Chinese Journal of Tissue Engineering Research ›› 2020, Vol. 24 ›› Issue (16): 2585-2591.doi: 10.3969/j.issn.2095-4344.2270
Previous Articles Next Articles
Application of bioprinting in reconstruction of musculoskeletal interface
Zhang Junwei1, Cui Yutao2, Li Zuhao2, Wang Zhonghan2, Liu He2, Luo Wenbin2
- 1Department of Orthopedics, Xifeng District People’s Hospital of Qingyang City, Qingyang 745000, Gansu Province, China; 2Department of Orthopedics, the Second Hospital of Jilin University, Changchun 130041, Jilin Province, China
-
Received:2019-10-21Revised:2019-10-24Accepted:2019-12-05Online:2020-06-08Published:2020-03-26 -
Contact:Luo Wenbin, MD, Department of Orthopedics, the Second Hospital of Jilin University, Changchun 130041, Jilin Province, China -
About author:Zhang Junwei, Associate chief physician, Department of Orthopedics, Xifeng District People’s Hospital of Qingyang City, Qingyang 745000, Gansu Province, China -
Supported by:the National Natural Science Foundation of China, No. 81671804 and 81772456
CLC Number:
Cite this article
Zhang Junwei, Cui Yutao, Li Zuhao, Wang Zhonghan, Liu He, Luo Wenbin. Application of bioprinting in reconstruction of musculoskeletal interface[J]. Chinese Journal of Tissue Engineering Research, 2020, 24(16): 2585-2591.
share this article
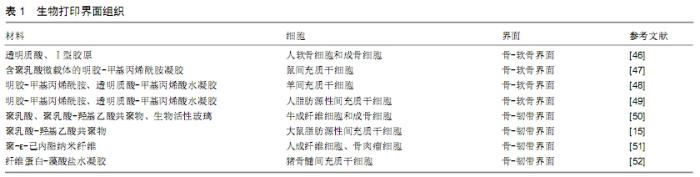
2.1 生物打印方法 用搭载细胞的水凝胶作为生物墨水,使用喷墨打印、激光辅助生物打印和微挤压式生物打印的方法成为制备界面组织工程支架的主流选择[28-30]。 喷墨打印是目前最为常用的打印类型,工作原理与普通的2D打印机相似,由生物材料和相应细胞成分组成的生物墨水通过打印机喷头在垂直方向上运动,从而打印出3D组织工程支架[31]。喷墨打印虽然具有打印速度快、成本低、操作便捷、应用广泛等优势,但热喷墨技术使生物材料和细胞在打印过程中需要承受较大的热和机械应力,同时存在喷头堵塞、细胞密度不理想和液滴尺寸难以控制等缺点。而声控喷墨技术虽解决了热喷墨技术的一些缺点,但其对打印材料的黏度具有较高的要求,这些缺点限制了喷墨打印技术在生物界面组织制备领域的应用[32]。 激光辅助生物打印是将激光聚焦于吸收性底物,底物吸收激光后为打印提供驱动力,并将生物材料挤压出打印喷口。激光辅助生物打印的喷头为开放式结构,同时对细胞产生的伤害较小,细胞存活率可达95%以上,确保生物支架中细胞具有良好的活性[33]。同时该技术还具有高分辨率和较高的打印精度,但是设备价格昂贵、打印过程耗时长及潜在的金属感染风险限制了其广泛应用[34]。 微挤压式生物打印是这些生物打印方法中最有前景的一种技术,因为其具有梯度打印的能力和与生物材料良好的兼容性[32]。在微挤压式生物打印中,生物墨水通过气动(高压)或机械力(活塞)连续地通过喷嘴以细丝状的形式从针状喷头中挤出,层层堆积,最终形成三维结构。与喷墨打印技术相比,微挤压式打印可更广泛的利用多种生物材料,具有更多样化的生物交联机制。同时,打印环境的温度适宜,避免持续高温影响细胞的活性[35]。以细胞球体作为基本的打印单位,可使得打印区域具有较高的细胞密度,也使得不同种类细胞的均匀分布成为可能[36-37]。提高生物打印的分辨率和细胞存活率是微挤压式生物打印面临的重大挑战[38]。 2.2 骨-软骨界面的生物打印 骨-软骨界面广泛存在于各种骨末端的关节部位,原生骨-软骨界面组织在胶原纤维结构、力学性能、细胞力学和生物化学成分方面存在显著差异[39]。在结构上,骨-软骨界面由3层结构组成,即关节软骨层、钙化软骨层和软骨下骨,这些复杂的结构使得这种界面组织在应力分布、滑动力学和负荷转移方面具有独特的性能[40]。 针对骨-软骨界面的梯度性质结构和组成特征,为更好地模拟自然微环境和这种异质性组织的机械特性,界面支架需具备以下特征:①合适的多孔性变化的结构,为不同界面层中的多种类型细胞提供理想的生存坏境。