Chinese Journal of Tissue Engineering Research ›› 2020, Vol. 24 ›› Issue (4): 596-605.doi: 10.3969/j.issn.2095-4344.1930
Previous Articles Next Articles
Application and research of non-coding RNA in bone tissue engineering with cells and scaffold
Huang Hui, Dai Yao, Li Yongsheng, Chen Wei, Tang Fang, Huang Yuting, Zhou Zheng, Liu Hairong
- Hunan University, Changsha 410082, Hunan Province, China
-
Received:2019-04-17Revised:2019-04-22Accepted:2019-06-22Online:2020-02-08Published:2020-01-07 -
Contact:Zhou Zheng, Associate professor, Hunan University, Changsha 410082, Hunan Province, China Liu Hairong, Associate professor, Hunan University, Changsha 410082, Hunan Province, China -
About author:Huang Hui, Master candidate, Hunan University, Changsha 410082, Hunan Province, China -
Supported by:the National Key Research & Development Program of China, No. 2018YFC1105800; the National Natural Science Foundation of China, No. 31520103905
CLC Number:
Cite this article
Huang Hui, Dai Yao, Li Yongsheng, Chen Wei, Tang Fang, Huang Yuting, Zhou Zheng, Liu Hairong. Application and research of non-coding RNA in bone tissue engineering with cells and scaffold[J]. Chinese Journal of Tissue Engineering Research, 2020, 24(4): 596-605.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
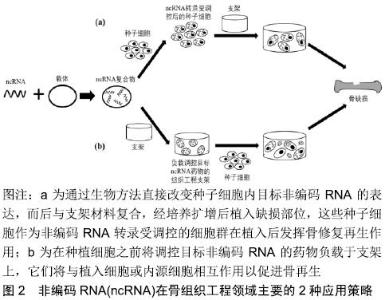
