Chinese Journal of Tissue Engineering Research ›› 2020, Vol. 24 ›› Issue (19): 3010-3016.doi: 10.3969/j.issn.2095-4344.2070
Previous Articles Next Articles
Urine-derived stem cells differentiate into urothelial cells and smooth muscle cells in vitro
Zhu Haizhou1, 2, Zhao Zhankui1, Wang Xinzhe1, Liu Deqian1, Yu Honglian3
- 1Department of Urology, Affiliated Hospital of Jining Medical University, Jining 272029, Shandong Province, China; 2Department of Surgery Pandect, Clinical Medical School, Jining Medical University, Jining 272067, Shandong Province, China; 3Key Laboratory of Collaborative Innovation Center, Jining Medical University, Jining 272029, Shandong Province, China
-
Received:2019-07-08Revised:2019-07-09Accepted:2019-10-09Online:2020-07-08Published:2020-04-08 -
Contact:Zhao Zhankui, MD, PhD, Associate professor, Department of Urology, Affiliated Hospital of Jining Medical University, Jining 272029, Shandong Province, China -
About author:Zhu Haizhou, Master, Professor, Department of Urology, Affiliated Hospital of Jining Medical University, Jining 272029, Shandong Province, China; Department of Surgery Pandect, Clinical Medical School, Jining Medical University, Jining 272067, Shandong Province, China -
Supported by:the National Natural Science Foundation of China, No. 81500517; the Young Teacher Scientific Research Support Fund of Jining Medical College, No. JYFC2018FKJ009 and JYFC2018KJ004
CLC Number:
Cite this article
Zhu Haizhou, Zhao Zhankui, Wang Xinzhe, Liu Deqian, Yu Honglian. Urine-derived stem cells differentiate into urothelial cells and smooth muscle cells in vitro[J]. Chinese Journal of Tissue Engineering Research, 2020, 24(19): 3010-3016.
share this article
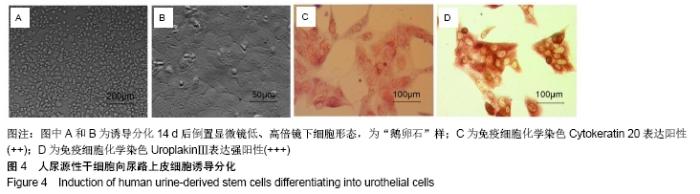
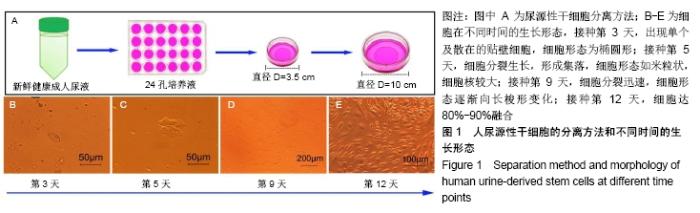
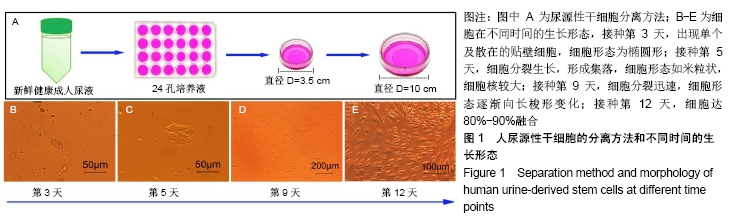
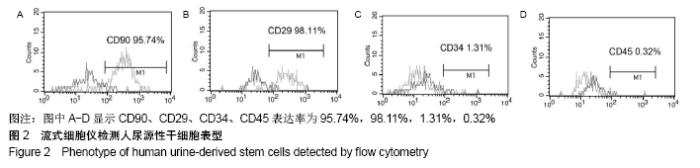
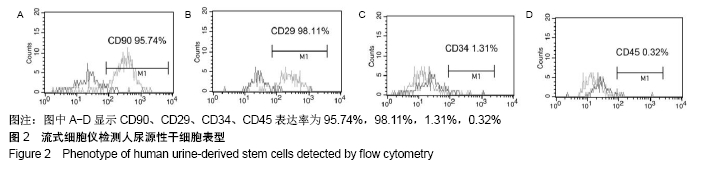
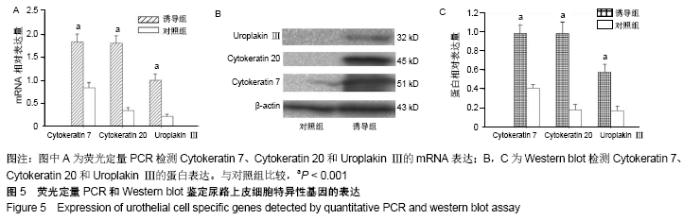
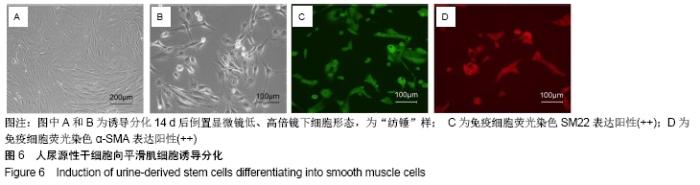
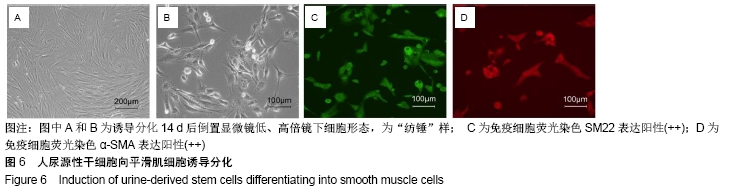
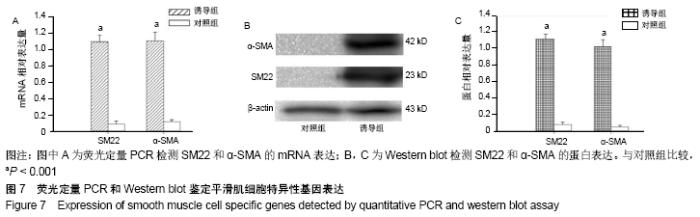
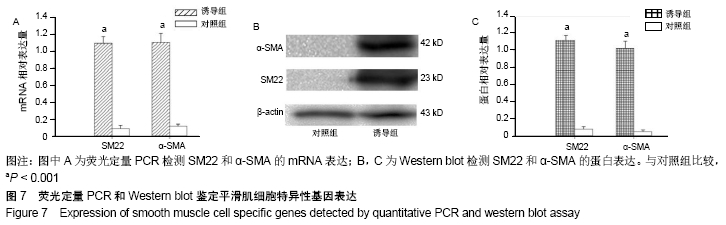
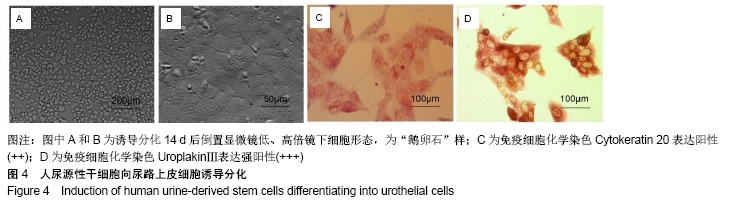
2.4 尿源性干细胞向尿路上皮细胞诱导分化结果 尿源性干细胞经尿路上皮诱导专用培养基诱导分化14 d后,倒置显微镜下可见细胞形态变成“鹅卵石”样,见图4A,B。免疫细胞化学染色可见诱导组Cytokeratin 20表达阳性(++)和Uroplakin Ⅲ蛋白表达强阳性(+++),见图4C,D。荧光定量PCR结果显示诱导组尿路上皮细胞特异性基因Cytokeratin 7、Cytokeratin 20和Uroplakin Ⅲ表达显著高于对照组(P < 0.001),见图5A。Western blot结果显示诱导组Cytokeratin 7、Cytokeratin 20和Uroplakin Ⅲ蛋白表达水平显著高于对照组,见图5B。以上证实尿源性干细胞经体外诱导后可以表达尿路上皮细胞特异性表型蛋白,初步向尿路上皮细胞诱导分化成功。 "
