与细胞外基质分离而产生的一种独特形式细胞凋亡,在保护生物体方面起着至关重要的作用,可以防止分离的隔室重新黏附到替代底物上,从而导致异常增殖[142]。失巢凋亡相关标志物白细胞介素6、血清/糖皮质激素调节激酶1和周期素依赖性激酶抑制因子1A,在缺血性脑卒中男性患者中显著上调,与肥大细胞活化呈正相关,白细胞介素6和血清/糖皮质激素调节激酶1与树突状细胞静息和单核细胞呈负相关,嗅素4、丙酮酸脱氢酶激酶4、癌胚抗原相关细胞黏附分子6和白细胞介素6在缺血性脑卒中女性患者中与健康人有显著差异,白细胞介素6与肥大细胞活化和肥大细胞静息呈正负相关,嗅素4和癌胚抗原相关细胞黏附分子6与巨噬细胞M0呈强正相关,与巨噬细胞M2和自然杀伤细胞活化呈负相关[66]。在缺血性脑卒中患者中,浸润免疫细胞幼稚B细胞、嗜酸性粒细胞、M0巨噬细胞、M2巨噬细胞和静息肥大细胞等与失巢凋亡调节因子密切相关,在不同失巢凋亡簇中存在不同的免疫特征。基因本体论与基因组百科全书富集分析显示,失巢凋亡相关基因蛋白激酶B1、乳腺癌转移抑制因子1、线粒体肽酰-tRNA水解酶2、转录因子Dp1和Ⅰ型分裂蛋白转导素样增强子富集于碳水化合物消化吸收及Notch信号通路等途径,主要参与失巢凋亡和脱氧核糖核酸结合转录因子活性的调节,并与免疫浸润有显著相关性[143]。
缺血性脑卒中非凋亡型调节性细胞死亡的致病机制,见表1。
2.2 缺血性脑卒中中调节性死亡途径的相互作用 自噬通过复杂的方式与细胞死亡过程相互作用,具体取决于所涉及的特定细胞环境和信号通路。HOU等[144]证明铁蛋白的自噬降解促进了铁死亡,这一过程被称为铁蛋白自噬。LI等[145]在大脑中动脉闭塞小鼠模型中发现,介导铁蛋白自噬的选择性受体——核受体共激活因子4上调可促进神经元铁死亡,而核受体共激活因子4下调可通过阻断铁蛋

白自噬来保护神经元免于铁死亡;此外,泛素特异性肽酶14被鉴定为从核受体共激活因子4中切割泛素的去泛素酶,抑制泛素特异性肽酶14可降低核受体共激活因子4诱导的铁死亡,具有神经保护作用。铜离子通过与谷胱甘肽过氧化酶4的半胱氨酸残基C107和C148结合来诱导铁死亡,促进谷胱甘肽过氧化酶4与自噬受体Tax1结合蛋白1的结合,从而增加谷胱甘肽过氧化酶4的泛素化[146]。坏死性凋亡也可引起溶酶体依赖性死亡的发生,敲低受体相互作用丝氨酸/苏氨酸蛋白激酶1可防止糖氧剥夺诱导的星形胶质细胞损伤,抑制糖氧剥夺介导的星形胶质细胞溶酶体膜通透性以及大脑中动脉闭塞诱导的缺血大脑皮质星形胶质细胞溶酶体数量增加[147]。
铜通过腺苷酸活化蛋白激酶-哺乳动物雷帕霉素靶蛋白轴调节自噬水平,也直接结合并激活自噬相关激酶Unc-51样自噬激活激酶1/2。在脑缺血中,由铜驱动的自噬不仅可以对抗细胞损伤,还可以通过选择性降解抗凋亡或抗氧化因子谷胱甘肽过氧化酶4加剧神经元损失[148]。在短暂性大脑中动脉闭塞模型中,化合物23a可有效抑制受体相互作用丝氨酸/苏氨酸蛋白激酶1激酶活性,显著降低脑梗死体积并提高了神经功能评分,证明了对缺血半暗带组织中氧化应激、坏死性凋亡和铁死亡的多靶点作用[149]。线粒体是缺血性脑卒中中调节坏死之间交叉调节的核心介质,缺血性脑卒中引起的线粒体功能障碍表现为腺嘌呤核苷三磷酸缺乏、线粒体通透性转换孔开放、活性氧生成、钙超负荷和细胞色素C释放。亲环素D介导的线粒体通透性转换孔开放,对线粒体通透性转换孔介导的坏死、离子稳态破坏和活性氧生成至关重要,它与坏死性凋亡、铁死亡和聚ADP核糖聚合酶1依赖性细胞死亡密切相关[150]。在缺血再灌注损伤中,受体相互作用丝氨酸/苏氨酸蛋白激酶1和受体相互作用丝氨酸苏氨酸激酶3的上调导致亲环素D介导的线粒体通透性转换孔开放、钙离子内流和活性氧生成,而抑制亲环素D介导的线粒体通透性转换孔开放可以减轻缺血性脑卒中诱导的坏死性凋亡[151]。在缺血性脑卒中,长链酰基辅酶A合成酶4过表达、谷胱甘肽过氧化酶4抑制和谷胱甘肽下调导致脂氧合酶激活、钙离子内流、线粒体膜破裂和铁死亡抑制,有效减弱线粒体通透性转换孔开放引起的线粒体功能障碍[152]。此外,铁过载通过催化活性氧的产生并破坏脑卒中中的血脑屏障,进而导致膜过氧化物的过度产生、线粒体通透性转换孔开放和坏死性凋亡[153]。作为聚ADP核糖聚合酶1依赖性细胞死亡的机制之一,凋亡诱导因子显示出与细胞核中的受体相互作用丝氨酸苏氨酸激酶3相互作用并诱导脑出血小鼠坏死性凋亡的能力,而环孢霉素A干预通过抑制线粒体通透性转换孔开放来阻断受体相互作用丝氨酸苏氨酸激酶3-凋亡诱导因子复合物[154]。铁死亡和中性粒细胞胞外陷阱相关死亡在缺血性脑卒中损伤中可能发挥关联作用,谷胱甘肽的直接前体 γ-谷氨酰半胱氨酸治疗可有效减轻血脑屏障损伤、减少中性粒细胞浸润、抑制中性粒细胞胞外陷阱的释放,最终改善缺血性损伤[155]。细胞焦亡同样在促进中性粒细胞胞外陷阱形成、增加缺血性脑卒中神经损伤中发挥作用,含NLR家族Pyrin域蛋白3炎性体是中性粒细胞胞外陷阱和血栓形成的介质,抑制含NLR家族Pyrin域蛋白3可能具有减轻血栓事件的潜力[156]。
2.3 非凋亡型调节性细胞死亡在缺血性脑卒中中的临床应用 多数治疗缺血性脑卒中的非凋亡型调节性细胞死亡相关靶向药物还处于临床前研究。右美托咪定是一种选择性α2-肾上腺素能受体激动剂,是重症监护病房和麻醉中有用的镇静剂,研究表明右美托咪定在体内外对脑缺血再灌注损伤均具有保护作用,它可以通过低氧诱导因子1α抑制神经元自噬,增加氧-葡萄糖剥夺模型中原代培养的神经元活力、抑制神经细胞凋亡[157]。右美托咪定还可通过结节硬化2蛋白/哺乳动物雷帕霉素靶蛋白通路诱导星形胶质细胞自噬,防止氧糖剥夺诱导的细胞损伤[158]。
在药理学上,哺乳动物雷帕霉素靶蛋白复合体1可被雷帕霉素抑制,雷帕霉素是一种广泛用作免疫抑制剂的大环内酯类化合物,它的主要临床应用是移植后免疫抑制。雷帕霉素可穿过细胞膜和血脑屏障,因此,雷帕霉素可以通过多种机制保护血脑屏障,从而减少啮齿动物模型中局灶性脑缺血再灌注后的脑水肿,诱导自噬,减少内皮细胞死亡[159]。组织型纤溶酶原激活剂是缺血性脑卒中临床治疗中最重要的溶栓药物,具有神经保护作用,它诱导的FUN14结构域包含蛋白1依赖性线粒体自噬能够保护神经元免受脑缺血再灌注损伤[160]。通过组织型纤溶酶原激活剂溶栓可增加血栓栓塞性脑卒中小鼠血清游离胰岛素样生长因子1水平,减少脑缺血后有害自噬[161]。在大脑中动脉闭塞小鼠模型中,再灌注后给予组织型纤溶酶原激活剂治疗4 h后可引起出血并发症;而再灌注后给予抗高迁移率族蛋白B1抗体治疗4 h可抑制高迁移率族蛋白B1活性、减少梗死体积和肿胀、改善神经功能损伤和运动协调,无出血性并发症;此外,抗高迁移率族蛋白B1抗体可抑制脑缺血后继发性炎症反应的信号通路[162]。在多种缺血性和出血性脑卒中动物模型中,铁螯合剂甲磺酸去铁胺可通过抑制铁诱导的羟基自由基形成发挥神经保护作用。例如,经甲磺酸去铁胺治疗3 d后,脑卒中患者血清中过氧化物水平降低,总自由基捕获抗氧化能力水平升高[163]。羟氯喹是一种众所周知的抗疟疾和抗炎药物,通过抑制细胞焦亡在缺血性脑卒中具有潜在的神经保护作用。体内外实验表明,羟氯喹可以改善运动功能并减少脑梗死体积,凸显它作为神经保护剂的潜力[164]。虽然以上药物已经在临床上广泛应用,但是否是通过自噬、铁死亡及细胞焦亡等途径发挥缺血神经细胞保护作用还需进一步探索。
与缺血性脑卒中患者相比,疫苗诱导的免疫性血栓性血小板减少症患者血栓中的肽酰基精氨酸脱亚氨酶4表达更明显、中性粒细胞浸润和中性粒细胞胞外陷阱的形成更广泛[165]。急性缺血性脑卒中患者在接受非增强电子计算机断层扫描扫描和多模态磁共振成像后接受了机械支架和手动抽吸血栓切除术,并从脑动脉取出血栓栓塞,所有回收的血栓都显示出丰富的中性粒细胞胞外陷阱形成,在患者血栓富血小板区、富红细胞区和富血小板区交界处均显示中性粒细胞胞外陷阱[166]。目前有部分靶向调节性细胞死亡途径的相关蛋白被应用于缺血性脑卒中检查预测中,为未来缺血性脑卒中的治疗和预防提供了新的研究方向。
2.4 非凋亡型调节性细胞死亡应用于缺血性脑卒中领域的发展历程 见表2。

| [1] AJOOLABADY A, WANG S, KROEMER G, et al. Targeting autophagy in ischemic stroke: From molecular mechanisms to clinical therapeutics. Pharmacol Ther. 2021;225:107848. [2] LI Y, WU J, DU F, et al. Neuroprotective Potential of Glycyrrhizic Acid in Ischemic Stroke: Mechanisms and Therapeutic Prospects. Pharmaceuticals. 2024;17(11):1493. [3] LI C, JIANG M, FANG ZT, et al. Current evidence of synaptic dysfunction after stroke: Cellular and molecular mechanisms. CNS Neurrosci Ther. 2024; 30(5):e14744. [4] SMITH M, REDDY U, ROBBA C, et al. Acute ischaemic stroke: challenges for the intensivist. Intens Care Med. 2019;45(9):1177-1189. [5] COTTER TG. Apoptosis and cancer: the genesis of a research field. Nat rev Cancer. 2009;9(7): 501-507. [6] KOREN E, FUCHS Y. Modes of Regulated Cell Death in Cancer. Cancer discov. 2021;11(2):245-265. [7] VITALE I, PIETROCOLA F, GUILBAUD E, et al. Apoptotic cell death in disease-Current understanding of the NCCD 2023. Cell Death Differ. 2023;30(5):1097-1154. [8] 张思琦,郭朝瑞,董志强,等.调节性细胞死亡在心肌缺血再灌注损伤中的作用研究进展[J].中南药学,2024,22(7):1838-1844. [9] JIN X, JIN W, TONG L, et al. Therapeutic strategies of targeting non-apoptotic regulated cell death (RCD) with small-molecule compounds in cancer. Acta Pharm Sin B. 2024;14(7):2815-2853. [10] TIAN X, LI X, PAN M, et al. Progress of Ferroptosis in Ischemic Stroke and Therapeutic Targets. Cell Mol Neurobiol. 2024;44(1):25. [11] QIN R, XU W, QIN Q, et al. Identification of NETs-related genes as diagnostic biomarkers in ischemic stroke using RNA sequencing and single-cell analysis. Mamm Genome. 2025;36(2):651-664. [12] 邱伟彤,饶娆,韩永升.电针血清通过调控GSK-3β/Nrf2信号轴抑制OGD/R诱导的海马神经元铁死亡机制研究[J/OL].