2.1 在骨组织工程中作为生物活性因子的非编码RNA 2.1.1 生命活动中非编码RNA的调控作用 非编码RNA是一类不具有编码蛋白功能的RNA分子,最初被认为是转录的“噪音”,随着基因组技术的快速发展,大量研究表明非编码RNA广泛参与各种重要生命活动,如细胞增殖、分化和凋亡等过程,是生命过程的重要调节因子。非编码RNA根据长度可分为短链非编码RNA (<200 nt)和lncRNA(≥200 nt),其中短链非编码RNA主要包括miRNA和siRNA等。miRNA是一类长度为18-24个核苷酸的内源性小的单链非编码RNA,单个miRNA可以调节数百个mRNA,并且每个mRNA可以被不同的miRNA调节,从而在生命活动中产生复杂的调节网 络[7],miRNA的主要作用机制是通过完全或部分地与位于靶标mRNA的3'UTR中的互补位点结合来减弱其靶基因的表达,导致mRNA降解或翻译抑制,进而调控广泛的细胞功能,包括增殖、迁移、分化和凋亡等。siRNA是20 bp左右的短双链非编码RNA,能够通过RNA干扰(RNAi)的机制来调节基因的表达,RNAi的生物学机制是双链RNA(dsRNA)通过靶向互补mRNA进行降解而诱导基因沉默。相比于miRNA具有多个mRNA靶标,siRNA仅具有一个高度特异性的靶标。lncRNA大于200个核苷酸,甚至可能包含数千个核苷酸。lncRNA凭借其复杂结构以多种方式与DNA、RNA或蛋白质相互作用,lncRNA可通过染色质修饰、转录因子结合和有效的内源性机制等调节机制发挥功能[8]。 2.1.2 非编码RNA与骨形成 干细胞的成骨分化是骨形成的前提,受多种转录因子和信号通路的调控。转录因子如Runt相关转录因子2(Runx2)、成骨细胞特异性转录因子Osterix(Osx)和Smad家族成员蛋白(Smads)等与成骨分化密切相关。其中,Runx2作为成骨早期阶段负责成骨分化的关键转录因子,参与了骨形态发生蛋白、转化生长因子β和Wnt/β-catenin 等多条通路介导的成骨分化过程。同样,Smads可以通过参与转化生长因子β和骨形态发生蛋白信号通路调控成骨分化过程[9]。而Osx作为Runx2下游的一个重要转录因子,受Runx2的调控,影响成骨细胞的成熟。 信号通路如Wnt/β-catenin、骨形态发生蛋白、转化生长因子和MAPK等参与成骨分化信号传导,其中Wnt/β-catenin信号通路是成骨分化中的一条重要信号通路,主要是胞外Wnt蛋白与膜蛋白受体结合,形成二聚体促使β-catenin在胞内积累,而后β-catenin进入细胞核与T细胞转录因子、淋巴增强因子结合形成复合体,激活下游Runx2和Osx等转录因子,进而发挥对成骨分化的调控作用[10]。而骨形态发生蛋白介导的成骨分化途径为:首先骨形态发生蛋白与跨膜受体结合,随后通过Smad或MAPK信号通路传递信号,磷酸化Runx2和Osx等下游转录因子而调控成骨分化过程。MAPK信号通路可以通过保守的三级酶促级联反应激活特定转录因子,从而调控相关基因表达,进而在干细胞成骨分化过程中发挥重要的作用,其中研究最广泛的3条通路为细胞外信号调节激酶、c-Jun氨基末端激酶和p38 MAPK。 近年来,涉及骨相关疾病的非编码RNA的分子机制已被大量研究,越来越多的研究表明非编码RNA是维持骨稳态的重要调控因子,其在干细胞分化中发挥关键作用。已证实许多非编码RNA是参与调节复杂的成骨分化过程中的信号传导网络节点,其用作骨组织工程生物活性因子的研究也日益深入。 miRNA:研究表明越来越多的miRNA通过调控其靶基因的表达,影响各信号通路上游或下游调控因子的表达水平,进而影响成骨分化过程。Runx2受到许多miRNA的直接或间接调控,包括miR-221[11]、miR-467g[12]、miRNA-133a-5p等[13]。Osx的表达可被一些miRNA下调,进而抑制成骨分化过程,如miR-145和miR-143等[14]。 