Chinese Journal of Tissue Engineering Research ›› 2014, Vol. 18 ›› Issue (16): 2544-2549.doi: 10.3969/j.issn.2095-4344.2014.16.014
Previous Articles Next Articles
Constructing a controlled-release dexamethasone-loaded titania nanotube system
Wang Ming, Zhang He, Wang Lu, Deng Feng, Yang Sheng
- Department of Prosthodontics, Affiliated Hospital of Stomatology, Chongqing Medical University, Chongqing Key Laboratory for Oral Diseases and Biomedical Sciences, Chongqing 401147, China
-
Revised:2014-04-02Online:2014-04-16Published:2014-04-16 -
Contact:Yang Sheng, D.D.S., Ph.D., Attending physician, Lecturer Department of Prosthodontics, Affiliated Hospital of Stomatology, Chongqing Medical University, Chongqing Key Laboratory for Oral Diseases and Biomedical Sciences, Chongqing 401147, China -
About author:Wang Ming, Studying for master’s degree, Physician, Department of Prosthodontics, Affiliated Hospital of Stomatology, Chongqing Medical University, Chongqing Key Laboratory for Oral Diseases and Biomedical Sciences, Chongqing 401147, China -
Supported by:Doctoral Startup Fund of College of Stomatology in Chongqing Medical University, No. 2011; the General Program of Chongqing Municipal Health Bureau, No. 2012-2-121; the Natural Science Foundation of Chongqing Science & Technology Commission, No. CSTC2012jjA0178; Specialized Research Fund for the Doctoral Program of Higher Education in China, No. 20125503120009
CLC Number:
Cite this article
Wang Ming, Zhang He, Wang Lu, Deng Feng, Yang Sheng . Constructing a controlled-release dexamethasone-loaded titania nanotube system[J]. Chinese Journal of Tissue Engineering Research, 2014, 18(16): 2544-2549.
share this article

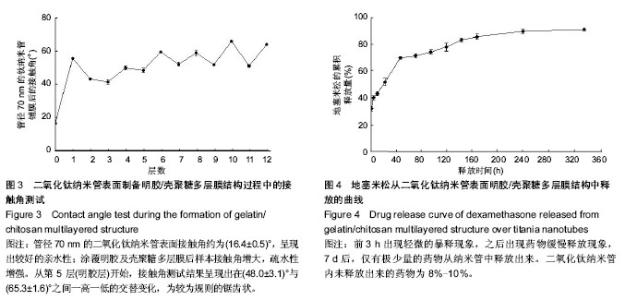
2.1 阳极氧化法制备的二氧化钛纳米管形貌 图1A示扫描电镜观察钛箔片的形貌,可见不规则微米级粗糙不平的结构。图1B示扫描电镜观察阳极氧化处理后的二氧化钛纳米管形貌,见垂直于钛基底的一端开口的纳米级管状结构,管径大小均匀,排列整齐,管径约为70 nm。 2.2 二氧化钛纳米管表面明胶/壳聚糖多层膜结构的形貌 图2A示二氧化钛纳米管表面已制备好的明胶/壳聚糖多层膜结构,已完全覆盖二氧化钛纳米管表面,封堵纳米管管口。图2B为二氧化钛纳米管表面已制备好的明胶/壳聚糖多层结构降解1 d后扫描电镜所观察到的形貌,先前完整的膜结构发生改变,逐渐降解,可见网状结构的形成。 2.3 接触角测试 图3所示为制备明胶/壳聚糖多层结构过程中的接触角测试的结果。管径70 nm的二氧化钛纳米管表面接触角约为(16.4±0.5)°,呈现出较好的亲水性;涂覆明胶及壳聚糖多层膜后样本接触角增大,疏水性增强。从第5层(明胶层)开始,接触角测试结果呈现出在(48.0±3.1)°与(65.3±1.6)°之间一高一低的交替变化,为较为规则的锯齿状。"

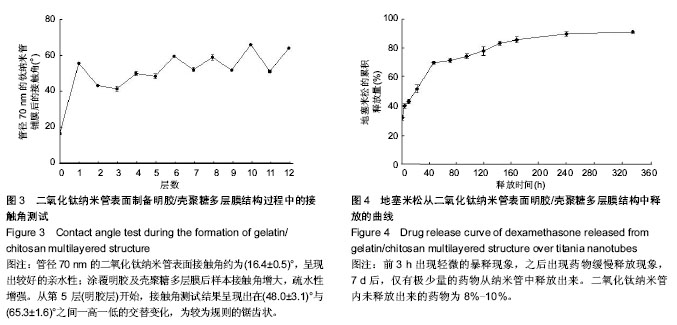
