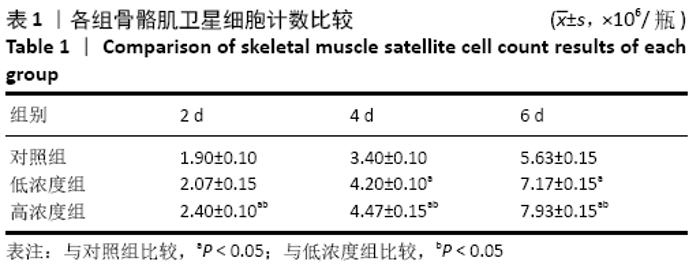
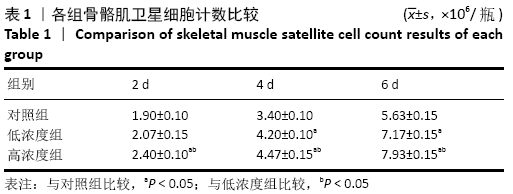
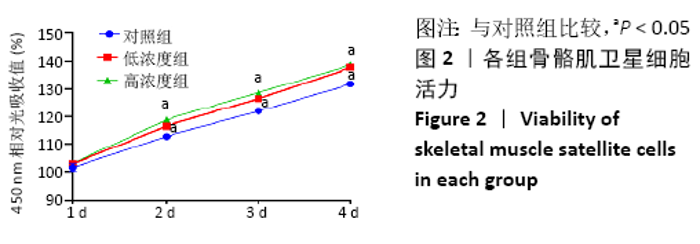
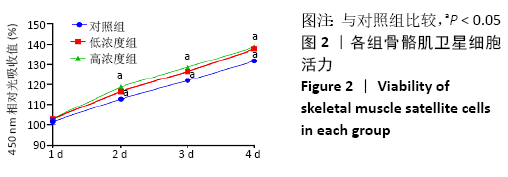
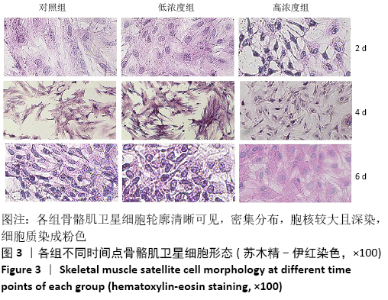
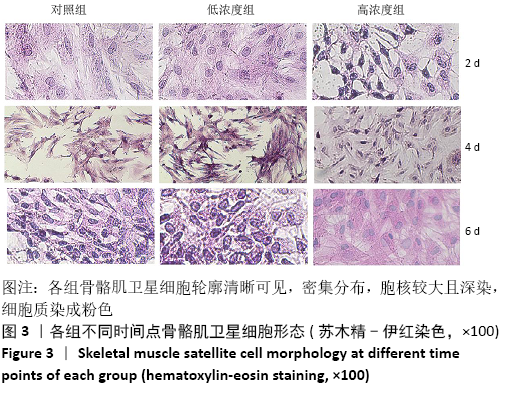
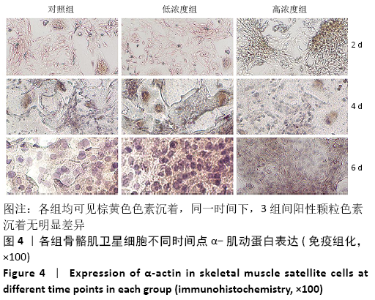
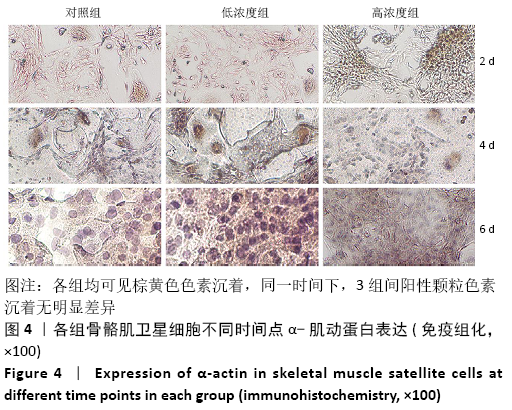
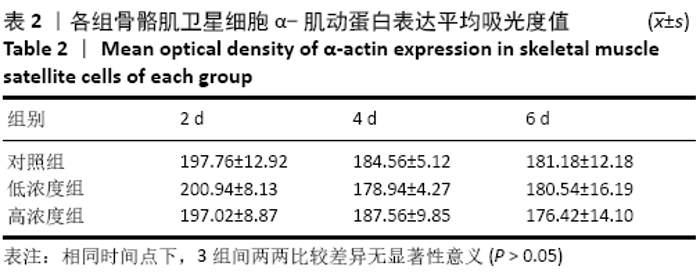
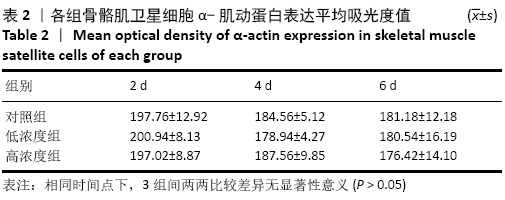
Chinese Journal of Tissue Engineering Research ›› 2021, Vol. 25 ›› Issue (13): 2030-2035.doi: 10.3969/j.issn.2095-4344.3513
Previous Articles Next Articles
Nerve growth factor interferes with proliferation and alpha-actin expression of skeletal muscle satellite cells in rats
Yan Nan, Si Xiaofeng, Zeng Liang, Tian Wei, Shan Guangdong, Xiong Lishuo, Yang Weijie, Wang Zhengdong#br#
- Shenyang Medical College, Shenyang 110034, Liaoning Province, China
-
Received:2020-05-28Revised:2020-05-29Accepted:2020-06-29Online:2021-05-08Published:2020-12-28 -
Contact:Wang Zhengdong, MD, Associate professor, Shenyang Medical College, Shenyang 110034, Liaoning Province, China -
About author:Yan Nan, MD, Associate professor, Shenyang Medical College, Shenyang 110034, Liaoning Province, China Si Xiaofeng, Physician, Shenyang Medical College, Shenyang 110034, Liaoning Province, China -
Supported by:the Science and Technology Program of Liaoning Province, No. 2017225060 (to WZD)
CLC Number:
Cite this article
Yan Nan, Si Xiaofeng, Zeng Liang, Tian Wei, Shan Guangdong, Xiong Lishuo, Yang Weijie, Wang Zhengdong. Nerve growth factor interferes with proliferation and alpha-actin expression of skeletal muscle satellite cells in rats[J]. Chinese Journal of Tissue Engineering Research, 2021, 25(13): 2030-2035.
share this article
| [1] WANG Y, XIAO X, WANG L. In vitro characterization of goat skeletal muscle satellite cells. Anim Biotechnol. 2020;31(2):115-121. [2] ZHAO Y, CHEN M, LIAN D, et al. Non-Coding RNA Regulates the Myogenesis of Skeletal Muscle Satellite Cells, Injury Repair and Diseases. Cells. 2019;8(9):988. [3] MUIR AR, KANJI AH, ALLBROOK D. The structure of the satellite cells in skeletal muscle. J Anat. 1965;99:435-444. [4] MANETTI M, TANI A, ROSA I, et al. Morphological evidence for telocytes as stromal cells supporting satellite cell activation in eccentric contraction-induced skeletal muscle injury. Sci Rep. 2019;9(1):14515. [5] WANG L, ZHU Y, LIU X, et al. Glycogen synthase kinase 3beta (GSK3beta) regulates the expression of MyHC2a in goat skeletal muscle satellite cells (SMSCs). Anim Sci J. 2019;90(8):1042-1049. [6] HAN S, CUI C, WANG Y, et al. FHL3 negatively regulates the differentiation of skeletal muscle satellite cells in chicken. Biotech. 