中国组织工程研究 ›› 2026, Vol. 30 ›› Issue (35): 9258-9268.doi: 10.12307/2026.278
• 组织构建综述 tissue construction review • 上一篇 下一篇
金属蛋白酶类组织抑制剂功能网络失衡驱动椎间盘退变的分子机制
肖 扬1,高梓博1,胡宇翔1,康智鑫1,张焯铉1,黄澄宇1,刘汯林1,陈 凯1,王洪伸2,李永津2
- 1广州中医药大学第二临床医学院,广东省广州市 510120;2广州中医药大学第二附属医院,广东省广州市 510120
-
收稿日期:2025-09-02修回日期:2025-12-12出版日期:2026-12-18发布日期:2026-04-29 -
通讯作者:王洪伸,主治中医师,博士后,广州中医药大学第二附属医院,广东省广州市 510120 通讯作者:李永津,主任中医师,副院长,广州中医药大学第二附属医院,广东省广州市 510120 -
作者简介:肖扬,男,2000年生,广东省广州市人,汉族,广州中医药大学在读硕士,主要从事脊柱疾病的基础与临床研究。 -
基金资助:广东省中医院拔尖人才资助专项(BJ2022YL07),项目负责人:李永津;国家自然科学基金面上项目(82274554),项目负责人:李永津;广东省科技计划项目(2023B1212060063),项目负责人:李永津;广州中医药大学第二附属医院(广东省中医院)中医药科学技术研究(YN2020QN14),项目负责人:王洪伸;广东省基础与应用基础研究基金面上项目(2024A1515010981),项目负责人:王洪伸
Molecular mechanism by which the imbalance of the functional network of tissue inhibitors of metalloproteinases drives intervertebral disc degeneration
Xiao Yang1, Gao Zibo1, Hu Yuxiang1, Kang Zhixin1, Zhang Chaoxuan1, Huang Chengyu1, Liu Honglin1, Chen Kai1, Wang Hongshen2, Li Yongjin2
- 1The Second Clinical College of Guangzhou University of Chinese Medicine, Guangzhou 510120, Guangdong Province, China; 2The Second Affiliated Hospital of Guangzhou University of Chinese Medicine, Guangzhou 510120, Guangdong Province, China
-
Received:2025-09-02Revised:2025-12-12Online:2026-12-18Published:2026-04-29 -
Contact:Wang Hongshen, Attending physician, MD, The Second Affiliated Hospital of Guangzhou University of Chinese Medicine, Guangzhou 510120, Guangdong Province, China Co-corresponding author: Li Yongjin, Chief physician, The Second Affiliated Hospital of Guangzhou University of Chinese Medicine, Guangzhou 510120, Guangdong Province, China -
About author:Xiao Yang, MS candidate, The Second Clinical College of Guangzhou University of Chinese Medicine, Guangzhou 510120, Guangdong Province, China -
Supported by:Top Talent Support Program of Guangdong Provincial Hospital of Chinese Medicine, No. BJ2022YL07 (to LYJ); National Natural Science Foundation of China (General Program), No. 82274554 (to LYJ); Guangdong Provincial Science and Technology Plan Project, No. 2023B1212060063 (to LYJ); Traditional Chinese Medicine Science and Technology Research Program of The Second Affiliated Hospital of Guangzhou University of Chinese Medicine (Guangdong Provincial Hospital of Chinese Medicine), No. YN2020QN14 (to WHS); Guangdong Basic and Applied Basic Research Foundation (General Program), No. 2024A1515010981 (to WHS)
摘要:
文题释义:
细胞外基质代谢:细胞外基质代谢通过动态平衡胶原蛋白等成分的合成、重塑与降解,维持组织结构和信号传递功能;靶向调控细胞外基质代谢正成为治疗脊柱退行性疾病的关键策略。
金属蛋白酶组织抑制剂:是基质金属蛋白酶的内源性特异性抑制因子,该家族目前包括金属蛋白酶组织抑制剂1、金属蛋白酶组织抑制剂2、金属蛋白酶组织抑制剂3、金属蛋白酶组织抑制剂4。通过调控基质金属蛋白酶活性参与细胞外基质代谢、组织重塑等生理病理过程,成为干预细胞外基质代谢紊乱的关键靶点。
背景:椎间盘退变是盘源性疾病的核心病理机制,核心特征为细胞外基质代谢失衡。金属蛋白酶组织抑制剂作为基质金属蛋白酶的内源性拮抗剂,通过调控细胞外基质稳态在椎间盘退变中发挥关键作用,但各亚型特异性功能、信号通路交互及表观遗传调控机制尚未系统阐明。
目的:综述金属蛋白酶组织抑制剂在椎间盘退变过程中的表达变化、功能异质性及调控网络,重点阐明金属蛋白酶组织抑制剂在氧化应激、机械负荷及炎症微环境中的分子机制和信号通路,并评估基于金属蛋白酶组织抑制剂基因治疗等策略的转化潜力。
方法:由第一作者检索PubMed、Web of Science、Embase、中国知网、万方等数据库,文献检索时限为各数据库建库至2025年3月,以“椎间盘退变,椎间盘退行性变,椎间盘退化,金属蛋白酶类组织抑制剂,信号通路”为中文检索词,以“Tissue Inhibitor of Metalloproteinases,Tissue Inhibitor of Metalloproteinase,TIMPs,Intervertebral disc degeneration,Disc degeneration,Degenerative Disc Disease,Degenerative Intervertebral Discs”为英文检索词,最终选取符合标准的76篇文献进行综述。
结果与结论:①金属蛋白酶组织抑制剂亚型功能:金属蛋白酶组织抑制剂1呈双向调节(早期保护/晚期耗竭);金属蛋白酶组织抑制剂2通过抑制基质金属蛋白酶活性维持细胞外基质稳态,其异常表达可激活促凋亡信号通路(如miR-185-5p/基质金属蛋白酶2轴及炎症因子介导的基质金属蛋白酶/金属蛋白酶组织抑制剂失衡);金属蛋白酶组织抑制剂3通过抑制基质金属蛋白酶活性、肿瘤坏死因子α转化酶/肿瘤坏死因子α轴及血管新生发挥多维度保护作用;金属蛋白酶组织抑制剂4受miR-155-5p/成纤维细胞生长因子2调控参与细胞外基质稳态;②表观遗传重编程机制:异常机械应力通过WTAP/YTH结构域家族蛋白2-m6A轴降解金属蛋白酶组织抑制剂3 mRNA,而miR-222靶向抑制金属蛋白酶组织抑制剂3协同加速细胞外基质降解;③多模式治疗策略:光生物调节(波长特异性调控金属蛋白酶组织抑制剂/基质金属蛋白酶)、干细胞外泌体(miR-199a/GREM1轴)及鸢尾素干预可重构基质代谢平衡。此综述总结“金属蛋白酶组织抑制剂功能网络失衡”理论框架,揭示其作为椎间盘退变核心驱动因素的多层次调控特性,为开发靶向表观遗传修饰、力学-生物学耦合干预的精准治疗提供理论依据。
https://orcid.org/0009-0009-9819-4758(肖扬);https://orcid.org/0000-0002-8712-1432(王洪伸);
https://orcid.org/0000-0001-8561-3380(李永津)
中国组织工程研究杂志出版内容重点:干细胞;骨髓干细胞;造血干细胞;脂肪干细胞;肿瘤干细胞;胚胎干细胞;脐带脐血干细胞;干细胞诱导;干细胞分化;组织工程
中图分类号:
引用本文
肖 扬, 高梓博, 胡宇翔, 康智鑫, 张焯铉, 黄澄宇, 刘汯林, 陈 凯, 王洪伸, 李永津. 金属蛋白酶类组织抑制剂功能网络失衡驱动椎间盘退变的分子机制[J]. 中国组织工程研究, 2026, 30(35): 9258-9268.
Xiao Yang, Gao Zibo, Hu Yuxiang, Kang Zhixin, Zhang Chaoxuan, Huang Chengyu, Liu Honglin, Chen Kai, Wang Hongshen, Li Yongjin. Molecular mechanism by which the imbalance of the functional network of tissue inhibitors of metalloproteinases drives intervertebral disc degeneration[J]. Chinese Journal of Tissue Engineering Research, 2026, 30(35): 9258-9268.
