中国组织工程研究 ›› 2026, Vol. 30 ›› Issue (35): 9248-9257.doi: 10.12307/2026.283
• 组织构建综述 tissue construction review • 上一篇 下一篇
淫羊藿苷防治骨质疏松症的分子机制
朱礼丰1,王文驰1,刘 强2,崔宪钦1,章震浩1,黄 杰1,吕柱成1,王磊航1,崔 伟3
- 1广西中医药大学,广西壮族自治区南宁市 530000;2江西中医药大学,江西省南昌市 330000;3广西中医药大学附属瑞康医院,广西壮族自治区南宁市 530000
-
收稿日期:2025-09-10修回日期:2025-12-03出版日期:2026-12-18发布日期:2026-04-29 -
通讯作者:崔伟,博士,主任医师,教授,硕士生导师,广西中医药大学附属瑞康医院,广西壮族自治区南宁市 530000 -
作者简介:朱礼丰,男,1999年生,江西省景德镇市人,汉族,广西中医药大学在读硕士,主要从事脊柱、骨关节创伤性疾病防治研究。 -
基金资助:广西中医药大学桂派中医药传承创新团队项目(2022A004),项目参与人:崔伟;广西壮族自治区中医药管理局自筹经费科研课题(20210579),项目负责人:崔伟;中国民族医药学会课题(2020MZ-040701),项目负责人:崔伟
Molecular mechanism of icariin in prevention and treatment of osteoporosis
Zhu Lifeng1, Wang Wenchi1, Liu Qiang2, Cui Xianqin1, Zhang Zhenhao1, Huang Jie1, Lyu Zhucheng1, Wang Leihang1, Cui Wei3
- 1Guangxi University of Chinese Medicine, Nanning 530000, Guangxi Zhuang Autonomous Region, China; 2Jiangxi University of Chinese Medicine, Nanchang 330000, Jiangxi Province, China; 3Affiliated Ruikang Hospital of Guangxi University of Chinese Medicine, Nanning 530000, Guangxi Zhuang Autonomous Region, China
-
Received:2025-09-10Revised:2025-12-03Online:2026-12-18Published:2026-04-29 -
Contact:Cui Wei, MD, Chief physician, Professor, Master’s supervisor, Affiliated Ruikang Hospital of Guangxi University of Chinese Medicine, Nanning 530000, Guangxi Zhuang Autonomous Region, China -
About author:Zhu Lifeng, MS candidate, Guangxi University of Chinese Medicine, Nanning 530000, Guangxi Zhuang Autonomous Region, China -
Supported by:Guizhou School of Traditional Chinese Medicine Inheritance and Innovation Team of Guangxi University of Chinese Medicine, No. 2022A004 (to CW); Guangxi Zhuang Autonomous Region Administration of Traditional Chinese Medicine Self-funded Scientific Research Project, No. 20210579 (to CW); Chinese Ethnic Medicine Association Project, No. 2020MZ-040701 (to CW)
摘要:
文题释义:
淫羊藿苷:是淫羊藿属植物中主要的活性类黄酮苷,具有抗骨质疏松、抗肿瘤、抗氧化、抗炎、保护心血管等多种药理活性等作用。
骨质疏松症:是一种由多种原因引发的全身性骨病,主要特征是骨量减少、骨微结构恶化,致使骨骼强度降低,发生骨折的概率增大。
背景:淫羊藿苷抗骨质疏松的药效学特征及作用机制正逐步获得学界认可,并且相关基础研究与临床转化探索日益成为研究焦点。
目的:总结淫羊藿苷发挥抗骨质疏松症方面的研究进展。
方法:检索中国知网及PubMed数据库收录的相关文献,中文检索词为“淫羊藿苷,骨质疏松症,中药复方,发病机制,信号通路,骨髓间充质干细胞,成骨细胞,破骨细胞”,英文检索词为“Icariin,Osteoporosis,Chinese Medicine compound,Pathogenesis,Signal path,BMSCs,Osteoblast,Osteoclast”,根据入选标准,最终纳入90篇文献进行综述。
结果与结论:淫羊藿苷处理可增加碱性磷酸酶活性,并以剂量依赖性方式诱导骨髓间充质干细胞中核心结合因子α1、骨形态发生蛋白2和骨形态发生蛋白4的表达。淫羊藿苷通过增加血清骨钙素、骨特异性碱性磷酸酶、Ⅰ型胶原蛋白N端肽、Ⅰ型胶原蛋白C端肽和抗酒石酸酸性磷酸酶5b水平,促进骨折部位的骨钙素分泌,加速骨折愈合。淫羊藿苷通过上调碱性磷酸酶和骨钙素水平及抑制Notch通路中Notch-1、CBF1、Jagged-1蛋白的表达,促进去卵巢骨质疏松症大鼠骨髓间充质干细胞的增殖和成骨细胞分化,达到防治骨质疏松症的作用。淫羊藿苷通过介导Wnt/β-catenin、丝裂原活化蛋白激酶、磷脂酰肌醇3-激酶/蛋白激酶B、骨保护素/核因子κB受体活化因子配体/核因子κB受体活化因子及Notch等多条信号轴实现骨代谢调控。其中Wnt/β-catenin通路与骨保护素/核因子κB受体活化因子配体/核因子κB轴构成核心调控机制,通过相互协同作用调节成骨-破骨动态平衡。淫羊藿苷可通过调控mRNAs表达修饰、氧化应激抑制及炎症微环境改善等多维度干预,影响成骨细胞、破骨细胞及骨髓间充质干细胞的生物学行为。
https://orcid.org/0009-0000-0510-3031(朱礼丰);https://orcid.org/0000-0003-2011-7508(崔伟)
中国组织工程研究杂志出版内容重点:干细胞;骨髓干细胞;造血干细胞;脂肪干细胞;肿瘤干细胞;胚胎干细胞;脐带脐血干细胞;干细胞诱导;干细胞分化;组织工程
中图分类号:
引用本文
朱礼丰, 王文驰, 刘 强, 崔宪钦, 章震浩, 黄 杰, 吕柱成, 王磊航, 崔 伟. 淫羊藿苷防治骨质疏松症的分子机制[J]. 中国组织工程研究, 2026, 30(35): 9248-9257.
Zhu Lifeng, Wang Wenchi, Liu Qiang, Cui Xianqin, Zhang Zhenhao, Huang Jie, Lyu Zhucheng, Wang Leihang, Cui Wei. Molecular mechanism of icariin in prevention and treatment of osteoporosis[J]. Chinese Journal of Tissue Engineering Research, 2026, 30(35): 9248-9257.
2.2 淫羊藿苷防治骨质疏松症的相关性研究 目前已有大量文献从骨组织形态学、生物力学、骨组织生化指标等方面证实了淫羊藿苷的抗骨质疏松作用[22-24],鉴于淫羊藿苷在骨质疏松防治中展现的独特优势,当前研究已聚焦于淫羊藿苷作用机制的深度解析,使之成为该领域的重要研究方向。淫羊藿苷的作用机制主要涉及对骨髓间充质干细胞、成骨细胞及破骨细胞分化与功能的调控。
2.2.1 淫羊藿苷对骨髓间充质干细胞的影响 骨髓间充质干细胞是起源于中胚层的一类非造血源性干细胞群体,具有长期自我更新能力及分化潜能[25],在特定信号调控下,该类细胞可实现向骨细胞、软骨细胞、脂肪细胞、肌细胞、神经细胞及心肌细胞等的定向转化[26]。现代药理学研究证实,淫羊藿苷可通过调控相关信号通路诱导骨髓间充质干细胞向成骨细胞分化,从而在骨代谢平衡调控中发挥干预作用,为骨质疏松症的防治提供了新的作用靶点。
HAN等[27]研究表明,经淫羊藿苷干预后,大鼠骨髓间充质干细胞中TAZ表达激活,成骨指标碱性磷酸酶、Runt相关转录因子2表达

升高,并且最佳干预浓度为1×10-7 mol/L;沉默小窝蛋白显著促进骨髓间充质干细胞的成骨分化,淫羊藿苷通过抑制小窝蛋白调控Hippo/TAZ信号通路,进而促进骨髓间充质干细胞的成骨分化。CHEN等[28]在卵巢切除大鼠模型中发现,淫羊藿苷增加大鼠的骨量并降低了骨髓脂肪含量,促进成骨分化并抑制骨髓间充质干细胞的成脂分化,并且淫羊藿苷也能通过减弱雌激素受体α的作用来降低骨硬化蛋白表达,表明淫羊藿苷通过增强骨硬化蛋白甲基化来改善骨量并降低骨髓脂肪含量。金东升等[29]在兔骨髓间充质干细胞模型中证实,载淫羊藿苷缓释微球的三维支架能促进骨髓间充质干细胞的增殖,增加细胞活性增加,以10 μmol/L淫羊藿苷组效果最佳;淫羊藿苷促进了Ⅰ型胶原、骨钙素、Runt相关转录因子2、血管内皮生长因子蛋白表达。蔡叶等[30]在经D-半乳糖诱导的衰老骨髓间充质干细胞模型中发现,淫羊藿苷增加了衰老骨髓间充质干细胞上清液中碱性磷酸酶含量,并降低了P53的表达水平,可能通过调节成骨相关信号通路中转录因子(Foxo3a)/β-catenin的表达改善衰老骨髓间充质干细胞的成骨分化能力。张锦明等[31]研究发现,在经过连续皮下注射地塞米松5周的小鼠骨质疏松模型中,淫羊藿苷促进小鼠骨髓间充质干细胞的成骨分化、抑制小鼠骨髓间充质干细胞的成脂分化,改善骨髓微环境,促进骨小梁形成,缓解骨质疏松症。TENG等[32]实验发现,miR-335-5p在骨质疏松症骨组织中低表达,淫羊藿苷处理逆转了miR-335-5p抑制剂对骨髓间充质干细胞基质矿化的抑制作用,此外,肿瘤抑制基因蛋白被证实是miR-335-5p的下游效应子,在淫羊藿苷诱导过程中,肿瘤抑制基因蛋白的过表达逆转了miR-335-5p模拟物对骨髓间充质干细胞成骨分化的促进作用。
