Chinese Journal of Tissue Engineering Research ›› 2020, Vol. 24 ›› Issue (22): 3504-3509.doi: 10.3969/j.issn.2095-4344.2295
Previous Articles Next Articles
Biological behaviors of fibroblasts on the surface of TiO2 nanotubes with different diameters after annealing treatment
Li Hongcai, Ma Zhuang, Guo Youling
- First Department of Stomatology, Zhuzhou Central Hospital, Zhuzhou 410002, Hunan Province, China
-
Received:2019-11-12Revised:2019-11-16Accepted:2020-01-02Online:2020-08-08Published:2020-04-26 -
Contact:Ma Zhuang, Chief physician, First Department of Stomatology, Zhuzhou Central Hospital, Zhuzhou 410002, Hunan Province, China -
About author:Li Hongcai, Master, Attending physician, First Department of Stomatology, Zhuzhou Central Hospital, Zhuzhou 410002, Hunan Province, China -
Supported by:Project of Hunan Provincial Health Department, No. c2013-051
CLC Number:
Cite this article
Li Hongcai, Ma Zhuang, Guo Youling. Biological behaviors of fibroblasts on the surface of TiO2 nanotubes with different diameters after annealing treatment[J]. Chinese Journal of Tissue Engineering Research, 2020, 24(22): 3504-3509.
share this article

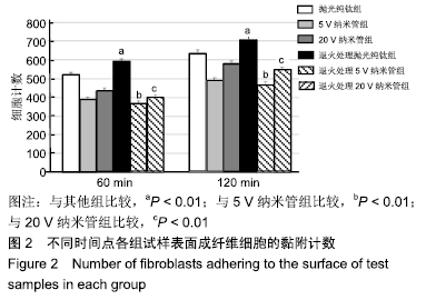
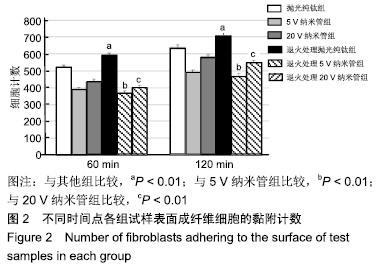
60 min时,抛光纯钛组与退火处理抛光纯钛组的黏附成纤维细胞数分别为520.78±11.56和590.67±13.18,两组间比较差异有显著性意义(P < 0.01);120 min时,抛光纯钛组与退火处理抛光纯钛组的黏附细胞数分别为633.00± 17.79和705.45±16.08,两组间比较差异有显著性意义(P < 0.01)。两个时间点,退火处理抛光纯钛表面黏附细胞数较退火处理前分别增加了13.42%和11.44%,退火处理促进了抛光纯钛表面成纤维细胞的黏附。 60 min时,5,20 V纳米管组黏附细胞数分别为 388.33±14.31和433.78±15.64,退火处理5,20 V纳米管组黏附细胞数分别为368.54±13.43和399.00±14.83,5,20 V纳米管退火后的黏附细胞数均少于退火前(P < 0.01)。 120 min时,5,20 V纳米管组成纤维细胞数分别为489.33±14.94和579.67±14.45,退火处理5,20 V纳米管组黏附细胞数分别为466.56±17.93和543.00±15.58,5,20 V纳米管退火后的黏附细胞数均少于退火前(P < 0.01)。退火处理抑制了钛纳米管表面成纤维细胞的黏附。60,120 min时,退火处理5 V纳米管组组黏附细胞数为各组中最少。 相同时间点下,5,20 V纳米管组黏附细胞数少于抛光纯钛组(P < 0.01)。 2.3 退火处理对材料表面成纤维细胞伸展形态的影响 培养1 d时各组试样表面成纤维细胞伸展形态,见图3。未行退火处理抛光纯钛表面的成纤维细胞多数呈现出典型长梭形,退火处理抛光纯钛表面的细胞呈现出更明显的板状伪足;与退火处理的纳米管相比,未行退火处理各纳米管表面的细胞伸展较宽,充满大量细胞质,板状伪足和丝状伪足更明显,呈现较强的细胞生物活性。 "

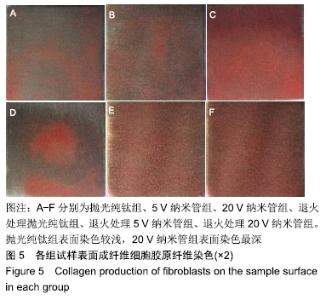
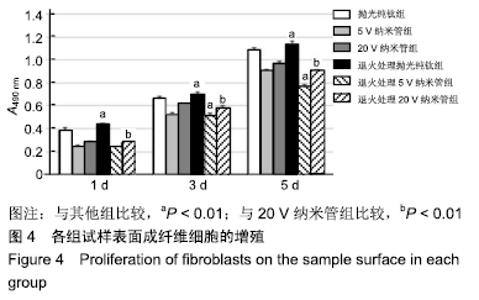
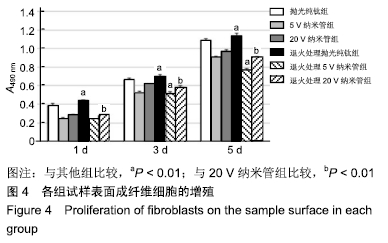
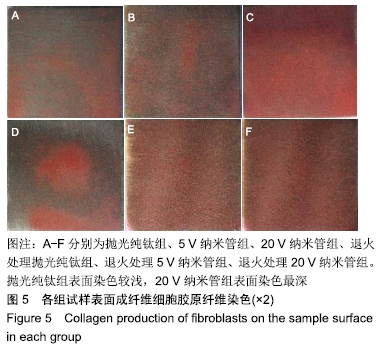
1,3,5 d时,退火处理5 V纳米管组A490 nm分别为0.24±0.01,0.52±0.02,0.77±0.01,均低于同时间点5 V纳米管组的0.25±0.01,0.53±0.01,0.91±0.01,且均低于同时间点其余各组,其中3,5 d时这种差异有显著性意义(P < 0.01);退火处理20 V纳米管组A490 nm分别为0.28±0.01,0.58±0.02,0.91±0.01,均显著低于同时间点20 V纳米管组的0.29±0.01,0.62±0.01,0.97±0.01(P < 0.01),说明退火处理抑制了TiO2纳米管表面成纤维细胞的增殖。与抛光纯钛相比,TiO2纳米管不同程度抑制了成纤维细胞的增殖,退火处理协同增强了这种抑制作用。 2.5 退火处理对材料表面成纤维细胞胶原分泌功能的影响 细胞培养3 d后各组试样表面成纤维细胞胶原纤维天狼星红/苦味酸液染色结果,见图5。抛光纯钛组表面染色较浅,20 V纳米管组表面染色最深。 "
