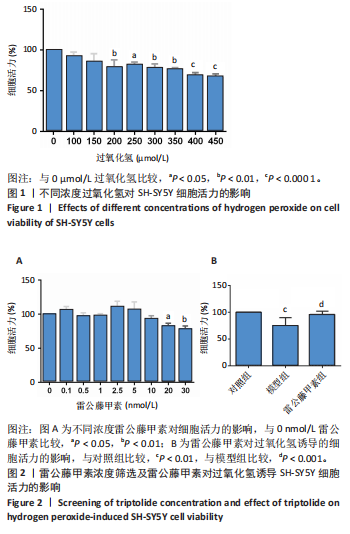
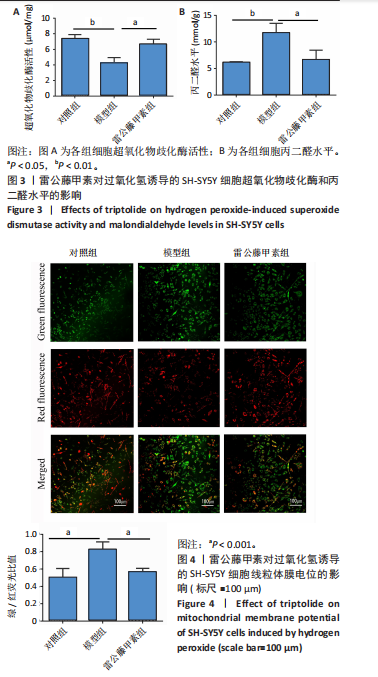
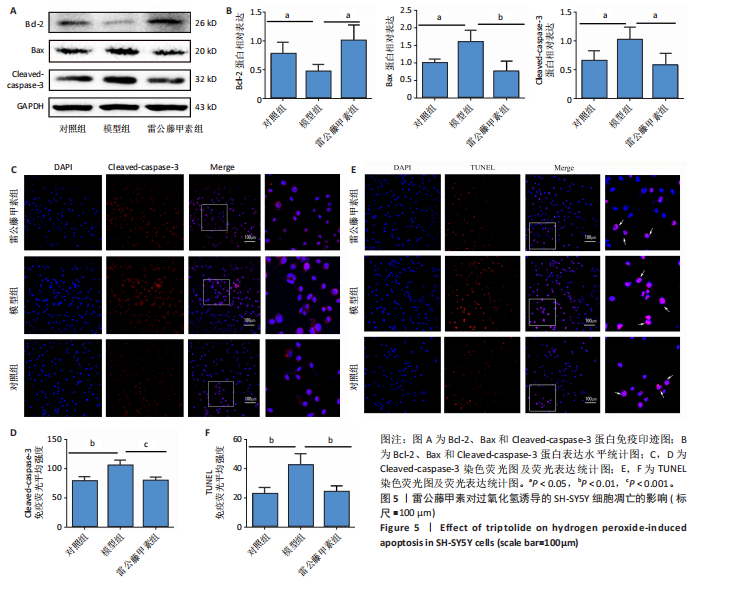
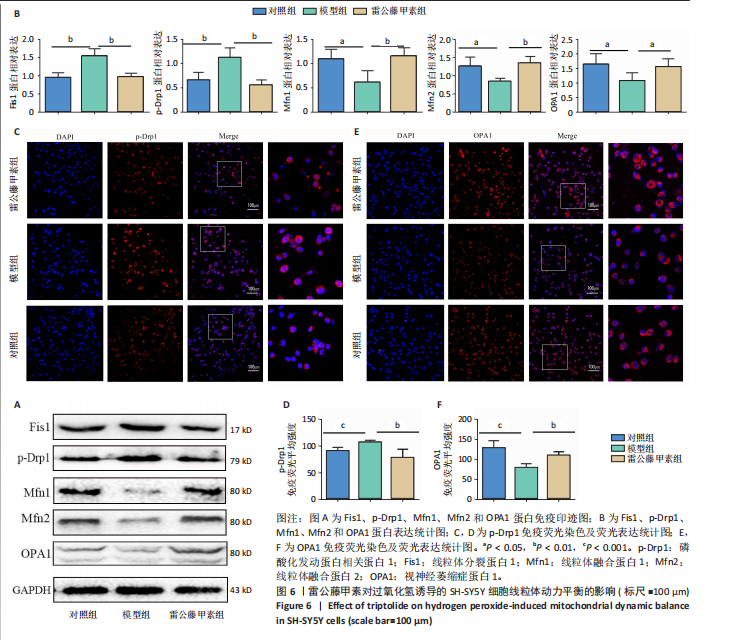
[1] ROSTAGNO AA. Pathogenesis of Alzheimer’s Disease. Int J Mol Sci. 2022;24(1): 107.
