[1] SUTTON EL. Insomnia. Ann Intern Med. 2021;174(3):ITC33-ITC48.
[2] PAUL AM, SALAS RE. Insomnia. Prim Care. 2024;51(2):299-310.
[3] PENG C, WANG K, WANG J, et al. Neural correlates of insomnia with depression and anxiety from a neuroimaging perspective: A systematic review. Sleep Med Rev. 2025;81:102093.
[4] 中华医学会神经病学分会,中华医学会神经病学分会睡眠障碍学组,中华医学会神经病学分会神经心理与行为神经病学学组.中国成人失眠伴抑郁焦虑诊治专家共识[J].中华神经科杂志,2020,53(8):564-574.
[5] 中华医学会神经病学分会睡眠障碍学组.中国成人失眠诊断与治疗指南(2023版)[J].中华神经科杂志,2024,57(6):560-584.
[6] BENJAFIELD AV, SERT KUNIYOSHI FH, MALHOTRA A, et al. Estimation of the global prevalence and burden of insomnia: a systematic literature review-based analysis. Sleep Med Rev. 2025;82:102121.
[7] ROSENQVIST TW, WIUM-ANDERSEN MK, WIUM-ANDERSEN IK, et al. Long-Term Use of Benzodiazepines and Benzodiazepine-Related Drugs: A Register-Based Danish Cohort Study on Determinants and Risk of Dose Escalation. Am J Psychiatry. 2024;181(3):246-254.
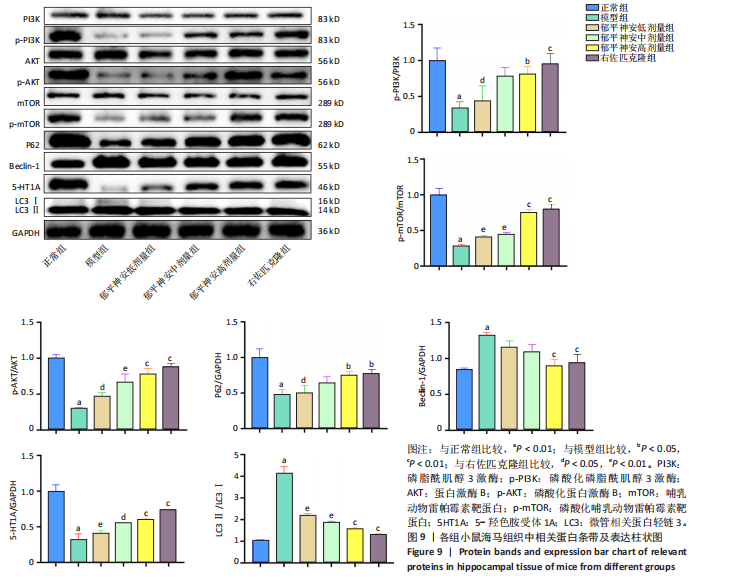
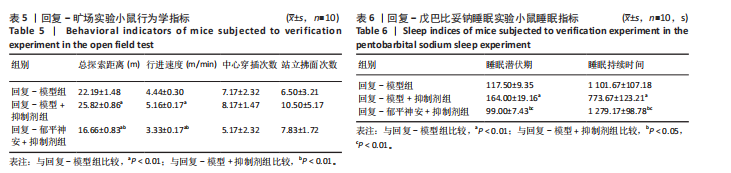
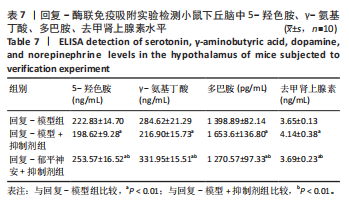
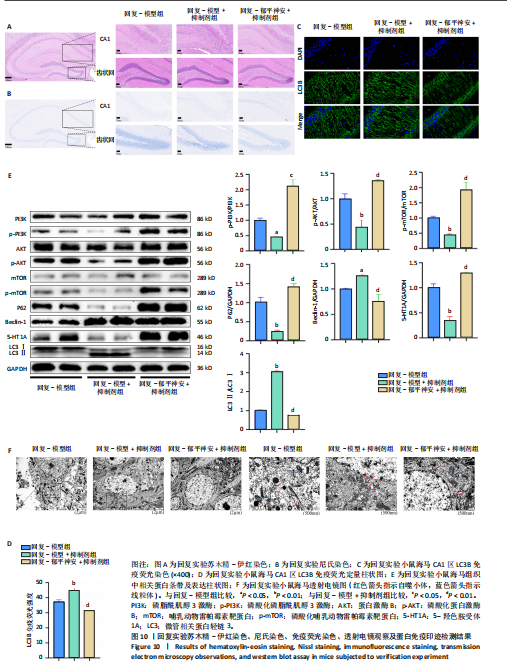
[8] 刘汇真,张旖旎,陈雨萌,等.安寐丹通过PI3K/Akt/mTOR信号通路调控细胞自噬改善睡眠剥夺小鼠学习记忆水平[J].中国实验方剂学杂志,2025, 31(14):1-9.