皖南医学院学报,1-11[2025-09-04].https://link.cnki.net/urlid/34.1068.R.20250609.1036.002. [13] KONG D, ZHU J, LIU Q, et al. Mesenchymal stem cells protect neurons against hypoxic-ischemic injury via inhibiting parthanatos, necroptosis, and apoptosis, but not autophagy. Cell Mol Neurobiol. 2017;37(2):303-313. [14] ZHANG X, YAN H, YUAN Y, et al. Cerebral ischemia-reperfusion-induced autophagy protects against neuronal injury by mitochondrial clearance. Autophagy. 2013;9(9):1321-1333. [15] GABRYEL B, KOST A, KASPROWSKA D, et al. AMP-activated protein kinase is involved in induction of protective autophagy in astrocytes exposed to oxygen-glucose deprivation. Cell Biol Int. 2017; 41(8):928-931. [16] JIANG T, YU J T, ZHU X C, et al. Ischemic preconditioning provides neuroprotection by induction of AMP-activated protein kinase-dependent autophagy in a rat model of ischemic stroke. Mol Neurobiol. 2015;51(1):220-229. [17] MELANIS K, STEFANOU MI, THEMISTOKLIS KM, et al. mTOR pathway - a potential therapeutic target in stroke. Ther Adv Neurol Diso. 2023; 16:17562864231187770. [18] LU X, ZHANG J, DING Y, et al. Novel Therapeutic Strategies for Ischemic Stroke: Recent Insights into Autophagy. Oxid Med Cell Longev. 2022; 2022:3450207. [19] WANG H, LIU Q, ZHANG X. C1q/tumor necrosis factor-related protein-1 attenuates microglia autophagy and inflammatory response by regulating the Akt/mTOR pathway. Life Sci. 2020; 256:117992. [20] XU D, WEN M, LEBOHANG ANESU B, et al. Genetically-engineered Salmonella typhimurium expressing FGF21 promotes neurological recovery in ischemic stroke via FGFR1/AMPK/mTOR pathway. J Neuroinflammation. 2025;22(1):170. [21] PANG Q, WU Y, JIN T, et al. bFGF Knockdown Inhibits mTOR Signaling by Suppressing Caveolin-1 and Aggravates Cognitive Damage After Arterial Ischemic Brain Injury in Juvenile Rats. Mol Neurobiol. 2025. doi: 10.1007/s12035-025-05108-9. [22] TANG L, ZHANG W, LIAO Y, et al. Autophagy: a double-edged sword in ischemia-reperfusion injury. Cell Mol Biol Lett. 2025;30(1):42. [23] HUA Y, ZHAI Y, WANG G, et al. Tong-Qiao-Huo-Xue decoction activates PI3K/Akt/mTOR pathway to reduce BMECs autophagy after cerebral ischemia/reperfusion injury. J Ethnopharmacol. 2022;298:115585. [24] YANG L, LI X, ZHAO Y, et al. NAT10 inhibition alleviates astrocyte autophagy by impeding ac4C acetylation of Timp1 mRNA in ischemic stroke. Acta Pharm Sin B. 2025;15(5):2575-2592. [25] SHI R, WENG J, ZHAO L, et al. Excessive autophagy contributes to neuron death in cerebral ischemia. CNS Neurosci Ther. 2012;18(3):250-260. [26] XU S, HUANG P, YANG J, et al. Calycosin alleviates cerebral ischemia/reperfusion injury by repressing autophagy via STAT3/FOXO3a signaling pathway. Phytomedicine. 2023;115: 154845. [27] WU S, YIN Y, DU L. FUS aggregation following ischemic stroke favors brain astrocyte activation through inducing excessive autophagy. Exp Neurol. 2022;355:114144. [28] ZHONG WJ, YANG XS, ZHOU H, et al. Role of Mitophagy in the Pathogenesis of Stroke: From Mechanism to Therapy. Oxid Med Cell Longev. 2022;2022:6232902. [29] SHEN Z, ZHENG Y, WU J, et al. PARK2-dependent mitophagy induced by acidic postconditioning protects against focal cerebral ischemia and extends the reperfusion window. Autophagy. 2017;13(3):473-485. [30] WANG H, CHEN S, ZHANG Y, et al. Electroacupuncture ameliorates neuronal injury by Pink1/Parkin-mediated mitophagy clearance in cerebral ischemia-reperfusion. Nitric Oxide-biol Ch. 2019;91:23-34. [31] WANG Y, XIE D, MA S, et al. Beta-asarone alleviated cerebral ischemia/reperfusion injury by targeting PINK1/Parkin-dependent mitophagy. Eur J Pharmacol. 2025;177831. [32] YU S, ZHENG S, LENG J, et al. Inhibition of mitochondrial calcium uniporter protects neurocytes from ischemia/reperfusion injury via the inhibition of excessive mitophagy. Neurosci Lett. 2016;628:24-29. [33] LEI L, YANG S, LU X, et al. Research Progress on the Mechanism of Mitochondrial Autophagy in Cerebral Stroke. Front Aging Neurosci. 2021; 13:698601. [34] YUAN Y, ZHENG Y, ZHANG X, et al. BNIP3L/NIX-mediated mitophagy protects against ischemic brain injury independent of PARK2. Autophagy. 2017;13(10):1754-1766. [35] SHI RY, ZHU SH, LI V, et al. BNIP3 interacting with LC3 triggers excessive mitophagy in delayed neuronal death in stroke. CNS Neurosci Ther. 2014;20(12):1045-1055. [36] FORCINA GC, DIXON SJ. GPX4 at the Crossroads of Lipid Homeostasis and Ferroptosis. Proteomics. 2019;19(18):e1800311. [37] ZHANG Y, SWANDA RV, NIE L, et al. mTORC1 couples cyst(e)ine availability with GPX4 protein synthesis and ferroptosis regulation. Nat Commun. 2021;12(1):1589. [38] LIU D, ZHU Y. Unveiling Smyd-2’s Role in Cytoplasmic Nrf-2 Sequestration and Ferroptosis Induction in Hippocampal Neurons After Cerebral Ischemia/Reperfusion. Cells. 2024;13(23):1969. [39] HOMMA T, KOBAYASHI S, FUJII J. Cysteine preservation confers resistance to glutathione-depleted cells against ferroptosis via CDGSH iron sulphur domain-containing proteins (CISDs). Free Radical Res. 2020;54(6):397-407. [40] SONG J, PARK J, OH Y, et al. Glutathione suppresses cerebral infarct volume and cell death after ischemic injury: involvement of FOXO3 inactivation and Bcl2 expression. Oxid Med Cell Longev. 