另外,miRNA 的差异性表达对成骨分化相关信号通路起着重要调控作用。DKK1和SFRP1是WNT信号通路典型的强效拮抗因子,miR-433-3p和miR-542-3p分别通过靶向下调DKK1和SFRP1的表达水平[15-16],增强 Wnt/β-catenin信号通路的传导,从而促进成骨分化。ACVR1(激活素A受体)是骨形态发生蛋白信号通路中的重要调控因子,负责骨髓系统的发育和修复,miR-208a-3p过表达将降低ACVR1的翻译水平,从而下调骨形态发生蛋白2及其下游靶标Smad 1/5和Smad 4的表达,骨形成受到抑制[17]。此外,转化生长因子β信号通路对 Smads蛋白(Smad2、Smad3)有关键的调控作用。研究发现miR-140可靶向结合 Smad3进而抑制转化生长因子β信号通路,而转化生长因子β信号通路也可抑制 miR-140的积累,表明miR-140/ Smad3/转化生长因子β信号通路3者以负反馈形式调节骨髓间充质干细胞的成骨分化[18]。 lncRNA:lncRNA已被证实可以通过表观遗传修饰与miRNA相互作用,以及与转录因子结合等方式参与调控成骨分化。表观遗传水平调控成骨分化的研究主要集中在组蛋白修饰方面,包括组蛋白的甲基化、乙酰化和泛素化等。哺乳动物的lncRNA中有大约20%可结合多梳抑制复合物2,EZH2是多梳抑制复合物2的核心亚基,可通过诱发组蛋白H3K27的甲基化来抑制基因转录[19]。研究发现lncRNA-HoxA-AS3和lncRNA ANCR均可与EZH2结合引发组蛋白H3K27的甲基化,使Runx2表达降低,进而抑制人间充质干细胞的成骨分化能力[20-21]。 另外,lncRNA-miRNA-mRNA调节模式在成骨分化中起着关键作用,即lncRNA可作为miRNA的竞争性内源RNA竞争miRNA的结合位点,从而减弱其对mRNA的直接影响。lncRNA MALAT1可竞争性地与miR-30结合,抑制miR-30与Runx2的3'UTR相互作用,进而上调Runx2的转录水平,促进脂肪间充质干细胞成骨分化[22]。研究发现lncRNA TUG1[23]、lncRNA PCAT1[24]、lncRNA HIF1A-AS2同样是通过吸附miRNA的方式来间接调节成骨相关信号分子的活性[25],进而调控成骨分化。 lncRNA也可直接与转录因子结合形成lncRNA-蛋白质复合物,且通过调节转录因子的表达水平在成骨分化中发挥作用。p38是MAPK通路的主要调控因子之一,lncRNA DANCR通过下调p38表达水平,致使p38-MAPK信号通路的失活,进而抑制人骨髓间充质干细胞的增殖和成骨分化[26]。另一种lncRNA MEG3则从骨形态发生蛋白4启动子解离转录因子SOX2以增加骨形态发生蛋白4基因的表达,证实MEG3过表达可通过靶向骨形态发生蛋白4的转录促进多发性骨髓瘤中间充质干细胞的成骨分化[27]。 2.2 基于非编码RNA的骨组织工程应用 目前非编码RNA在骨组织工程领域主要有2种应用策略[28]:一种是通过生物方法直接改变种子细胞内目标非编码RNA的表达,而后与支架材料复合,最后植入缺损部位以促进骨缺损修复;另一种是将调控目标非编码RNA的药物和支架材料组合构建基于非编码RNA的支架递送系统,进而调控种子细胞内目标非编码RNA的转录水平,促进骨缺损修复,见图2。"
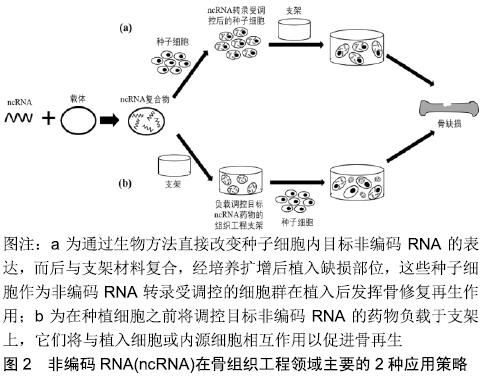