2019;9(6):206. [7] FORCINA L, MIANO C, PELOSI L, et al. An Overview about the Biology of Skeletal Muscle Satellite Cells. Curr Genomics. 2019;20(1):24-37. [8] KONING M, WERKER PM, VAN DER SCHAFT DW, et al. MicroRNA-1 and microRNA-206 improve differentiation potential of human satellite cells: a novel approach for tissue engineering of skeletal muscle. Tissue Eng Part A. 2012;18(9-10):889-898. [9] 吴森.牛原代肌肉卫星细胞和前体脂肪细胞不同混合比例共培养细胞活性的比较[J].青海畜牧兽医杂志,2019,49(2):14-20,50. [10] 吴森,孙永刚.血清浓度对共培养条件下牛原代肌肉卫星细胞及前体脂肪细胞活性影响[J].青海畜牧兽医杂志,2019,49(4):13-21. [11] SONG T, SADAYAPPAN S. Featured characteristics and pivotal roles of satellite cells in skeletal muscle regeneration. J Muscle Res Cell Motil. 2019. doi:10.1007/s10974-019-09553-7. [12] UMNOVA MM, SEENE TP. The effect of increased functional load on the activation of satellite cells in the skeletal muscle of adult rats. Int J Sports Med. 1991;12(5):501-504. [13] LI L, CHEN Y, NIE L, et al. MyoD-induced circular RNA CDR1as promotes myogenic differentiation of skeletal muscle satellite cells. Biochim Biophys Acta Gene Regul Mech. 2019;1862(8):807-821. [14] HUANG ZY, LI L, LI QQ, et al. The effect of lncRNA TCONS_00815878 on differentiation of porcine skeletal muscle satellite cells. Yi Chuan. 2019;41(12):1119-1128. [15] ZOU JL, SUN JH, QIU S, et al. Spatial distribution affects the role of CSPGs in nerve regeneration via the actin filament-mediated pathway. Exp Neurol. 2018;307:37-44. [16] MA F, XU F, LI R, et al. Sustained delivery of glial cell-derived neurotrophic factors in collagen conduits for facial nerve regeneration. Acta Biomater. 2018;69:146-155. [17] CAMERINO C, CONTE E, CALOIERO R, et al. Evaluation of Short and Long Term Cold Stress Challenge of Nerve Grow Factor, Brain-Derived Neurotrophic Factor, Osteocalcin and Oxytocin mRNA Expression in BAT, Brain, Bone and Reproductive Tissue of Male Mice Using Real-Time PCR and Linear Correlation Analysis. Front Physiol. 2017;8:1101. [18] JIAO G, PAN Y, WANG C, et al. A bridging SF/Alg composite scaffold loaded NGF for spinal cord injury repair. Mater Sci Eng C Mater Biol Appl. 2017;76:81-87. [19] REN Y, WU H, MA Y, et al. Potential of adipose-derived mesenchymal stem cells and skeletal muscle-derived satellite cells for somatic cell nuclear transfer mediated transgenesis in Arbas Cashmere goats. PLoS One. 2014;9(4):93583. [20] LIU Y, CHEN S, LI W, et al. Isolation and characterization of primary skeletal muscle satellite cells from rats. Toxicol Mech Methods. 