Chinese Journal of Tissue Engineering Research ›› 2023, Vol. 27 ›› Issue (20): 3188-3194.doi: 10.12307/2022.673
Previous Articles Next Articles
Effects of aerobic exercise on learning, memory, and hippocampal neuromorphology in mice with Alzheimer’s disease
Zhang Yeting1, 2, Fu Yan3, Li Xue4, Wei Cuilan5, Li Chuikun2, Yuan Qiongjia4
- 1Civil Aviation Flight University of China, Guanghan 618307, Sichuan Province, China; 2College of Physical Education, Chengdu University, Chengdu 610106, Sichuan Province, China; 3College of Physical Education, Southwest Minzu University, Chengdu 610041, Sichuan Province, China; 4Chengdu Sport University, Chengdu 610041, Sichuan Province, China; 5Sports Institute of Chengdu University of Technology, Chengdu 610059, Sichuan Province, China
-
Received:2021-06-29Accepted:2021-09-08Online:2023-07-18Published:2022-11-19 -
Contact:Yuan Qiongjia, Professor, Chengdu Sport University, Chengdu 610041, Sichuan Province, China -
About author:Zhang Yeting, PhD, Lecturer, Civil Aviation Flight University of China, Guanghan 618307, Sichuan Province, China; College of Physical Education, Chengdu University, Chengdu 610106, Sichuan Province, China -
Supported by:the Science and Technology Project of Sichuan Province, No. 2020YFH0184 (to ZYT)
CLC Number:
Cite this article
Zhang Yeting, Fu Yan, Li Xue, Wei Cuilan, Li Chuikun, Yuan Qiongjia. Effects of aerobic exercise on learning, memory, and hippocampal neuromorphology in mice with Alzheimer’s disease[J]. Chinese Journal of Tissue Engineering Research, 2023, 27(20): 3188-3194.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
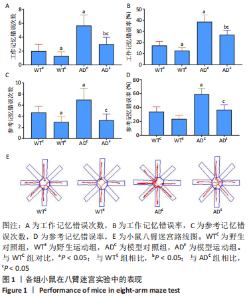
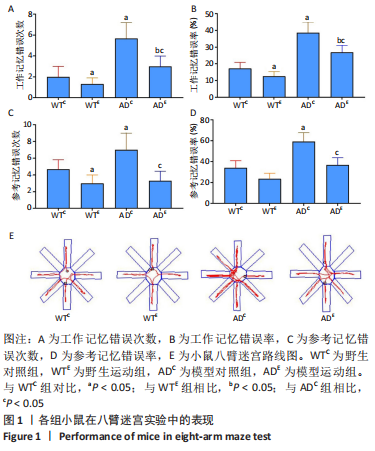
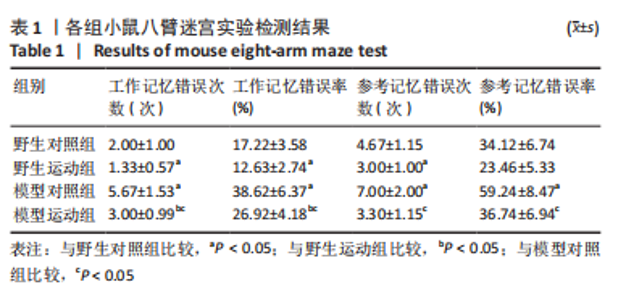
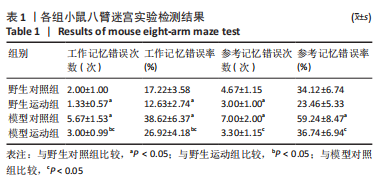
整体而言,阿尔茨海默症小鼠相较于野生型小鼠重复进入各臂的次数较多,吃完所有食物用时较长。通过两因素被试间方差分析工作记忆错误次数,干预方式的主效应显著[F(1,20)=7.143,P=0.028,ηP2=0.472],基因型的主效应显著[F(1,20)=3.681,P=0.003,ηP2=0.696];干预方式与基因型的交互作用不显著[F(1,20)=2.571,P=0.147,ηP2=0.243]。表明干预方式与基因型作为主效应均可以显著影响小鼠工作记忆错误次数,但是它们的交互作用并不显著。野生运动组小鼠的工作记忆错误次数显著低于野生对照组,模型运动组小鼠的工作记忆错误次数显著低于模型对照组(P=0.047,P=0.016);模型对照组小鼠的工作记忆错误次数显著高于模型运动组,模型运动组小鼠的工作记忆错误次数显著高于野生运动组(P=0.003,P=0.009)。 通过两因素被试间方差分析工作记忆错误率,干预方式的主效应显著[F(1,20)=10.146,P=0.013,ηP2=0.559],基因型的主效应显著[F(1,20)=48.78,P < 0.005,ηP2=0.859],干预方式与基因型的交互作用不显著[F(1,20)=1.936,P=0.202,ηP2=0.195]。