2.2.1 非编码RNA/干细胞疗法在骨组织工程中的应用 骨组织工程需要适当和丰富的种子细胞,多能干细胞可以自我更新并具有分化成多种细胞组织的潜能,从而提供潜在的无限的种子细胞。近年来,骨髓间充质干细胞和脂肪间充质干细胞等类型干细胞被用于修复骨缺损。在许多研究中,干细胞在植入前往往经基因工程改造,以表达特定类型的基因或预分化成特定细胞类型。由于非编码RNA在骨形成过程中发挥重要作用,直接改变干细胞内非编码RNA表达水平并结合适当支架的组织工程技术是一种新的、有前景的骨再生策略[29]。 骨髓间充质干细胞:是间充质干细胞中的一种,属于胚胎发育早期中胚层的一类多能干细胞,可以分化成骨、软骨、脂肪等细胞。在LIU等[30]的研究中,增强miR-26a表达后的骨髓间充质干细胞再结合传统的β-磷酸三钙支架,显著促进了小鼠颅骨缺损区域的骨再生。同样地,miR-210-3p转录上调的骨髓间充质干细胞与β-磷酸三钙支架的结合,可改善犬承重下颌骨缺损修复的效果[31]。骨质疏松症是一种常见的骨骼疾病,极易导致骨折并增加了骨修复的难度。LI等[32]开发一种基于miRNA的细胞/基因治疗方法,提取患骨质疏松症大鼠的骨髓间充质干细胞,让抑制miR-214的载体转染骨髓间充质干细胞,之后与明胶海绵支架结合再移植到因卵巢切除而患骨质疏松的大鼠股骨干骺端骨缺损处,不仅可促进骨愈合,而且还能改善骨质量。在植入物与周围骨组织的骨整合过程中,成骨生成和血管生成起到关键作用。Yan等[33]基于维生素C诱导方法制备由骨髓间充质干细胞组成的细胞膜片,通过将抑制miR-138的载体转染骨髓间充质干细胞膜片得到miR-138转录下调的骨髓间充质干细胞膜片,之后用这些骨髓间充质干细胞膜片紧密包裹Ti植入物,形成骨髓间充质干细胞膜片-植入物复合物,最后将其移植于骨缺损处,结果发现成骨-血管生成耦合作用得到增强,并促进了Ti植入物的骨整合。 脂肪间充质干细胞:相比于骨髓间充质干细胞,脂肪间充质干细胞能够在体外稳定增殖,并具有来源丰富、取材方便、多向分化潜能和安全性高等优点,是组织工程中理想的种子细胞之一。XIE等[34]通过慢病毒介导的方式得到miR-135转录上调的脂肪间充质干细胞,然后将这些脂肪间充质干细胞与聚脂酰二甘油酯支架复合,能显著促进大鼠颅骨缺损处新骨形成。另外,通过脂质体转染 miRNA模拟物方法过表达脂肪间充质干细胞中的miR-26a,之后将其种植到传统的多孔羟基磷灰石支架上,再植入大鼠临界尺寸胫骨缺损处,可显著改善新骨形成[35]。DENG等[36]的研究表明,通过基因操控使脂肪间充质干细胞中的miR-31表达受抑制,将这些脂肪间充质干细胞与β-磷酸三钙支架结合治疗大鼠颅骨临界尺寸骨缺损时表现出显著的骨再生效果。以上研究结果均表明,将种子细胞内非编码RNA转录有目的地改变后与骨组织工程支架结合,可以作为骨缺损修复的有效方法。 2.2.2 非编码RNA/支架递送系统在骨组织工程中的应用 骨组织工程的一个主要工作重点是开发可降解生物活性支架,尽可能地模拟组织天然环境下的三维结构、机械性能、细胞外基质成分及物理化学刺激。生物活性因子结合组织工程支架可进一步优化支架特征,从而促进骨缺损修复。为了有效地将生物活性因子递送给细胞或组织,近年来研究者设计了多种类型的骨组织工程支架(包括多孔支架、纳米纤维支架以及水凝胶支架[5],图3)来实现可控地、有目的地调控种子细胞内非编码RNA表达,以促进骨缺损修复。 "


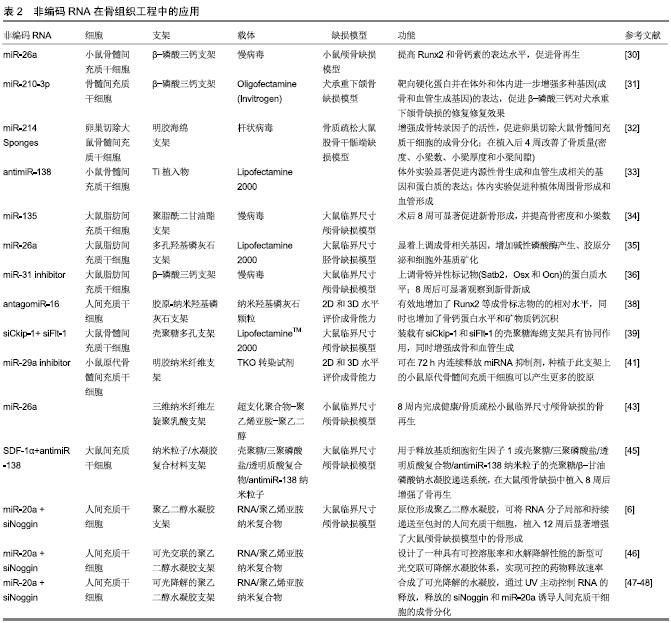
