Chinese Journal of Tissue Engineering Research ›› 2020, Vol. 24 ›› Issue (16): 2592-2598.doi: 10.3969/j.issn.2095-4344.2269
Previous Articles Next Articles
Repairing bone defects with heterogeneous calcined bone materials: characteristics, advantages and problems
Li Fang, Wu Chao, Shi Qianhui, Cheng Yuting, Zhou Qian, Huo Hua, Qi Yuhan, Dong Qiang, Liao Jian
- School/Hospital of Stomatology of Guizhou Medical University, Guiyang 550004, Guizhou Province, China
-
Received:2019-10-18Revised:2019-10-24Accepted:2019-11-29Online:2020-06-08Published:2020-03-27 -
Contact:Liao Jian, MD, Associate professor, Associate chief physician, Master’s supervisor, School/Hospital of Stomatology of Guizhou Medical University, Guiyang 550004, Guizhou Province, China -
About author:Li Fang, Master candidate, School/Hospital of Stomatology of Guizhou Medical University, Guiyang 550004, Guizhou Province, China -
Supported by:the National Natural Science Foundation of China, No. 81660179; the Science and Technology Program of Guizhou Province, No. LH[2016]7257
CLC Number:
Cite this article
Li Fang, Wu Chao, Shi Qianhui, Cheng Yuting, Zhou Qian, Huo Hua, Qi Yuhan, Dong Qiang, Liao Jian. Repairing bone defects with heterogeneous calcined bone materials: characteristics, advantages and problems[J]. Chinese Journal of Tissue Engineering Research, 2020, 24(16): 2592-2598.
share this article

2.1 煅烧骨材料的来源及制备 收取成年新鲜的牛 骨[14-15]、猪骨、羊骨[16]、鸡骨[17]、鱼骨[18-19] 、鱼鳞[20]、珊瑚等[21],手动去除黏附的组织。为了避免裂缝和烟灰在热处理过程中形成,故先将其放入蒸馏水中煮沸 12 h,再放入乙醇中脱水,之后在70 ℃下干燥3 d。将干燥的骨块在常规的Ni-Cr盘中以10 ℃/min的速率升至800 ℃下煅烧6 h,然后将煅烧的骨块置于铂片上,并以2.5 ℃/min的速率升至1 250,1 300,1 400 ℃各烧结1 h。最后缓慢冷却至室温,遂得到从黄白色逐渐变成白色的煅烧骨。当然,不同的煅烧方法中其温度可能稍有差异。 2.2 煅烧骨材料的性质 2.2.1 理化特性 生物羟基磷灰石的化学式为Ca10(PO4)6(OH)2,是天然煅烧骨的主要无机成分[13],也是众所周知的活性生物陶瓷材料[22]。煅烧骨通过热处理进行了脱脂、脱蛋白,抑制了受体中的免疫应答,也防止了病毒的传播,同时保留了天然骨骼的无机成分和三维多孔结构,因此具有骨传导性、间接的骨诱导性[23]、高度的生物相容性、可降解性、空间维持作用等[5]。煅烧骨中的天然骨架结构和相互连接的孔隙(图2)有利于氧气和营养物质传输及成骨细胞黏附、肉芽组织长入,促使材料内部的骨、软骨组织分化形成[24-26],然而多孔性却降低了煅烧骨的机械性能[27]。尽管如此,煅烧骨也能够维持到成骨细胞生长、增殖、分化并形成新骨的整个过程。不过,为了稳定其基本的机械性能,其孔径必须在100-500 μm之间[28],同时对其孔隙率也存在一定的限制[29]。SUNIL等[19]使用能量色散X射线光谱、X射线荧光和电感耦合等离子体在检测煅烧骨中发现了特征性的元素:阳离子有Na+、K+、Zn2+、Si4+、Mg2+、Ca2+等,阴离子如F-、OH-、CO32-等,它们在组织生化反应中发挥着重要作用。其中的Na+、Mg2+与骨状磷灰石层的形成有关[30],在牙齿和骨骼的发育中起着重要的作用,如果它们缺失则可能会导致骨质疏松甚至吸收;CO32-是大气中的CO2在羟基磷灰石中与OH-反应形成的碳酸根离子[31],由于碳酸盐是骨结构的组成部分,因此CO32-的存在可以改善羟基磷灰石生物活性;Ca2+和OH-可引起细胞外间质分泌酸性磷酸酯酶,促使成骨细胞分泌类骨质并形成骨组织。因此,煅烧骨被广泛用于生物医学领域[15,19,32-34]。不过,影响羟基磷灰石性质的最重要参数是热处理温度和时间[32,35]:当温度<560 ℃时,与有机化合物(脂肪和蛋白质)的降解和燃烧有关;>560 ℃时,与无机基质中的热转化有关[36];在1 250 ℃时,煅烧骨支架显示出结晶较好的羟基磷灰石;1 300 ℃是有利于在羟基磷灰石表面上形成生物矿化磷灰石的最佳温度[16]。此时,煅烧骨具有优异的细胞活力、血液相容性和增强磷灰石生长等特性。当然,温度越高、时间越久,羟基磷灰石晶体越致密、越不易吸收[37],且过度加热反而会降低其机械性能。因此,将羟基磷灰石单独用于整形外科和垂直骨增量的手术中仍然具有挑战性。 "


2.2.2 抗微生物特性 在骨增量手术当中偶尔会发生细菌感染导致手术失败的情况[38],这不仅会对患者造成再次创伤,同时还会给医疗系统带来较高的成本代价[39]。其中最常引起术区感染的细菌是金黄色葡萄球菌[40],因此寻找一种具有抗微生物特性的骨移植材料成为了当今的研究热点。有研究发现在1 000 ℃下煅烧牛骨和猪骨获得的羟基磷灰石具有抗微生物的特性,且对金黄色葡萄球菌生长的抑制率约为95%[37]。原理是金黄色葡萄球菌为革兰阳性菌,具有较厚肽聚糖组成的细胞壁、碳水化合物和带电荷的氨基酸聚合物,使其具有高度亲水性,利于羟基磷灰石与革兰阳性菌的细胞壁相互作用,使羟基磷灰石不规则的表面纹理对细菌细胞壁造成机械性损伤、羟基磷灰石颗粒表面上活性氧对其产生化学性损伤,从而达到预防细菌感染的效果。 2.3 复合支架材料 虽然煅烧骨具有以上的种种优点,但是其单独作为骨移植材料时由于其溶解度较低、吸收较慢、机械性能较差、没有或仅有轻微的骨诱导性使其应用受到限制。最近组织工程研究出的三维混合支架是将煅烧骨与陶瓷、聚合物、骨诱导因子或金属离子复合,它们可以综合并突显出各自材料的优点,使得此复合支架具有较高的骨诱导性、较强的机械性、较快的降解率等优点[41-43],更快更好地促进新骨生成,与材料吸收的速度相匹配,获得令人满意的骨增量效果[44]。 