淫羊藿苷的成骨诱导作用主要通过调节骨髓间充质干细胞的定向分化实现的,淫羊藿苷可显著上调骨髓间充质干细胞中成骨关键因子Runt相关转录因子2、血管内皮生长因子及碱性磷酸酶的表达水平。进一步机制解析表明,淫羊藿苷的细胞分化调控效应依赖多条信号通路的协同作用,但存在浓度依赖性特征:当淫羊藿苷的浓度超过特定阈值时,成骨诱导作用显著减弱,而适宜浓度范围是发挥最佳骨形成效应的关键决定因素,这种双向调节特性最终指向优化骨髓间充质干细胞成骨分化过程并提升骨密度的核心目标,为后续实验设计提供了重要的剂量效应参考依据。淫羊藿苷调控骨髓间充质干细胞增殖、分化的作用机制详见表1。
2.2.2 淫羊藿苷对成骨细胞的影响 骨骼内骨组织的稳态依赖于新骨形成与陈旧骨吸收之间的精密调控,这种动态平衡机制确保了骨组织的持续更新及骨骼生物力学性能的维持[33]。当这种平衡被打破时,骨代谢失衡随即发生,进而引发骨质疏松症[34]。淫羊藿苷可显著促进成骨细胞功能活化,该作用机制主要体现在对骨髓间充质干细胞成骨定向分化的诱导效应。淫羊藿苷能加速成骨前体细胞增殖周期进程,并刺激骨钙素等骨基质蛋白合成,最终通过优化骨形成与骨吸收的偶联关系,实现骨量积累与骨质量改善的治疗目标。
ZHANG等[35]研究发现,在经地塞米松刺激的小鼠成骨细胞中,淫羊藿苷通过上调SPI1增加SMAD5表达,并能缓解SPI1缺失对细胞自噬与成骨分化的抑制作用。ZHU等[36]实验发现,在骨质疏松大鼠模型中,锶-淫羊藿苷复合涂层植入体周围可见更显著的新骨形成,表明锶-淫羊藿苷复合涂层能有效增强钛植入体的骨整合作用。ZHOU等[37]研究发现,在前成骨细胞MC3T3-E1模型中,淫羊藿苷能快速诱导胰岛素样生长因子1受体与雌激素受体α的相互作用,激活胰岛素样生长因子1受体信号通路及其下游蛋白激酶B磷酸化,淫羊藿苷对MC3T3-E1细胞的这种促进作用及其对成熟成骨细胞碱性磷酸酶活性的成骨效应均呈现胰岛素样生长因子1受体依赖性。陈绎志等[38]在兔股骨缺损动物模型中发现,淫羊藿苷可促进兔成骨细胞增殖,提高成骨细胞活性,增加骨形成。刘婷等[39]研究发现,淫羊藿苷能明显促进hFOB1.19人成骨细胞的增殖并促进细胞分泌碱性磷酸酶,提示淫羊藿苷对成骨细胞活性的促进作用可能是其防治骨质疏松症的途径之一。
综合现有研究,淫羊藿苷通过促进成骨细胞增殖与分化活性发挥抗骨质疏松症作用。研究进一步揭示,淫羊藿苷可通过上调转录因子SPI1基因表达增强受体激活蛋白SMAD5蛋白水平,逆转骨桥蛋白、骨钙素及Runt相关转录因子2等成骨标志物的表达抑制,从而同时实现减弱细胞自噬水平与缓解成骨分化障碍的双重调控效应。值得注意的是,基于骨组织工程技术的进展,锶元素与淫羊藿苷的协同应用在骨质疏松防治领域展现出显著的应用前景,为临床转化研究奠定了重要基础。淫羊藿苷调控成骨细胞增殖、分化的作用机制详见表2。
2.2.3 淫羊藿苷对破骨细胞的影响 破骨细胞作为骨吸收过程的核心效应细胞,属于多核巨型终末分化细胞,直接参与骨吸收,是骨组织吸收的主要功能细胞,与成骨细胞、骨细胞等形成动态调控体系,在细胞因子、生长因子及激素等多重调控信号的协同作用下,借助精密的分子信号传导通路实现骨吸收与骨形成的耦合平衡[40-41]。
SI等[42]研究发现,在经核因子κB受体活化因子配体诱导的RAW264.7细胞中,淫羊藿苷抑制E3泛素连接酶表达,并可通过减少内源性核转录因子E2相关因子的泛素化来延迟核转录因子E2相关因子降解,并且这些作用可被E3泛素连接酶过表达显著逆转,表明淫羊藿苷通过靶向E3泛素连接酶/核转录因子E2相关因子信号通路抑制破骨细胞生成,从而缓解骨质疏松症。CHENG等[43]实验结果表明,在经硫代乙酰胺诱导的破骨细胞中,淫羊藿苷抑制核因子κΒ配体受体激活剂、核因子κB受体激活剂、蛋白激酶(P38)、细胞外信号调节激酶、原癌基因c-Fos和活化T细胞核因子1的蛋白表达,结果表明淫羊藿苷可能通过下调蛋白激酶/细胞外信号调节激酶信号通路来抑制破骨细胞分化,阻止硫代乙酰胺诱导的骨质流失。朱在师等[44]研究发现,在可溶性核因子κB受体活化因子配体诱导的RAW264.7前破骨细胞模型中,知母皂苷Ⅱ及淫羊藿苷单独及联合应用均能够抑制破骨细胞分化,而不影响破骨细胞对成骨细胞的耦联作用,并且知母皂苷Ⅱ+淫羊藿苷联合应用能增强破骨细胞-成骨细胞偶联作用,该作用机制可能与促进破骨细胞源性偶联因子的表达有关。贺龙刚等[45]在体外RAW264.7细胞诱导破骨细胞的模型中发现,淫羊藿中的主要有效成分淫羊藿次苷Ⅰ及其代谢产物淫羊藿次苷Ⅱ在体外能通过抑制二聚体转录因子AP-1/活化T细胞核转录因子c1信号通路相关基因表达,提示可能通过抑制c-Fos蛋白调控靶基因转录因子Fra-1、转录因子Fra-2和活化T细胞核转录因子c1表达,进而抑制破骨细胞生成。
现有证据表明,破骨细胞功能异常是导致骨代谢失衡的核心病理环节,其中核因子κB受体活化因子配体/核因子κB受体活化因子信号轴作为调控破骨细胞活性的关键通路,已成为干预骨吸收过程的重要靶点。淫羊藿苷可通过抑制性调节核因子κB受体活化因子配体信号通路下调活化T细胞核转录因子c1、c-Fos、肿瘤坏死因子受体相关因子6及转化生长因子β等促破骨生成因子的表达水平,从而阻断破骨细胞前体的增殖分化进程。尽管现有实验数据已初步揭示淫羊藿苷与“破骨细胞-成骨细胞偶联”机制的潜在关联,但在成骨细胞信号网络中多蛋白因子的交互作用需进一步深入探讨。淫羊藿苷调控破骨细胞增殖、分化的作用机制详见表3。
2.3 淫羊藿苷防治骨质疏松症的相关信号通路研究 在淫羊藿苷防治骨质疏松症的研究中,Wnt/β-catenin、Notch、磷脂酰肌醇3-激酶/蛋白激酶B、丝裂原活化蛋白激酶及核因子κB受体活化因子/核因子κB受体活化因子配体/骨保护素通路为研究的重点,这些通路分别参与成骨细胞分化、破骨细胞活化及炎症反应的核心调控,是骨重建失衡的关键节点。淫羊藿苷通过激活Wnt/β-catenin促进成骨,抑制核因子κB受体活化因子配体/骨保护素失衡以减少破骨,同时调控Notch、磷脂酰肌醇3-激酶/蛋白激酶B及丝裂原活化蛋白激酶通路协
同平衡骨代谢。相关研究已明确淫羊藿苷对这些通路的干预作用,并且这些通路间存在交叉调控网络,能全面阐释淫羊藿苷多靶点作用机制;此外,这些通路是现有骨质疏松药物研发的重要靶点,为淫羊藿苷的临床转化提供理论依据。
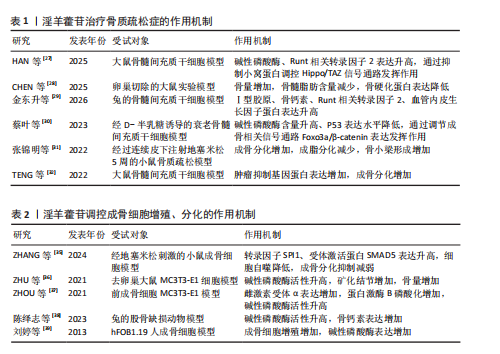
2.3.1 Wnt/β-catenin 信号通路 Wnt-β-catenin信号轴在骨骼发育过程中发挥核心调控作用,它通过级联反应调控多个骨骼代谢及组织重塑相关关键因子的表达。Wnt-β-catenin信号传导过程始于Wnt蛋白与细胞膜表面Frizzled受体家族及低密度脂蛋白受体相关蛋白5/6共受体的特异性结合,这种相互作用主要发生在受体富含半胱氨酸的结构域,配体-受体复合物形成后触发细胞质内无规则蛋白的构象改变及功能活化。在Wnt信号通路激活状态下,蓬乱蛋白通过特异性蛋白互作界面拮抗轴抑制蛋白的分子支架功能,进而干扰由腺瘤性息肉病蛋白、轴抑制蛋白、糖原合成酶激酶3β及酪蛋白激酶1α构成的β-catenin降解复合体组
装[46-47]。在这一过程中,直接阻断β-catenin的磷酸化修饰及其后续泛素-蛋白酶体降解途径,导致该关键信号分子在胞质内异常蓄积,积累的β-catenin随即发生核转位,与T细胞因子/淋巴增强因子转录复合物形成功能性转录调控单元,通过启动下游靶基因的转录激活程序,最终实现其对细胞增殖、分化的调控作用[48-49]。
GAO等[50]在12周龄雌性SD大鼠骨髓间充质干细胞中发现,硬化蛋白基因诱导可显著抑制骨髓间充质干细胞的增殖和成骨分化,经淫羊藿苷处理后不仅增加了骨髓间充质干细胞的数量,还增强了成骨诱导过程中碱性磷酸酶活性和钙结节的形成;硬化蛋白下调了包括Runt相关转录因子2、β-catenin和原癌基因c-Myc在内的成骨基因以及抗氧化因子(Prdx1、转录因子Cata和Nqo1)的表达,经淫羊藿苷处理后上述指标表达得以恢复;淫羊藿苷通过激活Wnt/β-catenin通路来促进骨髓间充质干细胞的原位增殖和成骨分化。ZHANG等[51]在大鼠骨髓间充质干细胞模型中发现,不同浓度淫羊藿苷处理增加了碱性磷酸酶活性,并以剂量依赖性方式提高核心结合因子α1、骨形态发生蛋白2和骨形态发生蛋白4表达;此外,淫羊藿苷增加血清中了骨钙素、骨特异性碱性磷酸酶、Ⅰ型胶原蛋白N端肽、Ⅰ型胶原蛋白C端肽和抗酒石酸酸性磷酸酶5b水平,促进骨折部位骨钙素分泌,加速大鼠骨折愈合,该作用机制与激活Wnt1/β-catenin成骨信号通路相关。另有研究发现,淫羊藿苷通过激活Wnt/β-catenin信号通路调节去卵巢大鼠骨髓基质细胞成骨分化与脂肪分化的平衡,这可能是淫羊藿苷治疗绝经后骨质疏松症的重要分子机制[52]。