此外,需要充分考虑支架的内部几何结构,从而理想地模拟自然组织的结构和特性,多孔的互连结构支持氧气、营养和代谢废物的运输与交换[41-42]。②模仿与天然骨和软骨区域相似的机械性能梯度,通过矿化和内部结构的梯度分布为骨区域提供合适的硬度梯度,并向软骨区逐渐降低硬度和增加耐磨性能[43]。③生物打印骨-软骨各层结构的比例应与原生骨-软骨组织相匹配。成人骨-软骨组织的整体厚度约为3 mm,其组成约为90%的关节软骨、5%的钙化软骨和5%的软骨下骨[41]。钙化软骨是关节软骨和软骨下骨中间厚度为20-250 μm一层。在任何成人中,钙化软骨的平均厚度和矿物质密度随年龄、关节面积和机械负荷而变化,而钙化软骨层的浅层与关节软骨通过一条约5 μm潮痕形成融合。考虑到患者之间变化范围广,不同层的厚度对界面精确重建至关重要,这也对生物打印分辨率提出了很高的要求[44-45]。④生物墨水材料应具有可生物降解性,适当的降解速率应与细胞外基质的生成速率相匹配,以补偿生物降解过程中强度的损失[45]。 由于水凝胶作为生物墨水的机械强度较差,生物打印骨-软骨界面的早期尝试主要集中在概念验证研究上。在一项比较研究中,利用生物打印技术制备了一个模拟的双层骨-软骨组织结构,对软骨细胞和成骨细胞在由透明质酸和Ⅰ型胶原组成的水凝胶中进行行为研究。结果表明与非天然细胞外基质水凝胶相比,细胞在由天然胞外基质组成的水凝胶上具有良好的增殖能力和维持活跃的细胞功能,从而证明了仿生组件设计对细胞活性具有重要意义[46]。为了模拟天然骨-软骨组织的带状成分,另一项研究应用了生物打印和微载体技术相结合的新策略制备了一种生物活性结构,即将间充质干细胞搭载于聚乳酸微载体中,然后用明胶-甲基丙烯酰胺凝胶作为生物墨水进行封装。该研究探讨了仿生结构设计,获得了含/不含搭载细胞的微载体的凝胶层的带状结构,通过整合搭载细胞的微载体,生物墨水的机械强度也得到了提高[47]。为了快速转化、应用于临床,研究者开发了一种新型手持生物打印装置,允许在单次手术中同时将生物支架和封装的细胞同轴挤压到体内软骨缺损中。将间充质干细胞包裹在明胶-甲基丙烯酰胺和透明质酸-甲基丙烯酸水凝胶中作为生物墨水,通过该装置打印的结构在绵羊全层厚度软骨缺损模型中显示出与宿主更好的整合效果[48]。此外,人类脂肪源性间充质干细胞是未来临床应用的一大细胞来源,在相关研究中的应用揭示了该装置应用于人体软骨生物制造的可行性[49]。各骨-软骨界面的相关研究见表1。 "
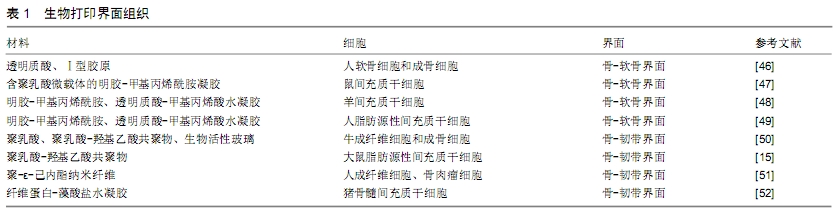

然而骨-软骨组织的组成和机械性能随深度变化很大,尽管在实用性和结构复杂性方面取得了令人欣喜的结果,但在水凝胶基的“纯”生物打印方法中,提供与其原生骨-软骨组织相近的适当的机械性能仍然没有得到解决。此外,利用生物打印技术对带状软骨组织进行仿生支架的制备也有待进一步研究。 2.3 骨-韧带/肌腱界面的生物打印 肌腱或韧带通过一种称为“附着”的特殊界面组织插入骨骼,该界面组织是不同生化和机械性能组织之间的过渡区域,对于肌肉骨骼运动中软组织和硬组织之间的相互作用是必不可少的[53]。骨-韧带/肌腱界面是一种多相过渡结构,由4个组成不同但结构互相连续的区域组成,即韧带/肌腱、纤维软骨、钙化纤维软骨和骨4部分,但是各个区域的细胞类型和细胞外基质具有显著区别。韧带区域主要存在的细胞是纤维母细胞,而细胞外基质以Ⅰ、Ⅲ型胶原为主;纤维软骨区域细胞为卵圆形的软骨细胞,而细胞外基质以蛋白聚糖、Ⅲ型胶原为主;钙化纤维软骨区域细胞为肥大的软骨细胞,而细胞外基质以X型胶原为主;骨区域的细胞为骨母细胞、成骨细胞和破骨细胞,钙化 的细胞外基质以Ⅰ型胶原为主。这样的多相过渡界面结构对于负荷在韧带/肌腱和骨组织间的传导非常重要,不仅能够有效避免应力集中,同时也有利于增强韧带插入骨组织的牢固程度,防止肌腱/韧带撕脱[54]。 骨-韧带/肌腱界面允许刚度逐渐增加,从而使应力集中最小化,并允许从韧带到骨的有效载荷转移。这些区域涉及2种不同成分的异质组织,即肌腱/韧带和骨。肌腱/韧带表现出承受张力的能力,而骨骼负责压缩负荷。骨-肌腱/韧带界面在结构组成和生物力学特性方面表现出相应的梯度,从而有效地转移不同材料之间的应力,并维持界面功能和稳态所需的异型细胞通信。这种特殊界面结构和独特的生物环境导致其在极为脆弱且易发生损伤[5]。在骨科重建手术中,实现肌 腱/韧带移植与软骨下骨的完全的功能性整合仍面临巨大挑战[55]。 SPALAZZI等[50]制备了一种3相支架用于前交叉韧带-骨界面的重建,A相由搭载成纤维细胞的聚乳酸形成,用于促进软组织形成;B相由聚乳酸-羟基乙酸共聚物组成,利于纤维软骨细胞和纤维软骨形成;C相由搭载成骨细胞的聚乳酸-羟基乙酸共聚物和生物活性玻璃组成,用于促进骨形成(图3)。为了促进附着体的再生,LIU等[15]开发了一种具有分级矿物涂层纳米纤维支架,以模拟自然肌腱到骨插入界面的矿物成分,这种支架具有诱导脂肪来源间充质干细胞分级成骨的能力。肌腱/韧带与骨界面的再生对于损伤后移植物的功能性整合至关重要。该研究目的是利用一站式静电纺丝制备的梯度支架来重建肌腱-骨界面,在聚-ε-己内酯纳米纤维支架上生长了2种细胞,该支架具有从随机到规律排列纳米纤维模式的逐渐转变,分别将骨肉瘤细胞和成纤维细胞接种在支架上培养96 h后,创造了一个随机的、一致的共培养组织界面,模拟了附着体成分的自然转变。结果表明,微结构梯度影响了细胞形态、组织拓扑结构,促进了细胞的生长[51]。设计一种多相结构、通过趋化间充质干细胞群的分化来模拟骨-韧带界面,这些间充质干细胞混合在纤维-海藻酸水凝胶中,通过在一个定制的双腔室培养系统中根据生长因子的空间分布呈现不同的分化结果,沿着软骨内、纤维软骨或韧带路径向下生长。这些间充质干细胞表现出在不同空间上的差异性分化,根据其所处的局部环境表现出软骨或韧带表型。这种方法为骨-韧带界面的制备提供了一种全新的工程策略[52]。 "


