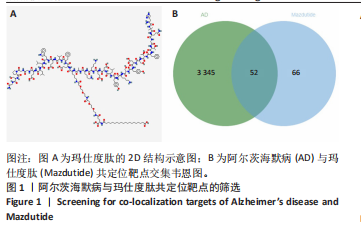
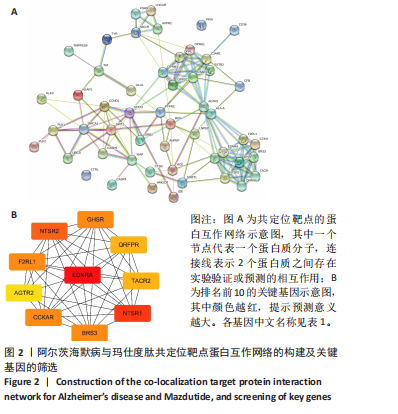
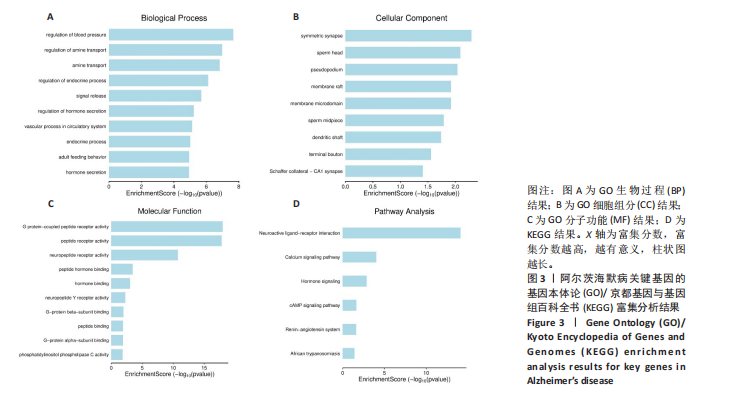
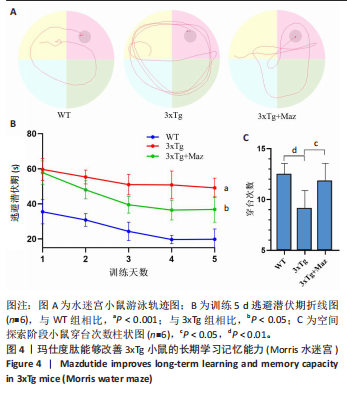
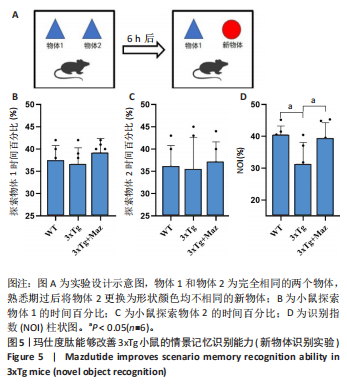
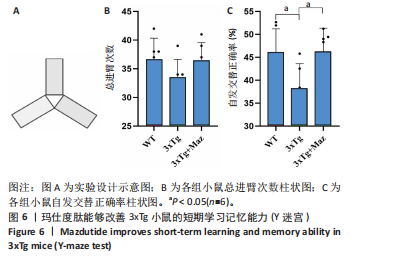
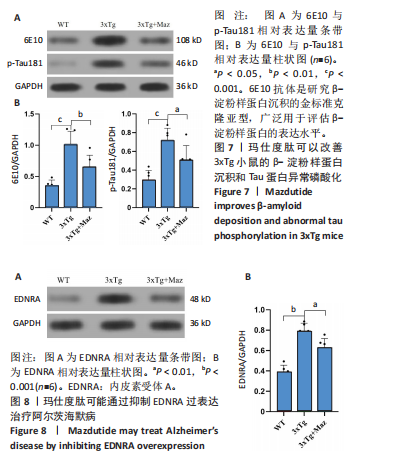
[1] ZHANG Z, XUE P, BENDLIN BB, et al. Melatonin: a potential nighttime guardian against Alzheimer’s. Mol Psychiatry. 2025;30(1):237-250.