表明干预方式与基因型作为主效应均可以显著影响小鼠工作记忆错误率,但是它们的交互作用并不显著。野生运动组小鼠的工作记忆错误次率显著低于野生对照组,模型运动组小鼠的工作记忆错误次率显著低于模型对照组(P=0.024,P=0.012);模型对照组小鼠的工作记忆错误率显著高于野生对照组,模型运动组小鼠的工作记忆错误率显著高于野生运动组(P < 0.005,P=0.004)。 通过两因素被试间方差分析参考记忆错误次数,干预方式的主效应显著[F(1,20)=11.130,P=0.010,ηP2=0.582],基因型的主效应不显著[F(1,20)=2.783,P=0.134,ηP2=0.696],干预方式与基因型的交互作用不显著[F(1,20)=1.565,P=0.246,ηP2=0.164]。表明干预方式对小鼠参考记忆错误次数的改变并不受基因型的影响,且基因型并未对小鼠的参考记忆错误次数产生显著影响。野生运动组小鼠的参考记忆错误次数显著低于野生对照组,模型运动组小鼠的参考记忆错误次数显著低于模型对照组(P=0.017,P=0.012);模型对照组小鼠的参考记忆错误次数显著高于野生对照组,模型运动组小鼠的参考记忆错误次数与野生运动组相比无明显差异(P=0.022,P=0.148)。 通过两因素被试间方差分析参考记忆错误率,干预方式的主效应显著[F(1,20)=17.027,P=0.003,ηP2=0.68],基因型的主效应显著[F(1,20)=22.851,P=0.001,ηP2=0.741],干预方式与基因型的交互作用不显著[F(1,20)=2.169,P=0.179,ηP2=0.213]。表明干预方式与基因型作为主效应均可以显著影响小鼠参考记忆错误率,但是它们的交互作用并不显著。野生运动组小鼠的参考记忆错误率与野生对照组相比无明显差异,模型运动组小鼠的参考记忆错误率显著低于模型对照组(P=0.097,P=0.004) ;模型对照组小鼠的参考记忆错误率显著高于野生对照组,模型运动组小鼠的参考记忆错误率与野生运动组相比无明显差异(P=0.002,P=0.238)。 2.3 各组小鼠海马神经形态 见图2。"
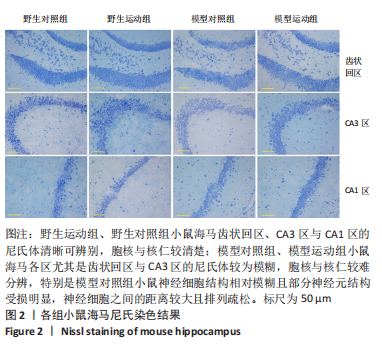
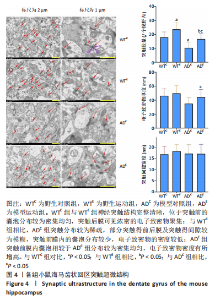
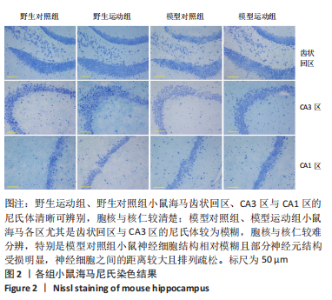
通过海马尼氏染色可以看出,相对于阿尔茨海默症小鼠,野生型小鼠海马齿状回区、CA3区与CA1区的尼氏体清晰可辨别,胞核与核仁较清楚,甚至可以区分出树突,表明其神经元结构较好,神经元之间的排列较为紧密有层次。相对而言,阿尔茨海默症小鼠海马各区尤其是齿状回区与CA3区的尼氏体颜色较淡且较为模糊,胞核与核仁也相对较难分辨,特别是模型对照组小鼠的神经细胞结构相对模糊且部分神经元结构受损明显,神经细胞之间的距离较大且排列疏松;相对于模型对照组来说,模型运动组小鼠海马各区尼氏体颜色较深,胞核与核仁也相对较容易分辨。这说明阿尔茨海默症小鼠海马神经细胞合成蛋白质的功能可能相对比较弱,其神经元出现了一定程度的受损,而运动可以在一定程度上改善阿尔茨海默症小鼠海马神经细胞结构。 2.4 各组小鼠海马神经细胞超微结构 见图3,4。"
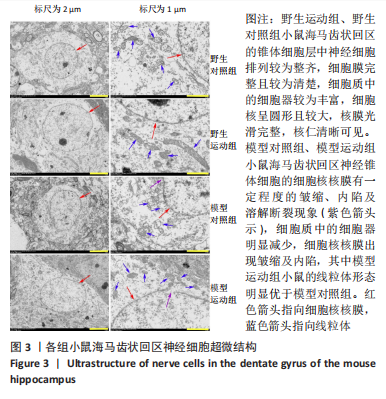

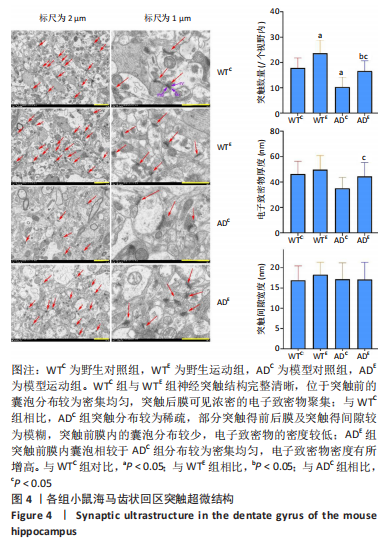
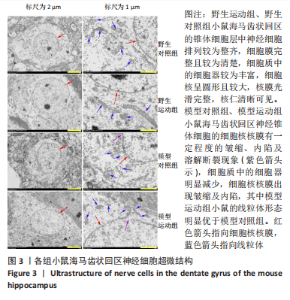
电镜下可观察到,野生型小鼠海马齿状回区的锥体细胞层中神经细胞排列较为整齐,细胞膜完整且较为清楚,细胞质中的细胞器如线粒体、粗面内质网、核糖体、高尔基复合体等较为丰富,甚至可以区分出溶酶体;细胞核呈现形状较为规则的圆形且比较大,核膜光滑完整,核仁清晰可见。阿尔茨海默症小鼠海马齿状回区神经锥体细胞的细胞核核膜有一定程度的皱缩、内陷及溶解断裂现象,细胞质中的细胞器如线粒体、内质网、高尔基复合体等的数量较野生型小鼠明显减少,其中有的线粒体出现了肿胀与空泡等现象,甚至有的线粒体的嵴出现溶解消失的情况,内质网出现了轻度扩张及液泡化的现象,高尔基的囊泡也有扩张现象发生;细胞核核膜出现皱缩及内陷呈锯齿样,而染色质则有固缩边聚现象。虽然模型对照组及模型运动组小鼠海马齿状回区神经细胞细胞核核膜都出现了一定程度的皱缩、内陷、溶解断裂现象,但是模型运动组小鼠的线粒体形态明显优于模型对照组小鼠。以上这些变化表明,APP/PS1双转基因小鼠的海马齿状回区神经细胞出现了受损及退化现象,而运动对这种受损及退化过程可能具有一定的预防作用。 电镜下还可观察到,野生对照组与野生运动组小鼠海马齿状回区分子层的神经突触大多结构完整清晰,位于突触前的囊泡分布也较为密集均匀,在突触后膜处能够观察到浓密的电子致密物聚集,电子致密物的密度高;与野生对照组相比,模型对照组突触的分布较为稀疏,而部分突触前后膜及突触间隙较为模糊,突触前膜内的囊泡分布较少,电子致密物的密度较低;模型运动组突触前膜内的囊泡相较于模型对照组分布较为密集均匀,电子致密物密度有所增高。 通过两因素被试间方差分析小鼠海马齿状回区突触的数量,干预方式的主效应显著[F(1,20)= 11.745,P=0.003,ηP2=0.37],基因型的主效应显著[F(1,20)=16.632,P=0.001,ηP2=0.454],干预方式与基因型的交互作用不显著[F(1,20)=0.016,P=0.900,ηP2=0.001]。