2015;2015:426069. [41] TUO QZ, MASALDAN S, SOUTHON A, et al. Characterization of Selenium Compounds for Anti-ferroptotic Activity in Neuronal Cells and After Cerebral Ischemia-Reperfusion Injury. Neurotherapeutic. 2021;18(4):2682-2691. [42] GOWTHAM A, CHAUHAN C, RAHI V, et al. An update on the role of ferroptosis in ischemic stroke: from molecular pathways to Neuroprotection. Expert Opin Ther Tar. 2024; 28(12):1149-1175. [43] FAN G, LIU M, LIU J, et al. The initiator of neuroexcitotoxicity and ferroptosis in ischemic stroke: Glutamate accumulation. Front Mol Neurosci. 2023;16:1113081. [44] DOLL S, PRONETH B, TYURINA YY, et al. ACSL4 dictates ferroptosis sensitivity by shaping cellular lipid composition. Nat Chem Biol. 2017; 13(1):91-98. [45] LIU M, XU Z, WANG L, et al. Cottonseed oil alleviates ischemic stroke injury by inhibiting the inflammatory activation of microglia and astrocyte. J Neuroinflamm. 2020;17(1):270. [46] WANG K, YANG L, LI Q, et al. Long-Term Alcohol Exposure Aggravates Ischemic Stroke-Induced Damage by Promoting Pericyte NLRP3 Inflammasome Activation via Pre-Activating the TLR4/NF-κB Pathway in Rats. J Inflamm Res. 2024; 17:4791-4810. [47] ZHAO J, MA M, LI L, et al. Oxysophoridine protects against cerebral ischemia/reperfusion injury via inhibition of TLR4/p38MAPK‑mediated ferroptosis. Mol Med Rep. 2023;27(2):44. [48] MA Y, WANG X, LI Y, et al. Mechanisms Associated with Mitophagy and Ferroptosis in Cerebral Ischemia-reperfusion Injury. J Integr Neurosci. 2025;24(3):26458. [49] XU S, LI X, LI Y, et al. Neuroprotective effect of Dl-3-n-butylphthalide against ischemia-reperfusion injury is mediated by ferroptosis regulation via the SLC7A11/GSH/GPX4 pathway and the attenuation of blood-brain barrier disruption. Front In Aging Neurosci. 2023;15:1028178. [50] KOPPULA P, ZHUANG L, GAN B. Cystine transporter SLC7A11/xCT in cancer: ferroptosis, nutrient dependency, and cancer therapy. Protein Cell. 2021;12(8):599-620. [51] DOMERCQ M, SZCZUPAK B, GEJO J, et al. PET Imaging with [(18)F]FSPG Evidences the Role of System xc(-) on Brain Inflammation Following Cerebral Ischemia in Rats. Theranostics. 2016; 6(11):1753-1767. [52] LIU H, YUE Q, ZHANG W, et al. Xinglou Chengqi Decoction Protects against Cerebral Ischemia/Reperfusion Injury by Inhibiting Ferroptosis via SLC7A11/GPX4 Signaling. Adv Biol (Weinh). 2024;8(11):e2400180. [53] HU X, BAO Y, LI M, et al. The role of ferroptosis and its mechanism in ischemic stroke. Exp Neurol. 2023;372:114630. [54] ZHAO Y, LIU Y, XU Y, et al. The Role of Ferroptosis in Blood-Brain Barrier Injury. Cell Mol Neurobiol. 2023;43(1):223-236. [55] WU Y, JIAO H, YUE Y, et al. Ubiquitin ligase E3 HUWE1/MULE targets transferrin receptor for degradation and suppresses ferroptosis in acute liver injury. Cell Death Differ. 2022;29(9):1705-1718. [56] ABE C, MIYAZAWA T, MIYAZAWA T. Current Use of Fenton Reaction in Drugs and Food. Molecules. 2022;27(17):5451. [57] HU X, LI Y, CHENG P, et al. Serum Level of Transferrin Unique Peptide Is Decreased in Patients With Acute Ischemic Stroke. Front Neurol. 2021;12:619310. [58] LUO Q, ZHENG J, FAN B, et al. Enriched environment attenuates ferroptosis after cerebral ischemia/reperfusion injury by regulating iron metabolism. Brain Res Bull. 2023;203:110778. [59] ZHU Z, SONG M, REN J, et al. Copper homeostasis and cuproptosis in central nervous system diseases. Cell Death Dis. 2024; 15(11):850. [60] ATKIN MA, GASPER A, ULLEGADDI R, et al. Oxidative susceptibility of unfractionated serum or plasma: response to antioxidants in vitro and to antioxidant supplementation. Clin Chem. 2005;51(11):2138-2144. [61] JIANG Y, WANG LP, DONG X H, et al. Trace Amounts of Copper in Drinking Water Aggravate Cerebral Ischemic Injury via Impairing Endothelial Progenitor Cells in Mice. CNS Neurosci Ther. 2015;21(8):677-680. [62] CUNNANE SC, TRUSHINA E, MORLAND C, et al. Brain energy rescue: an emerging therapeutic concept for neurodegenerative disorders of ageing. Nat Rev Drug Discov. 2020;19(9):609-633. [63] GUO Q, MA M, YU H, et al. Dexmedetomidine enables copper homeostasis in cerebral ischemia/reperfusion via ferredoxin 1. Ann Med. 2023;55(1):2209735. [64] LIN MC, LIU CC, LIAO CS, et al. Neuroprotective Effect of Quercetin during Cerebral Ischemic Injury Involves Regulation of Essential Elements, Transition Metals, Cu/Zn Ratio, and Antioxidant Activity. Molecules. 2021;26(20):6128. [65] YANG S, LI X, YAN J, et al. Disulfiram downregulates ferredoxin 1 to maintain copper homeostasis and inhibit inflammation in cerebral ischemia/reperfusion injury. Sci Rports. 2024;14(1):15175. [66] CHEN W, CHEN Y, WU L, et al. Identification of cell death-related biomarkers and immune infiltration in ischemic stroke between male and female patients. Front Immunol. 2023;14:1164742. [67] QIN R, LIANG X, YANG Y, et al. Exploring cuproptosis-related molecular clusters and immunological characterization in ischemic stroke through machine learning. Heliyon. 