| [1] ROGERS EH, HUNT JA, PEKOVIC-VAUGHAN V. Adult stem cell maintenance and tissue regeneration around the clock: do impaired stem cell clocks drive age-associated tissue degeneration? Biogerontology. 2018;19(6):497-517. [2] BHARADWAJ S, LIU G, SHI Y, et al. Multipotential differentiation of human urine-derived stem cells: potential for therapeutic applications in urology. Stem Cells. 2013;31(9):1840-1856. [3] ZHANG D, WEI G, LI P, et al. Urine-derived stem cells: A novel and versatile progenitor source for cell-based therapy and regenerative medicine. Genes Dis. 2014;1(1):8-17. [4] GAO P, JIANG D, LIU W, et al. Urine-derived Stem Cells, A New Source of Seed Cells for Tissue Engineering. Curr Stem Cell Res Ther. 2016;11(7):547-553. [5] PAVATHUPARAMBIL ABDUL MANAPH N, AL-HAWWAS M, BOBROVSKAYA L, et al. Urine-derived cells for human cell therapy. Stem Cell Res Ther. 2018;9(1):189. [6] BODIN A, BHARADWAJ S, WU S, et al. Tissue-engineered conduit using urine-derived stem cells seeded bacterial cellulose polymer in urinary reconstruction and diversion. Biomaterials. 2010;31(34): 8889-8901. [7] WU S, LIU Y, BHARADWAJ S, et al. Human urine-derived stem cells seeded in a modified 3D porous small intestinal submucosa scaffold for urethral tissue engineering. Biomaterials. 2011;32(5):1317-1326. [8] LANG R, LIU G, SHI Y, et al. Self-renewal and differentiation capacity of urine-derived stem cells after urine preservation for 24 hours. PLoS One. 2013;8(1):e53980. [9] ATALA A, BAUER SB, SOKER S, et al. Tissue-engineered autologous bladders for patients needing cystoplasty. Lancet. 2006;367(9518): 1241-1246. [10] RAYA-RIVERA A, ESQUILIANO DR, YOO JJ, et al. Tissue-engineered autologous urethras for patients who need reconstruction: an observational study. Lancet. 2011;377(9772):1175-1182. [11] DAVIS NF, CUNNANE EM, QUINLAN MR, et al. Biomaterials and Regenerative Medicine in Urology. Adv Exp Med Biol. 2018;1107: 189-198. [12] XIE M, XU Y, SONG L, et al. Tissue-engineered buccal mucosa using silk fibroin matrices for urethral reconstruction in a canine model. J Surg Res. 2014;188(1):1-7. [13] LIAO W, YANG S, SONG C, et al. Tissue-engineered tubular graft for urinary diversion after radical cystectomy in rabbits. J Surg Res. 2013;182(2):185-191. [14] MENG L, LIAO W, YANG S, et al. Tissue-engineered tubular substitutions for urinary diversion in a rabbit model. Exp Biol Med (Maywood). 2016;241(2):147-156. [15] LIU G, WU R, YANG B, et al. Human Urine-Derived Stem Cell Differentiation to Endothelial Cells with Barrier Function and Nitric Oxide Production. Stem Cells Transl Med. 2018;7(9):686-698. [16] CHOI JY, CHUN SY, HA YS, et al. Potency of Human Urine-Derived Stem Cells for Renal Lineage Differentiation. Tissue Eng Regen Med. 2017;14(6):775-785. [17] GUAN J, ZHANG J, GUO S, et al. Human urine-derived stem cells can be induced into osteogenic lineage by silicate bioceramics via activation of the Wnt/β-catenin signaling pathway. Biomaterials. 