2.4 其他界面 在肌肉骨骼界面中除了研究较为广泛的骨-软骨界面和骨-韧带/肌腱界面,还存在着骨-半月板界面和肌肉-肌腱界面,这2个界面的研究相对较少。 半月板是一种位于膝关节股骨和胫骨之间的新月形楔形软骨样组织,为股骨和胫骨提供缓冲的结构。与其他界面组织一样,半月板组织也呈现带状排列的胶原纤维,其主要细胞类型是纤维软骨细胞。干燥的半月板组织约含有80%的Ⅰ型胶原,同时也存在少量的Ⅱ、Ⅲ和Ⅴ型胶原。与骨-软骨界面的生物打印相比,涉及骨-半月板界面生物打印领域的方法很少。近年来的研究方法主要集中在半月板组织本身的生物制备上,骨-半月板界面的重要作用尚未被强调[56-57]。然而,考虑到与 骨-软骨界面成分和结构的相似性,骨-软骨界面生物打印的方法可以借鉴地应用于骨-半月板界面的生物打印。因此,近年来人们对半月板的生物打印越来越重视,这是今后研究的一个重要课题[58]。 肌肉-肌腱界面的组织学特征是界面处的肌纤维细胞膜过度折叠,通过含有成纤维细胞的结缔组织小桥桥接肌腱和肌肉之间的间隙,该结缔组织将肌腱的致密胶原纤维斜向连接到肌纤维[59]。考虑到肌肉-肌腱界面在界面组织中作为肌腱/韧带和肌肉之间的桥梁作用,骨-韧带/肌腱界面组织的现有组织工程策略可能适合肌肉-肌腱界面生物制造。多种生物支架已被应用于肌肉-肌腱界面的重建,其中一些可在市场上买到。考虑到肌肉-肌腱界面组织的生物力学环境,最近开发的生物打印方法结合加固结构呈现特定区域的生物和机械特性,使用多种类型的活细胞用于肌肉-肌腱界面工程[4,50]。 "

| [1] CROSS LM, THAKUR A, JALILI NA, et al. Nanoengineered biomaterials for repair and regeneration of orthopedic tissue interfaces. Acta Biomater.2016;42:2-17. [2] REY-RICO A, CUCCHIARINI M. Smart and Controllable rAAV Gene Delivery Carriers in Progenitor Cells for Human Musculoskeletal Regenerative Medicine with a Focus on the Articular Cartilage. Curr Gene Ther.2017;17(2):127-138. [3] SAHOO S, TEH T, HE P, et al. Interface tissue engineering: next phase in musculoskeletal tissue repair.Ann Acad Med Singapore.2011;40(5):245-251. [4] MERCERON TK, BURT M, SEOL YJ, et al. A 3D bioprinted complex structure for engineering the muscle-tendon unit. Biofabrication.2015;7(3):035003. [5] ANDARAWIS-PURI N, FLATOW EL, SOSLOWSKY LJ. Tendon basic science: Development, repair, regeneration, and healing. J Orthop Res.2015;33(6):780-784. [6] PETRIGLIANO FA, MCALLISTER DR, WU BM. Tissue engineering for anterior cruciate ligament reconstruction: a review of current strategies.Arthroscopy.2006;22(4):441-451. [7] SILVA MJ, THOMOPOULOS S, KUSANO N, et al. Early healing of flexor tendon insertion site injuries: Tunnel repair is mechanically and histologically inferior to surface repair in a canine model. J Orthop Res.2006;24(5):990-1000. [8] CHEN MH, WANG LL, CHUNG JJ, et al. Methods To Assess Shear-Thinning Hydrogels for Application As Injectable Biomaterials. Acs Biomater Sci Eng.2017;3(12):3146-6310. [9] BOUSHELL MK, HUNG CT, HUNZIKER EB,et al. Current strategies for integrative cartilage repair. Connect Tissue Res. 2017; 58(5):393-406. [10] MAKRIS EA, GOMOLL AH, MALIZOS KN, et al. Repair and tissue engineering techniques for articular