[2] LANE CA, HARDY J, SCHOTT JM, et al. Alzheimer’s disease. Eur J Neurol. 2018; 25(1):59-70.
[3] ZHENG Q, WANG X. Alzheimer’s disease: insights into pathology, molecular mechanisms, and therapy. Protein Cell. 2025;16(2):83-120.
[4] HARDY J. Alzheimer’s Disease: Treatment Challenges for the Future. J Neurochem. 2025;169(8):e70176.
[5] WANG R, REDDY PH. Role of Glutamate and NMDA Receptors in Alzheimer’s Disease. J Alzheimers Dis. 2017;57(4):1041-1048.
[6] LIANG Y, DORE V, ROWE CC, et al. Clinical Evidence for GLP-1 Receptor Agonists in Alzheimer’s Disease: A Systematic Review. J Alzheimers Dis Rep. 2024;8(1):777-789.
[7] JI L, JIANG H, BI Y, et al. Once-Weekly Mazdutide in Chinese Adults with Obesity or Overweight. N Engl J Med. 2025;392(22):2215-2225.
[8] VARGAS-SORIA M, CARRANZA-NAVAL MJ, DEL MARCO A, et al. Role of liraglutide in Alzheimer’s disease pathology. Alzheimers Res Ther. 2021;13(1):112.
[9] WANG W, WANG Q, QI X, et al. Associations of semaglutide with first-time diagnosis of Alzheimer’s disease in patients with type 2 diabetes: Target trial emulation using nationwide real-world data in the US. Alzheimers Dement. 2024; 20(12):8661-8672.
[10] WANG ZJ, LI XR, CHAI SF, et al. Semaglutide ameliorates cognition and glucose metabolism dysfunction in the 3xTg mouse model of Alzheimer’s disease via the GLP-1R/SIRT1/GLUT4 pathway. Neuropharmacology. 2023;240:109716.
[11] SALAMEH TS, RHEA EM, TALBOT K, et al. Brain uptake pharmacokinetics of incretin receptor agonists showing promise as Alzheimer’s and Parkinson’s disease therapeutics. Biochem Pharmacol. 2020;180:114187.
[12] DONG W, BAI J, YUAN Q, et al. Mazdutide, a dual agonist targeting GLP-1R and GCGR, mitigates diabetes-associated cognitive dysfunction: mechanistic insights from multi-omics analysis. EBioMedicine. 2025;117:105791.
[13] DING K, ZHANG Z, HAN Z, et al. Liver ALKBH5 regulates glucose and lipid homeostasis independently through GCGR and mTORC1 signaling. Science. 2025;387(6737):eadp4120.
[14] ZIMMERMANN T, THOMAS L, BAADER-PAGLER T, et al. BI 456906: Discovery and preclinical pharmacology of a novel GCGR/GLP-1R dual agonist with robust anti-obesity efficacy. Mol Metab. 2022;66:101633.
[15] PINERO J, SAUCH J, SANZ F, et al. The DisGeNET cytoscape app: Exploring and visualizing disease genomics data. Comput Struct Biotechnol J. 2021;19:2960-2967.
[16] WANG Y, XIAO J, SUZEK TO, et al. PubChem’s BioAssay Database. Nucleic Acids Res. 2012;40(Database issue):D400-D412.
[17] SHANNON P, MARKIEL A, OZIER O, et al. Cytoscape: a software environment for integrated models of biomolecular interaction networks. Genome Res. 2003; 13(11):2498-2504.
[18] CHIN CH, CHEN SH, WU HH, et al. CytoHubba: identifying hub objects and sub-networks from complex interactome. BMC Syst Biol. 2014;8 Suppl 4(Suppl 4):S11.