多孔支架:目前为止,用于骨组织工程的多孔支架材料的研究最多,主要包括陶瓷及其复合材料。作为一种无机非金属材料,陶瓷具有高硬度、高熔点和脆性等特点,其中磷酸三钙和羟基磷灰石作为牙齿和骨骼的成分之一,具有生物相容性、可降解性、骨传导性以及骨诱导性。目前更多的是通过将羟基磷灰石等磷酸钙盐与其他天然生物材料(如胶原、壳聚糖等)复合制成多孔复合支架,克服陶瓷脆性的同时提高了天然材料的机械强度,其中纳米羟基磷灰石可作为非编码RNA的递送载体。CASTANO等[37]首次报道了使用纳米羟基磷灰石递送载体开发新型生物活性支架,利用纳米羟基磷灰石颗粒充当miRNA的递送载体构建纳米羟基磷灰石-miRNA (nanomiR)系统,能以高效和低毒的方式将nanomiR-mimics和nanoantagomiRs递送至人间充质干细胞,从而调控人间充质干细胞中miRNA的转录水平。另外,MENCIA等[38]将纳米羟基磷灰石颗粒作为antagomiR-16的非病毒载体,与特异性设计的胶原-纳米羟基磷灰石支架组合,然后将人间充质干细胞种植于此递送平台,结果表明antagomiR-16/胶原-纳米羟基磷灰石支架递送系统有效地下调了人间充质干细胞内miR-16的转录,增加了Runx2等成骨标志物的相对水平,同时显著增加了骨钙蛋白水平及矿物质钙沉积,表明该药物递送支架在骨组织工程中具有显著的治疗潜力。天然生物材料壳聚糖具有无免疫原性、生物相容性、生物可降解性及抗菌性等性能,且能以可控的方式递送生物活性因子,适合作为骨组织工程支架材料。JIA等[39]将壳聚糖多孔支架同时作为Ckip-1和sFlt-1的siRNA药物储库,这2种siRNAs可以在该支架中持续释放2周以上,而在溶菌酶存在下快速降解,释放出的siCkip-1和siFlt-1发挥协同作用,可增强成骨和血管生成。研究表明这种负载双siRNAs壳聚糖支架显著促进了大鼠颅骨缺损模型中的骨再生。此外,有意思的是骨组织工程支架材料对特定成骨相关的非编码RNA也具有调节作用。例如,纳米生物玻璃陶瓷颗粒可诱导人成骨细胞内miR-30c的表达,从而抑制其靶标转化生长影响因子2和组蛋白脱乙酰基酶4的表达,促进成骨分化[40]。因此研究非编码RNA如何受生物材料调控,将有助于进一步理解并促进基于非编码RNA的药物递送支架在骨组织工程中的应用。 纳米纤维支架:具有类似天然细胞外基质的结构,其高的比表面积及交错的纳米纤维排列结构有利于细胞黏附、增殖、迁移和分化。此外,通过在纤维内包封或在表面负载生物活性因子等方式制备的纳米纤维支架,能够通过局部递送治疗剂来调节细胞功能。JAMES等[41]通过静电纺丝制备负载miR-29a抑制剂(miR-29家族成员是多种组织类型中细胞外基质合成的有效负调节因子)的明胶纳米纤维支架,该支架可在72 h内连续释放miRNA抑制剂,种植于此支架上的小鼠原代骨髓间充质干细胞可以产生更多的胶原,表明负载miR-29a抑制剂的明胶纳米纤维支架可促进细胞外基质的合成,从而促进骨修复。此外,PINESE等[42]设计了一种聚己内酯纳米纤维复合支架,可将siRNA/介孔二氧化硅纳米粒子-聚乙烯亚胺复合物负载于纳米纤维基材的表面或直接包封在纳米纤维中。这2种方法均可实现药物的持续释放,但特点各异:表面吸附法通常会导致药物的爆发释放,使其在体外转染时有更高的效率;而纳米纤维包封提供更持久的药物释放,同时可以保护这些不稳定分子免于酶促降解,是体内应用的理想选择。此外,药物/基因的双重或顺序递送可以更好地调控细胞命运和组织再生。