杨志烈等[53]研究发现,在环磷酰胺诱导的骨髓间充质干细胞成骨分化障碍模型中,经一定浓度的淫羊藿苷处理后细胞内Wnt4、β-catenin及细胞周期蛋白D1的mRNA水平显著升高,同时伴随β-catenin、细胞周期蛋白D1与c-Myc蛋白表达增强,而Dickkopf 相关蛋白1表达呈现下调趋势,该作用可能通过激活Wnt/β-catenin信号通路实现的。曾建春等[54]在人骨髓间充质干细胞模型中发现,含40 μg/L淫羊藿苷诱导液诱导细胞向成骨细胞分化,并且显著提高Wnt3a、β-catenin蛋白表达,该作用机制是通过激活Wnt/β-catenin信号通路实现的。ZENG等[55]在乙醇干预下的MC3T3-E1细胞模型中发现,淫羊藿苷增加了细胞内相对碱性磷酸酶面积,上调被酒精抑制的成骨相关蛋白和mRNAs表达水平,促进成骨分化;此外淫羊藿苷还降低了酒精升高的活性氧水平,减少了酒精干预诱导的成骨细胞凋亡,该作用机制是通过调节Wnt/β-catenin信号通路实现的。淫羊藿苷介导 Wnt/β-catenin信号通路调控骨质疏松症的作用机制,详见图5。
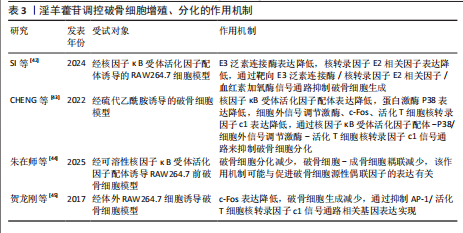
2.3.2 Notch信号通路 Notch信号通路在细胞增殖、分化及凋亡的调控网络中发挥关键作用,尤其在成骨分化早期阶段呈现促分化效应,而分化后期则表现为对成骨细胞增殖的抑制作用。当骨髓间充质干细胞的Notch通路被激活时,该通路通过维持干细胞未分化状态来促进细胞增殖,而阻断骨髓间充质干细胞内的Notch信号传导可诱导干细胞向成骨或软骨细胞定向分化[56]。Notch信号通路配体Jagged-1的过表达可通过延缓细胞衰老进程加速骨折早期修复,并显著提升成骨前体细胞的分化潜能[57]。通过抑制Notch信号通路活性可增强破骨细胞增殖活性并减少细胞凋亡;而Notch-1受体过度活化通过上调δ样配体3及Jagged-1表达水平发挥对破骨细胞的抑制效应[58-60]。
BIAN等[61]在去卵巢术诱导大鼠骨质疏松模型中发现,淫羊藿苷可通过调控Delta样配体1及肿瘤坏死因子α转化酶的表达水平激活Notch信号传导通路,该过程进一步诱导骨钙素、Ⅰ型胶原蛋白及Runt相关转录因子2分泌量增加,促进骨髓间充质干细胞向成骨分化。邓宇等[62]在大鼠骨髓间充质干细胞模型中发现,淫羊藿苷可显著上调HES、Runt相关转录因2 mRNA表达,同时促进Jagged-1、Notch-1、CBF1蛋白表达水平,进一步激活Notch信号通路,促进骨髓间充质干细胞向成骨细胞分化,进而改善骨质疏松症。徐娅等[63]在大鼠骨髓间充质干细胞的体外模型中发现,淫羊藿苷能够促进骨髓间充质干细胞向成骨分化,通过下调 Notch信号核心结合蛋白CBF1与通路中关键蛋白Notch-1的表达抑制Notch信号通路,从而发挥抗骨质疏松症作用。周亚妮等[64]在去卵巢骨质疏松症大鼠骨髓间充质干细胞模型中发现,淫羊藿苷通过上调碱性磷酸酶和骨钙素水平及抑制Notch通路中Notch-1、CBF1、Jagged-1蛋白表达,促进骨髓间充质干细胞的增殖和成骨细胞分化,从而达到防治骨质疏松症作用。淫羊藿苷介导Notch 信号通路干预骨质疏松症的作用机制,详见图6。
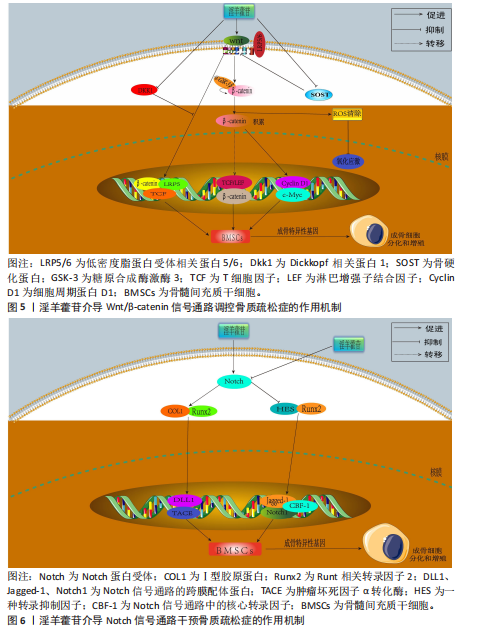
2.3.3 磷脂酰肌醇3-激酶/蛋白激酶B信号通路 磷脂酰肌醇3-激酶/蛋白激酶B信号传导系统普遍存在于各类细胞中,作为细胞内外信号整合枢纽,通过多层级调控网络参与细胞生长调控、增殖维持、分化定向及细胞骨架动态重排等关键生物学过程[65]。蛋白激酶B蛋白家族包含蛋白激酶B1、蛋白激酶B2和蛋白激酶B3三种亚型。作为磷脂酰肌醇信号通路的关键效应分子,磷脂酰肌醇3-激酶通过与蛋白激酶B的N端PH结构域结合诱导其构象重塑,使Thr308和Ser473两个关键磷酸化位点暴露并发生双重磷酸化修饰,这种共价修饰是蛋白激酶B活性激活的分子基础[66]。激活态蛋白激酶B通过磷酸化级联反应调控下游效应蛋白,包括哺乳动物雷帕霉素靶蛋白复合物1、MDM2原癌蛋白、Bcl-2相关死亡促进因子、核因子κB、糖原合成酶激酶3β、半胱氨酸天冬氨酸蛋白酶9、核糖体蛋白S6激酶及FoxO转录因子家族等,进而促进细胞新陈代谢、生长、增殖、凋亡和血管生成等[67-68]。
YAO等[69]在经柠檬酸铁铵诱导的铁过载骨髓间充质干细胞模型中发现,caspase-3和BAX蛋白表达升高,Bcl-2蛋白表达降低,淫羊藿苷处理可降低caspase-3和BAX蛋白表达、升高Bcl-2蛋白表达,表明淫羊藿苷能够防止铁过载诱导的骨髓间充质干细胞功能障碍,该作用机制与淫羊藿苷减弱铁过载诱导磷脂酰肌醇3-激酶/蛋白激酶B通路失活相关。JI等[70]在经地塞米松诱导的骨髓间充质干细胞模型中发现,淫羊藿苷通过激活磷脂酰肌醇3-激酶/蛋白激酶B信号传导上调组蛋白去甲基化酶并抑制组蛋白甲基转移酶2表达,促进地塞米松处理骨髓间充质干细胞的成骨分化。范凯健等[71]在MC3T3-E1细胞模型中发现,淫羊藿苷干预可以促进钙沉积、提高碱性磷酸酶活性,提高SMAD4、骨形态发生蛋白2、转化生长因子β蛋白表达,明显促进细胞的增殖与迁移,作用机制可能是淫羊藿苷通过促进磷脂酰肌醇3-激酶/蛋白激酶B信号通路的磷酸化有关。郭晓宇等[72]在大鼠骨髓间充质干细胞模型中发现,淫羊藿苷处理24 h后细胞内碱性磷酸酶基因表达水平、蛋白激酶B磷酸化表达水平升高,表明淫羊藿苷通过激活磷脂酰肌醇3-激酶/蛋白激酶B途径来促进大鼠骨髓间充质干细胞的成骨分化。淫羊藿苷介导磷脂酰肌醇3-激酶/蛋白激酶B信号通路调控骨质疏松症的作用机制,详见图7。
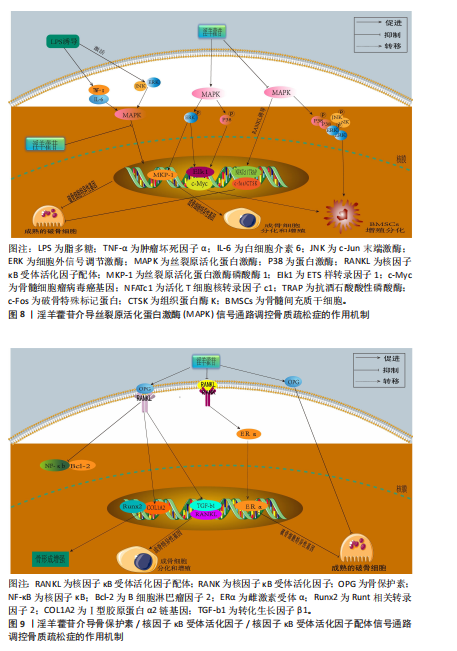
2.3.4 丝裂原活化蛋白激酶信号通路 丝裂原活化蛋白激酶信号通路作为细胞生理学调控及免疫应答的核心信号模块,是真核生物信号转导系统的关键结构单元,该通路通过级联反应介导并调控细胞分化、增殖、细胞凋亡、炎症反应和氧化应激等[73]。现有研究表明,丝裂原活化蛋白激酶信号通路在骨代谢调控网络中发挥核心调控作用,该通路通过精确调控破骨细胞生成关键下游效应分子c-Fos和活化T细胞核转录因子c1的表达活性形成级联调控效应,最终驱动破骨细胞分化进程[74-75]。丝裂原活化蛋白激酶信号转导系统由细胞外信号调节激酶、c-Jun末端激酶和P38丝裂原活化蛋白激酶等平行信号轴构成,其磷酸化参与调控核因子κB受体活化因子配体诱导的破骨细胞生成及骨吸收过程,而丝裂原活化蛋白激酶特异性抑制剂可有效阻断核因子κB受体活化因子配体介导的破骨细胞分化效应[76]。
HSIEH等[77]在脂多糖诱导的破骨细胞模型中发现,淫羊藿苷抑制脂多糖诱导的骨吸收和白细胞介素6和肿瘤坏死因子α表达,并且抑制脂多糖介导的P38和c-Jun末端激酶激活,表明淫羊藿苷通过丝裂原活化蛋白激酶通路抑制破骨细胞分化,防止炎症性骨质流失。DENG等[78]在苯和环磷酰胺诱导再生障碍性贫血大鼠骨髓间充质干细胞模型中发现,淫羊藿苷以剂量依赖性方式促进骨髓间充质干细胞的增殖,下调骨髓间充质干细胞中磷酸化P38/P38、磷酸化c-Jun末端激酶/c-Jun末端激酶和磷酸化细胞外信号调节激酶/细胞外信号调节激酶的蛋白水平,抑制骨髓间充质干细胞的脂肪分化,表明淫羊藿苷通过抑制丝裂原活化蛋白激酶通路促进骨髓间充质干细胞的增殖并抑制细胞凋亡和脂肪生成。QIN等[79]在大鼠骨髓间充质干细胞模型中发现,不同浓度淫羊藿苷处理可显著提高骨髓间充质干细胞中细胞外信号调节激酶和P38激酶的磷酸化,并且上调丝裂原活化蛋白激酶下游转录因子Elk1和骨髓细胞瘤病毒癌基因c-Myc的表达,并且最佳处理质量浓度度为 320 μg/L,表明淫羊藿苷通过激活细胞外信号调节激酶和P38丝裂原活化蛋白激酶信号传导促进大鼠骨髓间充质干细胞的增殖。XU等[80]在核因子κB受体活化因子配体诱导RAW264.7细胞向破骨细胞分化的实验中发现,经用不同浓度(0,10,10和10 mol/L)淫羊藿苷处理后,淫羊藿苷以剂量依赖性方式显著抑制了核因子κB受体活化因子配体诱导的活化T细胞核转录因子c1、c-Fos、抗酒石酸酸性磷酸酶和组织蛋白酶K的表达,表明淫羊藿苷在体外抑制核因子κB受体活化因子配体诱
导的破骨细胞形成和功能,该作用机制是通过抑制丝裂原活化蛋白激酶信号通路的激活来实现的。刘尚全等[81]在经地塞米松诱导的成骨细胞模型中发现,地塞米松促进丝裂原活化蛋白激酶磷酸酶1的表达、抑制丝裂原活化蛋白激酶的磷酸化、减弱丝裂原活化蛋白激酶信号途径的活性,从而抑制了成骨细胞的增殖和分化;淫羊藿苷处理使细胞外信号调节激酶磷酸化增加,同时抑制丝裂原活化蛋白激酶磷酸酶1的表达,表明淫羊藿苷通过丝裂原活化蛋白激酶信号途径促进了细胞的增殖分化,逆转了地塞米松对成骨细胞增殖分化的抑制作用。淫羊藿苷介导丝裂原活化蛋白激酶信号通路调控骨质疏松症的作用机制,详见图8。
2.3.5 骨保护素/核因子κB受体活化因子/核因子κB受体活化因子配体通路 骨保护素作为成骨细胞及骨髓间充质干细胞分泌的关键骨形成调控蛋白,通过拮抗核因子κB受体活化因子配体生物学活性参与破骨细胞分化成熟调控及骨代谢稳态维持。骨保护素/核因子κB受体活化因子配体/核因子κB受体活化因子信号轴构成破骨细胞生成及骨重塑过程的核心调控通路,在破骨细胞功能活化与骨组织动态平衡中发挥枢纽作用[82]。作为骨保护素/核因子κB受体活化因子配体/核因子κB受体活化因子信号轴的核心调控节点,核因子κB受体活化因子配体与其膜受体核因子κB受体活化因子的特异性结合可激活破骨细胞分化成熟程序,通过维持破骨细胞存活状态维持骨吸收活性。在该通路调控网络中,骨保护素作为可溶性诱骗受体,通过竞争性结合核因子κB受体活化因子配体分子有效阻断核因子κB受体活化因子配体-核因子κB受体活化因子信号转导,从而抑制破骨细胞功能活性并诱导其发生程序性死亡,最终实现骨吸收过程的负向调控[83]。因此,骨保护素/核因子κB受体活化因子配体比值是破骨细胞分化、成熟的重要指标,也是保持骨吸收和骨形成平衡状态的关键[84-85]。
ZHANG等[86]在双侧卵巢切除诱导的绝经后骨质疏松性骨折大鼠中发现,淫羊藿苷通过骨保护素/核因子κB受体活化因子配体通路促进Runt相关转录因子2和Ⅰ型胶原蛋白α2链基因的表达水平,抑制血清骨翻转生物标志物的表达水平,表明淫羊藿苷可通过骨保护素/