2.3.1 煅烧骨与β-磷酸三钙复合 众所周知,羟基磷灰石在体内的降解性较差,而β-磷酸三钙具有比羟基磷灰石更快的降解速率,且β-磷酸三钙还具有高热稳定性、较强的硬度、抗压强度及骨传导性。但也有研究报道称β-磷酸三钙的降解速度太快,远超过了新骨形成速度,以至于不能获得满意的成骨效果[45]。最近FRAYSSINET等[46]发现将质量比为30/70或40/60的β-磷酸三钙和羟基磷灰石混合组成双相磷酸钙陶瓷,能综合两者的降解速度之差,控制其降解速度。PICCIRILLO等[47]在鱼骨中加入脱水磷酸氢钙或磷酸二氢铵后进行煅烧,发现部分羟基磷灰石转化为β-磷酸三钙,并且在800-900 ℃时产生的双相磷酸钙最多[48]。该研究还发现此复合物具有互锁孔的天然骨结构,为血液渗入提供了良好条件,在骨缺损修复方面的效果优于单独使用羟基磷灰石或纯β-磷酸三钙陶瓷。此外,双相磷酸钙还具有均匀的孔隙尺寸、高结晶度、良好的生物相容性及骨诱导性,能够在较短的时间内将钙和磷离子吸附到支架表面[49],为新骨形成创造良好条件。虽然双相磷酸钙材料拥有独特的组成结构而具有良好的医学应用前景,但是其在临床治疗骨缺损的效果还有待进一步研究。 2.3.2 煅烧骨与纳米六方氮化硼复合 六方氮化硼是一种结晶的多层材料,具有较强的共价键和较弱的范德华力,因此拥有较好的机械强度、热传导性及抗热冲击性。近年来由于其无毒、透明、化学惰性及其它优异的性能,使得六方氮化硼成为化妆品中的功能性填料。此外,由于良好的生物相容性、生物降解性及促进骨组织钙化的功能使六方氮化硼在医学临床中同样具有重要的作用。BAKAN等[50]将羟基磷灰石与六方氮化硼以质量比200∶3混合,在1 300 ℃左右共烧结后经X射线衍射和傅里叶红外光谱检测发现,获得的复合材料同时含有六方氮化硼、β-磷酸三钙和羟基磷灰石;在此复合材料中培养人骨肉瘤细胞,结果发现其增殖及细胞活力未出现不良反应;抗菌实验发现,通过六方氮化硼/煅烧骨对粪肠球菌具有良好的抑制作用,表明其可用作骨组织工程中的安全生物材料。由于六方氮化硼/煅烧骨复合材料具有良好的机械性能[51]、生物学效应及可促进骨组织愈合、预防术后感染等,在临床上具有很大的开发潜力和应用价值。 2.3.3 煅烧骨与30%Bioglass?-45S5复合 在生物活性玻璃中,Bioglass?-45S5由45%SiO2、24.5%Na2O、24.5%CaO和6%P2O5组成。有学者将天然墨鱼骨在煅烧温度为900 ℃时加入30%的Bioglass?-45S5粉末共烧结后得到混合生物活性支架,此支架新生成的产物有磷酸钠、β-磷酸三钙、无定形二氧化硅和大量的磷灰石[52]。其中的钙离子在骨组织重塑过程中参与细胞内成骨机制的激活;硅离子参与新骨的形成及矿化。另外,在煅烧骨/Bioglass?-45S5的烧结过程中形成了较强的化学键,并且促进了部分羟基磷灰石转化为β-磷酸三钙。经压缩应力和极限抗压强度检测证明了Bioglass?-45S5的添加显著提高了此生物活性支架的平均抗压强度;生物学试验表明其对MG63细胞的体外增殖代谢活性产生了积极影响;在Dulbecco改良的Eagle培养基中促进了磷灰石层的形成;此混合支架比单一材料能更有效地促进碱性磷酸酶的产生。这些结果证明了此复合材料对种植手术区骨量不足的治疗提供了新思路。 2.3.4 煅烧骨与锶、聚己内酯复合 锶(Sr)是人体骨骼中不可缺少的微量元素,在抑制骨吸收和刺激新骨形成方面起着重要作用[53],具有促进骨缺损修复能力。KIM等[54]通过在猪煅烧骨表面上按顺序涂覆SrCl2和聚己内酯获得了含锶的复合支架,此支架可改善煅烧骨的机械性能,并可控制锶的释放速率以调节新骨形成速度。CHENG等[55]详细研究了煅烧骨、煅烧骨/己内酯和煅烧 骨/己内酯/锶对间充质干细胞的体外行为、增殖、成骨基因表达和碱性磷酸酶活性的影响,综合结果表明,虽然聚己内酯涂层略微影响了煅烧骨的骨修复潜力,但相比之下,煅烧骨/己内酯/锶能更好地促进间充质干细胞的成骨分化,体内实验也证实了煅烧骨/己内酯/锶加速骨缺损部位的血管化和骨化能力均优于煅烧骨和煅烧骨/己内酯,从而更好地完成骨缺损修复。 2.3.5 煅烧骨与硫酸钙复合 硫酸钙是一种来源广泛、成本低廉、吸收较快的骨移植材料,同时还具有一定的骨诱导能力[5],因此通常被当作颌骨的骨增量材料[56]。将煅烧骨与硫酸钙复合后能综合调节材料的降解速度。 DE TULLIO等[1]将硫酸钙与生物羟基磷灰石复合后植入人体拔牙窝中,检测其拔牙位点保存的效果。此试验中共有10名受试者,纳入标准为体健、上颌和/或下颌有一颗或多颗牙齿因残根或外伤等需要拔除,排除标准是存在可能影响手术伤口愈合的系统性危险因素患者。整个测试过程中采用双盲、随机原则,立即在16个新鲜拔牙窝内依次植入羟基磷灰石、硫酸钙、50%羟基磷灰石与50%硫酸钙复合物和空白对照,然后用富血小板纤维蛋白膜覆盖、缝合,5个月后发现无明显炎症的角化牙龈覆盖了所有植骨部位,其中空白对照组牙槽骨在水平向和垂直向上都存在显著骨吸收,而羟基磷灰石与硫酸钙复合组未见明显的骨吸收,且比植入单一材料组呈现出更多的新生骨量。然后从每个植骨部位用取骨钻(内径至少为2 mm)进行种植窝的初期预备并收取出取骨钻里的骨组织,随后再逐级预备种植窝,放置种植体,并对取出的骨块进行检测,结果发现部分硫酸钙颗粒转化为了磷酸钙,吸收较快的硫酸钙使羟基磷灰石颗粒充分发挥了空间维持作用,并促进血管、成骨细胞向羟基磷灰石内生长,使此复合材料的成骨速度与移植材料吸收速度相匹配,便于牙槽窝周围骨壁进行吸收和改建,最终基本上恢复了拔牙前牙槽骨的宽度与高度。虽然此复合物可显著刺激骨质生长,增加骨缺损的愈合率,但在临床上的使用尚未普及。 2.3.6 煅烧骨与壳聚糖复合 壳聚糖是一种来自甲壳素部分脱乙酰化的线性多糖,其结构式见图3,常存在于海洋甲壳类动物、昆虫和真菌细胞壁中,由于其较好的生物活性已被广泛应用于伤口覆盖膜、隐形眼镜和人造皮肤等。壳聚糖是亲水性的,可通过溶菌酶在人体内降解[22]。在壳聚糖基质中,添加煅烧骨来源的羟基磷灰石可得到的互连孔性复合材料[57],改善与骨组织之间的化学键、增强机械性能、加快降解速率。经检测此复合材料的pH值约为7.0,非常接近人类血浆的pH值。另外,它还无毒、不致畸,具有良好的热稳定性和细胞相容性,可促进细胞增殖等;其抗压强度为26.2-94.8 MPa,高于人体椎体,但低于桡骨和胫骨,表明天然羟基磷灰 石/壳聚糖复合材料可用于负载不高的骨缺损中。另外,羟基磷灰石的钙离子可通过配位键与壳聚糖的氨基结合,所以在一定范围内,壳聚糖含量越高复合材料的机械强度越强。因此,此复合物在骨替代材料中是一种较好的选择,有望应用于临床进行骨缺损修复[58]。 "

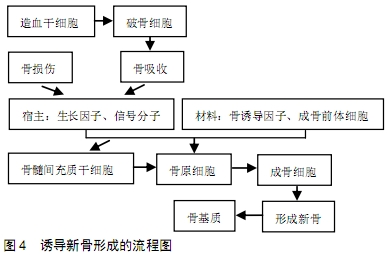