[9] 何钰珺,闫鹏轩,谢道俊.谢道俊教授用“轻可去实、重可去怯”法治疗失眠伴焦虑抑郁状态经验[J].吉林中医药,2023,43(6):656-661.
[10] 黄辰烨.郁平神安方治疗肝郁气滞型慢性失眠障碍合并焦虑抑郁状态的临床疗效观察与静息态功能磁共振研究[D].合肥:安徽中医药大学,2025.
[11] LIN H, XU Y, XIONG H, et al. Mechanism of action of Panax ginseng alcohol extract based on orexin-mediated autophagy in the treatment of sleep and cognition in aged sleep-deprived rats. J Ethnopharmacol. 2025;337(Pt 2):118907.
[12] MAO JQ, CHENG L, ZHANG YD, et al. Chinese formula Guben-Jiannao Ye alleviates the dysfunction of circadian and sleep rhythms in APP/PS1 mice implicated in activation of the PI3K/AKT/mTOR signaling pathway. J Ethnopharmacol. 2024;335:118696.
[13] BAO Y, ZHOU H, FU Y, et al. Zhumian Granules improves PCPA-induced insomnia by regulating the expression level of neurotransmitters and reducing neuronal apoptosis. J Ethnopharmacol. 2024;327:118048.
[14] ZHANG W, ZHANG X, YAN D, et al. Establishment of insomnia model of chronic unpredictable stress in rats. Heliyon. 2023;9(7):e18338.
[15] 闫德祺,张星平,张文慧,等.桂枝加龙骨牡蛎汤对肺不藏魄型不寐模型大鼠睡眠情况、血清及脑组织5羟色胺水平的影响[J].中医杂志,2022,63(19): 1865-1871.
[16] WANG Z, LI D, CHEN M, et al. A comprehensive study on the regulation of Compound Zaoren Granules on cAMP/CREB signaling pathway and metabolic disorder in CUMS-PCPA induced insomnia rats. J Ethnopharmacol. 2024;332: 118401.
[17] DU L, YANG D, WU L, et al. Integration of Gut Microbiota, Serum Metabolomic, and Network Pharmacology to Reveal the Anti Insomnia Mechanism of Mongolian Medicine Sugemule-4 Decoction on Insomnia Model Rats. Drug Des Devel Ther. 2024;18:2617-2639.
[18] WANG D, WU T, JIN J, et al. Periostracum Cicadae Extract and N-Acetyldopamine Regulate the Sleep-Related Neurotransmitters in PCPA-Induced Insomnia Rats. Molecules. 2024;29(15):3638.
[19] FU X, YAN S, HU Z, et al. Guhan Yangsheng Jing mitigates hippocampal neuronal pyroptotic injury and manifies learning and memory capabilities in sleep deprived mice via the NLRP3/Caspase1/GSDMD signaling pathway. J Ethnopharmacol. 2024;326:117972.