2.4 药物释放 图4为地塞米松从二氧化钛纳米管明胶/壳聚糖复合结构中释放出来的药物释放曲线。前3 h累积释放药物为初始负载量的(32.7±1.5)%,前6 h累积释放(40.7±1.5)%,前12 h累积释放(43.7±1.5)%,前24 h(1 d)累积释放(52.3±2.5)%,前48 h(2 d)累积释放(70±1)%,前72 h(3 d)累积释放(71.7±1.5)%,前96 h(4 d)累积释放(74.3%±1.5)%,前120 h(5 d)累积释放(78±3)%,前144 h (6 d)累积释放(83.3±1.5)%,前168 h(7 d)累积释放(85.7±2.1)%,前240 h(10 d)累积释放(89.7±1.5)%,前 336 h(4 d)累积释放(91±1)%。前3 h出现轻微的暴释现象,之后出现药物缓慢释放现象,7 d后,仅有极少量的药物从纳米管中释放出来。二氧化钛纳米管内未释放出来的药物为8%-10%。"
| [1] Hu Y,Cai K,Luo Z,et al.Layer-by-layer assembly of b-estradiol loaded mesoporous silica nanoparticles on titanium substrates and its implication for bone homeostasis.Adv Mater. 2010;22:4146-4150.
[2] Shi J,Votruba AR,Farokhzad OC,et al.Nanotechnology in drug delivery and tissue engineering: from discovery to applications. Nano Lett.2010;10(9):3223-3230.
[3] Lai M,Cai K,Zhao L,et al.Surface functionalization of TiO2 nanotubes with bone morphogenetic protein 2 and its synergistic effect on the differentiation of mesenchymal stem cells.Biomacromolecules.2011;12(4):1097-1105.
[4] Hu Y,Cai K,Luo Z,et al.TiO2 nanotubes as drug nanoreservoirs for the regulation of mobility and differentiation of mesenchymal stem cells. Acta Biomaterialia. 2012;8(1): 439-448.
[5] Tan AW,Pingguan-Murphy B,Ahmad R,et al.Review of titania nanotubes: Fabrication and cellular response.Ceramics International.2012;38(6):4421-4435.
[6] van Kooten C,Stax AS,Woltman AM,et al.Handbook of Experimental Pharmacology “Dendritic Cells”: the use of dexamethasone in the induction of tolerogenic DCs.Handb Exp Pharmacol.2009;(188):233-249.
[7] Baeuerle PA,Baichwal VR.NF-kappa B as a frequent target for immunosuppressive and anti-inflammatory molecules.Adv Immunol.1997;65:111-137.
[8] Cameron K,Travers P,Chander C,et al.Directed osteogenic differentiation of human mesenchymal stem/precursor cells on silicate substituted calcium phosphate.Journal of Biomedical Materials Research Part A.2013;101(1):13-22.
[9] Fratoddi I,Venditti I,Cametti C,et al.Functional polymeric nanoparticles for dexamethasone loading and release.Colloids Surf B Biointerfaces. 2012;93:59-66.
[10] Su Y,Su Q,Liu W,et al.Controlled release of bone morphogenetic protein 2 and dexamethasone loaded in core–shell PLLACL–collagen fibers for use in bone tissue engineering.Acta Biomaterialia.2012;8(2):763-771.