2.3.7 煅烧骨与骨形态发生蛋白复合 骨形态发生蛋白是一种骨诱导生长因子,能够促进血管和骨组织形成,如骨形态发生蛋白2和骨形态发生蛋白7可用于上颌窦底骨增量和拔牙位点保存[59]。其中研究最多的是骨形态发生蛋白2[60],它是促进骨缺损和骨折愈合中最显著的生长因子之一,将其逐级涂覆到植骨材料上可以改善骨再生的过程。然而,骨形态发生蛋白2的制备过程复杂、生产周期长、半衰期短,且骨形态发生蛋白2的爆发式释放可以诱导炎症反应,这种情况虽然被认为有利于促进骨形成,但是它也可以引起较多的不良反应[61],如:可能导致局部骨质过度生长、软组织肿胀、骨质溶解和异位骨形成,甚至导致远处癌症的发生等。虽然减少骨形态发生蛋白2的剂量可以减少这些不良反应,但通常都需要较高剂量的骨形态发生蛋白2来诱导显著的新骨形成。因此,要想使骨形态发生蛋白2既能挥发骨诱导能力又要避免并发症的形成,这就需要较低溶解度的载体来控制骨形态发生蛋白2的释放速率以达到理想的生物学效应。有学者发现煅烧骨的空心羟基磷灰石微球与骨形态发生蛋白2组合可以控制骨形态发生蛋白2的释放速度,同时还能拥有可观的骨再生效果[62]。因为骨形态发生蛋白2对羟基磷灰石具有较高亲和力,所以骨形态发生蛋白2可以通过静电相互作用及氢键结合牢固地吸附在羟基磷灰石表面,这种强烈的相互作用使得骨形态发生蛋白2难以被来自其他蛋白质的竞争性吸附置换,因此体外骨形态发生蛋白2的释放速率得以控制。另外,空心羟基磷灰石微球表面有大量的介孔壳等独特结构,使解吸的骨形态发生蛋白2可以很容易再次吸附在羟基磷灰石表面上。此外,由于表面壳壁的曲折性使骨形态发生蛋白2从孔到微球的外表面迁移变得困难,这些都将显著控制骨形态发生蛋白2的释放量。KIEKER等[63]曾将此组合物植入绵羊股骨缺损处,4周后便出现了新骨形成的迹象,在12周后组织学评估显示其编织骨和板层骨已形成。 FALCIGNO等[64]又发现了一种新型骨形态发生蛋白2模拟肽能更好地再现成骨特性,但骨形态发生蛋白2模拟肽的突出缺点是溶解性差,这就增加了临床应用难度。E7为骨结合蛋白中一种可以与钙结合的带正电荷氨基酸基序,主要负责蛋白质结合骨内的钙,还可以将骨形态发生蛋白2模拟肽锚定在异体材料的表面,保留其生物学活性,促进骨形成、矿化和骨整合。WU等[65]创新性地将E7与骨形态发生蛋白2模拟肽及煅烧骨复合起来成为一种新型骨增量材料,同时拥有煅烧骨的骨传导性和骨形态发生蛋白2肽的骨诱导性,不仅使骨形态发生蛋白2肽的溶解度得到改善,而且还被加载到煅烧骨的表面上持续发挥作用,诱导更快的新骨形成。与单独使用骨形态发生蛋白2肽相比,复合材料的碱性磷酸酶活性和矿化能力更高,表明此复合材料进一步促进了间充质干细胞的成骨分化。XI等[3]将此复合材料植入兔颅骨缺损处,发现在第4周时它们形成了相互连接的骨小梁,在第12周时观察到新骨髓组织之间形成了骨髓腔,缺损区域内新形成的骨组织完全矿化,其外观与正常骨相似,并且在骨厚度和生物力学强度方面都获得了令人满意的效果。由于此新型复合替代材料同时具有优异的骨诱导性和骨传导性,因此可考虑用于临床上进行骨增量手术。 2.3.8 煅烧骨与F1蛋白复合 乳胶F1是从巴西橡胶树中提取的天然乳胶[66],具有潜在的促进缺损骨组织恢复的能力。在60 ℃下将聚赖氨酸涂敷到胶乳中可以获得F1蛋白。已有研究证明,F1蛋白在不同物种(例如鼠、兔、狗和人)体内无毒、不致敏,表现出良好的生物相容性、新血管生成活性,促使成骨细胞黏附及外基质形成的能力,具有较高的临床应用潜力,已经用于不同的临床医学用途[67]。有学者将F1蛋白与煅烧骨复合后植入Wistar大鼠颅骨严重缺损处,6周后发现缺损处明显有新骨生成,经检测骨形态发生蛋白2、骨钙蛋白和骨桥蛋白有明显的增加趋势,还观察到大量血管和炎性细胞的存在,其中骨髓和骨小梁充满了缺损区域[4]。说明此复合物的骨诱导性较高,可显著刺激骨质生长,增加骨缺损愈合率,具有极大的临床应用价值。 2.4 煅烧骨的成骨原理 尽管煅烧骨去除了有机物质,但它留下的天然骨空隙和优异的生物学活性为血运重建和新骨形成提供了有利的空间维持作用[68],与宿主细胞因子或其他材料复合后形成骨磷灰石加速了煅烧骨与宿主骨之间的结合,诱导新骨在煅烧骨支架上形成并矿化,其成骨原理见图4所示:骨损伤刺激机体在骨缺损局部产生和骨改建中相同作用的生长因子、信号分子,与复合材料中所含有的成骨前体细胞、骨诱导因子一起使间充质干细胞、成纤维细胞和骨原细胞增殖、细胞外基质沉积、血管形成,其中骨原细胞将转化为成骨细胞,在受损的骨质表面分泌类骨质形成新骨,经矿化后形成骨基质。整个骨再生的过程中有3个机制相互调控:骨生成、骨诱导和骨传导,主要是通过成骨细胞和破骨细胞的相互作用发生的。因为骨增量材料的降解主要是通过溶解发生的,其中破骨细胞逐渐吸收周围骨质和移植材料,促进成骨细胞聚集并加速其活动形成新骨[69],所以此种形式又称为“爬行替代”的模式。 "
| [1] DE TULLIO I, CAPUTI S, PERFETTI G, et al. A Human Clinical and Histomorphometrical Study on Different Resorbable and Non-Resorbable Bone Substitutes Used in Post-Extractive Sites. Preliminary Results.Materials(Basel).2019;12(15).pii: E2408. doi: 10.3390/ma12152408. [2] TRAINI T, PIATTELLI A, CAPUTI S, et al. Regeneration of human bone using different bone substitute biomaterials.Clin Implant Dent Relat Res.2015;17(1):150-162. [3] XI Y, MIAO X, LI Y, et al. BMP2-mimicking peptide modified with E7 coupling to calcined bovine bone enhanced bone regeneration associating with activation of the Runx2/SP7 signaling axis. 2020; 108(1):80-93.doi:10.1002/jbm.b.34368. [4] SANTOS KOTAKE BG, GONZAGA MG, COUTINHO-NETTO J, et al. Bone repair of critical-sized defects in Wistar rats treated with autogenic, allogenic or xenogenic bone grafts alone or in combination with natural latex fraction F1.Biomed Mater.2018;13(2):025022. [5] STACCHI C, LOMBARDI T, OREGLIA F, et al. Histologic and Histomorphometric Comparison between Sintered Nanohydroxyapatite and Anorganic Bovine Xenograft in Maxillary Sinus Grafting: A Split-Mouth Randomized Controlled Clinical Trial.Biomed Res Int.2017;2017:9489825. [6] RAMIREZ FERNANDEZ MP, GEHRKE SA, PEREZ ALBACETE MARTINEZ C, et al.SEM-EDX Study of the Degradation Process of Two Xenograft Materials Used in Sinus Lift Procedures.Materials (Basel).2017;10(5).pii: E542. doi: 10.3390/ma10050542. [7] WANG W, YEUNG KWK. Bone grafts and biomaterials substitutes for bone defect repair: A review.Bioact Mater.2017;2(4):224-247. [8] BORG TD, MEALEY BL. Histologic healing following tooth extraction with ridge preservation using mineralized versus combined mineralized-demineralized freeze-dried bone allograft: a randomized controlled clinical trial. J Periodontol.2015;86(3):348-355. [9] MCALLISTER BS, HAGHIGHAT K. Bone augmentation techniques.J Periodontol.2007;78(3):377-396. [10] YAMAMURA H, DA SILVA VHP, RUIZ PLM, et al. Physico-chemical characterization and biocompatibility of hydroxyapatite derived from fish waste.J Mech Behav Biomed Mater.2018;80:137-142. [11] PRAKASAM M, LOCS J, SALMA-ANCANE K, et al. Fabrication, Properties and Applications of Dense Hydroxyapatite: A Review.J Funct Biomater.2015;6(4):1099-1140. [12] SZCZEŚ A, YAN Y, CHIBOWSKI E, et al. Properties of natural and synthetic hydroxyapatite and their surface free energy determined by the thin-layer wicking method.ApplSurf Sci.2018;434:1232-1238. [13] LOWE B, VENKATESAN J, ANIL S, et al. Preparation and characterization of chitosan-natural nano hydroxyapatite-fucoidan nanocomposites for bone tissue engineering.Int J Biol Macromol. 2016;93(Pt B):1479-1487. [14] ESMAEILKHANIAN A, SHARIFIANJAZI F, ABOUCHENARI A, et al. Synthesis and Characterization of Natural Nano-hydroxyapatite Derived from Turkey Femur-Bone Waste.Appl Biochem Biotechnol. 2019;189(3):919-932. [15] RAMÍREZ FERNÁNDEZ MP, MAZÓN P, GEHRKE SA, et al. Comparison of Two Xenograft Materials Used in Sinus Lift Procedures: Material Characterization and In Vivo Behavior.Materials (Basel). 2017; 10(6).pii: E623. doi:10.3390/ma10060623. [16] AARTHY S, THENMUHIL D, DHARUNYA G, et al.Exploring the effect of sintering temperature on naturally derived hydroxyapatite for bio-medical applications. J Mater Sci Mater Med.2019;30(2):21. [17] HEIDARI F, BAHROLOLOOM ME, VASHAEE D, et al. In situ preparation of iron oxide nanoparticles in natural hydroxyapatite/chitosan matrix for bone tissue engineering application.Ceram Int.2015;41(2):3094-3100. [18] APALANGYA V, RANGARI V, JEELANI S, et al. Rapid microwave synthesis of needle-liked hydroxyapatite nanoparticles via template directing ball-milled spindle-shaped eggshell particles.Ceram Int. 2018;44(6):7165-7171. [19] SUNIL BR,JAGANNATHAM M.Producing hydroxyapatite from fish bones by heat treatment.Mater Lett.2016;185:411-414. [20] PAUL S, PAL A, CHOUDHURY AR, et al. Effect of trace elements on the sintering effect of fish scale derived hydroxyapatite and its bioactivity. Ceram Int.2017;43(17):15678-15684. [21] FERNANDES HR, GADDAM A, REBELO A, et al. Bioactive Glasses and Glass-Ceramics for Healthcare Applications in Bone Regeneration and Tissue Engineering.Materials (Basel).2018;11(12). pii: E2530.doi: 10.3390/ma11122530. [22] BIGHETTI ACC, CESTARI TM, SANTOS PS, et al. In vitro and in vivo assessment of CaP materials for bone regenerative therapy. The role of multinucleated giant cells/osteoclasts in bone regeneration.J Biomed Mater Res B Appl Biomater.2020;108(1):282-297. [23] TSAI WC, LIAO CJ, WU CT, et al. Clinical result of sintered bovine hydroxyapatite bone substitute: analysis of the interface reaction between tissue and bone substitute.J Orthop Sci.2010;15(2):223-232. [24] FIENITZ T, MOSES O, KLEMM C, et al. Histological and radiological evaluation of sintered and non-sintered deproteinized bovine bone substitute materials in sinus augmentation procedures. A prospective, randomized-controlled, clinical multicenter study.Clin Oral Investig. 