cartilage.Nat Rev Rheumatol.2015;11:21-34. [11] NUKAVARAPU SP, DORCEMUS DL. Osteochondral tissue engineering: current strategies and challenges.Biotechnol adv. 2013;31(5):706-721. [12] SEIDI A, RAMALINGAM M, ELLOUMI-HANNACHI I, et al. Gradient biomaterials for soft-to-hard interface tissue engineering.Acta Biomater.2011;7:1441-1451. [13] MIR TA, NAKAMURA M. Three-Dimensional Bioprinting: Toward the Era of Manufacturing Human Organs as Spare Parts for Healthcare and Medicine.Tissue Eng Part B Rev. 2017;23(3):245-256. [14] CRISCENTI G, LONGONI A, DI LUCA A, et al. Triphasic scaffolds for the regeneration of the bone-ligament interface. Biofabrication. 2016;8(1):015009. [15] LIU W, LIPNER J, XIE J, et al. Nanofiber scaffolds with gradients in mineral content for spatial control of osteogenesis. ACS Appl Mater Interfaces. 2014;6(4):2842-2849. [16] NGUYEN QT, JACOBSEN TD, CHAHINE NO. Effects of Inflammation on Multiscale Biomechanical Properties of Cartilaginous Cells and Tissues.Acs Biomater Sci Eng. 2017; 3(11):2644-2656. [17] PIMENTEL CR, KO SK, CAVIGLIA C, et al. Three-dimensional fabrication of thick and densely populated soft constructs with complex and actively perfused channel network.Acta Biomater.2018;65:174-184. [18] SUNTORNNOND R, TAN EYS, AN J, et al. A highly printable and biocompatible hydrogel composite for direct printing of soft and perfusable vasculature-like structures.Sci Rep. 2017; 7(1):16902. [19] MEKHILERI NV, LIM KS, BROWN GCJ, et al. Automated 3D bioassembly of micro-tissues for biofabrication of hybrid tissue engineered constructs.Biofabrication. 2018;10(2): 024103. [20] KOO Y, KIM G. New strategy for enhancing in situ cell viability of cell-printing process via piezoelectric transducer-assisted three-dimensional printing. Biofabrication.2016;8(2):025010. [21] LIU X, YUK H, LIN S, et al. 3D Printing of Living Responsive Materials and Devices.Adv Mater.2018;30(4):201704821. [22] NAGARAJAN N,DUPRET-BORIES A,KARABULUT E,et al. Enabling personalized implant and controllable biosystem development through 3D printing.Biotechnol Adv. 2018;36(2): 521-533. [23] ZHANG YS, YUE K, ALEMAN J, et al. 3D Bioprinting for Tissue and Organ Fabrication.Ann Biomed Eng.2017;45(1):148-163. [24] XIONGFA J, HAO Z, LIMING Z, et al. Recent advances in 3D bioprinting for the regeneration of functional cartilage.Regen Med.2018;13(1):73-87. [25] GAO G, HUANG Y, SCHILLING AF, et al. Organ Bioprinting: Are We There Yet? Adv Healthc Mater. 