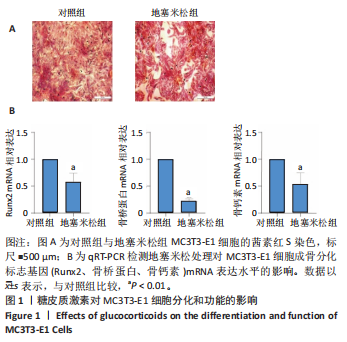
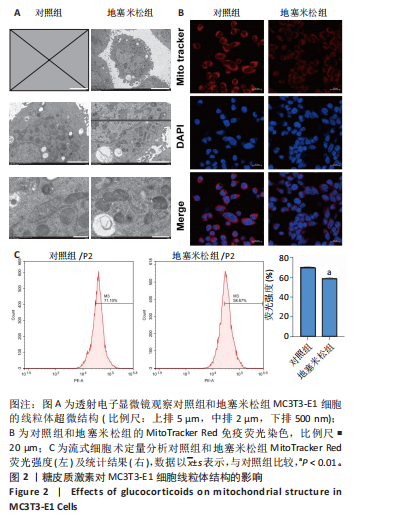
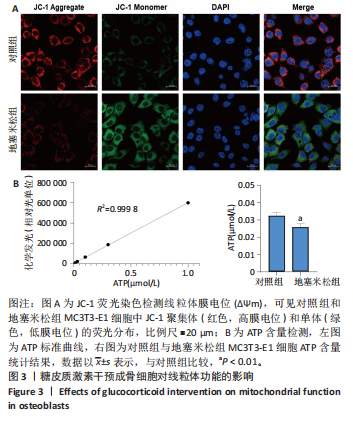
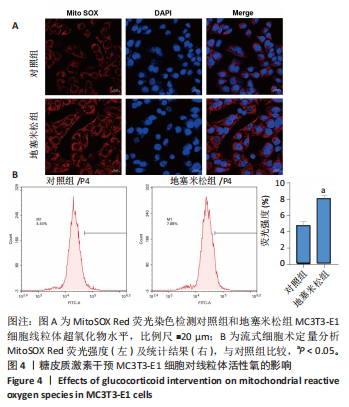
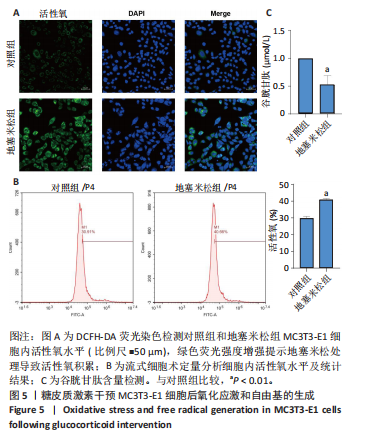
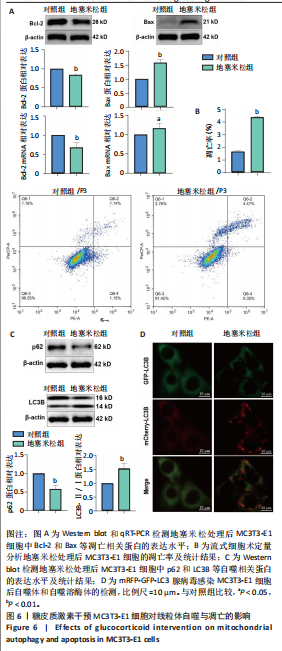
[1] 朱正杰,彭昊.成骨细胞在激素性股骨头坏死发病机制中的作用研究进展[J].生物骨科材料与临床研究,2025,22(1):76-81.
[2] GUO S, MAO L, JI F, et al. Activating AMP-activated protein kinase by an α1 selective activator compound 13 attenuates dexamethasone-induced osteoblast cell death. Biochem Biophys Res Commun. 2016;471(4):545-552.
[3] CHEN K, LIU Y, HE J, et al. Steroid-induced osteonecrosis of the femoral head reveals enhanced reactive oxygen species and hyperactive osteoclasts. Int J Biol Sci. 2020;16(11):1888-1900.
[4] 梁朝鑫,周安远,王伟,等.骨髓间充质干细胞治疗骨科疾病研究进展[J].陕西医学杂志,2022,51(1):121-124+129.
[5] LU C, QI H, XU H, et al. Global research trends of steroid-induced osteonecrosis of the femoral head: A 30-year bibliometric analysis. Front Endocrinol (Lausanne). 2022;13:1027603.
[6] LIU Q, WU Y, LI S, et al. Ursolic acid alleviates steroid-induced avascular necrosis of the femoral head in mouse by inhibiting apoptosis and rescuing osteogenic differentiation. Toxicol Appl Pharmacol. 2023;475:116649.
[7] 康少平,刘淑艳,李永升,等.激素性股骨头坏死患者骨髓间充质干细胞体外增殖分化能力的研究[J].中国细胞生物学学报,2015,37(11):1490-1496.
[8] ZHANG X, YANG Z, XU Q, et al. Dexamethasone Induced Osteocyte Apoptosis in Steroid-Induced Femoral Head Osteonecrosis through ROS-Mediated Oxidative Stress. Orthop Surg. 2024;16(3):733-744.