2012; 22(9):721-725. [21] 孙剑瑞,殷德涛,周金桥.神经生长因子对神经干细胞增殖、分化和凋亡的影响[J].中华实验外科杂志,2011,28(8):1350-1352. [22] 王雪峰.神经生长因子和钙拮抗剂联合使用对周围神经损伤再生的影响[D].长春:吉林大学,2012. [23] 代阳,王轶敏,刘新峰,等.小鼠骨骼肌卫星细胞的分离培养和鉴定[J].天津农学院学报,2014,21(1):1-4. [24] GULATI AK, REDDI AH, ZALEWSKI AA. Changes in the basement membrane zone components during skeletal muscle fiber degeneration and regeneration. J Cell Biol. 1983;97(4):957-962. [25] 柯棠山,董进,廖娟,等.兔骨骼肌卫星细胞的分离、培养及成肌特性[J].遵义医学院学报,2015,38(2):150-153,159. [26] 冯永强,李峥,褚万立,等.大鼠骨骼肌卫星细胞的原代培养鉴定和体外分化特点[J].中华医学杂志,2016,96(12):971-974. [27] 陈玉佩,王淑艳,刘通,等.大鼠腰多裂肌卫星细胞的分离培养与鉴定[J].细胞与分子免疫学杂志,2018, 34(8):714-717. [28] 刘通,于佳妮,陈玉佩,等.单根肌纤维法分离培养多裂肌卫星细胞及鉴定[J].中国组织工程研究,2018,22(25):4065-4070. [29] CACIALLI P, LUCINI C. Adult neurogenesis and regeneration in zebrafish brain: are the neurotrophins involved in? Neural Regen Res. 2019;14(12):2067-2068. [30] CHO HK, KIM W, LEE KY, et al. Beta-nerve growth factor gene therapy alleviates pyridoxine-induced neuropathic damage by increasing doublecortin and tyrosine kinase A in the dorsal root ganglion. Neural Regen Res. 2020;15(1):162-168. [31] SYVERUD BC, VANDUSEN KW, LARKIN LM. Effects of Dexamethasone on Satellite Cells and Tissue Engineered Skeletal Muscle Units. Tissue Eng Part A. 2016;22(5-6):480-489. [32] CHEN X, ZHU W. A Mathematical Model of Regenerative Axon Growing along Glial Scar after Spinal Cord Injury. Comput Math Methods Med. 2016;3030454. [33] TRAVAGLIA A, PIETROPAOLO A, DI MARTINO R, et al. A small linear peptide encompassing the NGF N-terminus partly mimics the biological activities of the entire neurotrophin in PC12 cells. ACS Chem Neurosci. 2015;6(8):1379-1392. [34] DUMONT NA, RUDNICKI MA. Characterizing Satellite Cells and Myogenic Progenitors During Skeletal Muscle Regeneration. Methods Mol Biol.2017;1560:179-188. [35] ZHAO J, SHEN X, CAO X, et al. HDAC4 Regulates the Proliferation, Differentiation and Apoptosis of Chicken Skeletal Muscle Satellite Cells. Animals (Basel). 2020;10(1):84. [36] FRANCO I, FERNANDEZ-GONZALO R, VRTACNIK P,et al. Healthy skeletal muscle aging: The role of satellite cells, somatic mutations and exercise. Int Rev Cell Mol Biol. 2019;346:157-200. [37] PAYNE S C, BELLEVILLE P J, KEAST J R. Regeneration of sensory but not motor axons following visceral nerve injury.Exp Neurol. 2015;266: 127-142. [38] SHI Q, GAO W, HAN X, et al. Collagen scaffolds modified with collagen-binding bFGF promotes the neural regeneration in a rat hemisected spinal cord injury model. Sci China Life Sci. 2014;57(2):232-40. [39] 陈立.