2.2 金属蛋白酶组织抑制剂各型作用机制的特点
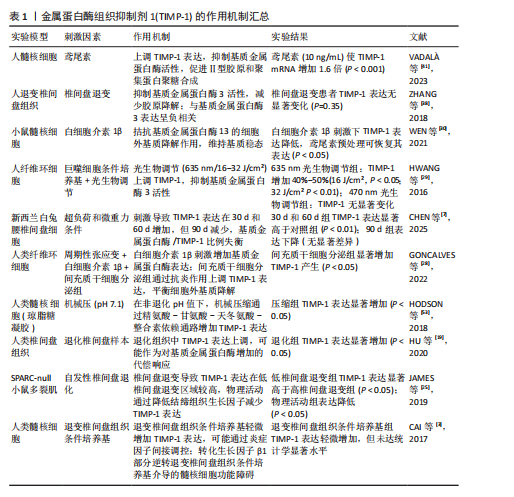
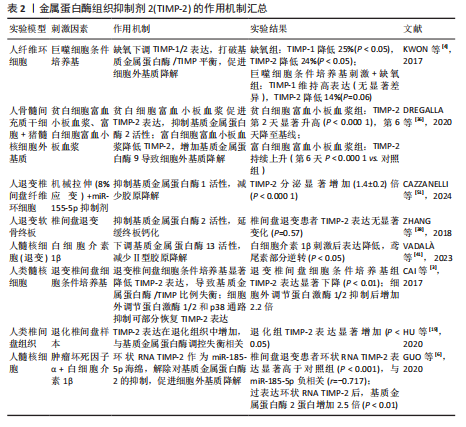
2.2.1 金属蛋白酶组织抑制剂1的双向调节特征 金属蛋白酶组织抑制剂1作为基质金属蛋白酶的抑制剂,最主要的功能是抑制细胞外基质降解。研究显示,通过基因治疗提高椎间盘内金属蛋白酶组织抑制剂1的表达,能够有效减缓椎间盘退变的进程。例如,一项兔模型体内研究发现,通过慢病毒载体介导的转化生长因子β3-金属蛋白酶组织抑制剂1基因转导,能够显著减缓椎间盘退变[18]。另一项研究进一步证实了慢病毒载体递送survivin-转化生长因子β3-金属蛋白酶组织抑制剂1基因,可以减缓椎间盘退变[14]。这表明金属蛋白酶组织抑制剂1在维持细胞外基质稳定性方面发挥着保护作用。尽管金属蛋白酶组织抑制剂1通常被认为是细胞外基质的保护者,但最新研究揭示了金属蛋白酶组织抑制剂1在椎间盘退变中可能存在双重角色,尤其是在氧化应激和炎症背景下。一项研究表明,YTH结构域家族蛋白2依赖的叉头转录因子O3 mRNA的m6A修饰介导了金属蛋白酶组织抑制剂1的表达,并促进了活性氧刺激后的椎间盘退变。在衰老小鼠的椎间盘中活性氧水平升高,而局部施用过氧化氢也会诱导活性氧积累和椎间盘退变。当活性氧水平升高时,YTH结构域家族蛋白2依赖的叉头转录因子O3 mRNA m6A修饰增加,进而促进金属蛋白酶组织抑制剂1的表达。这意味着,在氧化应激条件下,金属蛋白酶组织抑制剂1的表达可能不是单纯的保护性反应,而是参与到活性氧诱导的退变级联中,这种情况下金属蛋白酶组织抑制剂1可能通过影响细胞的凋亡或炎症反应来间接促进退变,形成一个复杂的反馈循环[8]。综上所述,金属蛋白酶组织抑制剂1在椎间盘退变中表现出保护性抑制细胞外基质降解与氧化应激下潜在促退变作用的双重角色,同时金属蛋白酶组织抑制剂1在椎间盘退变进程中呈现动态变化特征[7]。研究指出,在退变椎间盘组织中金属蛋白酶组织抑制剂1表达显著升高,进而抑制基质金属蛋白酶活性,减少细胞外基质的过度降解,从而延缓椎间盘退变进程。生物信息学分析显示金属蛋白酶组织抑制剂1作为蛋白互作网络中的核心基因,与细胞外基质重构和磷脂酰肌醇3激酶/蛋白激酶B信号通路密切相关,表明它参与椎间盘退变的关键调控机制[19]。在人退变椎间盘组织样本中进一步证实,金属蛋白酶组织抑制剂1水平与患者年龄及退变程度呈正相关[20]。有研究通过免疫组化检测157例腰椎手术患者髓核组织发现,金属蛋白酶组织抑制剂1表达与Videman评分呈负相关(r=-0.961,P < 0.05),提示金属蛋白酶组织抑制剂1水平与椎间盘退变程度呈负相关[21]。椎间盘退变患者椎间盘髓核及纤维环中基质金属蛋白酶1、金属蛋白酶组织抑制剂1的表达明显增高,对协助诊断椎间盘退变具有重要临床意义[22]。在腰椎间盘突出症患者中,术前血清基质金属蛋白酶9水
平显著低于健康对照组,而金属蛋白酶组织抑制剂1水平显著升高,导致基质金属蛋白酶9/金属蛋白酶组织抑制剂1比值明显降低。术后3个月随访显示,金属蛋白酶组织抑制剂1水平进一步升高,而基质金属蛋白酶9水平无显著变化,基质金属蛋白酶9/金属蛋白酶组织抑制剂1比值持续下降,表明在疾病进程中金属蛋白酶组织抑制剂1的代偿性升高能通过抑制基质金属蛋白酶9活性参与了椎间盘退变过程[23]。还有研究发现金属蛋白酶组织抑制剂1作为抗分解代谢蛋白,能够抑制基质金属蛋白酶3的酶活性。在衰老髓核组织中,免疫组化和Western blot检测显示衰老相关分泌表型(基质金属蛋白酶3、基质金属蛋白酶13、白细胞介素6、白细胞介素8)表达显著升高。蛋白互作分析揭示了基质金属蛋白酶3与金属蛋白酶组织抑制剂1等蛋白的相互作用关系[24]。还有研究从力学角度证实,椎间盘退变伴随多裂肌中金属蛋白酶组织抑制剂1/2降低和结缔组织生长因子介导的纤维化,但物理活动可通过抑制结缔组织生长因子/基质金属蛋白酶2/金属蛋白酶组织抑制剂2通路改善细胞外基质代谢失衡[25]。
多项研究探索调控金属蛋白酶组织抑制剂/基质金属蛋白酶平衡改善椎间盘退变的具体途径。有研究通过电针刺激腰椎夹脊穴,可显著上调大鼠退变椎间盘组织中的金属蛋白酶组织抑制剂1 mRNA及蛋白表达,并抑制基质金属蛋白酶3活性,证明针灸调控金属蛋白酶组织抑制剂/基质金属蛋白酶平衡保护细胞外基质的机制[26]。另一项研究发现术后复发患者的髓核组织金属蛋白酶组织抑制剂1表达水平显著低于未复发组,表明金属蛋白酶组织抑制剂1表达降低与椎间盘退变存在显著相关性。研究进一步证实,金属蛋白酶组织抑制剂1作为基质金属蛋白酶组织抑制因子,其水平下降可能通过破坏细胞外基质代谢平衡,加剧胶原降解,从而导致椎间盘突出术后复发,低表达金属蛋白酶组织抑制剂1难以抑制基质金属蛋白酶的活性失控,加速基质降解,是椎间盘突出术后复发的独立预测因子[27]。类似地,研究者发现白细胞介素1β联合机械负荷可显著上调人纤维环细胞中的基质金属蛋白酶1、基质金属蛋白酶3及炎症因子(白细胞介素6、白细胞介素8、前列腺素E2)表达,同时抑制Ⅰ型胶原α1表达。还有实验表明,间充质干细胞通过促进金属蛋白酶组织抑制剂1的表达,选择性抑制基质金属蛋白酶1/2/3/9的活性,而非直接调控炎症因子,从而维持细胞外基质稳态[28]。光生物调节治疗显示波长特异性效应:生物调节疗法在635 nm波长和16,32 J/cm2能量密度下显著促进金属蛋白酶组织抑制剂1合成,而在470 nm波长和16,32,64 J/cm2能量密度下显著下调基质金属蛋白酶1表达,表明光具备调节金属蛋白酶组织抑制剂1与基质金属蛋白酶1表达的能力,有助于重构金属蛋白酶组织抑制剂与基质金属蛋白酶之间平衡状态;生物调节疗法能够降低细胞外基质降解速度,从而延缓椎间盘退变[29]。研究表明,骨髓间充质干细胞来源细胞外囊泡通过传递miR-199a靶向抑制GREM1表达,进而阻断转化生长因子β信号通路激活,从而显著降低基质金属蛋白酶2和基质金属蛋白酶6的表达水平,同时促进细胞外基质关键成分Ⅱ型胶原和蛋白聚糖的合成。值得注意的是,实验数据显示细胞外囊泡处理组的金属蛋白酶组织抑制剂1表达亦呈下降趋势,提示骨髓间充质干细胞来源细胞外囊泡对椎间盘退变的保护作用可能存在独立于金属蛋白酶组织抑制剂1调控分子机制[30]。TSENG等[31]指出,金属蛋白酶组织抑制剂1和基质金属蛋白酶9在外周神经损伤模型中显著上调,显示金属蛋白酶组织抑制剂在调节细胞外基质的降解和修复过程中发挥重要作用。MISHRA等[32]研究表明,金属蛋白酶组织抑制剂1在脊髓损伤后显著上调,并通过基质金属蛋白酶依赖和非依赖路径发挥作用,直接减少细胞外基质的炎症和破坏,帮助维持血脊髓屏障,减少免疫细胞浸润,这进一步强调了金属蛋白酶组织抑制剂在细胞外基质代谢中的关键角色。QUAN等[33]研究使用佛波酯处理显著增加了金属蛋白酶组织抑制剂1的表达,但基质金属蛋白酶13的表达未发生改变。尽管金属蛋白酶组织抑制剂1水平升高,但由于β-catenin通过瞬时感受器电位香草酸受体4通道激活导致基质金属蛋白酶1和含血小板反应蛋白基序的解整合素样金属蛋白酶5表达增加,同时核因子κB介导的白细胞介素6和前列腺素E2升高,最终未能有效抑制髓核细胞的退变。
2.2.2 金属蛋白酶组织抑制剂2的促凋亡机制解析 金属蛋白酶组织抑制剂2在椎间盘退变中展现出独特的双向调控特性,其核心功能通过基质金属蛋白酶/金属蛋白酶组织抑制剂网络实现:通过抑制基质金属蛋白酶活性维持细胞外基质稳态,而异常表达时则触发促凋亡信号通路。值得注意的是,研究发现环状RNA 金属蛋白酶组织抑制剂2可通过吸附miR-185-5p解除对基质金属蛋白酶2的抑制,导致细胞外基质降解加速[6],这种miRNA介导的调控机制在炎症微环境中尤为显著。基质金属蛋白酶/金属蛋白酶组织抑制剂动态平衡在病理条件下更易被打破,如研究者观察到缺氧环境会显著降低金属蛋白酶组织抑制剂1/2表达,同时促使基质金属蛋白酶1水平上升[4],提示微环境调控网络的关键性作用。也有研究探讨脊髓刺激慢性背痛患者基质金属蛋白酶2和金属蛋白酶组织抑制剂2水平显著升高,提示这些因子在神经性疼痛中具有重要作用[34]。还有研究发现基质金属蛋白酶2和金属蛋白酶组织抑制剂2的活性变化与椎间盘退变及慢性背痛相关,进一步证明基质金属蛋白酶2和金属蛋白酶组织抑制剂2在椎间盘退变中具有重要作用[35]。
针对金属蛋白酶组织抑制剂2的双向调控特征,研究者开发出多种靶向干预手段。研究者证实470 nm的波长特异性光可选择性增强金属蛋白酶组织抑制剂2表达,抑制基质金属蛋白酶1生成[29],这种物理干预策略为精准调节基质金属蛋白酶/金属蛋白酶组织抑制剂平衡提供新思路。在生物治疗领域,研究者发现低白细胞富集血小板能促进间充质干细胞合成透明质酸,并通过上调基质金属蛋白酶2实现组织修复[36],该过程可能涉及金属蛋白酶组织抑制剂2在局部微环境中的动态再平衡机制。胶原酶联合注射通过提升金属蛋白酶组织抑制剂2水平,抑制基质金属蛋白酶9活性,为纤维环修复提供新策略[37]。这些研究均表明调控金属蛋白酶组织抑制剂家族成员的表达平衡可以实现椎间盘退变的靶向治疗。
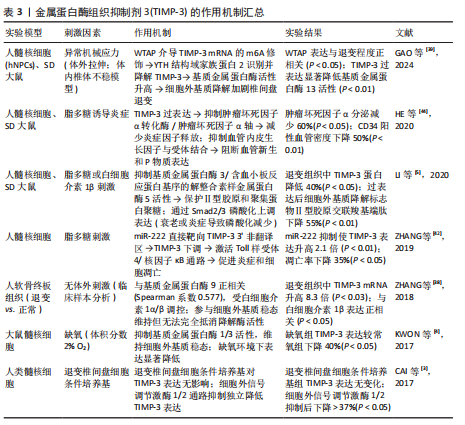