[20] WANG M, WANG G, ZHAO M, et al. Jujuboside A in ameliorating insomnia in mice via GABAergic modulation of the PVT. J Ethnopharmacol. 2025;349:119939.
[21] GONG Z, LAO D, WU Y, et al. Inhibiting PI3K/Akt-Signaling Pathway Improves Neurobehavior Changes in Anti-NMDAR Encephalitis Mice by Ameliorating Blood-Brain Barrier Disruption and Neuronal Damage. Cell Mol Neurobiol. 2023; 43(7):3623-3637.
[22] 田鸿芳,赵吉平.强弱刺激量针刺对肝郁化火型原发性失眠伴随抑郁焦虑情绪及生活质量影响的随机对照研究[J].中华中医药杂志,2022,37(2):1193-1197.
[23] 王业群,方无杰,项尚,等.解郁清心配方颗粒联合远程交互式CBT-I治疗肝郁化火型慢性失眠的临床疗效[J].中国实验方剂学杂志,2024,30(20):120-126.
[24] 徐金铭,王芳芳,曹迪,等.柴胡及其药对的药理研究进展[J/OL].中华中医药学刊,1-15[2025-07-30].https://link.cnki.net/urlid/21.1546.R.20250728.1644.012.
[25] 卜俊文,邵明国,杨元丰,等.白芍研究进展及其质量标志物(Q-Marker)预测[J/OL].中华中医药学刊,1-21[2025-07-30].https://link.cnki.net/urlid/21.1546.R.20250718.1119.010.
[26] HUANG Y, MIAO Q, CHEN N, et al. Tissue distribution of seven coptidis alkaloids in normal and insomnia rats affected by Cinnamomi cortex (Cinnamomum cassia) using liquid chromatography-tandem mass spectrometry. Fitoterapia. 2025;185:106723.
[27] LAVENDER I, MCGREGOR IS, SURAEV A, et al. Cannabinoids, Insomnia, and Other Sleep Disorders. Chest. 2022;162(2):452-465.
[28] KRAMER DJ, JOHNSON AA. Apigenin: a natural molecule at the intersection of sleep and aging. Front Nutr. 2024;11:1359176.
[29] HU Y, HE X, WU Y, et al. Sedative-Hypnotic Effect and Mechanism of Carbon Nanofiber Loaded with Essential Oils of Ligusticum chuanxiong (Ligusticum chuanxiong Hort.) and Finger Citron (Citrus medica L. var. sarcodactylis) on Mice Models of Insomnia. Biomolecules. 2024;14(9):1102.
[30] PRONIER É, MORICI JF, GIRARDEAU G. The role of the hippocampus in the consolidation of emotional memories during sleep. Trends Neurosci. 2023; 46(11):912-925.
[31] OLSEN LC, GALLER M, WITTER MP, et al. Transcriptional development of the hippocampus and the dorsal-intermediate-ventral axis in rats. Hippocampus. 2023;33(9):1028-1047.
[32] GIRI B, KINSKY N, KAYA U, et al. Sleep loss diminishes hippocampal reactivation and replay. Nature. 2024;630(8018):935-942.
[33] BECKER LA, PENAGOS H, FLORES FJ, et al. Eszopiclone and Zolpidem Produce Opposite Effects on Hippocampal Ripple Density. Front Pharmacol. 2022;12:792148.
[34] ZHANG K, WANG F, ZHAI M, et al. Hyperactive neuronal autophagy depletes BDNF and impairs adult hippocampal neurogenesis in a corticosterone-induced mouse model of depression. Theranostics. 2023;13(3):1059-1075.
[35] LI H, SUN J, WU Y, et al. Honokiol relieves hippocampal neuronal damage in Alzheimer’s disease by activating the SIRT3-mediated mitochondrial autophagy. CNS Neurosci Ther. 2024;30(8):e14878.
[36] LIU S, YAO S, YANG H, et al. Autophagy: Regulator of cell death. Cell Death Dis. 2023;14(10):648.