[2] KNAPSKOG AB, ENGEDAL K, SELBÆK G, et al. Alzheimer’s disease – diagnosis and treatment. Tidsskr Nor Laegeforen. 2021;141(7). doi: 10.4045/tidsskr.20.0919.
[3] XU X, PANG Y, FAN X. Mitochondria in oxidative stress, inflammation and aging: from mechanisms to therapeutic advances. Signal Transduct Target Ther. 2025; 10(1):190.
[4] KLEMMENSEN MM, BORROWMAN SH, PEARCE C, et al. Mitochondrial dysfunction in neurodegenerative disorders. Neurotherapeutics. 2024;21(1):e00292.
[5] ATTWELL D, LAUGHLIN SB. An energy budget for signaling in the grey matter of the brain. J Cereb Blood Flow Metab. 2001;21(10):1133-1145.
[6] COYLE JT, PUTTFARCKEN P. Oxidative stress, glutamate, and neurodegenerative disorders. Science. 1993;262(5134):689-695.
[7] RAI SN, SINGH C, SINGH A, et al. Mitochondrial Dysfunction: a Potential Therapeutic Target to Treat Alzheimer’s Disease. Mol Neurobiol. 2020;57(7): 3075-3088.
[8] MACDONALD R, BARNES K, HASTINGS C, et al. Mitochondrial abnormalities in Parkinson’s disease and Alzheimer’s disease: can mitochondria be targeted therapeutically? Biochem Soc Trans. 2018;46(4):891-909.
[9] EBADPOUR N, MAHMOUDI M, KAMAL KHEDER R, et al. From mitochondrial dysfunction to neuroinflammation in Parkinson’s disease: Pathogenesis and mitochondrial therapeutic approaches. Int Immunopharmacol. 2024;142(Pt A): 13015.
[10] EO H, YU SH, CHOI Y, et al. Mitochondrial transplantation exhibits neuroprotective effects and improves behavioral deficits in an animal model of Parkinson’s disease. Neurotherapeutics. 2024;21(4):e00355.
[11] WEI YM, WANG YH, XUE HQ, et al. Triptolide, A Potential Autophagy Modulator. Chin J Integr Med. 2019;25(3):233-240.
[12] JIA L, NIE XQ, JI HM, et al. Multiple-Coated PLGA Nanoparticles Loading Triptolide Attenuate Injury of a Cellular Model of Alzheimer’s Disease. Biomed Res Int. 2021;2021:8825640.
[13] XU P, WU Z, PENG Y, et al. Neuroprotection of Triptolide against Amyloid-Beta1-42-induced toxicity via the Akt/mTOR/p70S6K-mediated Autophagy Pathway. An Acad Bras Cienc. 2022;94(2):e20210938.
[14] XU P, WANG H, LI Z, et al. Triptolide attenuated injury via inhibiting oxidative stress in Amyloid-Beta25-35-treated differentiated PC12 cells. Life Sci. 2016;145:19-26.
[15] HE Z, BOTCHWAY BOA, ZHANG Y, et al. Triptolide activates the Nrf2 signaling pathway and inhibits the NF-κB signaling pathway to improve Alzheimer disease. Metab Brain Dis. 2024;39(1):173-182.
[16] ZHENG Y, ZHANG WJ, WANG XM. Triptolide with potential medicinal value for diseases of the central nervous system. CNS Neurosci Ther. 2013;19(2):76-82.
[17] SONG W, LIU M, WU J, et al. Preclinical Pharmacokinetics of Triptolide: A Potential Antitumor Drug. Curr Drug Metab. 2019;20(2):147-154.
[18] CHEN P, ZHONG X, SONG Y, et al. Triptolide induces apoptosis and cytoprotective autophagy by ROS accumulation via directly targeting peroxiredoxin 2 in gastric cancer cells. Cancer Lett. 2024;587:216622.
[19] LI W, YANG Y, HU Z, et al. Neuroprotective effects of DAHP and Triptolide in focal cerebral ischemia via apoptosis inhibition and PI3K/Akt/mTOR pathway activation. Front Neuroanat. 2015;9:48.