2.2.3 金属蛋白酶组织抑制剂3的多维调控网络 金属蛋白酶组织抑制剂3在椎间盘退变中的功能调控呈现多维度特征,其表达水平受表观遗传和分子信号网络的精细调节。研究人员研究了退变软骨终板样本中基质金属蛋白酶和金属蛋白酶组织抑制剂的表达,发现基质金属蛋白酶3、基质金属蛋白酶9和金属蛋白酶组织抑制剂3的升高与椎间盘退变密切相关,提示这些分子在细胞外基质降解中起重要作用[38]。在异常应力条件下,椎间盘细胞中的WTAP表达上调,导致金属蛋白酶组织抑制剂3 mRNA的m6A修饰增加,这种修饰让YTH结构域家族蛋白2能够识别并结合金属蛋白酶组织抑制剂3 mRNA,从而促进其降解。金属蛋白酶组织抑制剂3表达降低导致基质金属蛋白酶活性增加,进而加速了细胞外基质的降解,促进了椎间盘退变[39]。实验证实,Fibulin-3通过上调金属蛋白酶组织抑制剂3的表达显著抑制基质金属蛋白酶活性,减少胶原和蛋白聚糖降解,从而延缓髓核细胞衰老,表明金属蛋白酶组织抑制剂3是椎间盘退变的关键调控因子[40]。另外有研究证实鸢尾素能够显著增加人椎间盘细胞的增殖、糖胺聚糖含量和代谢活性,并促进金属蛋白酶组织抑制剂3基因表达,进而减少基质金属蛋白酶13和白细胞介素1β的mRNA水平,从而抑制细胞外基质降解[41],有助于维持椎间盘细胞的基质合成和抑制炎症反应。值得注意的是,金属蛋白酶组织抑制剂3的表达还受miR-222和P14ARF的双向调控,前者证明miR-222在椎间盘退变组织中高表达,并与临床MRI分级呈正相关。miR-222通过直接靶向金属蛋白酶组织抑制剂3的3’非翻译区,降低金属蛋白酶组织抑制剂3的表达,从而促进炎症因子(如肿瘤坏死因子α、白细胞介素1β和白细胞介素6)的产生和细胞凋亡。这些结果表明,miR-222通过靶向金属蛋白酶组织抑制剂3,促进了椎间盘退变中的炎症和细胞凋亡[42],后者证明金属蛋白酶组织抑制剂3在椎间盘源性疼痛中表达下调,与炎症和血管新生密切相关[43]。研究表明,通过腺病毒载体上调金属蛋白酶组织抑制剂3的表达,能够抑制内皮细胞的迁移和管状结构形成,减少血管内皮生长因子的分泌,从而抑制血管新生[44]。金属蛋白酶组织抑制剂3还能够降低肿瘤坏死因子α和P物质的表达,减轻炎症反应。研究人员发现,局部炎症活动失调与多裂肌内脂肪积累相关,可能导致椎间盘退变及损伤,揭示了炎症在椎间盘退变中的作用[45]。椎间盘退变的分子机制研究揭示:在异常机械应力作用下,WTAP介导的m6A甲基化修饰通过促进YTH结构域家族蛋白2依赖性金属蛋白酶组织抑制剂3 mRNA降解,导致基质金属蛋白酶活性失控,进而引发细胞外基质过度分解。值得注意的是,miR-222通过靶向金属蛋白酶组织抑制剂3的3’非翻译区域协同激活核因子κB炎症通路,而P14ARF基因表现出促凋亡(caspase-3激活)和促血管生成(血管内皮生长因子上调)双重效应。临床相关性分析显示多裂肌脂肪浸润程度与退变分级显著正相关,提示力学微环境调控的重要性。基于此,采用CRISPR-Cas9介导的miR-222基因沉默、重组鸢尾素靶向给药系统(聚乳酸-羟基乙酸共聚物纳米载体)及3D打印仿生水凝胶支架(甲基丙烯酰化明胶/甲基丙烯酸酯透明质酸复合体系)3种技术路径策略,为椎间盘退变的精准治疗提供理论依据和转化路径。研究指出,在未暴露促炎细胞因子的情况下,金属蛋白酶组织抑制剂1和金属蛋白酶组织抑制剂2的表达显著低于金属蛋白酶组织抑制剂3,而促炎因子(白细胞介素1β和肿瘤坏死因子)的暴露会削弱金属蛋白酶组织抑制剂对基质金属蛋白酶3的抑制作用,尤其在营养缺乏条件下更为明显。金属蛋白酶组织抑制剂3对含血小板反应蛋白基序的解整合素样金属蛋白酶4的抑制特异性更强,提示其在维持细胞外基质稳态中起关键作用[46]。金属蛋白酶组织抑制剂家族,尤其是金属蛋白酶组织抑制剂3通过选择性抑制含血小板反应蛋白基序的解整合素样金属蛋白酶4/5的活性,调控蛋白聚糖降解过程。在椎间盘退变中,含血小板反应蛋白基序的解整合素样金属蛋白酶与金属蛋白酶组织抑制剂3的表达失衡加剧细胞外基质分解,而靶向金属蛋白酶组织抑制剂的调控可能成为干预椎间盘退变新策略[47]。
在炎症调控方面,金属蛋白酶组织抑制剂3通过多重机制发挥保护作用[48]。金属蛋白酶组织抑制剂3表达下调会导致肿瘤坏死因子α转化酶/肿瘤坏死因子α轴激活,退变组织中基质金属蛋白酶3与白细胞介素1α存在正相关,而金属蛋白酶组织抑制剂3与基质金属蛋白酶9及白细胞介素1β呈现协同上调现象,证实炎症因子可通过重塑基质金属蛋白酶/金属蛋白酶组织抑制剂平衡激活凋亡通路[38]。另有研究同样证实金属蛋白酶组织抑制剂3缺失导致肿瘤坏死因子α转化酶/肿瘤坏死因子α轴的表达上调,减弱了对基质金属蛋白酶3的抑制作用,从而促进了炎症反应和基质降解[49]。另有研究发现miR-222抑制剂能够上调金属蛋白酶组织抑制剂3的表达,减少炎症反应和细胞凋亡[42]。还有研究发现这种炎症调控与细胞外基质稳态维持密切相关:金属蛋白酶组织抑制剂3不仅直接抑制基质金属蛋白酶9/13活性延缓基质降解[39,41],还能通过Smad2/3磷酸化调控维持基质合成[5]。
金属蛋白酶组织抑制剂3的另一重要功能维度是抑制血管新生。金属蛋白酶组织抑制剂3能够抑制内皮细胞的迁移和管状结构形成,减少血管内皮生长因子与其受体的结合,从而抑制血管新生。上述发现表明,金属蛋白酶组织抑制剂3的功能失调能引起椎间盘退变,有望成为潜在治疗靶点[5]。
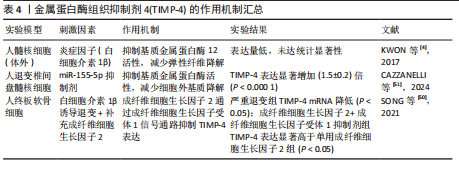
2.2.4 金属蛋白酶组织抑制剂4:被忽视的调节者 以往研究主要集中于金属蛋白酶组织抑制剂1、金属蛋白酶组织抑制剂2和金属蛋白酶组织抑制剂3的生物学作用,而对金属蛋白酶组织抑制剂4的调控机制及其在细胞外基质动态平衡中的作用仍缺乏深入探讨。近年研究表明,金属蛋白酶组织抑制剂4在软骨细胞的合成和分解代谢中起关键作用。成纤维细胞生长因子2通过增加金属蛋白酶组织抑制剂4的表达,抑制基质金属蛋白酶13的活性,从而维持细胞外基质的稳定性。这些发现为理解成纤维细胞生长因子2调控金属蛋白酶组织抑制剂4在软骨细胞退变中的作用重要依据[50]。同时,金属蛋白酶组织抑制剂4的表达可受多种因子调控,miR-155-5p通过介导炎症反应与机械应力的协同作用加剧椎间盘退变,抑制miR-155-5p可显著上调金属蛋白酶组织抑制剂4表达, 从而抑制基质金属蛋白酶活性并减少细胞外基质降解,其靶向抑制或为延缓退变的潜在治疗策略[51] 。这些发现表明,金属蛋白酶组织抑制剂4表达水平不仅与退变程度负相关,并可能通过miR-155-5p-金属蛋白酶组织抑制剂4-基质金属蛋白酶轴和成纤维细胞生长因子2-金属蛋白酶组织抑制剂4-基质金属蛋白酶13通路双重机制,对细胞外基质重塑和软骨细胞功能维持产生重要调节作用。未来研究需进一步阐明金属蛋白酶组织抑制剂4在椎间盘退变中的时空特异性表达规律及其与不同基质金属蛋白酶亚型的相互作用网络,为开发靶向细胞外基质代谢的干预策略提供依据。
2.2.5 金属蛋白酶组织抑制剂各亚型的关系与协同效应 椎间盘退变基质代谢失衡与金属蛋白酶组织抑制剂/基质金属蛋白酶动态平衡破坏具有重要关联,在炎症因子(如白细胞介素1β、肿瘤坏死因子α)持续刺激下,金属蛋白酶组织抑制剂1的表达虽短期内反馈性上调,但最终因氧化应激和促分解代谢信号(如核因子κB通路激活)导致功能代偿失调[20],这种动态失衡在缺氧模型中得到验证:基质金属蛋白酶1在纤维环细胞中增加,而金属蛋白酶组织抑制剂1/2降低;基质金属蛋白酶3在髓核细胞中增加,而金属蛋白酶组织抑制剂1/2降低,这种微环境改变可产生级联效应[4]。还有研究者发现退变细胞可以通过旁分泌肿瘤坏死因子α/白细胞介素1β来上调健康髓核细胞的基质金属蛋白酶1/3/13表达,而转化生长因子β1能逆转此效应并激活金属蛋白酶组织抑制剂1/2/3[3]。在治疗策略研究领域,天然活性分子展现出多靶点调控优势。例如,研究者证实鸢尾素可显著上调人类髓核细胞中金属蛋白酶组织抑制剂1和金属蛋白酶组织抑制剂3的mRNA表达,同时下调基质金属蛋白酶13和白细胞介素1β的mRNA表达,从而抑制细胞外基质分解代谢并促进合成代谢延缓椎间盘退变[41]。类似地,淫羊藿苷通过上调金属蛋白酶组织抑制剂1/2抑制基质金属蛋白酶3/9活性,多靶点调控细胞外基质代谢平衡[20]。研究发现,犬退变椎间盘中金属蛋白酶组织抑制剂1/2/3表达均显著升高,而突出椎间盘中金属蛋白酶组织抑制剂1持续上调但金属蛋白酶组织抑制剂3无变化。金属蛋白酶组织抑制剂1的异常升高可能与含血小板反应蛋白基序的解整合素样金属蛋白酶4/5活性失衡相关,提示不同金属蛋白酶组织抑制剂成员在退变和突出阶段具有差异性的调控作用[52]。这些研究为开发基于金属蛋白酶组织抑制剂/基质金属蛋白酶稳态调控网络的精准治疗靶点提供了重要依据。
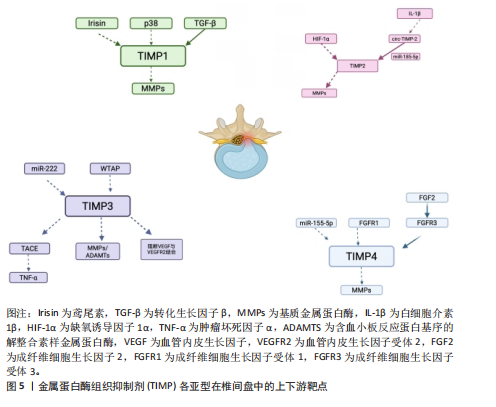
2.2.6 椎间盘退变的治疗新策略 深入研究金属蛋白酶组织抑制剂机制能够为开发新的椎间盘退变治疗策略提供基础。例如,基因疗法用于减缓椎间盘退变进程,其中转化生长因子β3、结缔组织生长因子和金属蛋白酶组织抑制剂1等能够促进退变椎间盘的细胞外基质合成,从而延缓退变[14,18],同时miRNA也在椎间盘细胞的炎症反应和机械感知中发挥重要作用,为靶向治疗提供了新的思路。此外,有研究表明,胞磷胆碱能降低基质金属蛋白酶2和基质金属蛋白酶9水平和活性,并显著提高金属蛋白酶组织抑制剂1和金属蛋白酶组织抑制剂3水平,为理解椎间盘退变的信号通路提供了新的视角[12]。骨髓间充质干细胞分泌的细胞外囊泡已被证实可以通过调节椎间盘细胞凋亡来保护椎间盘,促进组织修复[30]。这些椎间盘退变治疗策略的有效性依赖于对椎间盘退变机制的深入研究,实现从基础研究到临床应用的转化,最终实现提高患者临床疗效提高的目标[53]。金属蛋白酶组织抑制剂各亚型在椎间盘中的上下游靶点,见图5。
2.3 金属蛋白酶组织抑制剂的信号通路机制
2.3.1 核因子κB信号通路 研究显示,核因子κB信号通路在椎间盘退变中发挥着重要的调控作用[54-58]。研究通过TMT定量蛋白质组学分析发现,退变髓核组织中金属蛋白酶组织抑制剂1表达显著上调,同时核因子κB转录因子被鉴定为差异表达蛋白相关的关键分子。退变髓核细胞中基质金属蛋白酶3表达同步升高,而金属蛋白酶组织抑制剂1作为内源性抑制剂可能通过抑制基质金属蛋白酶3活性调节细胞外基质代谢[24]。相反,病原体感染,如痤疮丙酸杆菌通过激活核因子κB信号通路显著上调基质金属蛋白酶1的表达,并抑制金属蛋白酶组织抑制剂1的合成,该机制导致细胞外基质核心成分聚集蛋白聚糖和Ⅱ型胶原合成减少,基质金属蛋白酶1/金属蛋白酶组织抑制剂1表达失衡,而核因子κB抑制剂BAY11-7082可特异性逆转此病理过程[59],这种矛盾的调控模式提示金属蛋白酶组织抑制剂的功能可能依赖于微环境中的特定刺激因子,有待深入研究。
炎症因子网络通过激活核因子κB通路,显著上调基质金属蛋白酶1/3/13并下调金属蛋白酶组织抑制剂1/2表达,形成促进基质降解的正反馈循环[3]。值得注意的是,针对该通路的干预策略显示出治疗潜力:通过调节纤维化相关基因(结缔组织生长因子、P物质、金属蛋白酶组织抑制剂1/2)并抑制核因子κB活性,
有效减轻退变相关的肌肉纤维化[25];而骨髓间充质干细胞的旁分泌效应可通过阻断核因子κB及线粒体凋亡通路,降低炎症因子白细胞介素1β诱导的纤维环细胞凋亡,同时恢复金属蛋白酶组织抑制剂1的表达水平[60]。补肝健腰方通过抑制核因子κB通路下调基质金属蛋白酶3和金属蛋白酶组织抑制剂1,避免单一靶点干预导致的代谢失调[61]。上述研究系统揭示了核因子κB在金属蛋白酶组织抑制剂介导基质代谢平衡中的作用 。