2017;21(3):787-794. [25] LONDONO-RESTREPO SM, JERONIMO-CRUZ R, RUBIO-ROSAS E, et al. The effect of cyclic heat treatment on the physicochemical properties of bio hydroxyapatite from bovine bone.J Mater Sci Mater Med. 2018;29(5):52. [26] JOSCHEK S, NIES B, KROTZ R, et al. Chemical and Physicochemical Characterization of Porous Hydroxapatite Ceramics Made of Natural Bone.Biomaterials.2000;21:1645-1658. [27] ZHANG J, BARBIERI D, TEN HOOPEN H, et al.Microporous calcium phosphate ceramics driving osteogenesis through surface architecture.J Biomed Mater Res A.2015;103(3):1188-1199. [28] JIN QM, TAKITA H, KOHGO T, et al. Effects of Geometry of Hydroxyapatite as Cell Substratum in BMP-Induced Ectopic Bone Formation.J Biomed Mater Res.2016;51(2000):491-499. [29] DEWIDAR MM, LIM JK. Properties of solid core and porous surface Ti–6Al–4V implants manufactured by powder metallurgy.J Alloys Compd.2008;454(1-2):442-446. [30] RINCÓN-LÓPEZ JA, HERMANN-MUÑOZ JA, GIRALDO-BETANCUR AL, et al. Synthesis Characterization and In Vitro Study of Synthetic and Bovine Derived Hydroxyapatite Ceramics A Comparison.Materials (Basel).2018;11(3).pii: E333. [31] ZHANG YJ, LU JJ. A Mild and Efficient Biomimetic Synthesis of Rodlike Hydroxyapatite Particles with a High Aspect Ratio Using Polyvinylpyrrolidone As Capping Agent.Cryst Growth Des.2008;8(7): 2101-2107. [32] XU AT, QI WT, LIN MN, et al. The optimization of sintering treatment on bovine-derived bone grafts for bone regeneration: in vitro and in vivo evaluation.J Biomed Mater Res B Appl Biomater. 2020;108(1):272-281. [33] MATÉ SÁNCHEZ DE VAL J, MAZÓN P, PIATTELLI A, et al. Comparison among the physical properties of calcium phosphate-based bone substitutes of natural or synthetic origin.Int J Appl Ceram Technol. 2018;15(4):930-937. [34] RAMIREZ FERNANDEZ MP, GEHRKE SA, MAZON P, et al. Implant Stability of Biological Hydroxyapatites Used in Dentistry. Materials(Basel).2017;10(6).pii: E644. doi: 10.3390/ma10060644.. [35] MATE SANCHEZ DE VAL JE, CALVO-GUIRADO JL, GOMEZ-MORENO G, et al. Influence of hydroxyapatite granule size, porosity, and crystallinity on tissue reaction in vivo. Part B: a comparative study with biphasic synthetic biomaterials.Clin Oral Implants Res.2018;29(11):1077-1084. [36] LONDOÑO-RESTREPO SM, RAMIREZ-GUTIERREZ CF, DEL REAL A, et al. Study of bovine hydroxyapatite obtained by calcination at low heating rates and cooled in furnace air.J Mater Sci. 2016;51(9): 4431-4441. [37] RESMIM CM, DALPASQUALE M, VIELMO NIC, et al. Study of physico-chemical properties and in vitro antimicrobial activity of hydroxyapatites obtained from bone calcination.Prog Biomater. 