[11] Yu WQ,Zhang YL,Jiang XQ,et al.In vitro behavior of MC3T3-E1 preosteoblast with different annealing temperature titania nanotubes.Oral Dis.2010;16 (7):624-630.
[12] Hu X,Shen H,Shuai K,et al.Surface bioactivity modification of titanium by CO2 plasma treatment and induction of hydroxyapatite: in vitro and in vivo studies. Appl Surf Sci. 2011; 257(6):1813-1823.
[13] Bajgai MP,Parajuli DC,Park SJ,et al.In vitro bioactivity of sol-gel-derived hydroxyapatite particulate nanofiber modified titanium.J Mater Sci Mater Med.2010;21(2):685-694.
[14] Bai Y,Park IS,Park HH,et al.The effect of annealing temperatures on surface properties, hydroxyapatite growth and cell behaviors of TiO2 nanotubes. Surf Interf Analysis. 2011;43(6):998-1005.
[15] Yu WQ,Jiang XQ,Zhang FQ,et al.The effect of anatase TiO2 nanotube layers on MC3T3-E1 preosteoblast adhesion, proliferation, and differentiation.J Biomed Mater Res A.2010; 94(4):1012-1022.
[16] Wang CY,Zhao BH,Ai HJ,et al.Comparison of biological characteristics of mesenchymal stem cells grown on two different titanium implant surfaces.Biomed Mater. 2008;3(1): 015004.
[17] Mellado-Valero A,Buitrago-Vera P,Solá-Ruiz MF,et al.Decontamination of dental implant surface in peri-implantitis treatment: A literature review.Med Oral Patol Oral Cir Bucal.2013;18(6):e869-e876.
[18] Wennerberg A,Albrektsson T,Johansson C,et al.Experimental study of turned and grit-blasted screw-shaped implants with special emphasis on effects of blasting material and surface topography.Biomaterials.1996;17(1):15-22.
[19] Kim HW,Koh YH,Li LH,et al.Hydroxyapatite coating on titanium substrate with titania buffer layer processed by solgel method.Biomaterials.2004;25 (13):2533-2538.
[20] Mamalis A,Silvestros S.Modified titanium surfaces alter osteogenic differentiation: a comparative microarray-based analysis of human mesenchymal cell response to commercial titanium surfaces.J Oral Implantol. 2013;39(5):591-601.
[21] Kim MJ,Kim CW,Lim YJ,et al.Microrough titanium surface affects biologic response in MG63 osteoblast‐like cells.J Biomed Mater Res A. 2006;79(4):1023-1032.
[22] Saito T,Hayashi H,Kameyama T,et al.Suppressed proliferation of mouse osteoblast-like cells by a rough-surfaced substrate leads to low differentiation and mineralization.Mater Sci Eng C.2010;30(1):1-7.
[23] Schwartz Z,Olivares-Navarrete R,Wieland M,et al. Mechanisms regulating increased production of osteoprotegerin by osteoblasts cultured on microstructured titanium surfaces.Biomaterials.2009;30(20):3390-3396.
[24] Liu H,Webster TJ.Nanomedicine for implants: A review of studies and necessary experimental tools. Biomaterials. 2007;28(2):354-369.
[25] Chen ZX,Takao Y,Wang WX,et al.Surface characteristics and in vitro biocompatibility of titanium anodized in a phosphoric acid solution at different voltages.Biomed. Mater.2009;4(6):
[26] Yu WQ,Qiu J,Zhang FQ.In vitro corrosion study of different TiO2 nanotube layers on titanium in solution with serum proteins.Colloids Surf B Biointerfaces.2011;84(2):400-405.
[27] Swami N,Cui Z,Nair LS.Titania Nanotubes, Novel nanostructures for improved osseointegration.J Heat Transfer. 2011;133(3):034002.
[28] Kim HM,Chae WP,Chang KW,et al.Composite nanofiber mats consisting of hydroxyapatite and titania for biomedical applications.J Biomed Mater Res B Appl Biomater. 2011;94(2): 380-387.