2024; 10(17):e36559. [68] ZHENG P, ZHOU C, DING Y, et al. Disulfidptosis: a new target for metabolic cancer therapy. J Exp Clin Cancer Res. 2023;42(1):103. [69] CHANG J, LIU D, XIAO Y, et al. Disulfidptosis: a new target for central nervous system disease therapy. Front Neurosci. 2025;19:1514253. [70] RONGXING Q, LIJUAN H, WEI X, et al. Identification of disulfidptosis-related genes and analysis of immune infiltration characteristics in ischemic strokes. Math Biosci Eng. 2023;20(10): 18939-18959. [71] LIU S P, LIU C, XU B, et al. Disulfidptosis and its Role in Peripheral Blood Immune Cells after a Stroke: A New Frontier in Stroke Pathogenesis. Curr Neurovasc Res. 2024;20(5):608-622. [72] LIU S, WU Q, XU C, et al. Ischemic Postconditioning Regulates New Cell Death Mechanisms in Stroke: Disulfidptosis. Biomolecules. 2024;14(11):1390. [73] LI S, CHEN N, HE J, et al. NDUFA11 may be the disulfidptosis-related biomarker of ischemic stroke based on integrated bioinformatics, clinical samples, and experimental analyses. Front Neurosci. 2024;18:1505493. [74] XIAOGUANG L, KELLEN O, YILEI Z, et al. Cystine transporter regulation of pentose phosphate pathway dependency and disulfide stress exposes a targetable metabolic vulnerability in cancer. Nat Cell Biol. 2020;22(4):476-486. [75] ZHAO S, ZHUANG H, JI W, et al. Identification of Disulfidptosis-Related Genes in Ischemic Stroke by Combining Single-Cell Sequencing, Machine Learning Algorithms, and In Vitro Experiments. Neuromolecular Med. 2024;26(1):39. [76] YUAN J, AMIN P, OFENGEIM D. Necroptosis and RIPK1-mediated neuroinflammation in CNS diseases. Nat Rev Neurosci. 2019;20(1):19-33. [77] LI C, LUO Y, LI S. Mechanistic insights of neuronal death and neuroprotective therapeutic approaches in stroke. Neural Regen Res. 2025; 21(3):869-886. [78] REN K, PEI J, GUO Y, et al. Regulated necrosis pathways: a potential target for ischemic stroke. Burns Trauma. 2023;11:tkad016. [79] ZHANG YY, LIU WN, LI YQ, et al. Ligustroflavone reduces necroptosis in rat brain after ischemic stroke through targeting RIPK1/RIPK3/MLKL pathway. Naunyn Schmiedebergs Arch Pharmacol. 2019;392(9):1085-1095. [80] LI J, ZHANG J, ZHANG Y, et al. TRAF2 protects against cerebral ischemia-induced brain injury by suppressing necroptosis. Cell Death Dis. 2019; 10(5):328. [81] YUAN Z, YI-YUN S, HAI-YAN Y. Triad3A displays a critical role in suppression of cerebral ischemic/reperfusion (I/R) injury by regulating necroptosis. Biomed Pharmacother. 2020;128:110045. [82] NAITO MG, XU D, AMIN P, et al. Sequential activation of necroptosis and apoptosis cooperates to mediate vascular and neural pathology in stroke. Proc Natl Acad Sci U S A. 2020;117(9):4959-4970. [83] WANG JJ, LIU F, YANG F, et al. Disruption of auto-inhibition underlies conformational signaling of ASIC1a to induce neuronal necroptosis. Nat Commun. 2020;11(1):475. [84] ZHU YM, LIN L, WEI C, et al. The Key Regulator of Necroptosis, RIP1 Kinase, Contributes to the Formation of Astrogliosis and Glial Scar in Ischemic Stroke. Transl Stroke Res. 2021;12(6):991-1017. [85] CHEN AQ, FANG Z, CHEN XL, et al. Microglia-derived TNF-α mediates endothelial necroptosis aggravating blood brain-barrier disruption after ischemic stroke. Cell Death Dis. 2019;10(7):487. [86] COOKSON BT, BRENNAN MA. Pro-inflammatory programmed cell death. Trends Microbiol. 2001; 9(3):113-114. [87] ZHAO L, LI S, WANG X, et al. The AGEs/RAGE Signaling Pathway Regulates NLRP3-Mediated Neuronal Pyroptosis After MCAO Injury in Lepr-/- Obese Rats. J Inflamm Res. 2024;17:6935-6954. [88] HOU Y, YAN Z, WAN H, et al. A Combination of Astragaloside IV and Hydroxysafflor Yellow A Attenuates Cerebral Ischemia-Reperfusion Injury via NF-κB/NLRP3/Caspase-1/GSDMD Pathway. Brain Sci. 2024;14(8):781. [89] YE J, BI X, DENG S, et al. Hypoxanthine is a metabolic biomarker for inducing GSDME-dependent pyroptosis of endothelial cells during ischemic stroke. Theranostics. 2024;14(15):6071-6087. [90] LI L, SHI C, DONG F, et al. Targeting pyroptosis to treat ischemic stroke: From molecular pathways to treatment strategy. Int Immunopharmacol. 2024;133:112168. [91] ZHAO M, XIAN W, LIU W, et al. Maresin1 alleviates neuroinflammation by inhibiting caspase-3/ GSDME-mediated pyroptosis in mice cerebral ischemia-reperfusion model. J Stroke Cerebrovasc Dis. 2024;107789. [92] SUN R, LIAO W, LANG T, et al. Astrocyte-derived exosomal miR-378a-5p mitigates cerebral ischemic neuroinflammation by modulating NLRP3-mediated pyroptosis. Front Immunol. 2024;15:1454116. [93] FANG H, FAN LL, DING YL, et al. Pre-electroacupuncture Ameliorates Cerebral Ischemia-reperfusion Injury by Inhibiting Microglial RhoA/pyrin/GSDMD Signaling Pathway. Neurochem Res. 2024;49(11):3105-3117. [94] ZHANG Y, WU Q, PAN Y, et al. Intracerebroventricular calycosin attenuates cerebral ischemia-reperfusion injury in rats via HMGB1-dependent pyroptosis inhibition. Front Pharmacol. 2025;16:1596087. [95] ZHOU C, QIU SW, WANG FM, et al. Gasdermin D could be lost in the brain parenchyma infarct core and a pyroptosis-autophagy inhibition effect of Jie-Du-Huo-Xue decoction after stroke. Front Pharmacol. 