2018;7(1):201701018. [26] RAVNIC DJ, LEBERFINGER AN, KODURU SV, et al. Transplantation of Bioprinted Tissues and Organs: Technical and Clinical Challenges and Future Perspectives.Ann Surg. 2017;266(1):48-58. [27] YANG Y, SONG X, LI X, et al. Recent Progress in Biomimetic Additive Manufacturing Technology: From Materials to Functional Structures.Adv Mater.2018;19:e1706539. [28] LEVATO R, WEBB WR, OTTO IA, et al. The bio in the ink: cartilage regeneration with bioprintable hydrogels and articular cartilage-derived progenitor cells.Acta Biomater. 2017;61:41-53. [29] MOUSER VHM, LEVATO R, MENSINGA A, et al. Bio-ink development for three-dimensional bioprinting of hetero-cellular cartilage constructs.Connect Tissue Res. 2018:1-15. [30] YANG J, ZHANG YS, YUE K, et al. Cell-laden hydrogels for osteochondral and cartilage tissue engineering.Acta Biomater. 2017;57:1-25. [31] HUANG Y, ZHANG XF, GAO G, et al. 3D bioprinting and the current applications in tissue engineering.Biotechnol J. 2017; 12(8).doi:10.1002/biot.201600734. [32] BRACAGLIA LG, SMITH BT, WATSON E, et al.3D printing for the design and fabrication of polymer-based gradient scaffolds. Acta Biomater.2017;56:3-13. [33] MORA-BOZA A, LOPEZ-DONAIRE ML. Preparation of Polymeric and Composite Scaffolds by 3D Bioprinting.Adv Exp Med Biol.2018;1058:221-245. [34] PATI F, CHO DW. Bioprinting of 3D Tissue Models Using Decellularized Extracellular Matrix Bioink.Methods Mol Biol. 2017;1612:381-390. [35] DEMIRTAS TT, IRMAK G, GUMUSDERELIOGLU M. A bioprintable form of chitosan hydrogel for bone tissue engineering.Biofabrication.2017;9(3):035003. [36] MARTINEZ PR, GOYANES A, BASIT AW, et al. Fabrication of drug-loaded hydrogels with stereolithographic 3D printing.Int J Pharm.2017;532(1):313-317. [37] YOON S, PARK JA, LEE HR, et al. Inkjet-Spray Hybrid Printing for 3D Freeform Fabrication of Multilayered Hydrogel Structures.Adv Healthc Mater.2018;7(14):e1800050. [38] MARQUES CF, DIOGO GS, PINA S, et al. Collagen-based bioinks for hard tissue engineering applications: a comprehensive review.J Mater Sci Mater Med.2019;30(3):32. [39] DISCHER DE, MOONEY DJ, ZANDSTRA PW. Growth factors, matrices, and forces combine and control stem cells. Science. 