核因子κB受体活化因子配体信号传导促进去卵巢大鼠骨折愈合。ZHANG等[87]在小鼠成骨细胞MC3T3-E和小鼠破骨细胞RAW264.7共培养系统中发现,一定浓度的淫羊藿苷处理可提高骨保护素、核因子κB受体活化因子配体基因和蛋白的表达,降低核因子κB基因和蛋白的表达,增加碱性磷酸酶、转化生长因子β1和核因子κB受体活化因子配体基因的表达,降低核因子κB受体活化因子基因的表达,表明淫羊藿苷促进MC3T3-E1细胞的成骨活性、抑制RAW264.7 细胞的破骨细胞活性。李伟娟等[88]在核因子κB受体活化因子配体诱导小鼠RAW264.7细胞破骨分化的模型中发现,淫羊藿苷显著抑制核因子κB受体活化因子配体诱导RAW264.7细胞分化成破骨细胞,这种抑制作用可通过升高雌激素受体α表达而直接或间接降低核因子κB受体活化因子表达,进而降低核因子κB受体活化因子配体/核因子κB受体活化因子信号水平,最终降低破骨细胞形成数量和破骨细胞骨吸收活性。吴祖锋等[89]在大鼠双侧卵巢切除构建的骨质疏松症模型中发现,低、中、高剂量(200,400,800 mg/kg)淫羊藿苷灌胃干预可显著提升大鼠骨保护素蛋白表达水平、上调核因子κB受体活化因子配体/核因子κB受体活化因子信号通路活性,进而抑制破骨细胞分化,发挥防治骨质疏松作用。马小妮等[90]在大鼠骨髓间充质干细胞模型中发现,10-5 mol/L淫羊藿苷干预可显著提升骨保护素蛋白表达水平、骨保护素/核因子κB受体活化因子配体比值,通过阻断破骨细胞前体分化及成熟破骨细胞功能活性发挥抗骨吸收效应,表明淫羊藿苷通过调控骨保护素/核因子κB受体活化因子配体信号通路影响骨吸收,从而发挥抗骨质疏松症的作用。淫羊藿苷介导骨保护素/核因子κB受体活化因子/核因子κB受体活化因子配体信号通路调控骨质疏松症的作用机制,详见图9。
| [1] ZOU Z, LIU W, CAO L, et al. Advances in the occurrence and biotherapy of osteoporosis. Biochem Soc Trans. 2020;48(4):1623-1636. [2] ELONHEIMO H, LANGE R, TOLONEN H, et al. Environmental Substances Associated with Osteoporosis-A Scoping Review. Int J Environ Res Public Health. 2021;18(2):738. [3] 中国骨质疏松症流行病学调查及“健康骨骼”专项行动结果发布[J].中华骨质疏松和骨矿盐疾病杂志,2019,12(4):317-318. [4] 中华医学会骨质疏松和骨矿盐疾病分会,章振林.原发性骨质疏松症诊疗指南(2022)[J].中国全科医学,2023,26(14):1671-1691. [5] JOHNSTON CB, DAGAR M. Osteoporosis in Older Adults. Med Clin North Am. 2020;104(5): 873-884. [6] SLEEMAN A, CLEMENTS JN. Abaloparatide: A new pharmacological option for osteoporosis. Am J Health Syst Pharm. 2019;76(3):130-135. [7] MEGAPANOU E, FLORENTIN M, MILIONIS H, et al. Drug-Induced Hypophosphatemia: Current Insights. Drug Saf. 2020;43(3):197-210. [8] SUN P, ZHANG C, HUANG Y, et al. Jiangu granule ameliorated OVX rats bone loss by modulating gut microbiota-SCFAs-Treg/Th17 axis. Biomed Pharmacother. 2022;150:112975. [9] WANG Z, WANG D, YANG D, et al. The effect of icariin on bone metabolism and its potential clinical application. Osteoporos Int. 2018;29(3):535-544. [10] QIAN HQ, WU DC, LI CY, et al. A systematic review of traditional uses, phytochemistry, pharmacology and toxicity of Epimedium koreanum Nakai. J Ethnopharmacol. 2024; 318(Pt B):116957. [11] ZHAO M, XU P, SHI W, et al. Icariin exerts anti-tumor activity by inducing autophagy via AMPK/mTOR/ULK1 pathway in triple-negative breast cancer. Cancer Cell Int. 2024;24(1):74. [12] LEI X, WEN D, HUANG Z, et al. Icariin attenuates oxidative stress via SIRT1/PGC-1α pathway in SAH mice. Exp Neurol. 2025;390:115303. [13] ZHANG X, HAN N, LI G, et al. Local icariin application enhanced periodontal tissue regeneration and relieved local inflammation in a minipig model of periodontitis. Int J Oral Sci. 2018;10(2):19. [14] GAO L, ZHANG SQ. Antiosteoporosis Effects, Pharmacokinetics, and Drug Delivery Systems of Icaritin: Advances and Prospects. Pharmaceuticals (Basel). 2022;15(4):397. [15] SHA M, CAO A, YANG S, et al. [Determination of icariin in Epimedium koreanum nakai by high performance liquid chromatography]. Se Pu. 1997;15(2):166-177. [16] XIAO Q, CHEN A, GUO F. Effects of Icariin on expression of OPN mRNA and type I collagen in rat osteoblasts in vitro. J Huazhong Univ Sci Technolog Med Sci. 2005;25(6):690-702. [17] CHEN KM, GE BF, LIU XY, et al. Icariin inhibits the osteoclast formation induced by RANKL and macrophage-colony stimulating factor in mouse bone marrow culture. Pharmazie. 2007;62(5):388-891. [18] MOK SK, CHEN WF, LAI WP, et al. Icariin protects against bone loss induced by oestrogen deficiency and activates oestrogen receptor-dependent osteoblastic functions in UMR 106 cells. Br J Pharmacol. 2010;159(4):939-949. [19] LIANG W, LIN M, LI X, et al. Icariin promotes bone formation via the BMP-2/Smad4 signal transduction pathway in the hFOB 1.19 human osteoblastic cell line. Int J Mol Med. 2012;30(4):889-895. [20] ZHANG D, FONG C, JIA Z, et al. Icariin Stimulates Differentiation and Suppresses Adipocytic Transdifferentiation of Primary Osteoblasts Through Estrogen Receptor-Mediated Pathway. Calcif Tissue Int. 2016;99(2):187-198. [21] LIANG X, HOU Z, XIE Y, et al. Icariin promotes osteogenic differentiation of bone marrow stromal cells and prevents bone loss in OVX mice via activating autophagy. J Cell Biochem. 