2.3.2 丝裂原活化蛋白激酶信号通路 丝裂原活化蛋白激酶信号通路在椎间盘退变病理过程中发挥关键调控作用[62-64]。研究证实,miR-155-5p在退变椎间盘的髓核和纤维环细胞中显著上调,通过激活丝裂原活化蛋白激酶/p38信号通路促进白细胞介素6、白细胞介素8等促炎因子分泌,加剧炎症反应并加速细胞外基质降解。此外,抑制miR-155-5p可显著增加抗炎因子白细胞介素10和金属蛋白酶组织抑制剂4的表达,提示其作为椎间盘退变治疗靶点的潜在价值[51,65]。研究表明,白细胞介素1β通过激活核因子κB和丝裂原活化蛋白激酶信号通路显著上调基质金属蛋白酶1、基质金属蛋白酶13及含血小板反应蛋白基序的解整合素样金属蛋白酶5的表达,同时促进细胞外基质分解代谢并抑制合成代谢,导致椎间盘退变进程的恶化。尽管金属蛋白酶组织抑制剂1在白细胞介素1β诱导的炎症反应中出现代偿性上调,但对基质金属蛋白酶活性的抑制作用不足以抵消核因子κB和丝裂原活化蛋白激酶通路介导的基质降解效应,最终引发细胞外基质的代谢失衡[66-67]。上述机制揭示了miR-155-5p-p38-金属蛋白酶组织抑制剂轴在椎间盘退变中的调控作用,提示靶向抑制miR-155-5p可能成为干预椎间盘退变的新策略[68-69]。
2.3.3 磷脂酰肌醇3激酶/蛋白激酶B 信号通路和Wnt/β-catenin信号通路 磷脂酰肌醇3激酶/蛋白激酶B和Wnt/β-catenin作为调控细胞存活与代谢的关键通路,在椎间盘生理稳态和退变过程中均发挥重要作用[70-73]。研究发现,磷脂酰肌醇3激酶/蛋白激酶B通路激活通过双重机制维持细胞外基质稳态:一方面通过上调金属蛋白酶组织抑制剂1抑制基质金属蛋白酶活性,从而减少细胞外基质降解[19];另一方面通过促进SRY盒转录因子9表达增强髓核细胞外基质的合成,显著提升Ⅱ型胶原和聚集蛋白聚糖等关键成分的表达。值得注意的是,Wnt/β-catenin通路同样通过调控金属蛋白酶组织抑制剂/基质金属蛋白酶系统影响细胞外基质平衡,该通路激活可促进细胞外基质合成酶的表达,同时抑制分解代谢相关因子,形成与磷脂酰肌醇3激酶/蛋白激酶B通路的功能互补[24]。目前研究共识表明:①磷脂酰肌醇3激酶/蛋白激酶B主导椎间盘细胞存活信号,通过抗凋亡效应维持髓核细胞群稳态;② Wnt/β-catenin侧重维持细胞外基质合成代谢调控,其异常激活可能导致纤维环结构重塑;③金属蛋白酶组织抑制剂/基质金属蛋白酶系统作为共同下游靶点,成为通路交互的重要调控网络。未来的研究需着重解析两条通路在机械应力、炎症微环境等病理条件下的时空激活特征,并开发椎间盘退变靶向调控的精准干预策略[74-75]。金属蛋白酶组织抑制剂的作用除了作用于透明软骨祖细胞,ROBERTS等[76]还探讨了机械过载对骨骼肌肥大机制的影响,表明磷脂酰肌醇3激酶/蛋白激酶B信号通路在肌肉适应和生长中起着重要作用,这一机制的理解不仅有助于揭示机械负荷对细胞生存的影响,也为研究椎间盘退变中的肌肉适应提供了新的视角。
2.3.4 P14ARF/金属蛋白酶组织抑制剂3信号通路 近年研究发现,肿瘤抑制因子P14ARF的表达水平与椎间盘退变进程呈负相关,其在正常椎间盘组织中高表达,而在退变组织中显著下调。腺病毒介导的P14ARF过表达可有效抑制椎间盘退变模型中的促炎细胞因子释放,包括肿瘤坏死因子α、白细胞介素1β和白细胞介素6,并降低病理性血管生成水平,提示P14ARF通过调控炎症微环境与血管稳态参与退变进程。值得注意的是,P14ARF与金属蛋白酶组织抑制剂3构成协同调控轴:在椎间盘细胞中,P14ARF过表达可正向调控金属蛋白酶组织抑制剂3的表达,而金属蛋白酶组织抑制剂3缺失则会加剧细胞炎症反应和血管生成。P14ARF/金属蛋白酶组织抑制剂3信号调控轴通过双重机制延缓椎间盘退变:①抑制促炎因子级联反应,阻断核因子κB等炎症通路激活;② 维持细胞外基质稳态,减少基质金属蛋白酶介导的分解代谢。
上述发现揭示P14ARF/金属蛋白酶组织抑制剂3轴可能通过整合炎症抑制与基质保护功能,成为椎间盘退变治疗的新靶点,后期研究仍需明确该信号轴在机械负荷、衰老等微环境中的动态调控作用[43]。
2.3.5 YTH结构域家族蛋白2/叉头框蛋白O3通路 YTH结构域家族蛋白2 /叉头框蛋白O3通路在椎间盘退变中的作用主要通过调控金属蛋白酶组织抑制剂/基质金属蛋白酶平衡实现。作为关键m6A阅读蛋白,YTH结构域家族蛋白2通过识别特定m6A修饰调控靶mRNA稳定性。在氧化应激条件下,YTH结构域家族蛋白2被异常激活并加速叉头框蛋白O3 mRNA降解,抑制叉头框蛋白O3对金属蛋白酶组织抑制剂1的转录激活作用,叉头框蛋白O3招募组蛋白乙酰转移酶和中介复合物亚基维持金属蛋白酶组织抑制剂1表达,从而抑制基质金属蛋白酶活性,这种调控失衡最终导致细胞外基质降解加速[8]。
值得关注的是,YTH结构域家族蛋白2对金属蛋白酶组织抑制剂的调控存在多维度特征。在机械应力刺激下,WTAP介导的m6A修饰增强,促进了YTH结构域家族蛋白2与金属蛋白酶组织抑制剂3 mRNA的结合,导致基质金属蛋白酶活性升高。该研究首次揭示了WTAP-YTH结构域家族蛋白2轴在力学微环境失衡中的调节作用[39]。上述研究表明,YTH结构域家族蛋白2通过降解叉头框蛋白O3和金属蛋白酶组织抑制剂3 mRNA的双重机制,在氧化应激和异常机械应力等病理条件下显著影响金属蛋白酶组织抑制剂/基质金属蛋白酶稳态。这种多靶点调控特性提示,靶向干预YTH结构域家族蛋白2可能成为逆转椎间盘退变的新策略。
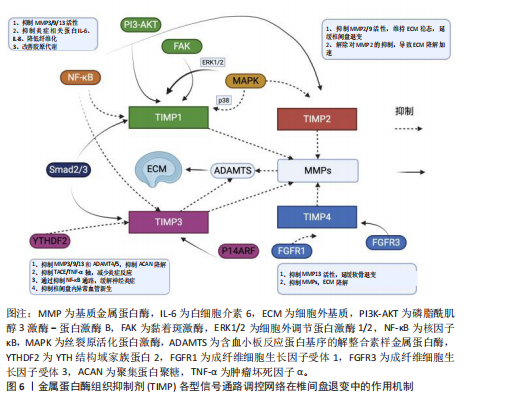
金属蛋白酶组织抑制剂各型信号通路调控网络在椎间盘退变中的作用机制,见图6。金属蛋白酶组织抑制剂的作用机制汇总见表1-4。
| [1] GÖKŞEN A, KOCAMAN H, MERVE ARMAN G, et al. Comprehensive analysis of muscles wasting in disc herniation. J Biomech. 2022; 145:111391. [2] ZHAO R, LIU W, XIA T, et al. Disordered Mechanical Stress and Tissue Engineering Therapies in Intervertebral Disc Degeneration. Polymers (Basel). 2019;11(7):1151. [3] CAI F, ZHU L, WANG F, et al. The Paracrine Effect of Degenerated Disc Cells on Healthy Human Nucleus Pulposus Cells Is Mediated by MAPK and NF-κB Pathways and Can Be Reduced by TGF-β1. DNA Cell Biol. 2017;36(2):143-158. [4] KWON WK, MOON HJ, KWON TH, et al. The Role of Hypoxia in Angiogenesis and Extracellular Matrix Regulation of Intervertebral Disc Cells During Inflammatory Reactions. Neurosurgery. 2017;81(5):867-875. [5] LI Y, ZHANG T, TIAN W, et al. Loss of TIMP3 expression induces inflammation, matrix degradation, and vascular ingrowth in nucleus pulposus: A new mechanism of intervertebral disc degeneration. FASEB J. 2020;34(4):5483-5498. [6] GUO W, ZHANG B, SUN C, et al. Circular RNA derived from TIMP2 functions as a competitive endogenous RNA and regulates intervertebral disc degeneration by targeting miR1855p and matrix metalloproteinase 2. Int J Mol Med. 2020;46(2):621-632. [7] CHEN X, LI Z, ZHENG C, et al. Expression of MMP1, MMP3, and TIMP1 in intervertebral discs under simulated overload and microgravity conditions. J Orthop Surg Res.2025;20(1):71. [8] WANG F, WANG Y, ZHANG S, et al. YTHDF2-dependent m6A modification of FOXO3 mRNA mediates TIMP1 expression and contributes to intervertebral disc degeneration following ROS stimulation. Cell Mol Life Sci. 2024;81(1):477. [9] 衡伟,钱济先,周程沛,等.飞行员椎间盘退行性疾病流行病学调查进展[J].中华全科医学,2023,21(3):473-476. [10] 刘磊,武建运,黄伟敏,等.腰椎退行性疾病手术患者椎间盘退变及责任间隙分布情况研究[J].中国全科医学,2021,24(5):585-591. [11] CHEN ZX, XU B, HUANG ZL, et al. Causal relationship between systemic circulatory inflammatory regulators and intervertebral disc degeneration: A bidirectional 2-sample Mendelian randomization study. Medicine (Baltimore). 