2009;324(5935):1673-1677. [40] VINING KH, MOONEY DJ. Mechanical forces direct stem cell behaviour in development and regeneration.Nat Rev Mol Cell Biol.2017;18(12):728-742. [41] DI LUCA A, VAN BLITTERSWIJK C, MORONI L. The osteochondral interface as a gradient tissue: from development to the fabrication of gradient scaffolds for regenerative medicine.Birth Defects Res C Embryo Today. 2015;105(1):34-52. [42] OLIVEIRA I, VIEIRA S, OLIVEIRA JM, et al. Nanoparticles-Based Systems for Osteochondral Tissue Engineering.Adv Exp Med Biol.2018;1059:209-217. [43] PEREIRA DR, REIS RL, OLIVEIRA JM. Layered Scaffolds for Osteochondral Tissue Engineering. Adv Exp Med Biol. 2018; 1058:193-218. [44] ZHANG Y, WANG F, TAN H, et al. Analysis of the mineral composition of the human calcified cartilage zone.Int J Med Sci.2012;9(5):353-360. [45] LONGLEY R, FERREIRA AM, GENTILE P. Recent Approaches to the Manufacturing of Biomimetic Multi-Phasic Scaffolds for Osteochondral Regeneration.Int J Mol Sci. 2018; 19(6).pii: E1755.doi:10.3390/ijms19061755. [46] PARK JY, CHOI JC, SHIM JH, et al. A comparative study on collagen type I and hyaluronic acid dependent cell behavior for osteochondral tissue bioprinting. Biofabrication. 2014;6(3): 035004. [47] LEVATO R, VISSER J, PLANELL JA, et al. Biofabrication of tissue constructs by 3D bioprinting of cell-laden microcarriers. Biofabrication.2014;6(3):035020. [48] DI BELLA C, DUCHI S, O'CONNELL CD, et al. In situ handheld three-dimensional bioprinting for cartilage regeneration.J Tissue Eng Regen Med.2018;12(3):611-621. [49] ONOFRILLO C, DUCHI S, O'CONNELL CD, et al. Biofabrication of human articular cartilage: a path towards the development of a clinical treatment.Biofabrication.2018;10(4): 045006. [50] SPALAZZI JP, DAGHER E, DOTY SB, et al. In vivo evaluation of a multiphased scaffold designed for orthopaedic interface tissue engineering and soft tissue-to-bone integration. J Biomed Mater Res A.2008;86(1):1-12. [51] NOWLIN J, BISMI MA, DELPECH B, et al. Engineering the hard-soft tissue interface with random-to-aligned nanofiber scaffolds.Nanobiomedicine.2018;5:1849543518803538. [52] HARRIS E, LIU Y, CUNNIFFE G, et al. Biofabrication of soft tissue templates for engineering the bone-ligament interface. Biotechnol Bioeng. 