2024;15:1449452. [96] LIANG Y, SONG P, CHEN W, et al. Inhibition of Caspase-1 Ameliorates Ischemia-Associated Blood-Brain Barrier Dysfunction and Integrity by Suppressing Pyroptosis Activation. Front Cell Neurosci. 2020;14:540669. [97] WANG Y, GUAN X, GAO CL, et al. Medioresinol as a novel PGC-1α activator prevents pyroptosis of endothelial cells in ischemic stroke through PPARα-GOT1 axis. Pharmacol Res. 2021;169: 105640. [98] FRANK D, VINCE JE. Pyroptosis versus necroptosis: similarities, differences, and crosstalk. Cell Death Differ. 2019;26(1):99-114. [99] SáNCHEZ-FERNáNDEZ A, SKOURAS DB, DINARELLO CA, et al. OLT1177 (Dapansutrile), a Selective NLRP3 Inflammasome Inhibitor, Ameliorates Experimental Autoimmune Encephalomyelitis Pathogenesis. Front Immunol. 2019;10:2578. [100] TIAN HY, LEI YX, ZHOU JT, et al. Insight into interplay between PANoptosis and autophagy: novel therapeutics in ischemic stroke. Front Mol Neurosci. 2024;17:1482015. [101] YIN N, WANG W, PEI F, et al. A Neutrophil Hijacking Nanoplatform Reprograming NETosis for Targeted Microglia Polarizing Mediated Ischemic Stroke Treatment. Adv Sci (Weinh). 2024;11(17):e2305877. [102] DENORME F, PORTIER I, RUSTAD JL, et al. Neutrophil extracellular traps regulate ischemic stroke brain injury. J Clin Invest. 2022;132(10): e154225. [103] WANG R, ZHU Y, LIU Z, et al. Neutrophil extracellular traps promote tPA-induced brain hemorrhage via cGAS in mice with stroke. Blood. 2021;138(1):91-103. [104] LI Z, SUN S, XIAO Q, et al. Neuron Derived Cold-Inducible RNA-Binding Protein Promotes NETs Formation to Exacerbate Brain Endothelial Barrier Disruption after Ischemic Stroke. Aging Dis. 2024; 16(1):520-539. [105] HU K, YE J, FAN P, et al. Targeting and reprogramming microglial phagocytosis of neutrophils by ginsenoside Rg1 nanovesicles promotes stroke recovery. Bioact Mater. 2025; 47:181-197. [106] ZHANG T, SUN Y, XIA J, et al. Targeting HPK1 inhibits neutrophil responses to mitigate post-stroke lung and cerebral injuries. EMBO Mol Med. 2025;17:1018-1040. [107] LI X, MA Y, WANG D. The role of P-selectin/PSGL-1 in regulating NETs as a novel mechanism in cerebral ischemic injury. Front Neurol. 2024;15: 1442613. [108] SEOL SI, OH SA, DAVAANYAM D, et al. Blocking peptidyl arginine deiminase 4 confers neuroprotective effect in the post-ischemic brain through both NETosis-dependent and -independent mechanisms. Acta Neuropathol Commun. 2025;13(1):33. [109] BAUMANN T, DE BUHR N, BLUME N, et al. Assessment of associations between neutrophil extracellular trap biomarkers in blood and thrombi in acute ischemic stroke patients. J Thromb Thrombolysis, 2024;57(6):936-946. [110] OH SA, SEOL SI, DAVAANYAM D, et al. Platelet-derived HMGB1 induces NETosis, exacerbating brain damage in the photothrombotic stroke model. Mol Med. 2025;31(1):46. [111] DI G, VáZQUEZ-REYES S, DÍAZ B, et al. Daytime DNase-I Administration Protects Mice From Ischemic Stroke Without Inducing Bleeding or tPA-Induced Hemorrhagic Transformation, Even With Aspirin Pretreatment. Stroke. 2025; 56(2):527-532. [112] DHANESHA N, JAIN M, TRIPATHI AK, et al. Targeting Myeloid-Specific Integrin α9β1 Improves Short- and Long-Term Stroke Outcomes in Murine Models With Preexisting Comorbidities by Limiting Thrombosis and Inflammation. Circ Res. 2020;126(12):1779-1794. [113] WEI N, LU T, GU J, et al. Lipoxin A4 suppresses neutrophil extracellular traps formation through the FPR2-dependent regulation of METTL3 in ischemic stroke. Brain Res Bull. 2024;220:111178. [114] TANG C, JIA F, WU M, et al. Elastase-targeting biomimic nanoplatform for neurovascular remodeling by inhibiting NETosis mediated AlM2 inflammasome activation in ischemic stroke. J Control Release. 2024;375:404-421. [115] HUANG T, GUO Y, XIE W, et al. Brain border-derived CXCL2+ neutrophils drive NET formation and impair vascular reperfusion following ischemic stroke. CNS Neurosci Ther. 2024;30(8):e14916. [116] KEUTERS MH, KEKSA-GOLDSTEINE V, RÕLOVA T, et al. Benserazide is neuroprotective and improves functional recovery after experimental ischemic stroke by altering the immune response. Sci Rep. 2024;14(1):17949. [117] HAUSENLOY DJ, YELLON DM. The mitochondrial permeability transition pore: its fundamental role in mediating cell death during ischaemia and reperfusion. J Mol Cell Cardiol. 2003;35(4):339-341. [118] YU J, ZHONG B, XIAO Q, et al. Induction of programmed necrosis: A novel anti-cancer strategy for natural compounds. Pharmacol Ther. 2020;214:107593. [119] LIU D, JI Q, CHENG Y, et al. Cyclosporine A loaded brain targeting nanoparticle to treat cerebral ischemia/reperfusion injury in mice. J Nanobiotechnology. 2022;20(1):256. [120] ZHAO J, LIU X, BLAYNEY A, et al. Intrinsically Disordered N-terminal Domain (NTD) of p53 Interacts with Mitochondrial PTP Regulator Cyclophilin D. J Mol Biol. 2022;434(9):167552. [121] OKAHARA A, KOGA JI, MATOBA T, et al. Simultaneous targeting of mitochondria and monocytes enhances neuroprotection against ischemia-reperfusion injury. Sci Rep. 2020;10(1):14435. [122] DENORME F, MANNE BK, PORTIER I, et al. Platelet necrosis mediates ischemic stroke outcome in mice. Blood. 