表明干预方式与基因型作为主效应均可以显著影响海马齿状回区突触的数量,但是它们的交互作用并不显著。野生运动组的突触数量显著高于野生对照组,模型运动组的突触数量显著高于模型对照组(P=0.03,P=0.021);模型对照组的突触数量显著低于野生对照组,模型运动组的突触数量显著低于野生运动组(P=0.002,P=0.048)。 通过两因素被试间方差分析小鼠海马齿状回区电子致密物厚度,干预方式的主效应不显著[F(1,20)=2.347,P=0.141,ηP2=0.105],基因型的主效应不显著[F(1,20)=3.95,P=0.061,ηP2=0.165],干预方式与基因型的交互作用不显著[F(1,20)=0.505,P=0.486,ηP2=0.025]。表明干预方式与基因型作为主效应均不能显著影响海马齿状回区电子致密物厚度,且它们的交互作用并不显著。 通过两因素被试间方差分析小鼠海马齿状回区突触间隙距离,干预方式的主效应不显著[F(1,20)=0.19,P=0.668,ηP2=0.009],基因型的主效应不显著[F(1,20)=0.084,P=0.775,ηP2=0.004],干预方式与基因型的交互作用不显著[F(1,20)=0.224,P=0.641,ηP2=0.011]。表明干预方式与基因型作为主效应均不能显著影响海马齿状回区突触间隙距离,且它们的交互作用并不显著。以上数据表明,长期有氧运动无论是对野生型小鼠,还是对APP/PS1转基因阿尔茨海默症小鼠而言,都可以显著增加其海马突触的数量。但是对其突触电子致密物厚度及突触间隙距离没有显著影响。 中国组织工程研究杂志出版内容重点:组织构建;骨细胞;软骨细胞;细胞培养;成纤维细胞;血管内皮细胞;骨质疏松;组织工程 "

| [1] MARTIN P, ANDERS W. World Alzheimer Report 2016. Alzheimer’s Disease International, 2016. [2] BROOKMEYER R, JOHNSON E, ZIEGLER-GRAHAM K, et al. Forecasting the global burden of Alzheimer’s disease. Alzheimers Dement. 2007; 3(3):186-191. [3] 赵祥,魏翠兰,张业廷.神经发生和炎性环境在运动条件下的改变与调节[J].中国组织工程研究,2021,25(5):813-820. [4] BITTNER T, BURGOLD S, DOROSTKAR MM, et al. Amyloid plaque formation precedes dendritic spine loss. Acta Neuropathol. 2012; 124(6):797-807. [5] TOBIN MK, MUSARACA K, DISOUKY A, et al. Human Hippocampal Neurogenesis Persists in Aged Adults and Alzheimer’s Disease Patients. Cell Stem Cell. 2019;24(6):974-978. [6] LAUTENSCHLAGER N, COX K, ELLIS K. Physical activity for cognitive health: what advice can we give to older adults with subjective cognitive decline and mild cognitive impairment? Dialogues Clin Neuro. 2019;21(1):61-68. [7] 张象,张业廷.运动改善阿尔茨海默症模型小鼠病程的剂量效应关系[J].中国组织工程研究,2021,25(17):2761-2766. [8] KURZINA NP, ARISTOVA IY, VOLNOVA AB, et al. Deficit in working memory and abnormal behavioral tactics in dopamine transporter knockout rats during training in the 8-arm maze. Behav Brain Res. 2020; 390:112642. [9] MCKHANN GM, KNOPMAN DS, CHERTKOW H, et al. The diagnosis of dementia due to Alzheimer’s disease: Recommendations from the National Institute on Aging-Alzheimer’s Association workgroups on diagnostic guidelines for Alzheimer’s disease. Alzheimers Dement. 