2024;103(36):e39521. [12] GUNDOGDU EB, BEKAR A, TURKYILMAZ M, et al. CDP-choline modulates matrix metalloproteinases in rat sciatic injury. J Surg Res. 2016;200(2):655-663. [13] LI Y, LI K, HAN X, et al. The imbalance between TIMP3 and matrix-degrading enzymes plays an important role in intervertebral disc degeneration. Biochem Biophys Res Commun. 2016;469(3):507-514. [14] YUE B, LIN Y, MA X, et al. Survivin-TGFB3-TIMP1 Gene Therapy Via Lentivirus Vector Slows the Course of Intervertebral Disc Degeneration in an In Vivo Rabbit Model. Spine (Phila Pa 1976). 2016;41(11):926-934. [15] LAPIÈRE CHM. Tadpole collagenase, the single parent of such a large family. Biochimie. 2005; 87(3-4):243-247. [16] RIES C. Cytokine functions of TIMP-1. Cell Mol Life Sci. 2014;71(4):659-672. [17] BREW K, NAGASE H. The tissue inhibitors of metalloproteinases (TIMPs): an ancient family with structural and functional diversity. Biochim Biophys Acta. 2010; 1803(1):55-71. [18] LIU Y, YU T, MA XX, et al. Lentivirus-mediated TGF-β3, CTGF and TIMP1 gene transduction as a gene therapy for intervertebral disc degeneration in an in vivo rabbit model. Exp Ther Med. 2016;11(4):1399-1404. [19] HU S, FU Y, YAN B, et al. Analysis of key genes and pathways associated with the pathogenesis of intervertebral disc degeneration. J Orthop Surg Res. 2020;15(1):371. [20] 赵继荣,史凡凡,赵宁,等.基质金属蛋白酶与基质金属蛋白酶组织抑制剂对椎间盘退变的调控及中医药干预的研究进展[J]. 中华中医药学刊,2023,41(9):16-20. [21] 蔡挪亚.TGF-β3、TIMP-1、Bcl-2在退变腰椎间盘髓核组织中的表达及其意义[D].青岛:青岛大学,2018. [22] 冯习坤,孙马玲,曾昭池,等.椎间盘病变椎间盘髓核及纤维环MMP-1及TIMP-1表达变化及意义[J].局解手术学杂志,2017, 26(8):573-576. [23] 曾涛,朱英,陶金,等.腰椎间盘突出症患者手术前后血清mmp-2和mmp-9水平的变化及其对疼痛感知的影响[J].颈腰痛杂志, 2024,45(4):611-615. [24] ZHANG G, LI L, YANG Z, et al. TMT-Based Proteomics Analysis of Senescent Nucleus Pulposus from Patients with Intervertebral Disc Degeneration. Int J Mol Sci. 2023;24(17): 13236. [25] JAMES G, KLYNE DM, MILLECAMPS M, et al. ISSLS Prize in Basic science 2019: Physical activity attenuates fibrotic alterations to the multifidus muscle associated with intervertebral disc degeneration. Eur Spine J. 2019;28(5):893-904. [26] 鲍杰伟,施凤飞,邱全河,等.电针对腰椎间盘退变模型大鼠炎症因子表达的影响[J].中国医药,2022,17(11):1671-1675. [27] 杨好,陈程程,宣以道,等.腰椎间盘突出症患者髓核组织timp-1、il-27水平对术后复发的预测价值[J].贵州医科大学学报, 2023,48(7):826-830. [28] GONÇALVES RM, SAGGESE T, YONG Z, et al. Interleukin-1β More Than Mechanical Loading Induces a Degenerative Phenotype in Human Annulus Fibrosus Cells, Partially Impaired by Anti-Proteolytic Activity of Mesenchymal Stem Cell Secretome. Front Bioeng Biotechnol. 2022;9:802789. [29] HWANG MH, KIM KS, YOO CM, et al. Photobiomodulation on human annulus fibrosus cells during the intervertebral disk degeneration: extracellular matrix-modifying enzymes. Lasers Med Sci. 2016; 31(4):767-777. [30] WEN T, WANG H, LI Y, et al. Bone mesenchymal stem cell-derived extracellular vesicles promote the repair of intervertebral disc degeneration by transferring microRNA-199a. Cell Cycle. 2021;20(3):256-270. [31] TSENG KY, WANG HC, CHENG KF, et al. Sciatic Nerve Intrafascicular Injection Induces Neuropathy by Activating the Matrix Modulators MMP-9 and TIMP-1. Front Pharmacol. 2022;13:859982. [32] MISHRA RR, NIELSEN BE, TRUDRUNG MA, et al. The Effect of Tissue Inhibitor of Metalloproteinases on Scar Formation after Spinal Cord Injury. Cells. 2024;13(18):1547. [33] QUAN H, KIM H. Protein Kinase C and Matrix Metalloproteinases Expression Using Phorbol Myristate Acetate in Degenerative Intervertebral Disc Cells. Clin Orthop Surg. 2024;16(5):827-835. [34] KAMIENIAK P, BIELEWICZ J, KURZEPA J, et al. Serum Level of Metalloproteinase-2 but not Metalloproteinase-9 Rises in Patients With Failed Back Surgery Syndrome After Spinal Cord Stimulation. Neuromodulation. 2019;22(3):262-268. [35] BIELEWICZ J, DANILUK B, KAMIENIAK P. Metalloproteinase-2 in failed back surgery syndrome caused by epidural fibrosis: can it play a role in persistent pain? Front Hum Neurosci. 2023;17:1248943. [36] DREGALLA RC, URIBE Y, BODOR M. Human mesenchymal stem cells respond differentially to platelet preparations and synthesize hyaluronic acid in nucleus pulposus extracellular matrix. Spine J. 2020; 20(11):1850-1860. [37] 汤华,陈少军.胶原酶不同注射方法对腰椎间盘突出症患者血清timp-2、mmp-9表达及疼痛评分的影响[J].颈腰痛杂志,2023, 44(5):836-838. [38] ZHANG JF, WANG GL, ZHOU ZJ, et al. Expression of Matrix Metalloproteinases, Tissue Inhibitors of Metalloproteinases, and Interleukins in Vertebral Cartilage Endplate. Orthop Surg. 2018;10(4):306-311. [39] GAO D, ZHAO Q, LIU C, et al. Abnormal stress promotes intervertebral disc degeneration through WTAP/YTHDF2-dependent TIMP3 m6A modification. J Cell Physiol. 