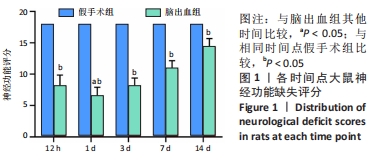
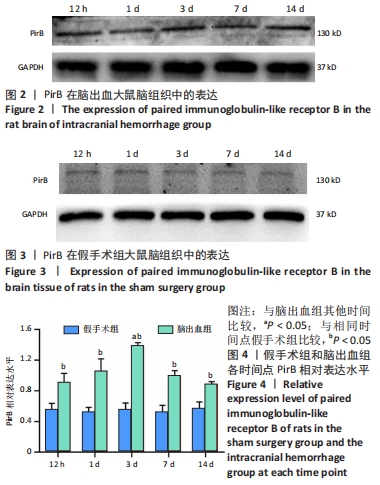
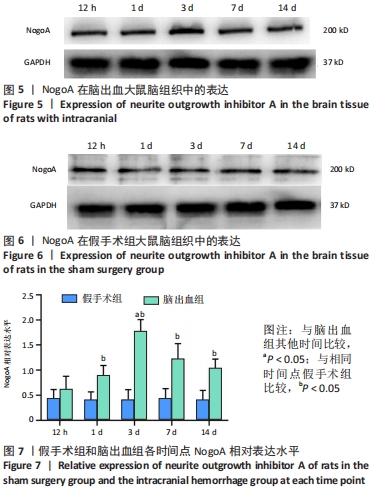
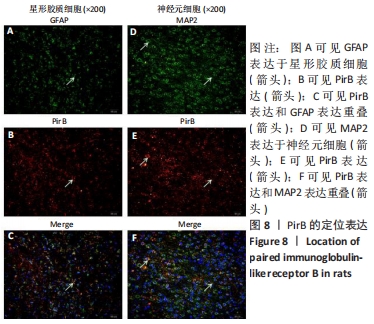
[1] LAN X, HAN X, LI Q, et al. Modulators of microglial activation and polarization after intracerebral haemorrhage. Nat Rev Neurol. 2017; 13(7):420-433.
[2] SCHWAB ME, CARONI P. Antibody against myelin-associated inhibitor of neurite growth neutralizes nonpermissive substrate properties of CNS white matter. Neuron. 2008;60(3):404-405.
[3] CARONI P, SCHWAB ME. Two membrane protein fractions from rat central myelin with inhibitory properties for neurite growth and fibroblast spreading. J Cell Biol. 1988;106(4):1281-1288.
[4] CHONG SY, ROSENBERG SS, FANCY SP, et al. Neurite outgrowth inhibitor Nogo-A establishes spatial segregation and extent of oligodendrocyte myelination. Proc Natl Acad Sci U S A. 2012;109(4):1299-1304.
[5] KUBAGAWA H, BURROWS PD, COOPER MD. A novel pair of immunoglobulin-like receptors expressed by B cells and myeloid cells. Proc Natl Acad Sci U S A. 1997;94(10):5261-5266.
[6] SYKEN J, GRANDPRE T, KANOLD PO, et al. PirB restricts ocular-dominance plasticity in visual cortex. Science. 2006;313(5794):1795-1800.
[7] ATWAL JK, PINKSTON-GOSSE J, SYKEN J, et al. PirB is a functional receptor for myelin inhibitors of axonal regeneration. Science. 2008; 322(5903):967-970.
[8] SCHMANDKE A, SCHMANDKE A, SCHWAB ME. Nogo-A: Multiple Roles in CNS Development, Maintenance, and Disease. Neuroscientist. 2014; 20(4):372-386.
[9] ZHONG J, FAN S, YAN Z, et al. Effects of Nogo-A Silencing on TNF-α and IL-6 Secretion and TH Downregulation in Lipopolysaccharide-Stimulated PC12 Cells. Biomed Res Int. 2015;2015:817914.
[10] INEICHEN BV, PLATTNER PS, GOOD N, et al. Nogo-A Antibodies for Progressive Multiple Sclerosis. CNS Drugs. 2017;31(3):187-198.
[11] DENG XH, AI WM, LEI DL, et al. Lipopolysaccharide induces paired immunoglobulin-like receptor B (PirB) expression, synaptic alteration, and learning-memory deficit in rats. Neuroscience. 2012;209:161-170.
[12] VANGUILDER STARKEY HD, VAN KIRK CA, BIXLER GV, et al. Neuroglial expression of the MHCI pathway and PirB receptor is upregulated in the hippocampus with advanced aging. J Mol Neurosci. 2012;48(1):111-126.