2019;120(8):13121-13132. [22] BALLHAUSE TM, JIANG S, XIE W, et al. Fracture healing in a mouse model of Hajdu-Cheney-Syndrome with high turnover osteopenia results in decreased biomechanical stability. Sci Rep. 2023;13(1):11418. [23] SANTOS TA, RIBEIRO JL, BATTISTELLI LS, et al. Limosilactobacillus reuteri with menaquinone-7 improves bone biomechanics and microarchitecture in ovariectomized mice: preliminary study. J Bone Miner Metab. 2025;43(4):335-347. [24] WANIONOK NE, COLAREDA GA, FERNANDEZ JM. Humulus lupulus Promoting Osteoblast Activity and Bone Integrity: Effects and Mechanisms. Biology (Basel). 2025;14(5):582. [25] CLAVIN NW, FERNANDEZ J, SCHÖNMEYR BH, et al. Fractionated doses of ionizing radiation confer protection to mesenchymal stem cell pluripotency. Plast Reconstr Surg. 2008;122(3):739-748. [26] BUDGUDE P, KALE V, VAIDYA A. Microvesicles and exosomes isolated from murine bone marrow-derived mesenchymal stromal cells primed with p38MAPK inhibitor differentially regulate hematopoietic stem cell function. Artif Cells Nanomed Biotechnol. 2025;53(1):122-137. [27] HAN YD, ZHANG HF, XU YT, et al.[Mechanism of icariin in promoting osteogenic differentiation of BMSCs and improving bone metabolism disorders through caveolin-1/Hippo signaling pathway]. Zhongguo Zhong Yao Za Zhi. 2025; 50(3):600-608. [28] CHEN X, LIU X, WAN J, et al. Icariin Facilitates Osteogenic Differentiation and Suppresses Adipogenic Differentiation of Bone Marrow Mesenchymal Stem Cells by Enhancing SOST Methylation in Postmenopausal Osteoporosis. J Gene Med. 2025;27(1):e70010. [29] 金东升,赵张红,朱子银,等.淫羊藿苷缓释微球三维支架对兔骨髓间充质干细胞成骨分化的影响[J].中国组织工程研究,2026, 30(7):1658-1668. [30] 蔡叶,王明飞,张磊,等.淫羊藿苷对衰老骨髓间充质干细胞成骨的影响[J].中国骨质疏松杂志,2023,29(2):162-167. [31] 张锦明,田滢舟,赵玲,等.淫羊藿苷促进骨髓间充质干细胞成骨分化缓解小鼠骨质疏松的机制[J].中国组织工程研究,2022, 26(19):2991-2996. [32] TENG JW, BIAN SS, KONG P, et al. Icariin triggers osteogenic differentiation of bone marrow stem cells by up-regulating miR-335-5p. Exp Cell Res. 2022;414(2):113085. [33] 王想福,孙凤歧,叶丙霖,等.破骨细胞与骨质疏松症的关系研究进展[J].中国骨质疏松杂志,2015,21(11):1420-1424. [34] CHU W, PENG W, LU Y, et al. PRMT6 Epigenetically Drives Metabolic Switch from Fatty Acid Oxidation toward Glycolysis and Promotes Osteoclast Differentiation During Osteoporosis. Adv Sci (Weinh). 2024; 11(40):e2403177. [35] ZHANG J, MAO Y, RAO J. The SPI1/SMAD5 cascade in the promoting effect of icariin on osteogenic differentiation of MC3T3-E1 cells: a mechanism study. J Orthop Surg Res. 2024;19(1):444. [36] ZHU Y, ZHENG T, WEN LM, et al. Osteogenic capability of strontium and icariin-loaded TiO2 nanotube coatings in vitro and in osteoporotic rats. J Biomater Appl. 2021;35(9): 1119-1131. [37] ZHOU L, POON CC, WONG KY, et al. Icariin ameliorates estrogen-deficiency induced bone loss by enhancing IGF-I signaling via its crosstalk with non-genomic ERα signaling. Phytomedicine. 2021;82:153413. [38] 陈绎志,董忠,杨俊,等.淫羊藿苷对兔成骨细胞的调控[J].中国老年学杂志,2023, 43(8):1937-1939. [39] 刘婷,曹春雨,郝然,等.黔岭淫羊藿总黄酮类成分对hFOB1.19人SV40转染成骨细胞活性的影响[J].中国实验方剂学杂志, 2013,19(2):267-270. [40] SONG L, ZHOU Y, QU L, et al. Exploring Effects and Mechanism of Ingredients of Herba Epimedii on Osteogenesis and Osteoclastogenesis In Vitro. Comb Chem High Throughput Screen. 2024;27(19): 2824-2837. [41] ZHANG D, ZHANG J, FONG C, et al. Herba epimedii flavonoids suppress osteoclastic differentiation and bone resorption by inducing G2/M arrest and apoptosis. Biochimie. 2012; 94(12):2514-2522. [42] SI Y, LI Y, GU K, et al. Icariin ameliorates osteoporosis in ovariectomized rats by targeting Cullin 3/Nrf2/OH pathway for osteoclast inhibition. Biomed Pharmacother. 2024;173:116422. [43] CHENG L, JIN X, SHEN H, et al. Icariin attenuates thioacetamideinduced bone loss via the RANKLp38/ERKNFAT signaling pathway. Mol Med Rep. 2022;25(4):126. [44] 朱在师,黄泽灵,蔡伟业,等.基于“阴阳平衡”探讨知母皂苷BⅡ-淫羊藿苷协同对破骨细胞-成骨细胞耦联功能的影响[J].中国实验方剂学杂志,2025,31(18):48-57. [45] 贺龙刚,高奥,邱煌沛,等.淫羊藿次苷Ⅰ及其代谢产物淫羊藿次苷Ⅱ通过AP-1/NFATc1信号通路调控破骨细胞生成[J].