ZHANG等[43]将具有高miRNA结合亲和力和低毒性的超支化聚合物作为miR-26a的载体,并将miR-26a/超支化聚合物复合物包封在可生物降解的聚合物微球中,且该聚合物微球被固定在三维纳米纤维支架上,从而实现2阶段递送miRNA:可控持续递送(第一阶段,聚合物微球)和高转染效率(第二阶段,miRNA/超支化聚合物复合物),最终可控地改变内源性细胞内非编码RNA的表达来修复临界大小的骨缺损。另有研究表明,研究非编码RNA和其所处环境的纳米结构之间的相互作用可能是增强成骨诱导的新策略。IZADPANAHI等[44]研究了纳米纤维的排列和取向对间充质干细胞中非编码RNA调节网络的影响,表明静电纺纳米纤维的几何力学或拓扑结构影响非编码RNA的表达,进而调控干细胞的增殖和分化能力,是骨组织工程有吸引力的研究和应用方向。 水凝胶支架:水凝胶支架是由亲水聚合物链发生物理或化学交联形成的,其三维网络结构可以吸收和保留大量的水,营养物质在其中可以自由进出。同时,水凝胶支架也可以将生物活性因子封装在内,随后以局部和受控的方式进行递送,因此被广泛应用于组织工程和药物释控等领域。相对于固体多孔支架和纳米纤维支架,水凝胶支架可用于包封细胞。NGUYEN等[6]提出了一种原位形成的聚乙二醇水凝胶系统,在大鼠颅骨缺损模型处,人间充质干细胞和RNA/聚乙烯亚胺纳米复合物被同时包封在聚乙二醇水凝胶中,其中siNoggin和miR-20a可局部并持续地被递送至人间充质干细胞及缺损周围的细胞中,促进大鼠颅骨缺损处的骨再生。骨再生是一个复杂和连续的过程,因此实现生物活性因子的可控连续释放,对促进骨缺损愈合至关重要。WU等[45]将基质细胞衍生因子1α和壳聚糖/三聚磷酸盐/透明质酸/antimiR-138纳米粒子(CTH/antimiR-138 NPs)包封在壳聚糖/β-甘油磷酸钠水凝胶中形成纳米粒子/水凝胶复合体系,其中分别从水凝胶基质中快速释放基质细胞衍生因子1α,从纳米粒子/水凝胶复合体系中缓慢释放antimiR-138,结果表明基质细胞衍生因子1α和CTH/antimiR-138 NPs在时空序列中的双重释放可有效抑制间充质干细胞内的miR-138表达,促进其迁移及成骨分化,增强大鼠临界尺寸颅骨缺损处的骨再生能力。NGUYEN等[46]则通过将葡聚糖与单(2-丙烯酰氧基乙基)琥珀酸酯发生酯化反应得到水凝胶单体,进而设计了一种具有可控溶胀率和水解降解性的新型水凝胶体系,可实现siRNA释放速率的定制化设计。虽然可以通过预先设计材料成分和结构、控制制备参数等方式来预设水凝胶的降解速率,进而实现药物的可控递送,然而一旦将这些负载药物的生物材料注射或植入体内,患者和临床医生都不能针对药物实时需求而改变药物释放速率。近期研究者构建了一种可光降解的水凝胶系统,可在指定时间点主动释放RNA。HUYNH等[47-48]将RNA/聚乙烯亚胺复合物加载到可光降解的聚乙二醇水凝胶中,当UV施加时,可光降解水凝胶中的RNA释放加速,从水凝胶中释放的siNoggin或miR-20a诱导人间充质干细胞成骨分化。该系统为生物医学应用提供了具有潜在应用价值的“按需”RNA递送平台。水凝胶的另一个优点,可原位形成复杂三维形状从而更有利于填充骨缺陷。然而间充质干细胞增殖、迁移和分化会受到体外细胞培养基质材料力学特性的影响:间充质干细胞在柔软的基底上趋向于分化成脂肪或神经元细胞,而硬的基底上趋向于成骨分化。当在可注射的、易成型的软水凝胶中包裹间充质干细胞时,水凝胶的易用性与间充质干细胞成骨分化能力的有效结合将被限制。FRITH等[49]鉴定了响应不同水凝胶硬度的miRNA差异表达,发现miR-100-5p和miR-143-3p可调节间充质干细胞的分化命运,证明了miRNA信号传导在人间充质干细胞对其物理微环境的响应过程中发挥关键作用,从而可用于指导人间充质干细胞的命运。