2015;55:1-11. [18] KIM JY, CHUN SY, PARK JS, et al. Laminin and Platelet-Derived Growth Factor-BB Promote Neuronal Differentiation of Human Urine-Derived Stem Cells. Tissue Eng Regen Med. 2017;15(2): 195-209. [19] KANG HS, CHOI SH, KIM BS, et al. Advanced Properties of Urine Derived Stem Cells Compared to Adipose Tissue Derived Stem Cells in Terms of Cell Proliferation, Immune Modulation and Multi Differentiation. J Korean Med Sci. 2015;30(12):1764-1776. [20] MISHRA PJ, BANERJEE D. Activation and Differentiation of Mesenchymal Stem Cells. Methods Mol Biol. 2017;1554:201-209. [21] LV FJ, TUAN RS, Cheung KM, et al. Concise review: the surface markers and identity of human mesenchymal stem cells. Stem Cells. 2014;32(6):1408-1419. [22] HE Q, YE Z, ZHOU Y, et al. Comparative study of mesenchymal stem cells from rat bone marrow and adipose tissue. Turk J Biol. 2018;42: 477-489. [23] ESPINOZA JL, ELBADRY MI, CHONABAYASHI K, et al. Hematopoiesis by iPSC-derived hematopoietic stem cells of aplastic anemia that escape cytotoxic T-cell attack. Blood Adv. 2018;2(4): 390-400. [24] MIGUELES RP, SHAW L, RODRIGUES NP, et al. Transcriptional regulation of Hhex in hematopoiesis and hematopoietic stem cell ontogeny. Dev Biol. 2017;424(2):236-245. [25] RAMOS-VARA JA, MILLER MA, BOUCHER M, et al. Immunohistochemical detection of uroplakin III, cytokeratin 7, and cytokeratin 20 in canine urothelial tumors. Vet Pathol. 2003;40(1): 55-62. [26] SLEDGE DG, PATRICK DJ, FITZGERALD SD, et al. Differences in expression of uroplakin III, cytokeratin 7, and cyclooxygenase-2 in canine proliferative urothelial lesions of the urinary bladder. Vet Pathol. 2015;52(1):74-82. [27] CHU P, WU E, WEISS LM. Cytokeratin 7 and cytokeratin 20 expression in epithelial neoplasms: a survey of 435 cases. Mod Pathol. 2000;13(9):962-972. [28] WU XR, KONG XP, PELLICER A, et al. Uroplakins in urothelial biology, function, and disease. Kidney Int. 2009;75(11):1153-1165. [29] MATUSZEWSKI MA, TUPIKOWSKI K, DOŁOWY Ł, et al. Uroplakins and their potential applications in urology. Cent European J Urol. 2016;69(3):252-257. [30] LEE G. Uroplakins in the lower urinary tract. Int Neurourol J. 2011; 15(1):4-12. [31] GUHA A, DESHPANDE A, JAIN A, et al. Uroplakin 3a+ Cells Are a Distinctive Population of Epithelial Progenitors that Contribute to Airway Maintenance and Post-injury Repair. Cell Rep. 2017;19(2): 246-254. [32] OSBORN SL, KURZROCK EA. In Vitro Differentiation and Propagation of Urothelium from Pluripotent Stem Cell Lines. Methods Mol Biol. 2018;1655:137-144. [33] ZUPANČIČ D, MRAK POLJŠAK K, KREFT ME. Co-culturing porcine normal urothelial cells, urinary bladder fibroblasts and smooth muscle cells for tissue engineering research. Cell Biol Int. 2018;42(4):411-424. [34] BOUMELHEM BB, FRASER ST, ASSINDER SJ. Differentiation of Urothelium