2011;7(3):263-269. [10] WAKHLOO D, OBERHAUSER J, MADIRA A, et al. From cradle to grave: neurogenesis, neuroregeneration and neurodegeneration in Alzheimer’s and Parkinson’s diseases. Neural Regen Res. 2022;17(12): 2606-2614. [11] VERMUNT L, SIKKES SAM, VAN DEN HOUT A, et al. Duration of preclinical, prodromal, and dementia stages of Alzheimer’s disease in relation to age, sex, and APOE genotype. Alzheimers Dement. 2019; 15(7):888-898. [12] HANSEN RA, GARTLEHNER G, WEBB AP, et al. Efficacy and safety of donepezil, galantamine, and rivastigmine for the treatment of Alzheimer’s disease: A systematic review and meta-analysis. Clin Interv Aging. 2008;3(2):211-225. [13] MONTGOMERY W, GOREN A, KAHLE-WROBLESKI K, et al. Detection, diagnosis, and treatment of Alzheimer’s disease dementia stratified by severity as reported by caregivers in Japan. Neuropsych Dis Treat. 2018;14:1843-1854. [14] PARANJPE MD, WANG JK, ZHOU Y. Sex, ApoE4 and Alzheimer’s disease: rethinking drug discovery in the era of precision medicine. Neural Regen Res. 2021;16(9):1764-1765. [15]HOFFMANN K, SOBOL NA, FREDERIKSEN KS, et al.Moderate-to-High Intensity Physical Exercise in Patients with Alzheimer’s Disease: A Randomized Controlled Trial. J Alzheimers Dis. 2016;50(2):443-453. [16] KE HC, HUANG HJ, LIANG KC, et al. Selective improvement of cognitive function in adult and aged APP/PS1 transgenic mice by continuous non-shock treadmill exercise. Brain Res. 2011;1403:1-11. [17] LIU HL, ZHAO G, ZHANG H, et al. Long-term treadmill exercise inhibits the progression of Alzheimer’s disease-like neuropathology in the hippocampus of APP/PS1 transgenic mice. Behav Brain Res. 2013;256:261-272. [18] SCARMEAS N, LUCHSINGER JA, SCHUPF N, et al. Physical Activity, Diet, and Risk of Alzheimer Disease. JAMA. 2009;302(6):627-637. [19] SMITH JC, NIELSON KA, WOODARD JL, et al. Physical activity and brain function in older adults at increased risk for Alzheimer’s disease. Brain Sci. 2013;3(1):54-83. [20] LAUTENSCHLAGER NT, COX KL, FLICKER L, et al. Effect of physical activity on cognitive function in older adults at risk for Alzheimer disease: a randomized trial. JAMA. 2008 ;300(9):1027-1037. [21] ETGEN T, SANDER D, HUNTGEBURTH U, et al. Physical Activity and Incident Cognitive Impairment in Elderly Persons The INVADE Study. Arch Intern Med. 2010;170(2):186-190. [22] KNOBLAUCH A, KÖRNER E, KÖRNER U, et al. Structural synaptic plasticity has high memory capacity and can explain graded amnesia, catastrophic forgetting, and the spacing effect. PloS One. 2014;9(5): e96485. [23] ALONSO-NANCLARES L, MERINO-SERRAIS P, GONZALEZ S, et al. Synaptic changes in the dentate gyrus of APP/PS1 transgenic mice revealed by electron microscopy. J Neuropath Exp Neur. 2013;72(5): 386-395. [24] SHENG C, XU P, ZHOU K, et al.