大鼠肌卫星细胞体外培养及移植延缓失神经骨骼肌萎缩的实验研究[D].上海:复旦大学,2012. [40] 陈静静,沈志森,巩长凤,等.骨骼肌卫星细胞的培养及其在骨骼肌组织工程研究中的应用[J].中国生物医学工程学报,2011,30(6): 955-960. [41] ZHANG Y, GAO F, WU D, et al. Lentiviral mediated expression of a NGF-soluble Nogo receptor 1 fusion protein promotes axonal regeneration. Neurobiol Dis. 2013;58:270-280. [42] ZUO E, ZHANG C, MAO J, et al. 2,5-Hexanedione mediates neuronal apoptosis through suppression of NGF via PI3K/Akt signaling in the rat sciatic nerve. Biosci Rep. 2019;39(2):BSR20181122. [43] SHE Y, DENG H, CAI H, et al. Regulation of the expression of key signalling molecules in mTOR pathway of skeletal muscle satellite cells in neonatal chicks: Effects of leucine and glycine-leucine peptide. J Anim Physiol Anim Nutr (Berl). 2019;103(3):786-90. [44] LI S, LIU D, FU Y, et al. Podocan Promotes Differentiation of Bovine Skeletal Muscle Satellite Cells by Regulating the Wnt4-beta-Catenin Signaling Pathway. Front Physiol. 2019;10:1010. |
| [1] | Xu Feng, Kang Hui, Wei Tanjun, Xi Jintao. Biomechanical analysis of different fixation methods of pedicle screws for thoracolumbar fracture [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1313-1317. |
| [2] | Jiang Yong, Luo Yi, Ding Yongli, Zhou Yong, Min Li, Tang Fan, Zhang Wenli, Duan Hong, Tu Chongqi. Von Mises stress on the influence of pelvic stability by precise sacral resection and clinical validation [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1318-1323. |
| [3] | Zhang Tongtong, Wang Zhonghua, Wen Jie, Song Yuxin, Liu Lin. Application of three-dimensional printing model in surgical resection and reconstruction of cervical tumor [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1335-1339. |
| [4] | Zhang Yu, Tian Shaoqi, Zeng Guobo, Hu Chuan. Risk factors for myocardial infarction following primary total joint arthroplasty [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1340-1345. |
| [5] | Wei Wei, Li Jian, Huang Linhai, Lan Mindong, Lu Xianwei, Huang Shaodong. Factors affecting fall fear in the first movement of elderly patients after total knee or hip arthroplasty [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1351-1355. |
| [6] | Wang Jinjun, Deng Zengfa, Liu Kang, He Zhiyong, Yu Xinping, Liang Jianji, Li Chen, Guo Zhouyang. Hemostatic effect and safety of intravenous drip of tranexamic acid combined with topical application of cocktail containing tranexamic acid in total knee arthroplasty [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1356-1361. |