2020;135(6):429-440. [123] VASEVA AV, MARCHENKO ND, JI K, et al. p53 opens the mitochondrial permeability transition pore to trigger necrosis. Cell. 2012;149(7):1536-1548. [124] ZHOU Z, LI W, NI L, et al. Icariin improves oxidative stress injury during ischemic stroke via inhibiting mPTP opening. Mol Med. 2024;30(1):77. [125] FRANKO J, POMFY M, PROSBOVÁ T. Apoptosis and cell death (mechanisms, pharmacology and promise for the future). Acta medica (Hradec Kralove). 2000;43(2):63-68. [126] LIU Y, CHE X, YU X, et al. Phosphorylation of STAT3 at Tyr705 contributes to TFEB-mediated autophagy-lysosomal pathway dysfunction and leads to ischemic injury in rats. Cell Mol Life Sci. 2023;80(6):160. [127] NI Y, GU WW, LIU ZH, et al. RIP1K Contributes to Neuronal and Astrocytic Cell Death in Ischemic Stroke via Activating Autophagic-lysosomal Pathway. Neuroscience. 2018;371:60-74. [128] HU K, PARK Y, OLIVAS Y, et al. Cathepsin B knockout confers significant brain protection in the mouse model of stroke. Exp Neurol. 2023;368:114499. [129] DONATI C, HASHIM, II, POZSONI NB, et al. Investigation of the in vitro anticancer potential of bis(imino)acenaphthene-N-heterocyclic carbene transition metal complexes revealed TrxR inhibition and triggering of immunogenic cell death (ICD) for allyl palladates. RSC Med Chem. 2025;16(6):2592-2602. [130] CAI J, YE Z, HU Y, et al. Identification of immunogenic cell death-related gene classification patterns and immune infiltration characterization in ischemic stroke based on machine learning. Front Cell Neurosci. 2022;16:1094500. [131] WANG M, WEI G, GU S, et al. A Machine Learning-Based Classification of Immunogenic Cell Death Regulators and Characterisation of Immune Microenvironment in Acute Ischemic Stroke. Int J Clin Pract. 2023;2023:9930172. [132] QIN L, LI S, CAO X, et al. Potential diagnostic biomarkers for immunogenic cell death in elderly female patients with ischemic stroke: identification and analysis. Sci Rep. 2024;14(1):14553. [133] ARTUS C, BOUJRAD H, BOUHARROUR A, et al. AIF promotes chromatinolysis and caspase-independent programmed necrosis by interacting with histone H2AX. EMBO J. 2010;29(9):1585-1599. [134] GERACE E, MASI A, RESTA F, et al. PARP-1 activation causes neuronal death in the hippocampal CA1 region by increasing the expression of Ca(2+)-permeable AMPA receptors. Neurobiol Dis. 2014; 70:43-52. [135] YUE CL, DING YX, CHEN M, et al. Medroxyprogesterone promotes neuronal survival after cerebral ischemic stroke by inhibiting PARthanatos. Front Pharmacol. 2025;16:1487436. [136] LI J, LIU X, WANG Z, et al. Ozone therapy mitigates parthanatos after ischemic stroke. Biol Res. 2024; 57(1):71. [137] CHEN HQ, ZHANG QG, ZHANG XY, et al. 4’-O-methylbavachalcone alleviates ischemic stroke injury by inhibiting parthanatos and promoting SIRT3. Eur J Pharmacol. 2024;972:176557. [138] JACKSON CW, XU J, ESCOBAR I, et al. Resveratrol Preconditioning Downregulates PARP1 Protein to Alleviate PARP1-Mediated Cell Death Following Cerebral Ischemia. Transl Stroke Res. 2023;15(1): 165-178. [139] PING N, ZUO K, CAI J, et al. Apigenin protects against ischemic stroke by increasing DNA repair. Front Pharmacol. 2024;15:1362301. [140] WU H, LI Y, ZHANG Q, et al. Crocetin antagonizes parthanatos in ischemic stroke via inhibiting NOX2 and preserving mitochondrial hexokinase-I. Cell Death Dis. 2023;14(1):50. [141] ZHANG J, LI X, KWANSA H, et al. Augmentation of poly(ADP-ribose) polymerase-dependent neuronal cell death by acidosis. J Cereb Blood Flow Metab. 2017;37(6):1982-1993. [142] CHEN S, GU J, ZHANG Q, et al. Development of Biomarker Signatures Associated with Anoikis to Predict Prognosis in Endometrial Carcinoma Patients. J Oncol. 2021;2021:3375297. [143] QIN X, YI S, RONG J, et al. Identification of anoikis-related genes classification patterns and immune infiltration characterization in ischemic stroke based on machine learning. Front Aging Neurosci. 2023;15:1142163. [144] HOU W, XIE Y, SONG X, et al. Autophagy promotes ferroptosis by degradation of ferritin. Autophagy. 2016;12(8):1425-1428. [145] LI C, SUN G, CHEN B, et al. Nuclear receptor coactivator 4-mediated ferritinophagy contributes to cerebral ischemia-induced ferroptosis in ischemic stroke. Pharmacol Res. 2021;174:105933. [146] DIAN H, YAN Z, JUNFEI S, et al. Identification of Potential Neddylation-related Key Genes in Ischemic Stroke based on Machine Learning Methods. Mol Neurobiol. 2023;61(5):2530-2541. [147] DU HP, GUO Y, ZHU YM, et al. RIPK1 inhibition contributes to lysosomal membrane stabilization in ischemic astrocytes via a lysosomal Hsp70.1B-dependent mechanism. Acta Pharmacol Sin. 2023;44(8):1549-1563. [148] LI L, LV L, WANG Z, et al. From copper homeostasis to cuproptosis: a new perspective on CNS immune regulation and neurodegenerative diseases. Front Neurol. 