中华中医药杂志,2017,32(3):1299-1302. [46] ZHAO Y, LIU J, ZHANG Y, et al. Mir-381-3p aggravates ovariectomy-induced osteoporosis by inhibiting osteogenic differentiation through targeting KLF5/Wnt/β-catenin signaling pathway. J Orthop Surg Res. 2024; 19(1):480. [47] ZHENG S, HU G, ZHENG J, et al. Osthole accelerates osteoporotic fracture healing by inducing the osteogenesis-angiogenesis coupling of BMSCs via the Wnt/β-catenin pathway. Phytother Res. 2024;38(8):4022-4035. [48] PUJIA A, RUSSO C, MAUROTTI S, et al. Bergamot Polyphenol Fraction Exerts Effects on Bone Biology by Activating ERK 1/2 and Wnt/β-Catenin Pathway and Regulating Bone Biomarkers in Bone Cell Cultures. Nutrients. 2018;10(9):1305. [49] HONG G, HE X, SHEN Y, et al. Chrysosplenetin promotes osteoblastogenesis of bone marrow stromal cells via Wnt/β-catenin pathway and enhances osteogenesis in estrogen deficiency-induced bone loss. Stem Cell Res Ther. 2019; 10(1):277. [50] GAO J, XIANG S, WEI X, et al. Icariin Promotes the Osteogenesis of Bone Marrow Mesenchymal Stem Cells through Regulating Sclerostin and Activating the Wnt/β-Catenin Signaling Pathway. Biomed Res Int. 2021;2021:6666836. [51] ZHANG XY, CHEN YP, ZHANG C, et al. Icariin Accelerates Fracture Healing via Activation of the WNT1/β-catenin Osteogenic Signaling Pathway. Curr Pharm Biotechnol. 2020;21(15): 1645-1653. [52] XU YX, WU CL, WU Y, et al. Epimedium-derived flavonoids modulate the balance between osteogenic differentiation and adipogenic differentiation in bone marrow stromal cells of ovariectomized rats via Wnt/β-catenin signal pathway activation. Chin J Integr Med. 2012;18(12):909-917. [53] 杨志烈,王成龙,赵东峰,等.淫羊藿苷对环磷酰胺化疗导致小鼠骨髓间充质干细胞成骨分化障碍的保护作用[J].中国组织工程研究,2016,20(6):777-784. [54] 曾建春,曾意荣,樊粤光,等.淫羊藿甙诱导MSCs向成骨细胞分化过程中对Wnt信号通路的影响[J].广州中医药大学学报,2014, 31(4):607-611,678. [55] ZENG Q, CHEN YP, SONG SL, et al. [Icariin promotes alcohol-inhibited osteogenic differentiation of MC3T3-1-E1 cells by regulating LAP autophagy]. Zhongguo Zhong Yao Za Zhi. 2025;50(3):590-599. [56] ZANOTTI S, CANALIS E. Notch Signaling and the Skeleton. Endocr Rev. 2016;37(3): 223-253. [57] TIAN Y, XU Y, XUE T, et al. Notch activation enhances mesenchymal stem cell sheet osteogenic potential by inhibition of cellular senescence. Cell Death Dis. 2017; 8(2):e2595. [58] TAO ZS, LI TL, XU HG, et al. Hydrogel contained valproic acid accelerates bone-defect repair via activating Notch signaling pathway in ovariectomized rats. J Mater Sci Mater Med. 2021;33(1):4. [59] XU C, DINH VV, KRUSE K, et al. Induction of osteogenesis by bone-targeted Notch activation. Elife. 2022;11:e60183. [60] ZHU Z, WANG Z, MA C, et al. Isopsoralen promotes osteogenic differentiation of human jawbone marrow mesenchymal cells through Notch signaling pathway. Ann Anat. 2023;250:152156. [61] BIAN Q, HUANG JH, LIU SF, et al. Different molecular targets of Icariin on bMSCs in CORT and OVX-rats. Front Biosci (Elite Ed). 2012;4(4): 1224-1236. [62] 邓宇,陈廖斌.淫羊藿苷通过激活Notch信号通路促进骨髓间充质干细胞向成骨细胞分化的实验研究[J].中医学报,2017,32(12): 2393-2398,2403. [63] 徐娅,王攀攀,许青青,等. 淫羊藿苷在促大鼠骨髓间充质干细胞骨向分化过程中对Notch信号通路Notch1、CBF1蛋白表达的影响[C]//第十三次全国中西医结合虚证与老年医学学术研讨会论文集,2013:103-111. [64] 周亚妮,张晓文,王丹.淫羊藿苷抑制Notch信号通路促进骨髓间充质干细胞分化研究[J].商洛学院学报,2024,38(4):70-75. [65] CHANG L, ZHAO D, LIU HB, et al. Activation of sonic hedgehog signaling enhances cell migration and invasion by induction of matrix metalloproteinase-2 and -9 via the phosphoinositide-3 kinase/AKT signaling pathway in glioblastoma. Mol Med Rep. 2015; 12(5):6702-6710. [66] SOLIT DB, BASSO AD, OLSHEN AB, et al. Inhibition of heat shock protein 90 function down-regulates Akt kinase and sensitizes tumors to Taxol. Cancer Res. 2003;63(9): 2139-2144. [67] HUANG H, JI F, QI G, et al. Rehmannioside A promotes the osteoblastic differentiation of MC3T3-E1 cells via the PI3K/AKT signaling pathway and inhibits glucocorticoid-induced bone loss in vivo. J Pharmacol Sci. 2024; 156(4):247-257. [68] XU K, FEI W, GAO W, et al. SOD3 regulates FLT1 to affect bone metabolism by promoting osteogenesis and inhibiting adipogenesis through PI3K/AKT and MAPK pathways. Free Radic Biol Med. 2024;212:65-79. [69] YAO X, JING X, GUO J, et al. Icariin Protects Bone Marrow Mesenchymal Stem Cells Against Iron Overload Induced Dysfunction Through Mitochondrial Fusion and Fission, PI3K/AKT/mTOR and MAPK Pathways. Front Pharmacol. 2019;10:163. [70] JI W, GONG G, LIU Y, et al. Icariin promotes osteogenic differentiation of bone marrow mesenchymal stem cells (BMSCs) by activating PI3K-AKT-UTX/EZH2 signaling in steroid-induced femoral head osteonecrosis. J Orthop Surg Res. 2025;20(1):290. [71] 范凯健,钮艾雯,吴辉辉.