[37] LI W, HE P, HUANG Y, et al. Selective autophagy of intracellular organelles: recent research advances. Theranostics. 2021;11(1):222-256.
[38] HWANG HJ, HA H, LEE BS, et al. LC3B is an RNA-binding protein to trigger rapid mRNA degradation during autophagy. Nat Commun. 2022;13(1):1436.
[39] LI J, WANG Y, LUO Y, et al. USP5-Beclin 1 axis overrides p53-dependent senescence and drives Kras-induced tumorigenicity. Nat Commun. 2022;13(1):7799.
[40] RONG Y, FAN J, JI C, et al. USP11 regulates autophagy-dependent ferroptosis after spinal cord ischemia-reperfusion injury by deubiquitinating Beclin 1. Cell Death Differ. 2022;29(6):1164-1175.
[41] HUANG X, YAO J, LIU L, et al. S-acylation of p62 promotes p62 droplet recruitment into autophagosomes in mammalian autophagy. Mol Cell. 2023;83(19):3485-3501.e11.
[42] KUMAR AV, MILLS J, LAPIERRE LR. Selective Autophagy Receptor p62/SQSTM1, a Pivotal Player in Stress and Aging. Front Cell Dev Biol. 2022;10:793328.
[43] LI R, PAN Y, JING N, et al. Flavonoids from mulberry leaves exhibit sleep-improving effects via regulating GABA and 5-HT receptors. J Ethnopharmacol. 2025;337(Pt 3):118734.
[44] LIU YM, LI JC, GU YF, et al. Cannabidiol Exerts Sedative and Hypnotic Effects in Normal and Insomnia Model Mice Through Activation of 5-HT1A Receptor. Neurochem Res. 2024;49(5):1150-1165.
[45] YU L, WEI J, LIU P. Attacking the PI3K/Akt/mTOR signaling pathway for targeted therapeutic treatment in human cancer. Semin Cancer Biol. 2022;85:69-94.
[46] CAO F, XU Y, ZHANG M, et al. Baihui (DU20), Shenmen (HT7) and Sanyinjiao (SP6) target the cAMP/CREB/BDNF and PI3K/Akt pathways to reduce central nervous system apoptosis in rats with insomnia. Heliyon. 2022;8(12):e12574.
[47] ZHU W, GONG A, ZHANG B, et al. The Chronobiological and Neuroprotective Mechanisms of Resveratrol in Improving Sleep. Mediators Inflamm. 2025; 2025:4954030.
[48] LI R, ZHENG Y, ZHANG J, et al. Gomisin N attenuated cerebral ischemia-reperfusion injury through inhibition of autophagy by activating the PI3K/AKT/mTOR pathway. Phytomedicine. 2023;110:154644.
[49] DING MR, QU YJ, HU B, et al. Signal pathways in the treatment of Alzheimer’s disease with traditional Chinese medicine. Biomed Pharmacother. 2022;152: 113208.
[50] WANG L, TIAN S, RUAN S, et al. Neuroprotective effects of cordycepin on MPTP-induced Parkinson’s disease mice via suppressing PI3K/AKT/mTOR and MAPK-mediated neuroinflammation. Free Radic Biol Med. 2024;216:60-77.
[51] SHI M, YANG J, LIU Y, et al. Huanglian Wendan Decoction Improves Insomnia in Rats by Regulating BDNF/TrkB Signaling Pathway Through Gut Microbiota-Mediated SCFAs and Affecting Microglia Polarization. Mol Neurobiol. 2025;62(1):1047-1066.
[52] BAI G, QIAO Y, LO PC, et al. Anti-depressive effects of Jiao-Tai-Wan on CORT-induced depression in mice by inhibiting inflammation and microglia activation. J Ethnopharmacol. 2022;283:114717.
[53] WU Y, YAO C, ZHANG L, et al. Sophora flavescens alcohol extract ameliorates insomnia and promotes PI3K/AKT/BDNF signaling transduction in insomnia model rats. Neuroreport. 2024;35(5):275-282.
|