[29] Simchi A,Tamjid E,Pishbin F,et al.Recent progress in inorganic and composite coatings with bactericidal capability for orthopaedic applications. Nanomedicine.2011;7(1):22-39.
[30] Gao L,Feng B,Wang J,et al.Micro/nanostructural porous surface on titanium and bioactivity.J Biomed Mater Res Part B Appl Biomater.2009;89(2):335-341.
[31] Zhao L,Mei S,Chu PK,et al.The influence of hierarchical hybrid micro/nano-textured titanium surface with titania nanotubes on osteoblast functions. Biomaterials. 2010;31(19): 5072-5082.
[32] Sato M,Sambito MA,Aslani A,et al.Increased osteoblast functions on undoped and yttrium-doped nanocrystalline hydroxyapatite coatings on titanium.Biomaterials. 2006;27(11): 2358-2369.
[33] Popat KC,Leoni L,Grimes CA,et al.Influence of engineered titania nanotubular surfaces on bone cells.Biomaterials. 2007; 28(21):3188-3197.
[34] Oh S,Brammer KS,Li Y S J,et al.Stem cell fate dictated solely by altered nanotube dimension.Proc Natl Acad Sci U S A. 2009; 106(7):2130-2135.
[35] Minagar S,Berndt CC,Wang J,et al.A review of the application of anodization for the fabrication of nanotubes on metal implant surfaces.Acta biomaterialia. 2012;8(8):2875-2888.
[36] Karla S,Seunghan O.Improved bone-forming functionality on diameter-controlled TiO2 nanotube surface.Acta Biomaterialia. 2009;5(8):3215-3223.
[37] Discher DE,Mooney DJ,Zandstra PW. Growth factors, matrices, and forces combine and control stem cells.Science. 2009;324(5935):1673-1677.
[38] Kang Y,Kim S,Khademhosseini A,et al.Creation of bony microenvironment with CaP and cell-derived ECM to enhance human bone-marrow MSC behavior and delivery of BMP-2. Biomaterials.2011;32(26):6119-6130.
[39] Macdonald ML,Samuel RE,Shah NJ,et al.Tissue integration of growth factor-eluting layer-by-layer polyelectrolyte multilayer coated implants. Biomaterials. 2011;32(5): 1446-1453.
[40] Balaur E,Macak JM,Tsuchiya H,et al.Wetting behaviour of layers of TiO2 nanotubes with different diameters.J Mater Chem.2005;15:4488-4491.
[41] Hu Y,Cai K,Luo Z,et al.Regulation of the differentiation of mesenchymal stem cells in vitro and osteogenesis in vivo by microenvironmental modification of titanium alloy surfaces. Biomaterials.2012;33(13):3515-3528.
[42] 关林波,但卫华,曾睿,等.明胶及其在生物材料中的应用[J].材料导报,2006,20(S2):380-383.
[43] 罗华丽,鲁在君.壳聚糖作为药物缓释载体的研究进展[J].高分子通报,2006,19(7):25-30.
[44] Son JS,Choi YA,Park EK,et al.Drug delivery from hydroxyapatite-coated titanium surfaces using biodegradable particle carriers.J Biomed Mater Res B Appl Biomater. 2013; 101(2):247-257.
[45] Cai KY,Hu Y,Luo Z,et al.Cell-speci?c gene transfection from a gene-functionalized poly(D,L-lactic acid) substrate fabricated by the layer-by-layer assembly technique.Angew Chem Int Ed. 2008’47;(39):7479-7481. |
| [1] | Zhang Qing, Fan Junbai, Zhao Xiaoyu. The best route and dose of dexamethasone for brachial plexus block: a systematic review and meta-analysis [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(29): 4751-4756. |
| [2] | Li Hongcai, Ma Zhuang, Guo Youling. Biological behaviors of fibroblasts on the surface of TiO2 nanotubes with different diameters after annealing treatment [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(22): 3504-3509. |
| [3] | Zhou Yimo, Zhang Jianning, Shan Zhongshu. Effects of psoralen corylifolia extract on bone mineral density and bone biomechanics in osteoporosis rats [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(2): 165-170. |
| [4] | Meng Chenyang, Xue Fei, Jia Yanfei, Tong Yanxiang, Zhang Lifeng, Yu Chengyong, Zhang Zhehan, Wang Wenxuan, Hao Ting, Feng Wei . MicroRNA-141 is regulated by serum of velvet antler to promote the proliferation of bone marrow mesenchymal stem cells in dexamethasone-induced cell model [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(19): 2991-2996. |
| [5] |
Li Qinxuan, Wang Yizhi, Xu Yan, Zhang Huayang, Lü Qixuan, Zhang Cheng, He Zhengyun, Zhang Xiao, Yang Zheng.