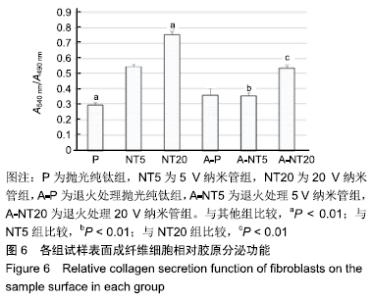
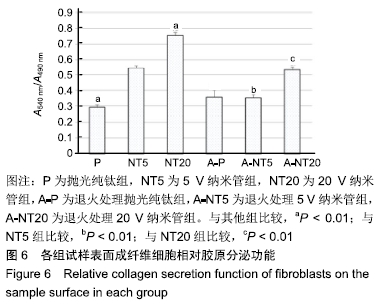
各组试样表面成纤维细胞的相对胶原分泌功能,见图6。细胞培养3 d后,退火处理抛光纯钛组细胞的相对胶原分泌为0.35±0.04,显著高于抛光纯钛组的0.29±0.01(P < 0.01),说明退火处理提高了抛光纯钛表面成纤维细胞的相对胶原分泌功能。退火处理5,20 V纳米管组细胞的相对胶原分泌分别为0.35±0.02和0.53±0.02,5,20 V纳米管组分别为0.54±0.01和0.75±0.02,退火处理各组的相对胶原分泌均低于对应的未退火处理组(P < 0.01),说明退火处理抑制了纳米管表面成纤维细胞的相对胶原分泌功能。抛光纯钛组的相对胶原分泌功能显著低于5,20 V纳米管组(P < 0.01),说明纳米管不同程度促进了成纤维细胞的胶原分泌功能。抛光纯钛组的相对胶原分泌功能最小(P < 0.01),20 V纳米管组的相对胶原分泌功能最大(P < 0.01)。 "
| [1] KULKARNI M, MAZARE A, GONGADZE E, et al.Titanium nanostructures for biomedical applications.Nanotechnology. 2015;26(6):062002. [2] 李红彩,马壮.TiO_2纳米管对种植体周细胞影响的研究进展[J].中国组织工程研究,2016,20(52):7899-7904. [3] ZHANG L, LIAO X, FOK A, et al. Effect of crystalline phase changes in titania (TiO2) nanotube coatings on platelet adhesion and activation. Mater Sci Eng C Mater Biol Appl.2018;82(1):91-101. [4] 杨修春,崔晓琳,任鹏,等.后处理工艺对阳极氧化钛生物活性的影响[J].功能材料,2015,46(1):1135-1140,1143. [5] PARK S, MIN D, LIM H, et al. Effect of heat treatment on phase transition of nanotubular titanium oxide arrays.J Nanosci Nanotechnol. 2011;11(2):1476-1479. [6] OH S, DARAIO C, CHEN LH, et al. Significantly accelerated osteoblast cell growth on aligned TiO2 nanotubes.J Biomed Mater Res A. 2006;78(1):97-103. [7] AN SH, NARAYANAN R, MATSUMOTO T, et al. Crystallinity of anodic TiO2 nanotubes and bioactivity.J Nanosci Nanotechnol. 2011;11(6): 4910-4918. [8] YU WQ, ZHANG YL, JIANG XQ, et al. In vitro behavior of MC3T3-E1 preosteoblast with different annealing temperature titania nanotubes. Oral Dis.2010;16(7):624-630. [9] MINAGAR S, WANG J, BERNDT CC, et al. Cell response of anodized nanotubes on titanium and titanium alloys.J Biomed Mater Res A. 2013;101(9):2726-2739. [10] NAIR M, ELIZABETH E. Applications of Titania Nanotubes in Bone Biology.J Nanosci Nanotechnol.2015;15(2):939-955. [11] SHOKUHFAR T, HAMLEKHAN A, CHANG JY, et al. Biophysical evaluation of cells on nanotubular surfaces: the effects of atomic ordering and chemistry.Int J Nanomedicine.2014;12(9):3737-3748. [12] ZHU Y, ZHANG CN, GU YX, et al. The responses of human gingival fibroblasts to magnesium-doped titanium.J Biomed Mater Res A. 2020;108(2):267-278. [13] 赖颖真,卢薛冠,蔡艺煌.钛及氧化锆表面微沟槽结构对人牙龈成纤维细胞生物学行为的影响[J]. 