from Mouse Embryonic Stem Cells in Chemically Defined Conditions. Methods Mol Biol. 2019;2029:103-115. [35] SUZUKI K, KOYANAGI-AOI M, UEHARA K, et al. Directed differentiation of human induced pluripotent stem cells into mature stratified bladder urothelium. Sci Rep. 2019;9(1):10506. [36] ZHANG Y, MCNEILL E, TIAN H, et al. Urine derived cells are a potential source for urological tissue reconstruction. J Urol. 2008; 180(5):2226-2233. [37] BHARADWAJ S, LIU G, SHI Y, et al. Characterization of urine-derived stem cells obtained from upper urinary tract for use in cell-based urological tissue engineering.Tissue Eng Part A. 2011;17(15-16): 2123-2132. [38] GUO X, CHEN SY. Transforming growth factor-β and smooth muscle differentiation. World J Biol Chem. 2012;3(3):41-52. [39] SINHA S, HOOFNAGLE MH, KINGSTON PA, et al. Transforming growth factor-beta1 signaling contributes to development of smooth muscle cells from embryonic stem cells. Am J Physiol Cell Physiol. 2004;287(6):C1560-1568. [40] XIE WB, LI Z, MIANO JM, et al. Smad3-mediated myocardin silencing: a novel mechanism governing the initiation of smooth muscle differentiation. J Biol Chem. 2011;286(17):15050-15057. [41] ALIMPERTI S, YOU H, GEORGE T, et al. Cadherin-11 regulates both mesenchymal stem cell differentiation into smooth muscle cells and the development of contractile function in vivo. J Cell Sci. 2014;127(Pt 12):2627-2638. [42] HAN Y, LI N, TIAN X, et al. Endogenous transforming growth factor (TGF) beta1 promotes differentiation of smooth muscle cells from embryonic stem cells: stable plasmid-based siRNA silencing of TGF beta1 gene expression. J Physiol Sci. 2010;60(1):35-41. |
| [1] | Li Cai, Zhao Ting, Tan Ge, Zheng Yulin, Zhang Ruonan, Wu Yan, Tang Junming. Platelet-derived growth factor-BB promotes proliferation, differentiation and migration of skeletal muscle myoblast [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1050-1055. |
| [2] | Wang Feng, Zhou Liyu, Saijilafu, Qi Shibin, Ma Yanxia, Wei Shanwen. CaMKII-Smad1 promotes axonal regeneration of peripheral nerves [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1064-1068. |
| [3] | Zhou Wu, Wang Binping, Wang Yawen, Cheng Yanan, Huang Xieshan. Transforming growth factor beta combined with bone morphogenetic protein-2 induces the proliferation and differentiation of mouse MC3T3-E1 cells [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(23): 3630-3635. |
| [4] | Yang Fan, Liu Baoyi, Liu Jiahe, Yang Jiahui, Qin Kairong, Zhao Dewei. Morphological characteristics of in vitro cultured articular chondrocytes from Sprague-Dawley rats at passage number from 0 to 3 [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(14): 2161-2165. |