2017;114(10):2400-2411. [53] YANG PJ, TEMENOFF JS. Engineering orthopedic tissue interfaces.Tissue Eng Part B Rev.2009;15:127-141. [54] SPALAZZI JP, DOTY SB, MOFFAT KL, et al. Development of controlled matrix heterogeneity on a triphasic scaffold for orthopedic interface tissue engineering.Tissue Eng. 2006; 12(12):3497-3508. [55] KIM HM, GALATZ LM, DAS R, et al. Musculoskeletal deformities secondary to neurotomy of the superior trunk of the brachial plexus in neonatal mice.J Orthop Res. 2010; 28(10):1391-1398. [56] WARREN PB, HUEBNER P, SPANG JT, et al. Engineering 3D-Bioplotted scaffolds to induce aligned extracellular matrix deposition for musculoskeletal soft tissue replacement. Connect Tissue Res. 2017;58(3-4):342-354. [57] ROMANAZZO S, VEDICHERLA S, MORAN C, et al. Meniscus ECM-functionalised hydrogels containing infrapatellar fat pad-derived stem cells for bioprinting of regionally defined meniscal tissue. J Tissue Eng Regen Med. 2018;12(3):e1826-e1835. [58] ONG CS, YESANTHARAO P, HUANG CY, et al. 3D bioprinting using stem cells.Pediatr Res. 2018;83:223-231. [59] MARKSTEDT K, MANTAS A, TOURNIER I, et al. 3D Bioprinting Human Chondrocytes with Nanocellulose-Alginate Bioink for Cartilage Tissue Engineering Applications. Biomacromolecules.2015;16(5):1489-1496. [60] STEINERT AF, PALMER GD, CAPITO R, et al. Genetically enhanced engineering of meniscus tissue using ex vivo deliVery of transforming growth factor-beta 1 complementary deoxylibonucleic acid. Tissue Eng.2007;13(9):2227-2237. |
| [1] | Han Ningning, Zuo Jinfu, Sun Miao, Tang Shengjian, Liu Fangjun. Application and progress of umbilical cord mesenchymal stem cells in bone tissue engineering [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(13): 2079-2086. |
| [2] | Cao Xuhan, Bai Zixing, Sun Chengyi, Li Yanle, Sun Weidong. Application of robot in orthopedic surgery: reliability and room for improvement [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(9): 1416-1421. |
| [3] | Xu Guofeng, Li Xuebin, Tang Yifan, Zhao Yin, Zhou Shengyuan, Chen Xiongsheng, Jia Lianshun. The role of autophagy in ossification of the human ligamentum flavum [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(8): 1174-1181. |
| [4] | Wang Gang, Li Donghui, Bai Zhiming. Evolution and progress of replacement therapy, materials and reconstruction of long ureteral injuries [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(8): 1299-1305. |
| [5] |
Wang Tiantian, Wang Jianzhong.
Application and prospect of bone marrow mesenchymal stem cells in the
treatment of early femoral head necrosis |