中华口腔医学杂志,2019,54(10):676-682. [14] PHAM MH, HAUGEN HJ, RINNA A, et al. Hydrofluoric acid treatment of titanium surfaces enhances the proliferation of human gingival fibroblasts.J Tissue Eng.2019;10:204173141982895. [15] 包崇云.牙种植体穿龈部位表面微结构对生物密封的影响[J].口腔材料器械杂志,2017,26(3):113-117. [16] 江瑶,黎红.钛表面粗糙度对成纤维细胞生长影响的研究进展[J].口腔医学研究,2019,35(8):738-740. [17] RADTKE A, BAL M, JĘDRZEJEWSKI T. Novel Titania Nanocoatings Produced by Anodic Oxidation with the Use of Cyclically Changing Potential: Their Photocatalytic Activity and Biocompatibility. Nanomaterials (Basel).2018;8(9).pii: E712. doi: 10.3390/nano8090712. [18] FADEYEV FA, KHRUNYK YY, BELIKOV SV, et al. The Adhesion of Human Dermal Fibroblasts on Anodized Nanotube-layered Titanium, Modified for Implantology Application.Dokl Biol Sci.2019;486(1):91-93. [19] 李宝娥,李军,梁春永,等.阳极氧化处理对钛表面粗糙度和亲水性的影响[J].稀有金属材料与工程,2016,45(4):858-862. [20] 李红彩,张玉梅,孙海平.不同管径Ti-TiO2纳米管对成纤维细胞和成骨细胞黏附的影响[J].中华口腔医学杂志,2012,47(2):122-126. [21] 李红彩,马壮.不同管径钛纳米管对成纤维细胞增殖、伸展和胶原分泌功能的影响[J].实用口腔医学杂志,2017,33(05):612-616. [22] 梁砚琴,杨贤金,崔振铎,等.钛和钛合金表面TiO2纳米管研究进展[J].科技导报,2009,27(14):94-101. [23] HYAM RS, LEE J, CHO E, et al. Effect of annealing environments on self-organized TiO2 nanotubes for efficient photocatalytic applications. J Nanosci Nanotechnol.2012;12(12):8908-8912. [24] 梅盛林,张玉梅,赵领洲,等.近β钛合金TLM表面阳极氧化对成骨细胞早期附着的影响[J].实用口腔医学杂志,2010,26(5):597-601. [25] PARK J, BAUER S, SCHMUKI P, et al. Narrow window in nanoscale dependent activation of endothelial cell growth and differentiation on TiO2 nanotube surfaces. Nano Lett. 2009; 9(9):3157-3164. [26] HUANG Q, YANG Y, ZHENG D, et al. Effect of construction of TiO2 nanotubes on platelet behaviors: Structure-property relationships.Acta Biomater.2017;51(15):505-512. [27] 石萍,于千.退火温度及TiO_2结构对羟基磷灰石的沉积诱导作用[J].材料热处理学报,2010,31(1):44-47. [28] 顾迎新,赖红昌,张志勇.钛基种植体表面纳米管改性的研究进展[J].口腔材料器械杂志,2012,21(3):145-148. [29] POPAT KC, ELTGROTH M, LATEMPA TJ, et al. Titania nanotubes: a novel platform for drug-eluting coatings for medical implants? Small. 2007;3(11):1878-1881. [30] WEI H, WU S, FENG Z, et al. Increased fibroblast functionality on CNN2-loaded titania nanotubes. Int J Nanomedicine.2012;7:1091-100. [31] LV L, LI K, XIE Y, et al. Enhanced osteogenic activity of anatase TiO2 film: Surface hydroxyl groups induce conformational changes in fibronectin. Mater Sci Eng C Mater Biol Appl.2017;78(1):96-104. [32] LEE SJ, OH TJ, BAE TS, et al. Effect of bisphosphonates on anodized and heat-treated titanium surfaces: an animal experimental study.J Periodontol.2011;82(7):1035-1042. [33] MORAVEC