[19] CHEN T, SUN T, BIAN Y, et al. The Design and Optimization of Monomeric Multitarget Peptides for the Treatment of Multifactorial Diseases. J Med Chem. 2022;65(5):3685-3705.
[20] BERTONI-FREDDARI C, SENSI SL, GIORGETTI B, et al. Decreased presence of perforated synapses in a triple-transgenic mouse model of Alzheimer’s disease. Rejuvenation Res. 2008;11(2):309-313.
[21] WANG ZJ, HAN WN, CHAI SF, et al. Semaglutide promotes the transition of microglia from M1 to M2 type to reduce brain inflammation in APP/PS1/tau mice. Neuroscience. 2024;563:222-234.
[22] MEKADA K, YOSHIKI A. Substrains matter in phenotyping of C57BL/6 mice. Exp Anim. 2021;70(2):145-160.
[23] CABRAL-MARQUES O, MARQUES A, GIIL LM, et al. GPCR-specific autoantibody signatures are associated with physiological and pathological immune homeostasis. Nat Commun. 2018;9(1):5224.
[24] XU Z, ZHOU Q, LIU C, et al. EDNRA affects susceptibility to large artery atherosclerosis stroke through potential inflammatory pathway. Sci Rep. 2024; 14(1):25173.
[25] WANG M, WANG L, LI X, et al. EDNRA regulates the tumour immune environment and predicts the efficacy and prognosis of cancer immunotherapy. J Cell Mol Med. 2024;28(22):e70172.
[26] LING W, JOHNSON SK, MEHDI SJ, et al. EDNRA-Expressing Mesenchymal Cells are Expanded in Myeloma Interstitial Bone Marrow and Associated with Disease Progression. Cancers (Basel). 2023;15(18):4519.
[27] LEE YJ, JUNG E, CHOI J, et al. The EDN1/EDNRA/beta‑arrestin axis promotes colorectal cancer progression by regulating STAT3 phosphorylation. Int J Oncol. 2023;62(1):13.
[28] PALMER JC, BARKER R, KEHOE PG, et al. Endothelin-1 is elevated in Alzheimer’s disease and upregulated by amyloid-beta. J Alzheimers Dis. 2012;29(4):853-861.
[29] MINERS JS, PALMER JC, TAYLER H, et al. Abeta degradation or cerebral perfusion? Divergent effects of multifunctional enzymes. Front Aging Neurosci. 2014;6:238.
[30] ZHANG J, WANG YJ, WANG X, et al. PKC-Mediated Endothelin-1 Expression in Endothelial Cell Promotes Macrophage Activation in Atherogenesis. Am J Hypertens. 2019;32(9):880-889.
[31] YANG W, LIU Y, XU QQ, et al. Sulforaphene Ameliorates Neuroinflammation and Hyperphosphorylated Tau Protein via Regulating the PI3K/Akt/GSK-3beta Pathway in Experimental Models of Alzheimer’s Disease. Oxid Med Cell Longev. 2020;2020:4754195.
[32] PAHLAVANI HA. Exercise therapy to prevent and treat Alzheimer’s disease. Front Aging Neurosci. 2023;15:1243869.
[33] RAJABLI F, BENCHEK P, TOSTO G, et al. Multi-ancestry genome-wide meta-analysis of 56,241 individuals identifies known and novel cross-population and ancestry-specific associations as novel risk loci for Alzheimer’s disease. Genome Biol. 2025;26(1):210.
[34] SINGH N, NANDY SK, JYOTI A, et al. Protein Kinase C (PKC) in Neurological Health: Implications for Alzheimer’s Disease and Chronic Alcohol Consumption. Brain Sci. 2024;14(6):554.
[35] VERMA A, CHAUDHARY S, SOLANKI K, et al. Exendin-4: a potential therapeutic strategy for Alzheimer’s disease and Parkinson’s disease. Chem Biol Drug Des. 2024;103(1):e14426.