[13] WANG J, ZHANG Y, XIA J, et al. Neuronal PirB Upregulated in Cerebral Ischemia Acts as an Attractive Theranostic Target for Ischemic Stroke. J Am Heart Assoc. 2018;7(3):e007197.
[14] DENG B, LI L, GOU X, et al. TAT-PEP Enhanced Neurobehavioral Functional Recovery by Facilitating Axonal Regeneration and Corticospinal Tract Projection After Stroke. Mol Neurobiol. 2018;55(1): 652-667.
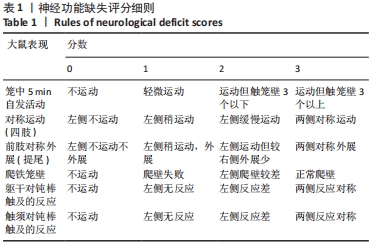
[15] HUA Y, SCHALLERT T, KEEP RF, et al. Behavioral tests after intracerebral hemorrhage in the rat. Stroke. 2002;33(10):2478-2484.
[16] DEINSBERGER W, VOGEL J, KUSCHINSKY W, et al. Experimental intracerebral hemorrhage: description of a double injection model in rats. Neurol Res. 1996;18(5):475-477.
[17] BEDERSON JB, PITTS LH, TSUJI M, et al. Rat middle cerebral artery occlusion: evaluation of the model and development of a neurologic examination. Stroke. 1986;17(3):472-476.
[18] GARCIA JH, WAGNER S, LIU KF, et al. Neurological deficit and extent of neuronal necrosis attributable to middle cerebral artery occlusion in rats. Statistical validation. Stroke. 1995;26(4):627-634.
[19] FEIGIN VL, LAWES CM, BENNETT DA, et al. Worldwide stroke incidence and early case fatality reported in 56 population-based studies: a systematic review. Lancet Neurol. 2009;8(4):355-369.
[20] SACCO S, MARINI C, TONI D, et al. Incidence and 10-year survival of intracerebral hemorrhage in a population-based registry. Stroke. 2009;40(2):394-399.
[21] QURESHI AI, MENDELOW AD, HANLEY DF. Intracerebral haemorrhage. Lancet. 2009;373(9675):1632-1644.
[22] KEEP RF, HUA Y, XI G. Intracerebral haemorrhage: mechanisms of injury and therapeutic targets. Lancet Neurol. 2012;11(8):720-731.
[23] SHEN H, CHEN Z, WANG Y, et al. Role of Neurexin-1β and Neuroligin-1 in Cognitive Dysfunction After Subarachnoid Hemorrhage in Rats. Stroke. 2015;46(9):2607-2615.
[24] ZHU W, GAO Y, WAN J, et al. Changes in motor function, cognition, and emotion-related behavior after right hemispheric intracerebral hemorrhage in various brain regions of mouse. Brain Behav Immun. 2018;69:568-581.
[25] SMEDFORS G, OLSON L, KARLSSON TE. A Nogo-Like Signaling Perspective from Birth to Adulthood and in Old Age: Brain Expression Patterns of Ligands, Receptors and Modulators. Front Mol Neurosci. 2018;11:42.
[26] TANG BL. Nogo-A and the regulation of neurotransmitter receptors. Neural Regen Res. 2020;15(11):2037-2038.
[27] XIE YX, ZHANG M, ZHANG CR, et al. Relationship between NogoA/NgR1/RhoA signaling pathway and the apoptosis of cerebral neurons after cerebral infarction in rats. Eur Rev Med Pharmacol Sci. 2020;24(1): 295-303.
[28] LIU Y, MA C, LI H, et al. Nogo-A/Pir-B/TrkB Signaling Pathway Activation Inhibits Neuronal Survival and Axonal Regeneration After Experimental Intracerebral Hemorrhage in Rats. J Mol Neurosci. 2019;69(3):360-370.
[29] WAGNER I, HAUER EM, STAYKOV D, et al. Effects of continuous hypertonic saline infusion on perihemorrhagic edema evolution. Stroke. 2011;42(6):1540-1545.
[30] VLIEG HC, HUIZINGA EG, JANSSEN BJC. Structure and flexibility of the extracellular region of the PirB receptor. J Biol Chem. 2019;294(12): 4634-4643.
[31] LI L, DENG B, LI S, et al. TAT-PEP, a novel blocker of PirB, enhances the recovery of cognitive function in mice after transient global cerebral ischemia. Behav Brain Res. 2017;326:322-330.
|