H, VANDROVCOVA M, CHOTOVA K, et al. Cell interaction with modified nanotubes formed on titanium alloy Ti-6Al-4V. Mater Sci Eng C Mater Biol Appl.2016;65(1):313-322. [34] FAN X, FENG B, LIU Z, et al. Fabrication of TiO2 nanotubes on porous titanium scaffold and biocompatibility evaluation in vitro and in vivo.J Biomed Mater Res A.2012; 100(12):3422-3427. [35] HUANG Q, YANG Y, HU R,et al. Reduced platelet adhesion and improved corrosion resistance of superhydrophobic TiO₂-nanotube- coated 316L stainless steel.Colloids Surf B Biointerfaces. 2015;125(1): 134-141. [36] TIAN A, QIN X, WU A, et al. Nanoscale TiO2 nanotubes govern the biological behavior of human glioma and osteosarcoma cells.Int J Nanomedicine.2015;10(25):2423-2439. [37] AMIN YAVARI S, CHAI YC, BÖTTGER AJ, et al. Effects of anodizing parameters and heat treatment on nanotopographical features, bioactivity, and cell culture response of additively manufactured porous titanium.Mater Sci Eng C Mater Biol Appl.2015;51:132-138. |
| [1] | Li Qin, Mao Shuangfa, Li Min, Cheng Jiyan. Protective effect and mechanism of dendrobium on fibroblasts damaged by ultraviolet B [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(8): 1228-1233. |
| [2] | Zhang Jinglin, Leng Min, Zhu Boheng, Wang Hong. Mechanism and application of stem cell-derived exosomes in promoting diabetic wound healing [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1113-1118. |
| [3] | Yang Sidi, Wang Qian, Xu Nuo, Wang Ronghan, Jin Chuanqi, Lu Ying, Dong Ming. Biodentine enhances the proliferation and differentiation of osteoblasts through upregulating bone morphogenetic protein-2 [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 516-520. |
| [4] | Chen Shuo, Xiao Dongqin, Li Xingping, Ran Bin, Shi Feng, Zhang Chengdong, Deng Li, Huang Nanxiang, Liu Kang, Feng Gang, Duan Ke. Preparation and characterization of tantalum functional coating on titanium implant [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 546-552. |
| [5] | Zheng Haisheng, Lan Yuqing, Zhong Xingwu, Ding Hui. Effect of curcumin nanoparticles on proliferation of human retinal pigment epithelial cells [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(27): 4335-4339. |
| [6] | Zhang Ansheng, Zhang Haiou, Ni Longxing. Regulating the expression of nucleotide-binding oligomerization domain-like receptor protein 3 inflammasome-related molecules in dental pulp fibroblasts under inflammation [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(26): 4107-4112. |