2019;8(1):1-9. [38] FEITOSA GT, SANTOS MVB, BARRETO HM, et al. Hydroxyapatites Obtained from Different Routes and their Antimicrobial Properties. Mater Sci Forum.2016;869:890-895. [39] LI B, WEBSTER TJ. Bacteria antibiotic resistance: New challenges and opportunities for implant-associated orthopedic infections.J Orthop Res.2018;36(1):22-32. [40] INZANA JA, SCHWARZ EM, KATES SL, et al. Biomaterials approaches to treating implant-associated osteomyelitis.Biomaterials. 2016;81:58-71. [41] ROSETI L, PARISI V, PETRETTA M, et al. Scaffolds for Bone Tissue Engineering: State of the art and new perspectives.Mater Sci Eng C Mater Biol Appl.2017;78:1246-1262. [42] LEE JW, CHU SG, KIM HT, et al. Osteogenesis of Adipose-Derived and Bone Marrow Stem Cells with Polycaprolactone/Tricalcium Phosphate and Three-Dimensional Printing Technology in a Dog Model of Maxillary Bone Defects.Polymers(Basel).2017;9(9).pii:E450. [43] YIN HM, QIAN J, ZHANG J, et al. Engineering Porous Poly(lactic acid) Scaffolds with High Mechanical Performance via a Solid State Extrusion/Porogen Leaching Approach.Polymers (Basel).2016;8(6).pii: E213. doi:10.3390/8060213. [44] YU X, TANG X, GOHIL SV, et al. Biomaterials for Bone Regenerative Engineering.Adv Healthc Mater.2015;4(9):1268-1285. [45] KOHRI M, MIKI K, WAITE DE, et al. In vitro stability of biphasic calcium phosphate ceramics. Biomaterials.1990;14(4):299-304. [46] FRAYSSINET P, TROUILLET JL, ROUQUET N, et al. Osseointegration of macroporous calcium phosphate ceramics having a different chemical composition.Biomaterials.1993;14(6):423-429. [47] PICCIRILLO C, SILVA MF, PULLAR RC, et al. Extraction and characterisation of apatite- and tricalcium phosphate-based materials from cod fish bones.Mater Sci Eng C Mater Biol Appl. 2013;33(1): 103-110. [48] ZHU Q, ABLIKIM Z, CHEN T, et al. The preparation and characterization of HA/β-TCP biphasic ceramics from fish bones. Ceramic Int. 2017;43(15):12213-12220. [49] PAL A, PAUL S, CHOUDHURY AR, et al.Synthesis of hydroxyapatite from Lates calcarifer fish bone for biomedical applications.Mater Lett. 2017;203:89-92. [50] BAKAN F, SEZEN M, GECGIN M, et al. Structural and Chemical Analysis of Hydroxyapatite (HA)-Boron Nitride (BN) Nanocomposites Sintered Under Different Atmospheric Conditions.Microsc Microanal. 2017;23(5):891-899. [51] UNAL S, EKREN N, SENGIL AZ, et al. Synthesis, characterization, and biological properties of composites of hydroxyapatite and hexagonal boron nitride.J Biomed Mater Res B Appl Biomater. 2018; 106(6):2384-2392. [52] COZZA N, MONTE F, BONANI W, et al. Bioactivity and mineralization of natural hydroxyapatite from cuttlefish bone and Bioglass((R)) co-sintered bioceramics.J Tissue Eng Regen Med.2018;12(2): e1131-e1142. [53] MEKA SRK, JAIN S, CHATTERJEE K. Strontium eluting nanofibers augment stem cell osteogenesis for bone tissue regeneration.Colloids Surf B Biointerfaces.2016;146:649-656. [54] KIM SS, SUN PARK M, JEON O, et al. Poly(lactide-co-glycolide)/ hydroxyapatite composite scaffolds for bone tissue engineering. Biomaterials.2006;27(8):1399-1409. [55] CHENG D, LIANG Q, LI Y, et al. Strontium incorporation improves the bone-forming ability of scaffolds derived from porcine bone.Colloids Surf B Biointerfaces.2018;162:279-287. [56] PETTINICCHIO M, TRAINI T, MURMURA G, et al. Histologic and histomorphometric results of