βIcariin Attenuates Synaptic and Cognitive Deficits in an A-Induced Rat Model of Alzheimer’s Disease. Biomed Res Int. 2017;2017:7464872. [25] YANG X, YAO C, TIAN T, et al. A novel mechanism of memory loss in Alzheimer’s disease mice via the degeneration of entorhinal-CA1 synapse. Mol Psychiatry. 2018;23(2):199-210. [26] GROSSBERG S. Acetylcholine Neuromodulation in Normal and Abnormal Learning and Memory: Vigilance Control in Waking, Sleep, Autism, Amnesia and Alzheimer’s Disease. Front Neural Circuits. 2017;11:82. [27] LI X, GUAN J, SUN T, et al. Circadian learning and memory changes in Abeta1-42 induced Alzheimer’s mice. Metab Brain Dis. 2020;35(3):463-471. [28] SAEEDI M, RASHIDY-POUR A. Association between chronic stress and Alzheimer’s disease: Therapeutic effects of Saffron. Biomed Pharmacother. 2021;133:110995. doi: 10.1016/j.biopha.2020.110995. [29] GÓMEZ-GALLEGO M, GÓMEZ-GARCÍA J. Stress and verbal memory in patients with Alzheimer’s disease: different role of cortisol and anxiety. Aging Ment Health. 2019;23(11):1496-1502. [30] MORENO-JIMENEZ EP, FLOR-GARCIA M, TERREROS-RONCAL J, et al. Adult hippocampal neurogenesis is abundant in neurologically healthy subjects and drops sharply in patients with Alzheimer’s disease. Nat Med. 2019;25(4):554. [31] CHEN X, WU X, WU H, et al. Phase separation at the synapse. Nat Neurosci. 2020;23(3):301-310. [32] KENNEDY M. The Protein Biochemistry of the Postsynaptic Density in Glutamatergic Synapses Mediates Learning in Neural Networks. Biochemistry. 2018;57(27):4005-4009. [33] ZENG M, CHEN X, GUAN D, et al. Reconstituted Postsynaptic Density as a Molecular Platform for Understanding Synapse Formation and Plasticity. Cell. 2018;174(5):1172-1187.e16. [34] ZENG M, SHANG Y, ARAKI Y, et al. Phase Transition in Postsynaptic Densities Underlies Formation of Synaptic Complexes and Synaptic Plasticity. Cell. 2016;166(5):1163-1175.e12. [35] BUONAGURO EF, MORLEY-FLETCHER S, AVAGLIANO C, et al. Glutamatergic postsynaptic density in early life stress programming: Topographic gene expression of mGlu5 receptors and Homer proteins. Prog Neuropsychopharmacol Biol Psychiatry. 