2024;239(5): e31219. [40] 王许可,张杨,罗刚,等.Fibulin-3调控组织金属蛋白酶抑制因子-3影响椎间盘髓核细胞衰老的机制研究[J].四川大学学报(医学版),2024,55(5):1217-1225. [41] VADALÀ G, DI GIACOMO G, AMBROSIO L, et al. Effect of Irisin on Human Nucleus Pulposus Cells: New Insights into the Biological Cross-talk Between Muscle and Intervertebral Disk. Spine (Phila Pa 1976). 2023;48(7):468-475. [42] ZHANG Y, YANG J, ZHOU X, et al. Knockdown of miR-222 inhibits inflammation and the apoptosis of LPS-stimulated human intervertebral disc nucleus pulposus cells. Int J Mol Med. 2019;44(4):1357-1365. [43] HE M, PANG J, SUN H, et al. P14ARF inhibits regional inflammation and vascularization in intervertebral disc degeneration by upregulating TIMP3. Am J Physiol Cell Physiol. 2020;318(4):C751-C761. [44] HE M, PANG J, SUN H, et al. Overexpression of TIMP3 inhibits discogenic pain by suppressing angiogenesis and the expression of substance P in nucleus pulposus. Mol Med Rep. 2020; 21(3):1163-1171. [45] JAMES G, CHEN X, DIWAN A, et al. Fat infiltration in the multifidus muscle is related to inflammatory cytokine expression in the muscle and epidural adipose tissue in individuals undergoing surgery for intervertebral disc herniation. Eur Spine J. 2021;30(4):837-845. [46] BAUMGARTNER L, WITTA S, NOAILLY J. Parallel Networks to Predict TIMP and Protease Cell Activity of Nucleus Pulposus Cells Exposed and Not Exposed to Pro-Inflammatory Cytokines. JOR Spine. 2025; 8(1):e70051. [47] LIANG H, LUO R, LI G, et al. The Proteolysis of ECM in Intervertebral Disc Degeneration. Int J Mol Sci. 2022;23(3):1715. [48] YE F, LYU FJ, WANG H, et al. The involvement of immune system in intervertebral disc herniation and degeneration. JOR Spine. 2022;5(1):e1196. [49] RAJASEKARAN S, CHITRAA T, DILIP CHAND RAJA S, et al. Subclinical infection can be an initiator of inflammaging leading to degenerative disk disease: evidence from host-defense response mechanisms. Eur Spine J. 2021;30(9):2586-2604. [50] SONG H, DU H, LI J, et al. Effect of fibroblast growth factor 2 on degenerative endplate chondrocyte: From anabolism to catabolism. Exp Mol Pathol. 2021;118:104590. [51] CAZZANELLI P, LAMOCA M, HASLER J, et al. The role of miR-155-5p in inflammation and mechanical loading during intervertebral disc degeneration. Cell Commun Signal. 2024; 22(1):419. [52] ISHINO H, SAKONJU I. Expression of metalloproteinases and their inhibitors in degenerated and extruded intervertebral disks in chondrodystrophic dogs. J Vet Med Sci. 2021;83(4):637-642. [53] HODSON NW, PATEL S, RICHARDSON SM, et al. Degenerate intervertebral disc-like pH induces a catabolic mechanoresponse in human nucleus pulposus cells. JOR Spine. 2018;1(1):e1004. [54] BURT KG, KIM MKM, VIOLA DC, et al. Nuclear factor κB overactivation in the intervertebral disc leads to macrophage recruitment and severe disc degeneration. Sci Adv. 2024;10(23): eadj3194. [55] CHEN S, HUANG Y, LEI L, et al. Daphnetin ameliorates intervertebral disc degeneration via the Keap1/Nrf2/NF-κB axis in vitro and in vivo. Int Immunopharmacol. 2025;145:113785. [56] WANG Y, ZHANG W, YANG Y, et al. Osteopontin deficiency promotes cartilaginous endplate degeneration by enhancing the NF-κB signaling to recruit macrophages and activate the NLRP3 inflammasome. Bone Res. 2024;12(1):53. [57] TENG Y, HUANG Y, YU H, et al. Nimbolide targeting SIRT1 mitigates intervertebral disc degeneration by reprogramming cholesterol metabolism and inhibiting inflammatory signaling. Acta Pharm Sin B. 2023;13(5):2269-2280. [58] HU H, WANG Z, YANG H, et al. Hypoxic Preconditional Engineering Small Extracellular Vesicles Promoted Intervertebral Disc Regeneration by Activating Mir-7-5p/NF-Κb/Cxcl2 Axis. Adv Sci (Weinh). 2023;10(35): e2304722. [59] ZHENG Y, LIN Y, CHEN Z, et al. Propionibacterium acnes induces intervertebral discs degeneration by increasing MMP-1 and inhibiting TIMP-1 expression via the NF-κB pathway. Int J Clin Exp Pathol. 2018;11(7):3445-3453. [60] HU J, YAN Q, SHI C, et al. BMSC paracrine activity attenuates interleukin-1β-induced inflammation and apoptosis in rat AF cells via inhibiting relative NF-κB signaling and the mitochondrial pathway. Am J Transl Res. 2017;9(1):79-89. [61] 仇湘中,刘栋,张信成,等.补肝健腰方对腰椎间盘退变大鼠椎间盘MMP-3、TIMP-1 mRNA表达的影响[J].中医药导报,2018, 24(14):21-25. [62] LI B, HU Y, CHEN Y, et al. Homoplantaginin alleviates intervertebral disc degeneration by blocking the NF-κB/MAPK pathways via binding to TAK1. Biochem Pharmacol. 