淫羊藿苷通过PI3K/AKT信号通路促进MC3T3-E1细胞的增殖分化[J].中南药学,2025,23(2):417-422. [72] 郭晓宇,李唯,陈克明,等.淫羊藿苷通过PI3K/AKT-eNOS信号途径促进大鼠骨髓基质细胞的成骨性分化[J].中国药理学通报, 2013,29(7):966-970. [73] MENG X, ZHANG S. MAPK cascades in plant disease resistance signaling. Annu Rev Phytopathol. 2013;51:245-266. [74] HTIKE K, YOSHIDA K, EGUCHI T, et al. Herbal medicine Ninjinyoeito inhibits RANKL-induced osteoclast differentiation and bone resorption activity by regulating NF-κB and MAPK pathway. J Oral Biosci. 2024;66(4):49-57. [75] LIU C, ZUO M, ZHAO J, et al. DPHB inhibits osteoclastogenesis by suppressing NF-κB and MAPK signaling and alleviates inflammatory bone destruction. Int Immunopharmacol. 2025;152:114377. [76] LI X, GAO C, ZHOU K, et al. Dendrobine Ameliorates Glucocorticoid-Induced Osteoporosis by Promoting Osteogenesis through JNK/p38 MAPK Pathway Activation and GR Nuclear Translocation Inhibition. J Agric Food Chem. 2024;72(30):16739-16748. [77] HSIEH TP, SHEU SY, SUN JS, et al. Icariin inhibits osteoclast differentiation and bone resorption by suppression of MAPKs/NF-κB regulated HIF-1α and PGE(2) synthesis. Phytomedicine. 2011;18(2-3):176-185. [78] DENG S, ZENG Y, XIANG J, et al. Icariin protects bone marrow mesenchymal stem cells in aplastic anemia by targeting MAPK pathway. Mol Biol Rep. 2022;49(9):8317-8324. [79] QIN S , ZHOU W, LIU S, et al. Icariin stimulates the proliferation of rat bone mesenchymal stem cells via ERK and p38 MAPK signaling. Int J Clin Exp Med. 2015;8(5):7125-7133. [80] XU Q, CHEN G, LIU X, et al. Icariin inhibits RANKL-induced osteoclastogenesis via modulation of the NF-κB and MAPK signaling pathways. Biochem Biophys Res Commun. 2019;508(3):902-906. [81] 刘尚全,杨颖,周丽斌,等.淫羊藿甙逆转地塞米松抑制成骨细胞分化及其机制[J].中华内分泌代谢杂志,2006,22(3):218-221. [82] NI X, WU B, LI S, et al. Equol exerts a protective effect on postmenopausal osteoporosis by upregulating OPG/RANKL pathway. Phytomedicine. 2023;108: 154509. [83] 叶子丰,戎宽,张信成,等.基于OPG/RANKL/RANK信号通路探讨益肾健骨方防治绝经后骨质疏松的作用机制[J].中国骨质疏松杂志,2023,29(1):23-28,57. [84] 谢忠建.从OPG-RANKL-RANK通路到骨质疏松症分子靶向治疗[J].中华骨质疏松和骨矿盐疾病杂志,2022,15(2):126-134. [85] 高城翰,刘晓炜,关雪峰.补肾法对去卵巢大鼠OPG/RANK/RANKL信号通路影响的Meta分析[J].中国实验方剂学杂志,2021, 27(20):172-179. [86] ZHANG Y, HAN B, WEI Y, et al. Icariin Promotes Fracture Healing in Ovariectomized Rats. Med Sci Monit. 2020;26:e924554. [87] ZHANG S, FENG P, MO G, et al. Icariin influences adipogenic differentiation of stem cells affected by osteoblast-osteoclast co-culture and clinical research adipogenic. Biomed Pharmacother. 2017;88:436-442. [88] 李伟娟,谢保平,石丽颖,等.从ERα/RANK通路探讨淫羊藿苷抑制破骨细胞分化作用[J].中国实验方剂学杂志,2017, 23(7):121-126. [89] 吴祖锋,袁垒,吴风晴,等.淫羊藿苷对骨质疏松症模型大鼠OPG/RANKL/RANK轴系统影响的实验研究[J].甘肃中医药大学学报, 2016,33(3):4-7. [90] 马小妮,葛宝丰,陈克明,等.淫羊藿苷通过OPG/RANKL信号途径调节骨吸收的机理研究[J].中国骨质疏松杂志,2013,19(1):1-5. |
| [1] | 刘文龙, 董 磊, 肖争争, 聂 宇. 骨质疏松患者行固定平台单髁置换后胫骨假体松动的有限元分析[J]. 中国组织工程研究, 2026, 30(9): 2191-2198. |
| [2] | 吴妍廷, 李 宇, 廖金凤. 氧化镁纳米粒调控成骨与血管生成相关基因表达促进骨缺损愈合[J]. 中国组织工程研究, 2026, 30(8): 1885-1895. |
| [3] | 蒋星海, 宋玉林, 李德津, 邵建敏, 徐军志, 刘华凯, 吴应国, 沈岳辉, 冯思诚. 血管内皮生长因子165基因转染骨髓间充质干细胞构建血管化两亲性肽凝胶模块[J]. 中国组织工程研究, 2026, 30(8): 1903-1911. |
| [4] | 陈豪杰, 王 黛, 沈 山. 种植体周围炎中的免疫炎症微环境机制[J]. 中国组织工程研究, 2026, 30(8): 2054-2062. |
| [5] | 胡雄科, 刘少华, 谭 谦, 刘 昆, 朱光辉. 紫草素干预骨髓间充质干细胞改善老年小鼠股骨的微结构[J]. 中国组织工程研究, 2026, 30(7): 1609-1615. |
| [6] | 宋浦蓁, 马贺宾, 陈宏广, 章亚东. 骨髓间充质干细胞外泌体联合转化生长因子β1对巨噬细胞的作用[J]. 中国组织工程研究, 2026, 30(7): 1616-1623. |
| [7] | 袁小霜, 杨 姁, 杨 波, 陈晓旭, 田 婷, 王飞清, 李艳菊, 刘 洋, 杨文秀. 弥漫性大B细胞淋巴瘤细胞条件培养液对人骨髓间充质干细胞增殖、凋亡的影响[J]. 中国组织工程研究, 2026, 30(7): 1632-1640. |
| [8] | 李镇宇, 张思明, 柏家祥, 朱 晨. 蛇床子素改善高糖环境下骨髓间充质干细胞的成骨分化功能[J]. 中国组织工程研究, 2026, 30(7): 1641-1648. |
| [9] | 韩念荣, 黄异飞, 艾克热木·吾斯曼, 刘岩路, 胡 炜. 高糖微环境中程序性细胞死亡受体1抑制大鼠骨髓间充质干细胞的成骨分化[J]. 中国组织工程研究, 2026, 30(7): 1649-1657. |
| [10] | 金东升, 赵张红, 朱子银, 张 森, 孙祖延, 邓 江. 淫羊藿苷缓释微球三维支架对兔骨髓间充质干细胞成骨分化的影响[J]. 中国组织工程研究, 2026, 30(7): 1658-1668. |
| [11] | 邹玉莲, 陈朝沛, 黄海霞, 兰玉燕, 刘 敏, 黄 婷. 白藜芦醇在炎症微环境下促进骨髓间充质干细胞的成骨分化[J]. 中国组织工程研究, 2026, 30(7): 1669-1678. |
| [12] | 刘安婷, 陆江涛, 张文杰, 贺 玲, 唐宗生, 陈晓玲. 血小板裂解物调控腺苷酸活化蛋白激酶抑制镉诱导的神经细胞凋亡[J]. 中国组织工程研究, 2026, 30(7): 1800-1807. |
| [13] | 彭志伟, 陈 雷, 佟 磊. 木犀草素促进糖尿病小鼠创面愈合的作用与机制[J]. 中国组织工程研究, 2026, 30(6): 1398-1406. |
| [14] | 朱奎成, 杜春燕, 章金涛. 无毛基因突变促进无毛小鼠白色脂肪组织褐变的作用机制[J]. 中国组织工程研究, 2026, 30(6): 1424-1430. |