three bone graft substitutes after sinus augmentation in humans.Clin Oral Investig.2012;16(1):45-53. [57] RUIXIN L, CHENG X, YINGJIE L, et al. Degradation behavior and compatibility of micro, nanoHA/chitosan scaffolds with interconnected spherical macropores.Int J Biol Macromol.2017;103: 385-394. [58] YUAN H, NING CHEN N, LÜ XY, et al. Experimental study of natural hydroxyapatite_chitosancomposite on reconstructin.J Nanjing Med Univ.2008;22(6):372-375. [59] SHEIKH Z, JAVAID MA, HAMDAN N, et al. Bone Regeneration Using Bone Morphogenetic Proteins and Various Biomaterial Carriers. Materials (Basel).2015;8(4):1778-1816. [60] ZHANG H, WANG F, DING L, et al. A meta analysis of lumbar spinal fusion surgery using bone morphogenetic proteins and autologous iliac crest bone graft.PLoS One.2014;9(6):e97049. [61] WILLIAMS BJ, SMITH JS, FU KM, et al. Does bone morphogenetic protein increase the incidence of perioperative complications in spinal fusion? A comparison of 55,862 cases of spinal fusion with and without bone morphogenetic protein.Spine(Phila Pa 1976). 2011;36(20): 1685-1691. [62] XIAO W, FU H, RAHAMAN MN, et al. Hollow hydroxyapatite microspheres: a novel bioactive and osteoconductive carrier for controlled release of bone morphogenetic protein-2 in bone regeneration. Acta Biomater.2013;9(9):8374-8383. [63] KIEKER CA, GERHART TN, SHELLING SH, et al. Long-Term Healing of Bone Using Recombinant Human Bone Morphogenetic Protein 2.Clin Orthop Relat Res.1995;(318):222-230. [64] FALCIGNO L, D'AURIA G, CALVANESE L, et al. Osteogenic properties of a short BMP-2 chimera peptide.J Pept Sci.2015;21(9):700-709. [65] WU M, CHEN G, LI YP. TGF-β and BMP signaling in osteoblast, skeletal development, and bone formation, homeostasis and disease. one Res.2016;4:16009. [66] DIAS FJ, ISSA JP, COUTINHO-NETTO J, et al. Morphometric and high resolution scanning electron microscopy analysis of low-level laser therapy and latex protein (Hevea brasiliensis) administration following a crush injury of the sciatic nerve in rats.J Neurol Sci. 2015;349(1-2):129-137. [67] MACHADO EG, ISSA JP, FIGUEIREDO FA, et al. A new heterologous fibrin sealant as scaffold to recombinant human bone morphogenetic protein-2 (rhBMP-2) and natural latex proteins for the repair of tibial bone defects.Acta Histochem.2015;117(3):288-296. [68] PATEL PP, BUCKLEY C, TAYLOR BL, et al. Mechanical and biological evaluation of a hydroxyapatite-reinforced scaffold for bone regeneration. J Biomed Mater Res A.2019;107(4):732-741. [69] SIMS A, MARTIN J. Coupling the activities of bone formation and resorption: a multitude of signals within the basic multicellular unit. Bonekey Rep.2014;3:1-10. |
| [1] | Zhang Tongtong, Wang Zhonghua, Wen Jie, Song Yuxin, Liu Lin. Application of three-dimensional printing model in surgical resection and reconstruction of cervical tumor [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1335-1339. |
| [2] | Liang Yan, Zhao Yongfei, Xu Shuai, Zhu Zhenqi, Wang Kaifeng, Liu Haiying, Mao Keya. Imaging evaluation of short-segment fixation and fusion for degenerative lumbar scoliosis assisted by highly selective nerve root block [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1423-1427. |