| [5] | Yan Nan, Si Xiaofeng, Zeng Liang, Tian Wei, Shan Guangdong, Xiong Lishuo, Yang Weijie, Wang Zhengdong. Nerve growth factor interferes with proliferation and alpha-actin expression of skeletal muscle satellite cells in rats [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(13): 2030-2035. |
| [6] | Hu Sheng, Yuan Haiyan, Hu Meng, Jin Shanhu. Effects of moderate treadmill exercise on alpha-smooth muscle actin and type IV collagen in the liver of type 2 diabetic rats [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(11): 1723-1727. |
| [7] | Chen Qiang, Zhuo Hongwu, Xia Tian, Ye Zhewei . Toxic effects of different-concentration isoniazid on newborn rat osteoblasts in vitro [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(8): 1162-1167. |
| [8] | Xu Guofeng, Li Xuebin, Tang Yifan, Zhao Yin, Zhou Shengyuan, Chen Xiongsheng, Jia Lianshun. The role of autophagy in ossification of the human ligamentum flavum [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(8): 1174-1181. |
| [9] |
Wang Tiantian, Wang Jianzhong.
Application and prospect of bone marrow mesenchymal stem cells in the
treatment of early femoral head necrosis |
| [10] | Zhang Wen, Lei Kun, Gao Lei, Li Kuanxin. Neuronal differentiation of rat bone marrow mesenchymal stem cells via lentivirus-mediated bone morphogenetic protein 7 transfection [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(7): 985-990. |
| [11] | Qin Xinyu, Zhang Yan, Zhang Ningkun, Gao Lianru, Cheng Tao, Wang Ze, Tong Shanshan, Chen Yu. Elabela promotes differentiation of Wharton’s jelly-derived mesenchymal stem cells into cardiomyocyte-like cells [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(7): 1046-1051. |
| [12] | Zhang Peigen, Heng Xiaolai, Xie Di, Wang Jin, Ma Jinglin, Kang Xuewen. Electrical stimulation combined with neurotrophin 3 promotes proliferation and differentiation of endogenous neural stem cells after spinal cord injury in rats [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(7): 1076-1082. |
| [13] | Huang Cheng, Liu Yuanbing, Dai Yongping, Wang Liangliang, Cui Yihua, Yang Jiandong. Transplantation of bone marrow mesenchymal stem cells overexpressing glial cell line derived neurotrophic factor gene for spinal cord injury [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(7): 1037-1045. |
| [14] | Huang Hui, Dai Yao, Li Yongsheng, Chen Wei, Tang Fang, Huang Yuting, Zhou Zheng, Liu Hairong. Application and research of non-coding RNA in bone tissue engineering with cells and scaffold [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(4): 596-605. |
| [15] | Tang Jingfeng, Zhang Jun, You Qi, Liu Yi. The role and mechanism of graphene and its derivatives-related composites in cartilage repair [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(4): 619-624. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||