2020;96:109725. [36] SCHAEFER ML, PEREZ PJ, WANG M, et al. Neonatal Isoflurane Anesthesia or Disruption of Postsynaptic Density-95 Protein Interactions Change Dendritic Spine Densities and Cognitive Function in Juvenile Mice. Anesthesiology. 2020;133(4):812-823. [37] DE BARTOLOMEIS A, AVAGLIANO C, VELLUCCI L, et al. Translating preclinical findings in clinically relevant new antipsychotic targets: focus on the glutamatergic postsynaptic density. Implications for treatment resistant schizophrenia. Neurosci Biobehav Rev. 2019;107:795-827. |
| [1] | Wang Ji, Zhang Min, Yang Zhongya, Zhang Long. A review of physical activity intervention in type 2 diabetes mellitus with sarcopenia [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(8): 1272-1277. |
| [2] | Zhang Yan, He Ruibo, Wang Qingbo, Pi Yihua, Lu Chunmin, Xu Chuanyi, Ma Gang, Peng Peng. Effects of aerobic exercises with different load volumes on inflammatory response and insulin signaling pathway of skeletal muscle in obese rats [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(8): 1237-1244. |
| [3] | Bao Wenxia, Zhan Dongge, Wang Nian, Yang Xianjun, Ding Chengbiao. Foot-type recognition algorithm based on plantar pressure images [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(4): 527-533. |
| [4] | Li Yangjie, Qi Rong, Zhang Xinyu, Cheng Jiaji, Zhou Ning, Cui Xue, Cheng Shuang, Wang Zhengdong, Yan Nan. Neuroprotective effects of sodium butyrate and acetylation proteomics analysis in fluorosis rats [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(20): 3151-3157. |
| [5] | Liu Peng, Ma Gang, He Ruibo, Peng Peng. Effect of aerobic exercise on renal fibrosis in rats with chronic renal failure [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(20): 3209-3215. |
| [6] | Liu Yunqin, Lin Li, Xiao Wenhao, Ji Qiuming, Liu Yanqin. Effects of icariin on NRG1-ErbB signaling pathways in hippocampus of schizophrenia rats [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(20): 3236-3241. |
| [7] | Shui Xiaoping, Li Chunying, Li Mingjuan, Li Shunchang, Sun Junzhi, Su Quansheng. Effects of aerobic and resistance exercise on antioxidant stress index and brain-derived neurotrophic factor expression in the hippocampus of type 2 diabetic rats [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(2): 264-269. |