2024;226:116389. [63] ZHANG C, LU Z, LYU C, et al. Andrographolide Inhibits Static Mechanical Pressure-Induced Intervertebral Disc Degeneration via the MAPK/Nrf2/HO-1 Pathway. Drug Des Devel Ther. 2023;17:535-550. [64] WANG T, ZHAO H, JING S, et al. Magnetofection of miR-21 promoted by electromagnetic field and iron oxide nanoparticles via the p38 MAPK pathway contributes to osteogenesis and angiogenesis for intervertebral fusion. J Nanobiotechnology. 2023;21(1):27. [65] FAN Z, LIU Y, SHI Z, et al. MiR-155 promotes interleukin-1β-induced chondrocyte apoptosis and catabolic activity by targeting PIK3R1-mediated PI3K/Akt pathway. J Cell Mol Med. 2020;24(15):8441-8451. [66] ZINGALE VD, GUGLIANDOLO A, MAZZON E. MiR-155: An Important Regulator of Neuroinflammation. Int J Mol Sci. 2021; 23(1):90. [67] LIAO Z, KE W, LIU H, et al. Vasorin-containing small extracellular vesicles retard intervertebral disc degeneration utilizing an injectable thermoresponsive delivery system. J Nanobiotechnology. 2022;20(1):420. [68] CAZZANELLI P, WUERTZ-KOZAK K. MicroRNAs in Intervertebral Disc Degeneration, Apoptosis, Inflammation, and Mechanobiology. Int J Mol Sci. 2020;21(10):3601. [69] MAHESH G, BISWAS R. MicroRNA-155: A Master Regulator of Inflammation. J Interferon Cytokine Res. 2019;39(6):321-330. [70] HUANG ZN, WANG ZY, CHENG XF, et al. Melatonin alleviates oxidative stress-induced injury to nucleus pulposus-derived mesenchymal stem cells through activating PI3K/Akt pathway. J Orthop Translat. 2023; 43:66-84. [71] ZHU Z, YU Q, LI H, et al. Vanillin-based functionalization strategy to construct multifunctional microspheres for treating inflammation and regenerating intervertebral disc. Bioact Mater. 2023;28:167-182. [72] RYU M, YURUBE T, TAKEOKA Y, et al. Gene-Silencing Therapeutic Approaches Targeting PI3K/Akt/mTOR Signaling in Degenerative Intervertebral Disk Cells: An In Vitro Comparative Study Between RNA Interference and CRISPR-Cas9. Cells. 2024;13(23):2030. [73] LI Y, ZHANG K, AI X, et al. A Biomimetic Peptide Functions as Specific Extracellular Matrix for Quiescence of Stem Cells against Intervertebral Disc Degeneration. Small. 2023; 19(44):e2300578. [74] HE X, LI Y, DENG B, et al. The PI3K/AKT signalling pathway in inflammation, cell death and glial scar formation after traumatic spinal cord injury: Mechanisms and therapeutic opportunities. Cell Prolif. 2022;55(9):e13275. [75] LI M, YU X, CHEN X, et al. Genkwanin alleviates intervertebral disc degeneration via regulating ITGA2/PI3K/AKT pathway and inhibiting apoptosis and senescence. Int Immunopharmacol. 2024;133:112101. [76] ROBERTS MD, MCCARTHY JJ, HORNBERGER TA, et al. Mechanisms of mechanical overload-induced skeletal muscle hypertrophy: current understanding and future directions. Physiol Rev. 2023;103(4):2679-2757. |
| [1] | 朱小龙, 张 玮, 杨 阳. 椎间盘再生与修复领域研究热点与前沿信息的可视化分析[J]. 中国组织工程研究, 2026, 30(9): 2391-2402. |
| [2] | 杨学涛, 朱梦菡, 张宸熙, 孙一民, 叶 玲. 抗氧化纳米材料在口腔中的应用和不足[J]. 中国组织工程研究, 2026, 30(8): 2044-2053. |
| [3] | 杨利霞, 刁立琴, 李 华, 冯亚婵, 刘 鑫, 于月欣, 窦茜茜, 谷辉峰, 徐兰举. 重组Ⅲ型人源化胶原蛋白改善大鼠光老化皮肤的调控机制[J]. 中国组织工程研究, 2026, 30(8): 1988-2000. |
| [4] | 董春阳, 周天恩, 莫孟学, 吕文权, 高 明, 朱瑞凯, 高志伟. 二甲双胍联合血水草敷料治疗深Ⅱ度烧伤创面的作用机制[J]. 中国组织工程研究, 2026, 30(8): 2001-2013. |
| [5] | 蔡子鸣, 于庆贺, 马鹏飞, 张 鑫, 周龙千, 张崇阳, 林文平. 血红素氧合酶1减轻脂多糖诱导髓核间充质干细胞的炎症反应[J]. 中国组织工程研究, 2026, 30(7): 1624-1631. |
| [6] | 夏林枫, 王 露, 龙乾发, 唐荣武, 罗浩东, 汤 轶, 钟 俊, 刘 阳. 人脐带间充质干细胞来源外泌体减轻脓毒症脑病小鼠血脑屏障损伤[J]. 中国组织工程研究, 2026, 30(7): 1711-1719. |
| [7] | 刘安婷, 陆江涛, 张文杰, 贺 玲, 唐宗生, 陈晓玲. 血小板裂解物调控腺苷酸活化蛋白激酶抑制镉诱导的神经细胞凋亡[J]. 中国组织工程研究, 2026, 30(7): 1800-1807. |
| [8] | 陈伊娴, 陈 晨, 卢立恒, 汤锦鹏, 于晓巍. 雷公藤甲素治疗骨关节炎的网络药理学分析与实验验证[J]. 中国组织工程研究, 2026, 30(4): 805-815. |
| [9] | 杨 肖, 白月辉, 赵甜甜, 王东昊, 赵 琛, 袁 硕. 颞下颌关节骨关节炎软骨退变:机制及再生的挑战[J]. 中国组织工程研究, 2026, 30(4): 926-935. |
| [10] | 郭 峰, 李 倩, 侯超文, 郭成吉. 运动改善神经病理性疼痛:精准运动处方及多模态协同推进临床应用[J]. 中国组织工程研究, 2026, 30(35): 9269-9280. |
| [11] | 刘洪伟, 张隆基. 慢病毒介导的基因治疗β-地中海贫血小鼠模型[J]. 中国组织工程研究, 2026, 30(34): 8978-8985. |
| [12] | 穆秉桃, 郭敏芳, 胡芬琦, 刘淇源, 贾 辉, 徐明源, 陈佳媛, 张慧宇, 孟 涛, 尉杰忠. 雷公藤甲素缓解过氧化氢诱导SH-SY5Y细胞凋亡的线粒体动力学机制[J]. 中国组织工程研究, 2026, 30(34): 8986-8993. |
| [13] | 张益瑞, 顾 叶, 钱正韬, 吴泽睿, 谢 恒, 唐奕涵, 顾赢楚, 方 涛, 王秋霏, 彭育沁, 耿德春, 徐耀增. 机械应力调控骨关节炎的分子机制与治疗靶点[J]. 中国组织工程研究, 2026, 30(34): 9048-9055. |
| [14] | 郑 颖, 李梦瑶, 郑帆帆, 何 昭, 张 宁, 邹嘉伦, 李优磊, 高 枫. 搭载生物材料细胞外泌体修复脊髓损伤的作用机制[J]. 中国组织工程研究, 2026, 30(32): 8544-8554. |
| [15] | 张一苇, 方 雅, 孙 鑫, 杨 涵, 林海扬, 陈周昊, 郑 悦, 符静珂, 王金武. 小球藻源多肽提取工艺优化对类风湿关节炎关键病理环节的调控作用[J]. 中国组织工程研究, 2026, 30(32): 8460-8470. |
中国组织工程研究杂志出版内容重点:干细胞;骨髓干细胞;造血干细胞;脂肪干细胞;肿瘤干细胞;胚胎干细胞;脐带脐血干细胞;干细胞诱导;干细胞分化;组织工程
1.1 资料来源
1.1.1 检索人及检索时间 第一作者在2025年3月进行检索。