[20] NIE J, ZHOU M, LÜ C, et al. Effects of triptolide on the synaptophysin expression of hippocampal neurons in the AD cellular model. Int Immunopharmacol. 2012;13(2):175-180.
[21] 宋哲,薛超,张小曼,等.脂联素通过激活PP2A减轻H2O2诱导的SH-SY5Y细胞损伤及tau蛋白过度磷酸化[J].中国病理生理杂志,2015,31(2):207-212.
[22] 于婧文,郭敏芳,李苏垚,等.黄芪甲苷通过调控线粒体功能抑制H2O2诱导的SH-SY5Y细胞凋亡[J].中国病理生理杂志,2022,38(9):1553-1560.
[23] ASHLEIGH T, SWERDLOW RH, BEAL MF. The role of mitochondrial dysfunction in Alzheimer’s disease pathogenesis. Alzheimers Dement. 2023;19(1):333-342.
[24] YAO MF, DANG T, WANG HJ, et al. Mitochondrial homeostasis regulation: A promising therapeutic target for Parkinson’s disease. Behav Brain Res. 2024; 459:114811.
[25] ELFAWY HA, DAS B. Crosstalk between mitochondrial dysfunction, oxidative stress, and age related neurodegenerative disease: Etiologies and therapeutic strategies. Life Sci. 2019;218:165-184.
[26] CAO H, ZUO C, GU Z, et al. High frequency repetitive transcranial magnetic stimulation alleviates cognitive deficits in 3xTg-AD mice by modulating the PI3K/Akt/GLT-1 axis. Redox Biol. 2022;54:102354.
[27] PERLUIGI M, DI DOMENICO F, BUTTERFIELD DA. Oxidative damage in neurodegeneration: roles in the pathogenesis and progression of Alzheimer disease. Physiol Rev. 2024;104(1):103-197.
[28] YULUG B, ALTAY O, LI X, et al. Multi-omics characterization of improved cognitive functions in Parkinson’s disease patients after the combined metabolic activator treatment: a randomized, double-blinded, placebo-controlled phase II trial. Brain Commun. 2025;7(1):fcae478.
[29] 于海杰,关启刚,郭亮,等.雷公藤内酯醇保护心肌H/R损伤的相关分子机制[J].贵州医科大学学报,2019,44(2):190-194.
[30] LV C, CHENG T, ZHANG B, et al. Triptolide protects against podocyte injury in diabetic nephropathy by activating the Nrf2/HO-1 pathway and inhibiting the NLRP3 inflammasome pathway. Ren Fail. 2023;45(1):2165103.
[31] ALQAHTANI T, DEORE SL, KIDE AA, et al. Mitochondrial dysfunction and oxidative stress in Alzheimer’s disease, and Parkinson’s disease, Huntington’s disease and Amyotrophic Lateral Sclerosis -An updated review. Mitochondrion. 2023;71:83-92.
[32] HASHIMI H, GAHURA O, PÁNEK T. Bringing together but staying apart: decisive differences in animal and fungal mitochondrial inner membrane fusion. Biol Rev Camb Philos Soc. 2025;100(2):920-935.
[33] PIRES M, REGO AC. Apoe4 and Alzheimer’s Disease Pathogenesis-Mitochondrial Deregulation and Targeted Therapeutic Strategies. Int J Mol Sci. 2023;24(1):778.
[34] GREL H, WOZNICA D, RATAJCZAK K, et al. Mitochondrial Dynamics in Neurodegenerative Diseases: Unraveling the Role of Fusion and Fission Processes. Int J Mol Sci. 2023;24(17):13033.
[35] BUCK SA, SANDERS LH. LRRK2-mediated mitochondrial dysfunction in Parkinson’s disease. Biochem J. 2025;482(11):721-739.
[36] 姚美桦,黄浩.线粒体呼吸链超级复合物研究进展[J].生物学教学,2024, 49(8):2-5.
[37] LÉVY E, EL BANNA N, BAÏLLE D, et al. Causative Links between Protein Aggregation and Oxidative Stress: A Review. Int J Mol Sci. 2019;20(16):3896.
[38] LIU G, YANG C, WANG X, et al. Oxygen metabolism abnormality and Alzheimer’s disease: An update. Redox Biol. 2023;68:102955.
[39] SONG T, SONG X, ZHU C, et al. Mitochondrial dysfunction, oxidative stress, neuroinflammation, and metabolic alterations in the progression of Alzheimer’s disease: A meta-analysis of in vivo magnetic resonance spectroscopy studies. Ageing Res Rev. 2021;72:101503.
|