| [3] | Zeng Yanhua, Hao Yanlei. In vitro culture and purification of Schwann cells: a systematic review [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1135-1141. |
| [4] | Liu Lihua, Sun Wei, Wang Yunting, Gao Fuqiang, Cheng Liming, Li Zirong, Wang Jiangning. Type L1 steroid-induced osteonecrosis of the femoral head through femoral head and neck junction decompression by fenestration: a single-center prospective clinical study [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(6): 906-911. |
| [5] | Hua Haotian, Zhao Wenyu, Zhang Lei, Bai Wenbo, Wang Xinwei. Meta-analysis of clinical efficacy and safety of antibiotic artificial bone in the treatment of chronic osteomyelitis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(6): 970-976. |
| [6] | Zhang Bin, Sun Lihua, Zhang Junhua, Liu Yusan, Cui Caiyun. A modified flap immediate implant is beneficial to soft tissue reconstruction in maxillary aesthetic area [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 707-712. |
| [7] | Xu Dongzi, Zhang Ting, Ouyang Zhaolian. The global competitive situation of cardiac tissue engineering based on patent analysis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 807-812. |
| [8] | Li Chenjie, Lü Linwei, Song Yang, Liu Jingna, Zhang Chunqiu. Measurement and statistical analysis of trabecular morphological parameters of titanium alloy peri-prosthesis under preload [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 516-520. |
| [9] | Wu Zijian, Hu Zhaoduan, Xie Youqiong, Wang Feng, Li Jia, Li Bocun, Cai Guowei, Peng Rui. Three-dimensional printing technology and bone tissue engineering research: literature metrology and visual analysis of research hotspots [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 564-569. |
| [10] | Chang Wenliao, Zhao Jie, Sun Xiaoliang, Wang Kun, Wu Guofeng, Zhou Jian, Li Shuxiang, Sun Han. Material selection, theoretical design and biomimetic function of artificial periosteum [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 600-606. |
| [11] | Liu Fei, Cui Yutao, Liu He. Advantages and problems of local antibiotic delivery system in the treatment of osteomyelitis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 614-620. |
| [12] | Li Xiaozhuang, Duan Hao, Wang Weizhou, Tang Zhihong, Wang Yanghao, He Fei. Application of bone tissue engineering materials in the treatment of bone defect diseases in vivo [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 626-631. |
| [13] | Zhang Zhenkun, Li Zhe, Li Ya, Wang Yingying, Wang Yaping, Zhou Xinkui, Ma Shanshan, Guan Fangxia. Application of alginate based hydrogels/dressings in wound healing: sustained, dynamic and sequential release [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 638-643. |
| [14] | Chen Jiana, Qiu Yanling, Nie Minhai, Liu Xuqian. Tissue engineering scaffolds in repairing oral and maxillofacial soft tissue defects [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 644-650. |
| [15] | He Jie, Chang Qi. Biological reconstruction of large bone defects after resection of malignant tumor of extremities [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(3): 420-425. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||