因此,调节种子细胞中这些机械敏感性非编码RNA的表达,可以增强其在柔软三维水凝胶中的成骨分化能力,从而增强水凝胶支架的骨修复能力。 多相复合支架:单一构型支架很难满足组织工程对支架材料的要求,因此将不同类型的支架相互复合,以充分发挥各自的优点。此外,随着骨相关生物活性因子及其释放方式的不断丰富,新型的药物递送系统不断被设计并制备出来,从而进一步促进骨缺损修复。有研究发现,骨形态发生蛋白2的骨诱导性质可以通过与新型骨诱导性双相磷酸钙支架的复合得到进一步增强[50]。然而将上述支架作为药物递送系统时,药物并不足以保持长期的释放[51]。通过将合适的聚合物水凝胶与多孔骨传导支架复合,可以构建更好的药物递送系统来促进骨愈合。SUKUL等[52]将纳米纤维素水凝胶掺入多孔双相磷酸钙支架内,其中生长因子(骨形态发生蛋白2-血管内皮生长因子)被物理包埋在可缓慢降解的纳米纤维素的纳米级纤维结构中。该系统可提供生长因子的持续释放,促进了种植在支架上的大鼠骨髓间充质干细胞的增殖和成骨分化。YAN等[53]则采用多层涂层技术制备多孔复合支架,其中可降解聚乳酸结合磷灰石纳米针状晶粒作为第一涂层,海藻酸钠/壳聚糖水凝胶复合物作为第二涂层并负载相关生长因子。该支架材料的降解可以非常好地匹配新骨形成,此外,联合使用低强度脉冲超声和生长因子,有效促进了血管和新骨形成。另外,还有直接制备多层多相支架,比如通过连续电纺丝法制备多层支架来提高药物控释[54],其中心层可作为药物载体,使支架既能提供结构支撑又能提供药物控释。GEBREMARIAM等[55]便开发了一种负载地塞米松的多层左旋聚乳酸/pluronic P123复合电纺纳米纤维支架。还有通过微流体结合静电纺丝制备微球纳米纤维交替多层支架,其中包裹种子细胞和生长因子的水凝胶微球与纳米纤维相互叠加,将纳米纤维的高比表面积及良好孔隙结构特性与水凝胶良好的渗透性及可包裹细胞和药物的特性很好地结合起来。例如DING等[56]制备的负载大鼠骨髓间充质干细胞和骨形态发生蛋白2的微球纳米纤维交替多层支架。此外在已成型支架表面涂覆或固定非编码RNA的方法,也在骨组织工程支架方面得到应用[57-58]。 近年来,基于计算机辅助设计的3D打印技术在组织工程领域的应用得到了广泛研究和快速发展,它可以精确构建具有个性化外形和孔隙结构的组织工程支架,也可在温和的条件下将由生物材料、细胞或生物活性因子组成的“生物墨水”直接打印成型,进而得到组织构建体。BAO等[59]采用CT引导下的熔融沉积3D打印技术制备了具有自体骨解剖结构的生物活性羟基磷灰石/聚己内酯多孔复合支架,然后将载有血管内皮生长因子165/骨形态发生蛋白2的热敏聚乳酸乙醇酸-聚乙二醇-聚乳酸乙醇酸水凝胶加载到支架中,该支架具有合适机械支撑能力并表现出骨传导性质,其中载有细胞因子的水凝胶具有良好的生物降解性,可以持续稳定地释放血管内皮生长因子165/骨形态发生蛋白2。这种采用“解剖-结构-功能”设计方式来构建功能性人工骨的策略,在骨缺损修复方面具有很大的潜力。而YOUNG PARK等[60]通过3D打印技术制备了载有牙髓干细胞、骨形态发生蛋白2和血管内皮生长因子的支架,实现了2种生长因子在支架中的可控分布与释放,其中在支架缺氧中心处“印刷”血管内皮生长因子,构建预血管化结构。结果表明,3D打印的预血管化结构可作为克服组织植入物尺寸限制和增强骨再生的一种有前景方法。