1.1.3 检索数据库 PubMed、Web of Science、Embase、万方、中国知网。
1.1.4 检索途径 采用主题词和自由词结合的方式进行检 索,以PubMed数据库检索策略为例,见图2。
1.1.6 检索文献量 初步检索700余篇文献。
1.2 入选标准
纳入标准:①金属蛋白酶组织抑制剂/基质金属蛋白酶调控网络失衡导致椎间盘退变的研究;②金属蛋白酶组织抑制剂1/2/3/4 在椎间盘退变中的作用机制研究;③金属蛋白酶组织抑制剂与丝裂原活化蛋白激酶、磷脂酰肌醇3激酶/蛋白激酶B、核因子κB 等信号通路相关的研究;④以金属蛋白酶组织抑制剂为作用靶点治疗椎间盘退变的相关研究。
排除标准:①与椎间盘退变无关的文献;②发表时间在 2015 年以前的文献;③研究内容、研究方法和实验设备过时及重复性文献。
1.3 质量评估及数据的提取 采用定性综述方法,通过计算机初步检索得到与研究目的相关的中英文文献700余篇,经资料收集人员根据纳入及排除标准进一步筛选,选择与此次综述内容相符的文献,最终确定纳入76篇符合标准的文献进行综述。文献筛选流程见图3。
3.1 既往他人研究贡献与局限 椎间盘退变的病理机制涉及多种分子调控网络异常,其中金属蛋白酶组织抑制剂功能网络失衡在细胞外基质降解中发挥关键作用。既往研究表明,金属蛋白酶组织抑制剂3作为聚集蛋白聚糖酶最有效的内源性抑制剂,其基因表达水平在退变椎间盘中显著降低,导致基质降解酶活性异常升高,加速胶原和聚集蛋白聚糖的分解[13]。同时,基质金属蛋白酶1在退变椎间盘中表达上调,而金属蛋白酶组织抑制剂1呈现动态波动,二者比例失衡可能破坏细胞外基质稳态。此外,金属蛋白酶组织抑制剂2等家族成员通过调控基质金属蛋白酶活性参与椎间盘退变进程,提示金属蛋白酶组织抑制剂功能网络的复杂性。然而,现有研究仍存在重要局限:金属蛋白酶组织抑制剂家族成员间相互作用及其整体网络失衡的分子机制尚未阐明;炎症因子、机械应力等微环境对金属蛋白酶组织抑制剂的调控途径仍不明确;当前研究成果多基于基础实验模型,临床和应用转化潜力受限,且缺乏针对金属蛋白酶组织抑制剂网络失衡的靶向治疗策略。值得注意的是,金属蛋白酶组织抑制剂与血管生成、神经损伤等多因素间的交互作用仍需深入探究。这些科学问题制约着基于细胞外基质稳态调控的椎间盘退变治疗策略发展。
3.2 该综述区别于他人他篇的特点 此综述突破传统单一亚型或通路的研究模式,构建金属蛋白酶组织抑制剂家族(金属蛋白酶组织抑制剂1/2/3/4)的亚型特异性功能图谱,系统解析功能异质性,如金属蛋白酶组织抑制剂1的动态双向调节、金属蛋白酶组织抑制剂3的多维调控网络及协同作用,填补了金属蛋白酶组织抑制剂4功能机制的认知空白;系统回顾表观遗传(如m6A修饰调控金属蛋白酶组织抑制剂3降解)、力学-生物学耦合(异常应力通过WTAP/ YTH结构域家族蛋白2轴破坏基质稳态)等跨学科关联,揭示椎间盘退变中“基因-环境-表型”的级联调控网络,弥补了既往研究对力学-表观交互机制的忽视;提出“金属蛋白酶组织抑制剂功能网络失衡”的理论假说,整合炎症-基质降解-血管新生-细胞衰老等多病理环节,突破传统“基质金属蛋白酶/金属蛋白酶组织抑制剂比例失衡”的简化模型;这些多维新视角使此综述成为连接金属蛋白酶组织抑制剂基础机制与精准治疗策略的文献依据。
3.3 综述的局限性 此综述系统阐述了金属蛋白酶组织抑制剂家族在椎间盘退变中的多维度调控机制,但仍存在若干亟待解决的科学问题:①现有研究对金属蛋白酶组织抑制剂亚型功能的时空特异性解析不足,如金属蛋白酶组织抑制剂1在退变早期保护作用和晚期功能代偿失调的动态转换机制尚未完全阐明,其与磷脂酰肌醇3激酶/蛋白激酶B信号通路的上下游调控关系仍存在争议。②亚型间协同/拮抗效应的研究局限于体外单一或较少干预因素模型,未能真实模拟退变微环境中力学异常、缺氧、炎症因子网络等多因素交互作用对金属蛋白酶组织抑制剂/基质金属蛋白酶平衡的影响。③表观遗传调控网络的复杂性被低估,例如m6A修饰对金属蛋白酶组织抑制剂3的调控可能受YTH结构域家族蛋白2之外阅读蛋白的补偿性调节,而miRNA-金属蛋白酶组织抑制剂互作网络在临床样本中呈现显著个体异质性。④现有椎间盘退变干预策略多聚焦于单一亚型或通路靶点,多靶点协同调控策略仍待进一步探索,多数动物模型未能复现人类退变的慢性进展特征,导致治疗机制的临床转化价值存疑。未来可以开发类器官模型以模拟人类疾病的复杂性,有望加速药物发现和开发过程,减少对动物实验的依赖,并提高临床试验的成功率。⑤金属蛋白酶组织抑制剂作为生物标志物的应用仍面临技术瓶颈,其表达水平与影像学分级、临床症状的非线性关系提示需建立多组学联合评价体系。未来研究需整合单细胞、时空转录组与力学生物学手段,揭示金属蛋白酶组织抑制剂在退变微环境中的时空异质性。
3.4 综述的重要意义 此综述系统回顾了金属蛋白酶组织抑制剂在椎间盘退变中的多维调控网络及转化潜力。在调控网络方面,文章总结了金属蛋白酶组织抑制剂4种亚型:金属蛋白酶组织抑制剂1通过动态双向调节参与早期基质保护及衰老相关功能耗竭;金属蛋白酶组织抑制剂2在椎间盘退变中双向调控细胞外基质稳态——生理性抑制基质金属蛋白酶维持结构,而病理异常则解除抑制,导致细胞外基质降解加速退变。金属蛋白酶组织抑制剂3作为核心枢纽,整合m6A表观遗传修饰(WTAP/YTH结构域家族蛋白2轴)、miRNA互作(miR-222靶向抑制)及抗血管新生功能,协同调控炎症-基质降解级联反应;金属蛋白酶组织抑制剂4则通过miR-155-5p/成纤维细胞生长因子2信号轴被确立为细胞外基质稳态的新型调节因子。在组织工程应用方面,可以通过精确调控基质金属蛋白酶和金属蛋白酶组织抑制剂的平衡,控制生物材料支架的降解速度和模式,使其更好地与新生组织匹配,促进组织整合和功能恢复。金属蛋白酶组织抑制剂的基质金属蛋白酶非依赖性功能可以促进细胞黏附、增殖和分化。在组织工程支架中引入金属蛋白酶组织抑制剂或调控其表达,可能直接影响植入细胞的行为,引导细胞向特定谱系分化,从而提高组织工程的效率。临床转化方面,靶向金属蛋白酶组织抑制剂治疗在椎间盘退变临床转化中具有显著潜力。有研究通过基因改造来增加金属蛋白酶组织抑制剂的表达,从而抑制基质金属蛋白酶的活性,减缓或逆转椎间盘退变,调节基质金属蛋白酶和金属蛋白酶组织抑制剂的表达,还有利用能够调节基质金属蛋白酶/金属蛋白酶组织抑制剂平衡的生物活性分子(如小分子抑制剂或外源性金属蛋白酶组织抑制剂)来抑制细胞外基质降解。另有研究使用智能材料与微环境调控,如水凝胶在椎间盘修复中展现出巨大潜力,它们可以通过递送生物活性物质或细胞来重建健康的微环境,从而影响金属蛋白酶组织抑制剂的表达和活性。例如生物仿生纳米囊泡结合机械响应性miR-1249被提出用于缓解椎间盘退变。还有研究利用干细胞或其他间充质细胞促进椎间盘再生,这些细胞可以分泌金属蛋白酶组织抑制剂,促进细胞外基质合成,并抑制炎症反应。通过整合生物力学、表观遗传学及代谢组学等多学科证据,此综述尝试性构建了“金属蛋白酶组织抑制剂功能网络失衡驱动椎间盘退变病理进程”的理论框架,为开发时序特异性靶向治疗提供了探索路径,为椎间盘退变研究从单一机制解析向系统化干预策略提供了基础,为组织工程修复椎间盘退变提供了新的靶点和理论依据。未来的研究应聚焦于开发真正意义上的多靶点协同干预策略,以期从根本上逆转或延缓退变进程。
3.5 课题专家组对未来的建议 ①深化金属蛋白酶组织抑制剂基础研究:椎间盘退变是脊柱退变性疾病的重要病理过程,金属蛋白酶组织抑制剂在其中发挥关键作用。未来研究应整合单细胞时空转录组与生物力学分析,解析金属蛋白酶组织抑制剂在椎间盘退变不同阶段的时空表达规律及功能转换机制,尤其是金属蛋白酶组织抑制剂1从早期保护到晚期功能耗竭的动态过程。通过多因素下的研究,揭示金属蛋白酶组织抑制剂亚型间的协同与拮抗效应及其对细胞外基质稳态的影响。此外,深入探讨异常机械应力通过表观遗传修饰(如m6A、DNA甲基化)调控金属蛋白酶组织抑制剂亚型表达的分子通路,明确其在疾病进展中的作用。②多靶点协同干预策略:设计针对金属蛋白酶组织抑制剂信号枢纽(如YTH结构域家族蛋白2/叉头框蛋白O3轴)的基因疗法,结合CRISPR-Cas9或腺病毒载体实现组织特异性调控。探索miRNA拮抗剂与金属蛋白酶组织抑制剂激动剂的联合应用,以及智能生物材料或物理干预设备(如光生物调节、动态力学加载装置)的应用,通过调控金属蛋白酶组织抑制剂/
基质金属蛋白酶平衡实现力学微环境重塑,为椎间盘退变治疗提供创新思路。③临床转化与精准医学:建立椎间盘退变患者队列的多组学数据库,筛选金属蛋白酶组织抑制剂相关生物标志物,用于疾病分期与疗效预测。利用人源椎间盘类器官和非人灵长类慢性退变模型,评估靶向金属蛋白酶组织抑制剂治疗的长期安全性与组织再生效果。基于遗传易感性分层,开发个性化治疗策略,如智能纳米递送系统,实现金属蛋白酶组织抑制剂激动剂/拮抗剂的按需释放,提高治疗精准性。④拓展研究与未来展望:探索椎间盘突出相关肌肉萎缩与金属蛋白酶组织抑制剂表达的潜在联系,以及磷脂酰肌醇3激酶/蛋白激酶B通路与金属蛋白酶组织抑制剂的交互作用。开展多中心临床试验,评估金属蛋白酶组织抑制剂相关疗法的中长期效果。通过这些研究方向,全面深化对金属蛋白酶组织抑制剂在椎间盘退变中作用机制的理解,为开发精准、高效的治疗策略提供科学依据。
中国组织工程研究杂志出版内容重点:干细胞;骨髓干细胞;造血干细胞;脂肪干细胞;肿瘤干细胞;胚胎干细胞;脐带脐血干细胞;干细胞诱导;干细胞分化;组织工程
椎间盘退变的病理特征在于细胞外基质合成与降解失衡。基质金属蛋白酶是关键降解酶,破坏胶原和蛋白聚糖,而金属蛋白酶组织抑制剂是其内源性抑制物。在椎间盘退变中,基质金属蛋白酶与金属蛋白酶组织抑制剂的稳态被破坏。当前研究热点聚焦于基质金属蛋白酶/金属蛋白酶组织抑制剂失衡的调控机制如炎症、氧化应激、表观遗传等,还有新型治疗策略如基因疗法、细胞疗法、小分子化合物机生物材料等方法。相较于早期单分子研究,当前综述更注重基质金属蛋白酶/金属蛋白酶组织抑制剂功能网络的系统性分析,整合多组学数据如基因组学、蛋白组学。未来个体差异的精准医疗、多模态分子影像学监测细胞外基质降解酶活性、应用人工智能挖掘数据深层关联、开发新型生物标志物用于早诊、预后将会成为热点。
中国组织工程研究杂志出版内容重点:干细胞;骨髓干细胞;造血干细胞;脂肪干细胞;肿瘤干细胞;胚胎干细胞;脐带脐血干细胞;干细胞诱导;干细胞分化;组织工程
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||