Effects of dexamethasone combined with estrogen on the
expression of interleukin-6, Caspase3 and Bcl-2 after spinal cord contusion in
rats |
| [6] | Li Ying, You Yapeng, Li Baoe, Song Yunjia, Ma Aobo, Chen Bo, Han Wen, Li Changyi. Type I collagen combined titanium dioxide nanotube composite coating modified titanium surface improves osteoblast adhesion and osseointegration [J]. Chinese Journal of Tissue Engineering Research, 2019, 23(14): 2169-2176. |
| [7] | Liu Wen-jing, Sun Yang-peng, Zheng You-hua, Zhang Zhi-guang. Combined use of 1,25-dihydroxyvitamin D3 and dexamethasone promotes osteogenic differentiation of synovial fluid mesenchymal stem cells from human temporomandibular joint [J]. Chinese Journal of Tissue Engineering Research, 2018, 22(9): 1443-1449. |
| [8] | Yang Ben, He Dong-ning, Qin Bo-heng, Zhang Bao-ping. Preparation and performance properties of functionalized multi-walled carbon nanotubes/chitosan-zinc composite scaffolds [J]. Chinese Journal of Tissue Engineering Research, 2018, 22(6): 889-895. |
| [9] | Meng Ai, Wang Jian, Sui Lei. Effect of mixed acid reflux time on purification and biocompatibility of large-inner-diameter multi-walled carbon nanotubes [J]. Chinese Journal of Tissue Engineering Research, 2018, 22(6): 896-901. |
| [10] | Zhang Xian-jun, Zhao Xi-jiang. In vitro bioactivity of silicon-incorporated titanium dioxide nanotubes [J]. Chinese Journal of Tissue Engineering Research, 2018, 22(30): 4864-4869. |
| [11] | Wang Ping-ting, Qi Xing-ying . Low-temperature synthesis and stabilization of carboxymethyl chitosan stabilized amorphous calcium phosphate [J]. Chinese Journal of Tissue Engineering Research, 2018, 22(22): 3467-3473. |
| [12] | Chen Dian, Hu Qing-lin, Zhou Mei-ling, Hong Xian, Sun Xiao-xi, Xi Jiao-ya. Advances in the construction of a scaffold for cardiac tissue engineering containing homogeneous and stable carbon nanotubes [J]. Chinese Journal of Tissue Engineering Research, 2018, 22(10): 1580-1585. |
| [13] | Wang Wei, Du Yuan-hong. Titanium surface modification by acidic oxidation treatment activates the Wnt signaling pathway in bone marrow mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2018, 22(1): 13-19. |
| [14] | Cao Liang-quan1, Du Bin2, Sun Guang-quan2, Liu Xin2, Chen Guo-qing3, Gu Lei1, Liu Bing4. Construction of a rabbit model of steroid-induced osteonecrosis of the femoral head by combining different concentrations of glucocorticoids with horse serum [J]. Chinese Journal of Tissue Engineering Research, 2017, 21(8): 1229-1235. |
| [15] | Lin Cheng-kai, Rong Li-min, Liu Bin. Biocompatibility of electrospun carbon nanotubes/poly(L-lactic acid) nanofiber scaffolds with mouse neural stem cells [J]. Chinese Journal of Tissue Engineering Research, 2017, 21(6): 940-945. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||