[9] YANG N, SUN H, XUE Y, et al. Inhibition of MAGL activates the Keap1/Nrf2 pathway to attenuate glucocorticoid-induced osteonecrosis of the femoral head. Clin Transl Med. 2021;11(6):e447.
[10] TAHRIR F G, LANGFORD D, AMINI S, et al. Mitochondrial quality control in cardiac cells: mechanisms and role in cardiac cell injury and disease. J Cell Physiol. 2019; 234(6):8122-8133.
[11] YANG H, DING N, QING S, et al. Knockdown of lncRNA XR_877193.1 suppresses ferroptosis and promotes osteogenic differentiation via the PI3K/AKT signaling pathway in SONFH. Acta Biochim Biophys Sin (Shanghai). 2025;57(8):1350-1362.
[12] 张杰,曹建泽,刘永飞,等.激素性股骨头坏死发病机制的研究进展[J].中国矫形外科杂志,2024,32(7):620-624+630.
[13] KOKKINOPOULOU I, MOUTSATSOU P. Mitochondrial Glucocorticoid Receptors and Their Actions. Int J Mol Sci. 2021;22(11):6054.
[14] 胡康一,曹林忠,万超超,等.基于线粒体功能障碍探讨中药防治激素性股骨头坏死的基础研究进展[J].中国临床药理学与治疗学,2023,28(6):688-696.
[15] YANG D, OKAMURA H, QIU L. Upregulated osterix expression elicited by Runx2 and Dlx5 is required for the accelerated osteoblast differentiation in PP2A Cα-knockdown cells. Cell Biol Int. 2018;42(4):403-410.
[16] CHEN L, SHI X, WENG SJ, et al. Vitamin K2 can rescue the dexamethasoneinduced downregulation of osteoblast autophagy and mitophagy thereby restoring osteoblast function in vitro and in vivo. Front Pharmacol. 2020;11:1209.
[17] HUANG X, JIE S, LI W, et al. miR-122-5p targets GREM2 to protect against glucocorticoid-induced endothelial damage through the BMP signaling pathway. Mol Cell Endocrinol. 2022;544:111541.
[18] LIU BH, XU CZ, LIU Y, et al. Mitochondrial quality control in human health and disease. Mil Med Res. 2024;11(1):32.
[19] 申屠路媚,牟艳玲.线粒体功能障碍机制及其相关疾病研究进展[J].生命科学,2018,30(1):87-93.
[20] YISANG Y,HAKJOO L,MARILEN F,et al.Non-conventional mitochondrial permeability transition:its regulation by mitochondrial dynamics. Biochim Biophys Acta Bioenerg. 2022;1864(1):148914.
[21] WANG, YUAN, LIU, et al. The role of mitochondria in aging, cell death, and tumor immunity. Front Immunol. 2024;15:1520072.
[22] GUO R, ZONG S, WU M, et al. Architecture of Human Mitochondrial Respiratory Megacomplex I(2)III(2)IV(2). Cell. 2017;170:1247-57 e12.
[23] 赵阳,李嘉麟,吴箫,等.胆碱激酶α沉默诱导线粒体功能障碍影响胶质瘤细胞的增殖和凋亡[J].中国组织工程研究,2026,30(1):130-138.
[24] REN GW, WEN SB, HAN J, et al. Network-Based Pharmacology and Bioinformatics Study on the Mechanism of Action of Gujiansan in the Treatment of Steroid-Induced Avascular Necrosis of the Femoral Head. Biomed Res Int. 2022;2022: 8080679.
[25] 唐建萍,毕泰瑜,魏士雄.心力衰竭中氧化应激的病理机制及治疗进展[J].广州医科大学学报,2025,53(1):54-60.
[26] ZHANG X, PANG R, ZHANG K, et al. Apocynin exerts cytoprotective effects on dexamethasone-induced osteoblasts by inhibiting oxidative stress through the Nrf2 signalling pathway. J Cell Mol Med. 2023;27(23):3911-3927.
[27] 毛筝,肖桂扬,梁淑芬,等.α-MSH调控Bcl-2线粒体途径在桃红四物汤防治激素性股骨头坏死中的作用机制[J].智慧健康,2023,9(34):40-45+49.
[28] YU L,CHEN Y,TOOZE SA.Autophagy pathway:Cellular and molecular mechanisms.Autophagy. 2018;14(2):207-215.
[29] TSUJIMOTO Y, SHIMIZU S. Another way to die: autophagic programmed cell death. Cell Death Differ. 2005;12 Suppl 2:1528-1534.
[30] ZHANG S, LIU Y, LIANG Q. Low-dose dexamethasone affects osteoblast viability by inducing autophagy via intracellular ROS. Mol Med Rep. 2018;17(3):4307-4316.
[31] 张勇杰,曹林忠,万超超,等.线粒体自噬在激素性股骨头坏死发病中的作用及研究进展[J].风湿病与关节炎,2023,12(10):66-70.
[32] LUO Z, XU X, SHO T, et al. ROS-induced autophagy regulates porcinetrophectoderm cell apoptosis, proliferation, and differentiation. Am J Physiol Cell Physiol. 2019;316(2):C198-C209.
[33] KISHIMOTO C, TOMIOKA N, NAKAYAMA Y, et al.Anti-oxidant effects of coenzyme Q10 on experimental viral myocarditis in mice. J Cardiovasc Pharmacol. 2003; 42(5):588-592. |