| [7] | Min Ziyang, Munire·Aili, Zheng Yunhao, Zeng Xingzhi, Bian Nanyan, Deng Shuangshan, Xie Jing. Mechanism and characteristics of mechanical microenvironment of extracellular matrix and intercellular interaction [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(25): 4034-4045. |
| [8] | Zhang Gaofei, Wang Di, Li Jiamei, Lou Hanxiao, Zeng Yueqin, Liu Wenjun. Mesenchymal stem cells promote wound healing by regulating the autophagy [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(25): 4058-4063. |
| [9] | Zhong Ruiying, Wang Fuke, Yang Guiran, Wang Guoliang, Hou Jianfei, Liao Xinyu. Co-culture of fibroblasts and vascular endothelial cells affects proliferation and osteogenesis of adipose stem cells [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(24): 3833-3839. |
| [10] | Chen Sheng, Yang Yan, Ding Liang, Ye Chuanjin, Zhang Lei, Xia Shu, Li Huiling, Huang Xiaofeng. Establishment and identification of an immortalized oral cancer-related fibroblast line [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(24): 3808-3813. |
| [11] | Zhou Fanqi, Li Baoying, Wang Kaitao, Tan Quanquan, Yun Chenxia, Leng Jing. Isolation and identification of fibroblast-like synoviocytes from tree shrews and establishment of TLR8 pathway related molecular detection methods [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(23): 3721-3727. |
| [12] | Meng Zengdong, Zhu Bin, Zhang Yanan, Luo Lilin, Zhang Yuqin. Mechanical properties and biocompatibility of porous ZnO/hydroxyapatite composites with different porosities [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(22): 3498-3504. |
| [13] | Feng Le, Qiu Peng, Liu Min, Zhou Hui. Characterization of chitosan-modified polyetheretherketone and its effect on MC3T3-E1 cell adhesion and proliferation [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(21): 3351-3356. |
| [14] | Feng Dongfei, He Hongxu, Xie Qi, Zhang Lili, Zhou Hui, Li Wei. Selection of key genes related to biological functions and regulation pathway in periodontal reconstruction [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(2): 253-259. |
| [15] | Huang Xiaoxiong, Chen Weikai, Liu Tao, Yang Huilin, He Fan. Hypoxic precondition rescues osteogenic potential of bone marrow mesenchymal stem cells derived from ovariectomized rats [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(13): 2034-2039. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||