| [15] | 李 豪, 陶红成, 曾 平, 刘金富, 丁 强, 牛驰程, 黄 凯, 康宏誉. 丝裂原活化蛋白激酶信号通路调控骨关节炎的发生发展:指导中药靶点治疗[J]. 中国组织工程研究, 2026, 30(6): 1476-1485. |
近年来,随着骨组织工程技术的突破性进展,中药活性成分与生物材料支架的复合应用
已成为再生医学领域的前沿方向,尤其在骨骼系统疾病防治中展现出显著转化价值。作为黄酮类化合物的代表性成分,淫羊藿苷在骨质疏松症干预中表现出多靶点调控优势:它的作用机制不仅体现在通过调控成骨-破骨细胞动态平衡来优化骨基质微结构,更可借助材料工程化策略显著增强新生骨组织的生物力学强度。尽管现有文献已对中药单体防治骨质疏松的分子机制进行了系统梳理,但针对淫羊藿苷特异性调控靶点及其在三维支架体系中的递送效能研究存在不足,亟待从维度开展深度解析。
尽管针对淫羊藿苷在骨质疏松症防治中的信号通路研究已积累一定实验数据,但当前研究尚缺乏对该活性成分调控骨代谢相关信号通路的系统性文献回顾。该文拟突破传统综述的横向罗列模式,从分子信号转导的纵向维度切入,重点解析淫羊藿苷通过多通路交互作用调控骨重建的分子机制,并创新性引入骨组织工程视角,探讨载药支架体系对淫羊藿苷靶向递送效率的促进作用及其在体内外实验中对成骨细胞分化的协同效应。这种研究设计既深化了传统中药药理学的机制解析维度,又为骨质疏松症治疗提供了“活性成分-生物材料-信号调控”三位一体的创新研究范式,有助于揭示淫羊藿苷抗骨质疏松作用的新靶点,为开发基于组织工程技术的中药复方制剂奠定一定的理论基础。
中国组织工程研究杂志出版内容重点:干细胞;骨髓干细胞;造血干细胞;脂肪干细胞;肿瘤干细胞;胚胎干细胞;脐带脐血干细胞;干细胞诱导;干细胞分化;组织工程
1.1 资料来源
1.1.1 检索人及检索时间 由第一作者在2025年1月进行检索。
1.1.2 文献检索时限 各数据库建库至2025年6月。
1.1.3 检索数据库 中国知网和PubMed数据库。
1.1.4 检索词 中文检索词为“淫羊藿苷,骨质疏松症,中药复方,发病机制,信号通路,骨髓间充质干细胞,成骨细胞,破骨细胞”,英文检索词为“ Icariin,Osteoporosis,Chinese Medicine compound,Pathogenesis,Signal path,Bmscs,Osteoblast,Osteoclast” ,各中文检索词之间用逻辑词“并且、或者”进行组合,各英文检索词之间用逻辑词“AND或OR”进行组合。
1.1.5 检索文献类型 研究性论文及著作、综述文章、基础实验等。
1.1.6 手工检索情况 无。
1.1.7 检索策略 中国知网和PubMed数据库检索策略详见图2。
1.1.8 检索文献量 初步检索到中英文文献
2 775篇,其中英文文献2 102篇、中文文献673篇。
1.2 入组标准
纳入标准:①实验方法和数据可靠,结果及结论与淫羊藿苷防治骨质疏松症的新方向相关;②涉及淫羊藿苷与Wnt/β-catenin、Notch、磷脂酰肌醇3-激酶/ 蛋白激酶B、丝裂原活化蛋白激酶、骨保护素 /核因子κB受体活化因子/核因子κB受体活化因子配体等相关信号通路的机制研究;③涉及淫羊藿苷防治骨质疏松症的相关性研究。
排除标准:①偏离研究目的的文献;②重复性文献;③详细资料及数据因文献年代久远而无法提供。
1.3 筛选流程与文献质量评价 经系统化文献管理流程,初始检索结果通过EndNote平台整合,剔除重复后获得英文文献823篇、中文文献409篇;基于研究主题相关性、学术时效性及方法学质量三重标准进行二次筛选,剔除不符合要求的文献,最终纳入符合标准的文献90篇(PubMed数据库66篇,中国知网24篇)。文献筛选详见图3。
3.1 既往他人在该领域的贡献和存在的问题 骨代谢作为贯穿生命全周期的动态过程,包含成骨细胞主导的骨形成与破骨细胞介导的骨吸收两个核心环节。当前,骨质疏松症及其并发症已对中老年人群健康构成严重威胁,该病导致的功能受限与医疗负担加剧,促使该疾病演变为全球性的重大健康议题。
随着对淫羊藿苷的研究和认识不断深入,发现淫羊藿苷影响成骨细胞的作用主要是通过调控Wnt/β-catenin、丝裂原活化蛋白激酶和磷脂酰肌醇3-激酶/蛋白激酶B信号通路来实现的。淫羊藿苷影响破骨细胞的作用则主要是通过调控骨保护素/核因子κB受体活化因子配体/核因子κB受体活化因子和Notch信号通路,延缓骨质吸收。此外,淫羊藿苷还可通过影响核因子κB受体活化因子配体-P38/细胞外信号调节激酶-活化T细胞核转录因子信号通路来抑制破骨细胞分化,从而达到保持骨内平衡。当前研究多集中于淫羊藿苷对成骨/破骨细胞标志物的调控作用,机制解析多局限于单一信号通路或特定蛋白表达层面,尚未系统考虑到多个信号途径、氧化应激和炎症反应之间的协同作用。
3.2 该综述区别于他人他篇的特点 通过对国内外相关文献的系统性检索与分类整合,该文全面综述了淫羊藿苷的生物学特性,深入探讨了它对成骨细胞、破骨细胞及骨髓间充质干细胞的调控作用及其相关信号传导机制,发现淫羊藿苷可通过多靶点、多通路协同作用改善骨代谢失衡,其活性成分在骨质疏松症防治领域展现出显著的药理活性,研究结果为该化合物作为新型骨代谢调节剂的临床转化提供了理论依据。通过系统解析淫羊藿苷抗骨质疏松的作用网络,发现它通过介导Wnt/β-catenin、丝裂原活化蛋白激酶、磷脂酰肌醇3-激酶/蛋白激酶B、骨保护素/核因子κB受体活化因子配体/核因子κB受体活化因子及Notch等多条信号轴实现骨代谢调控,其中Wnt/β-catenin通路与骨保护素/核因子κB受体活化因子配体/核因子κB受体活化因子轴构成核心调控机制,通过相互协同作用调节成骨-破骨动态平衡。研究揭示淫羊藿苷并非依赖单一信号通路,而是通过构建多通路协同作用发挥骨保护效应;此外,淫羊藿苷可通过调控mRNAs表达修饰、氧化应激抑制及炎症微环境改善等多维度干预,影响成骨细胞、破骨细胞及骨髓间充质干细胞的生物学行为。这些发现与既往关于淫羊藿苷抗肿瘤、抗炎等药理作用的报道形成有机联系,从骨代谢调控角度拓展了该天然产物的药理学内涵,为后续靶向干预策略的开发提供了多层次的理论参考。
3.3 该综述的局限性 针对淫羊藿主要活性成分淫羊藿苷防治骨质疏松症的研究仍存在以下局限:现有证据多局限于体外细胞模型,体内动物实验证据尚不充分,尤其在氧化应激调控机制的研究方面较为匮乏;淫羊藿苷发挥抗骨质疏松症作用的最适浓度阈值尚未明确,并且是否通过调控特定基因表达介导药效的作用路径仍需深入解析;在淫羊藿苷防治骨质疏松症的研究中,Wnt/β-catenin、Notch、磷脂酰肌醇3-激酶/蛋白激酶B、丝裂原活化蛋白激酶及核因子κB受体活化因子/核因子κB受体活化因子配体/骨保护素等信号通路之间存在复杂的串扰和相互调节机制,需深入解析串扰的分子开关(如关键蛋白磷酸化位点、转录因子竞争)及个体异质性等问题。上述机制性问题的阐明亟需通过构建多维研究体系加以突破,未来研究需进一步通过体内外实验验证及临床转化研究,为淫羊藿苷的临床应用提供科学依据。
3.4 该综述的重要意义 该文系统综述了淫羊藿苷通过靶向相关信号通路防治骨质疏松症的最新研究进展,发现淫羊藿苷可通过抑制骨细胞凋亡进程,并双向调控成骨-破骨细胞动态平衡,从而有效阻遏骨量丢失进程,为骨质疏松症临床干预提供了新型候选策略。值得关注的是,淫羊藿苷与骨组织工程支架等生物材料联用时可产生协同增效作用,在提升疗效的同时显著降低药物相关不良反应风险。上述发现不仅拓展了骨质疏松症治疗策略的研发维度,更提示淫羊藿苷具备向临床转化应用的潜力,但相关作用机制仍需通过多维度实验研究加以深入解析。
3.5 课题专家组建议 该文系统梳理了淫羊藿活性成分淫羊藿苷防治骨质疏松症的分子作用机制,从生物信号调控维度阐释其药效机制,为后续基础研究及临床转化提供理论依据,同时为中医药防治骨质疏松症的机制解析提供创新视角。未来研究需进一步通过体内外实验验证淫羊藿苷的作用路径,探索它与中医药复方或其他活性单体的协同应用潜力;在此基础上,应深入开发其与骨组织工程技术联用的综合干预方案,以期构建更具转化价值的骨质疏松症防治策略。
中国组织工程研究杂志出版内容重点:干细胞;骨髓干细胞;造血干细胞;脂肪干细胞;肿瘤干细胞;胚胎干细胞;脐带脐血干细胞;干细胞诱导;干细胞分化;组织工程
该综述系统阐明了淫羊藿苷通过多通路协同调控防治骨质疏松症的分子机制。研究聚焦五条核心信号通路:骨保护素/核因子κ B受体活化因子/核因子κ B受体活化因子配体、Wnt/β-catenin、磷脂酰肌醇3-激酶/蛋白激酶B、丝裂原活化蛋白激酶和Notch,揭示了淫羊藿苷"双向调控"的独特作用模式。在促进骨形成方面,淫羊藿苷通过激活Wnt/β-catenin通路上调Runx2、Osterix等成骨转录因子,同时激活磷脂酰肌醇3-激酶/蛋白激酶B通路增强成骨细胞存活;在抑制骨吸收方面,通过调节骨保护素/核因子κ B受体活化因子/核因子κ B受体活化因子配体平衡抑制破骨细胞分化,并下调NFATc1、c-Fos等关键转录因子。丝裂原活化蛋白激酶通路介导细胞增殖分化信号,Notch通路参与细胞命运调控,五条通路形成精密的调控网络。本研究创新性地构建了"单体成分-多靶点-信号网络-生物效应"的完整证据链,每条通路均配有清晰的机制图解和规范的三线表数据支撑,为中药单体防治骨质疏松症提供了分子水平的科学依据。研究整合了体外细胞实验、动物模型验证和初步临床观察,具有较强的转化应用价值,为开发新型抗骨质疏松药物和优化临床治疗方案提供了理论指导。
中国组织工程研究杂志出版内容重点:干细胞;骨髓干细胞;造血干细胞;脂肪干细胞;肿瘤干细胞;胚胎干细胞;脐带脐血干细胞;干细胞诱导;干细胞分化;组织工程
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||