| [6] | Huang Hui, Dai Yao, Li Yongsheng, Chen Wei, Tang Fang, Huang Yuting, Zhou Zheng, Liu Hairong. Application and research of non-coding RNA in bone tissue engineering with cells and scaffold [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(4): 596-605. |
| [7] | Liu Xiaoyin, , Zhong Lin, Zheng Bo, Wei Pan, Dai Chen, Hu Liangcong, Wang Tiantian, Liang Xiaolong, Zhang Sai, Wang Xiaoli. Diffusion tensor imaging predicting locomotor function recovery with 3D printing scaffold after spinal cord injury [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(28): 4547-4554. |
| [8] | Zhang Yifan, Zhang Jiaying, Xu Mingen, Wang Ling, Zhang He. Optimization of the coaxial cell printing performance of bioink and printing of tissue-engineered scaffolds with vascular-like structure [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(22): 3553-3558. |
| [9] | Zhu Haizhou, Zhao Zhankui, Wang Xinzhe, Liu Deqian, Yu Honglian. Urine-derived stem cells differentiate into urothelial cells and smooth muscle cells in vitro [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(19): 3010-3016. |
| [10] | Hu Kaiwei, Nie Ling, Zhang He, Li Yuzhou, Yang Sheng. Application of α-n-butyl 2-cyanoacrylate medical adhesive for repair of oral mucosal wound of rats [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(16): 2526-2530. |
| [11] | Liu Jin, Gao Lilan, Peng Yulin, Zhang Xizheng. Mechanical properties of rat tibia under high-G environment [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(11): 1654-1658. |
| [12] | He Yunfei, Wang Shuang, Ma Jun, Yu Lei, Wen Jiankun, Ye Xiaojian. P38/Akt pathway regulates the oriented differentiation of bone marrow mesenchymal stem cells in nanofiber annulus fibrosus scaffolds with different spatial structures [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(10): 1540-1546. |
| [13] | Li Shuhua, Li Meilan. Preparation of the surface patterned polyacrylamide-acrylic hydrogel [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(10): 1553-1556. |
| [14] | Wang Kai, Zheng Shuang, Pan Su, Zhang Heng, Zhang Wei, Wang Haosheng, Yang Xiaoyu, Fu Chuan. Preparation of 3D printed bone tissue engineering scaffold [J]. Chinese Journal of Tissue Engineering Research, 2019, 23(34): 5516-5522. |
| [15] | Shi Songyuan1, Peng Zhihui2. Current problems and potential treatment options for sports cartilage injury [J]. Chinese Journal of Tissue Engineering Research, 2019, 23(31): 5059-5064. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||