2025;16:1581045. [149] SONG Z, YE L, WANG Y, et al. Targeting RIPK1-mediated necroptosis, oxidative stress, and ferroptosis: A novel multitarget therapy for ischemic stroke. Eur J Med Chem. 2025;296:117884. [150] REN KD, LIU WN, TIAN J, et al. Mitochondrial E3 ubiquitin ligase 1 promotes brain injury by disturbing mitochondrial dynamics in a rat model of ischemic stroke. Eur J Pharmacol. 2019; 861:172617. [151] FAKHARNIA F, KHODAGHOLI F, DARGAHI L, et al. Prevention of Cyclophilin D-Mediated mPTP Opening Using Cyclosporine-A Alleviates the Elevation of Necroptosis, Autophagy and Apoptosis-Related Markers Following Global Cerebral Ischemia-Reperfusion. J Mol Neurosci. 2017;61(1):52-60. [152] HUANG Y, LIU J, HE J, et al. UBIAD1 alleviates ferroptotic neuronal death by enhancing antioxidative capacity by cooperatively restoring impaired mitochondria and Golgi apparatus upon cerebral ischemic/reperfusion insult. Cell Biosci. 2022;12(1):42. [153] NAKAMURA T, NAGURO I, ICHIJO H. Iron homeostasis and iron-regulated ROS in cell death, senescence and human diseases. Biochim Biophys Acta Gen Subj. 2019;1863(9):1398-409. [154] XU Y, WU X, HU W, et al. RIP3 facilitates necroptosis through CaMKII and AIF after intracerebral hemorrhage in mice. Neurosci Lett. 2021;749:135699. [155] GU X, DONG M, XIA S, et al. γ-Glutamylcysteine ameliorates blood-brain barrier permeability and neutrophil extracellular traps formation after ischemic stroke by modulating Wnt/β-catenin signalling in mice. Eur J Pharmacol. 2024:176409. [156] KUMAR R, PATIL G, DAYAL S. NLRP3-Induced NETosis: A Potential Therapeutic Target for Ischemic Thrombotic Diseases? Cells. 2023; 12(23):2709. [157] LUO C, OUYANG MW, FANG YY, et al. Dexmedetomidine Protects Mouse Brain from Ischemia-Reperfusion Injury via Inhibiting Neuronal Autophagy through Up-Regulating HIF-1α. Front Cell Neurosci. 2017;11:197. [158] ZHU C, ZHOU Q, LUO C, et al. Dexmedetomidine Protects Against Oxygen-Glucose Deprivation-Induced Injury Through Inducing Astrocytes Autophagy via TSC2/mTOR Pathway. Neuromolecular Med. 2020;22(2):210-217. [159] HADLEY G, BEARD DJ, COUCH Y, et al. Rapamycin in ischemic stroke: Old drug, new tricks?. J Cereb Blood Flow Metab. 2019;39(1):20-35. [160] CAI Y, YANG E, YAO X, et al. FUNDC1-dependent mitophagy induced by tPA protects neurons against cerebral ischemia-reperfusion injury. Redox Biol. 2021;38:101792. [161] THIEBAUT AM, BUENDIA I, GINET V, et al. Thrombolysis by PLAT/tPA increases serum free IGF1 leading to a decrease of deleterious autophagy following brain ischemia. Autophagy. 2022;18(6):1297-1317. [162] SUN JM, YEN TL, JAN JS, et al. Advances in Antibody-Based Therapeutics for Cerebral Ischemia. Pharmaceutics. 2022;15(1):145. [163] SELIM M. Treatment with the iron chelator, deferoxamine mesylate, alters serum markers of oxidative stress in stroke patients. Transl Stroke Res. 2010;1(1):35-39. [164] PENG W, GUO K, HU J, et al. Inhibition of Pyroptosis by Hydroxychloroquine as a Neuroprotective Strategy in Ischemic Stroke. eNeuro. 2024;12(1):ENEURO.0254-24.2024. [165] CARNEVALE R, LEOPIZZI M, DOMINICI M, et al. PAD4-Induced NETosis Via Cathepsin G-Mediated Platelet-Neutrophil Interaction in ChAdOx1 Vaccine-Induced Thrombosis-Brief Report. Arterioscler Thromb Vasc Biol. 2023; 43(10):e396-e403. [166] PIR GJ, PARRAY A, AYADATHIL R, et al. Platelet-Neutrophil Association in NETs-Rich Areas in the Retrieved AIS Patient Thrombi. Int J Mol Sci. 2022;23(22):14477. [167] LIPTON P. Lysosomal membrane permeabilization as a key player in brain ischemic cell death: a “lysosomocentric” hypothesis for ischemic brain damage. Transl Stroke Res. 2013;4(6):672-684. [168] NITATORI T, SATO N, WAGURI S, et al. Delayed neuronal death in the CA1 pyramidal cell layer of the gerbil hippocampus following transient ischemia is apoptosis. J Neurosci. 1995; 15(2):1001-1011. [169] ELIASSON MJ, SAMPEI K, MANDIR AS, et al. Poly(ADP-ribose) polymerase gene disruption renders mice resistant to cerebral ischemia. Nat Med. 1997;3(10):1089-1095. [170] DEGTEREV A, HUANG Z, BOYCE M, et al. Chemical inhibitor of nonapoptotic cell death with therapeutic potential for ischemic brain injury. Nat Chem Biol. 2005;1(2):112-119. [171] SCHINZEL AC, TAKEUCHI O, HUANG Z, et al. Cyclophilin D is a component of mitochondrial permeability transition and mediates neuronal cell death after focal cerebral ischemia. Proc Natl Acad Sci U S A. 2005;102(34):12005-12010. [172] AHMAD S, ELSHERBINY NM, HAQUE R, et al. Sesamin attenuates neurotoxicity in mouse model of ischemic brain stroke. Neurotoxicology. 2014;45:100-110. [173] DÁVALOS A, FERNANDEZ-REAL JM, RICART W, et al. Iron-related damage in acute ischemic stroke. Stroke. 1994;25(8):1543-1546. [174] DIXON SJ, LEMBERG KM, LAMPRECHT MR, et al. Ferroptosis: an iron-dependent form of nonapoptotic cell death. Cell. 2012;149(5): 1060-1072. [175] RUHNAU J, SCHULZE K, GAIDA B, et al. Stroke alters respiratory burst in neutrophils and monocytes. Stroke. 2014;45(3):794-800. [176] ZHANG C, WANG L, GUO Y, et al. Systematic analysis of brain and skull ischemic injury expression profiles reveals associations of the tumor immune microenvironment and cell death with ischemic stroke. Front Immunol. 2022;13:1082546. |