| [7] | Xiao Guoqing, Liu Xuanze, Yan Yuhao, Zhong Xihong. Influencing factors of knee flexion limitation after total knee arthroplasty with posterior stabilized prostheses [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1362-1367. |
| [8] | Huang Zexiao, Yang Mei, Lin Shiwei, He Heyu. Correlation between the level of serum n-3 polyunsaturated fatty acids and quadriceps weakness in the early stage after total knee arthroplasty [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1375-1380. |
| [9] | Zhang Chong, Liu Zhiang, Yao Shuaihui, Gao Junsheng, Jiang Yan, Zhang Lu. Safety and effectiveness of topical application of tranexamic acid to reduce drainage of elderly femoral neck fractures after total hip arthroplasty [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1381-1386. |
| [10] | Wang Haiying, Lü Bing, Li Hui, Wang Shunyi. Posterior lumbar interbody fusion for degenerative lumbar spondylolisthesis: prediction of functional prognosis of patients based on spinopelvic parameters [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1393-1397. |
| [11] | Lü Zhen, Bai Jinzhu. A prospective study on the application of staged lumbar motion chain rehabilitation based on McKenzie’s technique after lumbar percutaneous transforaminal endoscopic discectomy [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1398-1403. |
| [12] | Chen Xinmin, Li Wenbiao, Xiong Kaikai, Xiong Xiaoyan, Zheng Liqin, Li Musheng, Zheng Yongze, Lin Ziling. Type A3.3 femoral intertrochanteric fracture with augmented proximal femoral nail anti-rotation in the elderly: finite element analysis of the optimal amount of bone cement [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1404-1409. |
| [13] | Du Xiupeng, Yang Zhaohui. Effect of degree of initial deformity of impacted femoral neck fractures under 65 years of age on femoral neck shortening [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1410-1416. |
| [14] | Zhang Shangpu, Ju Xiaodong, Song Hengyi, Dong Zhi, Wang Chen, Sun Guodong. Arthroscopic suture bridge technique with suture anchor in the treatment of acromioclavicular dislocation [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1417-1422. |
| [15] | Liang Yan, Zhao Yongfei, Xu Shuai, Zhu Zhenqi, Wang Kaifeng, Liu Haiying, Mao Keya. Imaging evaluation of short-segment fixation and fusion for degenerative lumbar scoliosis assisted by highly selective nerve root block [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1423-1427. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||