| [8] | Peng Zifu, Guo Xiangying, Fang Hongbo, He Yimin, Jiang Ning. Exercise intervention improves hippocampal cognitive function by regulating insulin-like growth factor 1 [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(17): 2729-2737. |
| [9] | Ying Chunmiao, Ma Yucheng, Fan Feiyan, Gao Chen, Wang Shaona, Yang Xing, Zhang Yunke. Action mechanism of traditional Chinese medicine to regulate neural stem cells in treatment of Alzheimer’s disease [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(15): 2385-2394. |
| [10] | Wen Xiaoyu, Sun Yuhao, Xia Meng. Effects of serum containing Wuzang Wenyang Huayu Decoction on phosphorylated-tau protein expression in Alzheimer’s disease cell model [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1068-1073. |
| [11] | Tang Jiping, Zhang Yeting. Exercise regulates adult hippocampal neurogenesis in Alzheimer’s disease: mechanism and role [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(5): 798-803. |
| [12] | Shui Xiaoping, Li Chunying, Li Shunchang, Sun Junzhi, Su Quansheng . Effects of aerobic and resistance exercises on brain-derived neurotrophic factor, nuclear factor-kappa B and inflammatory cytokines in skeletal muscle of type II diabetic rats [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(5): 669-675. |
| [13] | She Jian, Zhao Jing, Zhang Jiaming, Xia Haisha, Zhong Dongling, Li Yuxi, Zheng Zhong, Li Juan, Jin Rongjiang. Visualization analysis of literature on eye tracking in cognitive behaviors of autistic patients [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(35): 5724-5732. |
| [14] | Wang Xue, Liu Yang, Xu Jianfeng, Long Qianfa, Wang Tong, Zhong Jun. Neuroprotective effect of umbilical cord mesenchymal stem cell-derived exosomes on hippocampal neurons in mice with intracerebral hemorrhage [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(31): 4928-4934. |
| [15] | Li Suyao, Guo Minfang, Yu Jingwen, Meng Tao, Mu Bingtao, Li Mengdi, Li Na, Song Lijuan, Ma Cungen, Yu Jiezhong. Effect of eriodictyol on the imbalance of mitochondrial dynamics and apoptosis in SH-SY5Y cells induced by hydrogen peroxide [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(31): 4975-4981. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||