| [1] | 吕国庆, 艾孜麦提江·肉孜, 熊道海. 鸢尾素抑制人关节软骨细胞中铁死亡的作用及其机制[J]. 中国组织工程研究, 2026, 30(6): 1359-1367. |
| [2] | 贾金文, 艾日法特·艾尼瓦尔, 张 娟. EP300对过敏性鼻炎大鼠相关自噬和凋亡的影响[J]. 中国组织工程研究, 2026, 30(6): 1439-1449. |
| [3] | 吕晓凡, 黄 懿, 丁留成. 糖尿病膀胱病的线粒体机制与干预治疗[J]. 中国组织工程研究, 2026, 30(6): 1508-1515. |
| [4] | 油惠娟, 吴姝臻, 荣 融, 陈立沅, 赵玉晴, 王清路, 欧小伟, 杨风英. 巨噬细胞自噬与肺部疾病:作用的两面性[J]. 中国组织工程研究, 2026, 30(6): 1516-1526. |
| [5] | 刘可新, 郝凯敏, 庄文越, 李正祎. 自噬相关基因在肺纤维化模型中的表达:生物信息学分析及实验验证[J]. 中国组织工程研究, 2026, 30(5): 1129-1138. |
| [6] | 胡 静, 朱 伶, 谢 娟, 孔德营, 刘豆豆. 自噬影响组蛋白修饰标记H3K4me3调控小鼠早期胚胎发育[J]. 中国组织工程研究, 2026, 30(5): 1147-1155. |
| [7] | 邹荣基, 喻芳芳, 王茂林, 贾卓鹏 . 雷公藤内酯酮抑制铁死亡改善大脑动脉闭塞/再灌注模型大鼠脑缺血再灌注损伤[J]. 中国组织工程研究, 2026, 30(4): 873-881. |
| [8] | 杨 肖, 白月辉, 赵甜甜, 王东昊, 赵 琛, 袁 硕. 颞下颌关节骨关节炎软骨退变:机制及再生的挑战[J]. 中国组织工程研究, 2026, 30(4): 926-935. |
| [9] | 朱礼丰, 王文驰, 刘 强, 崔宪钦, 章震浩, 黄 杰, 吕柱成, 王磊航, 崔 伟. 淫羊藿苷防治骨质疏松症的分子机制[J]. 中国组织工程研究, 2026, 30(35): 9248-9257. |
| [10] | 曹 珊, 王焱皙, 段凯旋, 祁 祥, 王昱涵. 消斑通脉方靶向miR-126-3p调控细胞自噬:防治动脉粥样硬化的生物信息学分析[J]. 中国组织工程研究, 2026, 30(35): 9355-9364. |
| [11] | 王玉河, 谢天宇, 马诗佳, 王玉娇, 李梦婷, 谢道俊. 郁平神安颗粒对失眠模型小鼠神经元自噬的调控作用[J]. 中国组织工程研究, 2026, 30(35): 9217-9230. |
| [12] | 李 瀛, 王全玉, 冯崇义, 常 顺, 杨春爱. 脑类器官区域特异性在缺血性脑卒中建模和药物开发中的应用[J]. 中国组织工程研究, 2026, 30(34): 9008-9016. |
| [13] | 刘雨潇, 黄思璟, 耿珑玉, 高蓓瑶, 杨 光, 葛瑞东, 高 颀. PIEZO离子通道在神经系统疾病中的作用及分子机制[J]. 中国组织工程研究, 2026, 30(34): 9017-9023. |
| [14] | 张益瑞, 顾 叶, 钱正韬, 吴泽睿, 谢 恒, 唐奕涵, 顾赢楚, 方 涛, 王秋霏, 彭育沁, 耿德春, 徐耀增. 机械应力调控骨关节炎的分子机制与治疗靶点[J]. 中国组织工程研究, 2026, 30(34): 9048-9055. |
| [15] | 马润秋, 杨慧霞, 李雪儿, 白志刚, 李桂忠, 郝银菊, 马胜超, 姜怡邓. 糖皮质激素诱导成骨细胞线粒体损伤在激素性股骨头坏死中的作用机制[J]. 中国组织工程研究, 2026, 30(34): 8845-8851. |
调节性细胞死亡是一种受控的细胞死亡形式[5]。根据不同的形态、分子机制和功能,调节性细胞死亡子程序可分为凋亡型细胞死亡和非凋亡型细胞死亡,在维持体内平衡、促进发育和调节免疫方面起着重要作用[6]。凋亡型调节性细胞死亡包括外源性途径、线粒体内源性途径和内质网应激途径。非凋亡型调节性细胞死亡的调控机制主要包括自噬、铁死亡、失巢凋亡、铜死亡、双硫死亡、坏死性凋亡、焦亡、氧死亡、碱死亡、聚ADP核糖聚合酶1依赖性细胞死亡、线粒体通透性转换驱动的坏死、中性粒细胞胞外陷阱相关死亡、溶酶体依赖性细胞死亡和免疫原性细胞死亡(图1)[7-9]。越来越多的非凋亡型调节性细胞死亡因诱导缺血性脑卒中神经细胞死亡受到广泛关注[10]。例如,脑缺血后,中性粒细胞外陷阱通过加剧延迟的免疫细胞浸润和血管损伤对脑损伤产生了显著贡献;在脑缺血再灌注小鼠脑组织样品中发现,癌胚抗原相关细胞附着分子3、肿瘤坏死因子、P选择素和低亲和力免疫球蛋白rFc区受体Ⅳ上调,CellChat分析揭示了缺血性脑卒中关键的细胞间相互作用,强调了中性粒细胞通过以血小板反应蛋白1和整合素相关蛋白作为关键介质的连接附着分子途径与小胶质细胞和T细胞交流[11]。邱伟彤等[12]应用大鼠电针血清作用氧糖剥夺2 h、复氧24 h的海马神经元细胞HT22,发现可充分缓解糖氧剥夺导致的铁离子积聚、增加铁死亡相关蛋白谷胱甘肽过氧化酶
4的表达、降低长链酰基辅酶A合成酶4的表达、增加细胞活力,这些结果和应用铁死亡抑制剂处理的糖氧剥夺海马神经元细胞HT22效果相一致。间充质干细胞通过抑制聚ADP核糖聚合酶1依赖性细胞死亡和坏死性凋亡来保护神经元免受缺氧缺血性损伤,下调坏死性凋亡中2种必需激酶受体相互作用蛋白激酶1和3的表达可减少氧糖剥夺诱导的神经元死亡[13]。该文全面总结非凋亡型调节性细胞死亡亚程序在缺血性脑卒中神经细胞损伤中发挥的作用及作用机制,为缺血性脑卒中的预测和治疗提供理论基础,为缺血性脑卒中靶向药的研发提供科研策略。 中国组织工程研究杂志出版内容重点:干细胞;骨髓干细胞;造血干细胞;脂肪干细胞;肿瘤干细胞;胚胎干细胞;脐带脐血干细胞;干细胞诱导;干细胞分化;组织工程
1.1 资料来源
1.1.1 检索人及检索时间 由第一作者在2025年1-4月进行检索。
1.1.2 文献检索时限 各数据库建库至2025年6月。
1.1.3 检索数据库 PubMed数据库、中国知网。
1.1.4 检索词 英文检索词为“ischemic
stroke,regulated cell death,autophagy,ferroptosis,cuproptosis,disulfidptosis,necroptosis,pyroptosis,alkaliptosis,oxeiptosis,parthanatos,mitochondrial permeability transition-driven necrosis,neutrophil extracellular trap-related death,lysosome-dependent cell death,immunogenic cell death,anoikis”,中文检索词为“缺血性脑卒中,调节性细胞死亡,铁死亡,坏死性凋亡,自噬,焦亡,铜死亡,双硫死亡,碱死亡,氧死亡,聚ADP核糖聚合酶1依赖性细胞死亡,溶酶体依赖性细胞死亡,失巢凋亡,线粒体通透性转变驱动的坏死,免疫原性细胞死亡,中性粒细胞胞外陷阱相关死亡”。
1.1.5 检索文献类型 研究原著及综述。
1.1.6 手工检索情况 无。
1.1.7 检索策略 以PubMed数据库检索策略为例,见图2。
1.1.8 检索文献量 共得到中、英文文献2 800余篇。
1.2 文章框架设计 见图3。
1.3 入组标准
纳入标准:①与缺血性脑卒中相关的文献;②与调节性细胞死亡相关的文献;③缺血性脑卒中分别与调节性细胞死亡亚型相互作用的文献(包括调节性细胞死亡亚型专有名词和或亚型相关的特定检测指标一同进行检索);④同一领域选择发表年份较新的文献;⑤筛选不同角度细胞死亡亚型在缺血性脑卒中的作用机制文献。
排除标准:①与研究主题无关的文献(如非凋亡型调节性细胞死亡亚型在其他疾病中的作用);②中英文中研究内容相同或相似的重复性文献;③存在争议或预警期刊发表的
文献。
1.4 文献检索结果 检索得到文献2 800余篇,根据入选标准,最终纳入176篇文献进行综述。文献检索流程见图4。
脑卒中以高发病率、高死亡率和高残疾率为主要特征,缺血性脑卒中是导致死亡的首要原因。已经确定了多种细胞死亡途径参与缺血性脑卒中神经损伤,深入探讨多种神经细胞死亡方式串扰的背后分子机制不仅会增强人们对细胞死亡机制的理解,还会为缺血性脑卒中提供潜在的新治疗靶点。自噬的神经保护作用归因于它能够减轻毒性蛋白和受损线粒体的积累,从而保持神经元的活力和功能。虽然缺血/再灌注损伤导致线粒体功能障碍、氧化应激反应增加和钙稳态失衡等,但用特定分子靶向线粒体自噬相关通路可能为某些缺血性脑卒中提供治疗方向,在临床应用中显示出巨大的前景[22]。铁死亡是由过量的铁离子触发的,在缺血性脑卒中期间这些离子在细胞中的积累除了直接伤害神经细胞外,还会加剧脑卒中后期的炎症,通过引发氧化应激、参与线粒体损伤和调控信号通路(如核转录因子)来加速神经元凋亡。铜死亡取决于铜离子对氧化还原反应的调节及其对线粒体功能的影响,它可促进神经炎症,同时与铁死亡相互作用在脑卒中后形成复杂的细胞死亡网络,因此,未来涉及探索抑制铁离子和铜离子过度积聚的方法对缺血性脑卒中治疗至关重要。参与损伤后血栓形成和炎症反应过程是中性粒细胞胞外陷阱死亡引起缺血性脑卒中神经损伤的主要原因。目前的研究表明,溶酶体依赖性细胞死亡、线粒体通透性转换驱动的坏死、铜死亡、中性粒细胞外陷阱相关细胞死亡在缺血性脑卒中神经损伤机制的研究涉及细胞和动物实验,但对相关机制、多种机制相互联系没有进行深入探讨,同时,通过靶向调控以上调节性细胞死亡缓解缺血性脑卒中神经损伤的治疗策略和针对性的药物开发研究较少,距离从基础研究向临床应用过渡还有很长的路要走。机器学习算法、聚类分析等生物信息技术手段证实,双硫死亡、免疫原性细胞死亡和失巢凋亡在缺血性脑卒中患者中存在显著的差异基因,这些基因和免疫细胞浸润及免疫反应存在显著相关性,可能在病程检测、临床筛查中具有重大意义。但目前对双硫死亡、免疫原性细胞死亡和失巢凋亡在缺血性脑卒中损伤中发挥作用的研究仅局限在基因预测阶段,没有在细胞和动物水平上进行验证及作用机制的深层探讨。
综上所述,非凋亡型调节性细胞死亡在缺血性脑卒中神经损伤中具有重要作用,具体的作用机制还有待今后更深层次的探讨,未来可能会优先考虑靶向药物研发、应用生物技术联合探讨多靶点药物作为潜在的研究重点,为多靶点药物开发和多靶点药物联合应用提供理论基础,为缺血性脑卒中的预防和增宽治疗窗口提供新的研究策略。
中国组织工程研究杂志出版内容重点:干细胞;骨髓干细胞;造血干细胞;脂肪干细胞;肿瘤干细胞;胚胎干细胞;脐带脐血干细胞;干细胞诱导;干细胞分化;组织工程
该文通过国内外数据库对非凋亡型调节性细胞死亡在缺血性脑卒中疾病中的应用进行了相关文献检索,文章内容概括了现阶段热点研究的自噬、铁死亡、铜死亡及坏死性凋亡等最新的研究进展,同时对新兴的调节性细胞死亡类型如双硫死亡、中性粒细胞胞外陷阱相关死亡、失巢凋亡、聚ADP核糖聚合酶1依赖性细胞死亡、线粒体通透性转换驱动的坏死和免疫原性细胞死亡等死亡形式在缺血性脑卒中神经细胞损伤机制中发挥的作用进行了综述总结。该文突出介绍了自噬对缺血性脑卒中的双重作用机制,通过基因调控、中草药及生物工程相结合等药物开发手段对自噬进行调控,缓解缺血性脑卒中神经细胞的损伤。对铁死亡、铜死亡和双硫死亡等细胞死亡方式在缺血性脑卒中神经细胞的损伤中发挥的作用机制进行总结。对同时出现的非凋亡型调节性细胞死亡亚型之间的相互作用关系进行了探讨。
中国组织工程研究杂志出版内容重点:干细胞;骨髓干细胞;造血干细胞;脂肪干细胞;肿瘤干细胞;胚胎干细胞;脐带脐血干细胞;干细胞诱导;干细胞分化;组织工程
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||