[36] KANG X, WANG D, ZHANG L, et al. Exendin-4 ameliorates tau hyperphosphorylation and cognitive impairment in type 2 diabetes through acting on Wnt/beta-catenin/NeuroD1 pathway. Mol Med. 2023;29(1):118.
[37] RAJABI H, AHMADI M, ASLANI S, et al. Exendin-4 as a Versatile Therapeutic Agent for the Amelioration of Diabetic Changes. Adv Pharm Bull. 2022;12(2):237-247.
[38] ESPARZA-SALAZAR FJ, LEZAMA-TOLEDO AR, RIVERA-MONROY G, et al. Exendin-4 for Parkinson’s disease. Brain Circ. 2021;7(1):41-43.
[39] WEI R, ZHANG L, HU W, et al. Zeb2/Axin2-Enriched BMSC-Derived Exosomes Promote Post-Stroke Functional Recovery by Enhancing Neurogenesis and Neural Plasticity. J Mol Neurosci. 2022;72(1):69-81.
[40] YANG G, PEI YN, SHAO SJ, et al. [Effects of electroacupuncture at “Baihui” and “Yongquan” on the levels of synaptic plasticity related proteins postsynaptic density-95 and synaptophysin in hippocampus of APP/PS1 mice]. Zhen Ci Yan Jiu. 2020;45(4):310-314.
[41] VE H, CABANA VC, GOUSPILLOU G, et al. Quantitative Immunoblotting Analyses Reveal that the Abundance of Actin, Tubulin, Synaptophysin and EEA1 Proteins is Altered in the Brains of Aged Mice. Neuroscience. 2020;442:100-113.
[42] WANG H, SHEN Z, WU CS, et al. Neuronal ablation of GHSR mitigates diet-induced depression and memory impairment via AMPK-autophagy signaling-mediated inflammation. Front Immunol. 2024;15:1339937.
[43] ZHANG LQ, ZHANG W, LI T, et al. GLP-1R activation ameliorated novel-object recognition memory dysfunction via regulating hippocampal AMPK/NF-kappaB pathway in neuropathic pain mice. Neurobiol Learn Mem. 2021;182:107463.
[44] ZHANG QH, HAO JW, LI GL, et al. Proinflammatory switch from Galphas to Galphai signaling by Glucagon-like peptide-1 receptor in murine splenic monocyte following burn injury. Inflamm Res. 2018;67(2):157-168.
[45] ATEF MM, ABOU HN, HAFEZ YM, et al. The potential protective effect of liraglutide on valproic acid induced liver injury in rats: Targeting HMGB1/RAGE axis and RIPK3/MLKL mediated necroptosis. Cell Biochem Funct. 2023;41(8):1209-1219.
[46] JI L, JIANG H, CHENG Z, et al. A phase 2 randomised controlled trial of mazdutide in Chinese overweight adults or adults with obesity. Nat Commun. 2023;14(1):8289.
[47] BHATTACHAR SN, THAM LS, LI Y, et al. Mazdutide reduces body weight in adults with overweight or obesity: a high-dose Phase 1 trial. Diabetes Obes Metab. 2025;27(11):6460-6469.
[48] AYESH H, AYESH S, NISWENDER K. Mazdutide Versus Dulaglutide for Weight Loss and Diabetes Management: Meta-Analysis of Randomized Clinical Trials. Am J Ther. 2024;31(5):e619-e622.
[49] DONG W, BAI J, YUAN Q, et al. Mazdutide, a dual agonist targeting GLP-1R and GCGR, mitigates diabetes-associated cognitive dysfunction: mechanistic insights from multi-omics analysis. EBioMedicine. 2025;117:105791.
[50] JI L, JIANG H, BI Y, et al. Once-Weekly Mazdutide in Chinese Adults with Obesity or Overweight. N Engl J Med. 2025;392(22):2215-2225.
|