此外,Paolini等[61]则通过在3D打印的聚乳酸支架上涂覆有荧光标记的聚酰氨基胺树枝状聚合物(一种新型无毒的递送载体),从而获得可用于细胞增殖和miRNA转染的生物相容性多功能支架。 非编码RNA作为生物活性因子在骨组织工程领域得到越来越多的研究与应用,然而在上述复杂体系中的应用还鲜有耳闻。可以预见的是,上述基于其他生物活性因子(如骨形态发生蛋白和血管内皮生长因子等)的复合支架和3D打印技术的研究基础将为非编码RNA/支架递送系统的构建提供借鉴和平台,推动其在骨组织工程中进一步应用。 2.3 在骨组织工程中有应用前景的非编码RNA 尽管基于非编码RNA的组织工程方法为骨缺损修复提供了一条新的有效途径,但目前应用于骨组织工程并取得良好骨修复效果的非编码RNA仍然有限。随着对非编码RNA在骨修复过程中调控机制的深入研究,大量具有骨组织工程应用前景的非编码RNA涌现而出。 近年来,与成骨分化相关的非编码RNA不断被发现并证实。研究表明miR-224[62]、miR-223[63]、miR-488[64]、miR-125b[65]、miR-98[66]、miR-145-5p等非编码RNA可抑制成骨分化[67]。相反,一些非编码RNA则促进成骨分化,它们包括miR-130a[68]、miR-27b[68]、miR-450b[69]、miR-335-5p等[70]。此外,lncRNA如linc-ROR[71]、lncRNA HIF1A-AS2[25]、LINC00707均可作为miRNA的竞争性内源RNA[72],促进脂肪间充质干细胞或骨髓间充质干细胞成骨分化。此外,已经证实机械刺激可通过机械信号传导途径促进成骨分化[73],研究表明miR-195-5p和miR-208a-3p都是机械敏感性非编码RNA[17,74],在机械负荷诱导的成骨分化和骨形成中起重要作用。 目前,非编码RNA在骨组织工程中的应用主要关注其成骨分化的促进作用,然而细胞增殖与迁移、细胞外基质的沉积及血管生成与重塑是创伤愈合的3个关键环节,以上环节的增强均能够进一步促进骨愈合。miR-217可促进骨髓间充质干细胞的增殖和成骨分化[75],而miR-133b和miR-125a-3p可作为间充质干细胞增殖和成骨分化的负调节因子[76-77]。另有研究表明,miR-21通过增强PI3K/Akt信号通路的活性进而显著促进骨髓间充质干细胞的迁移[78],miR-25和miR-221则可同时促进小鼠胚胎成骨细胞前体细胞MC3T3-E1的迁移、增殖和成骨分化[79-80],而miR-367则抑制了成骨细胞的增殖和迁移[81]。 已知miR-29家族是细胞外基质合成的负调控因子,通过抑制miR-29的表达可促进细胞外基质的合成[82]。另外,LI等[83]发现miR-214-5p通过靶向COL4A1负调控MC3T3-E1存活和细胞外基质形成。 研究表明,血管生成和成骨生成的相互耦合对于骨缺损修复至关重要。miR-26a已被应用于骨组织工程研究,以促进骨骼再生(骨生成和血管生成[84-85])。另外,ZHANG等[86]发现miR-378可同时增强骨髓间充质干细胞的成骨分化和血管生成。此外越来越多的证据表明,miRNA可以作为细胞间通信媒介,参与血管生成与骨生成间的相互耦合。FAN等[87]证实miR-200b可作为骨髓间充质干细胞和人脐静脉内皮细胞间通信的信号分子,调节成骨和血管生成。LIU等[88]首次提出生物材料可以引发外泌体分泌miR-130a促进骨髓间充质干细胞-内皮细胞通讯,间接促进体外人脐静脉内皮细胞的血管生成。此外,研究发现在新生和成熟破骨细胞中,lncRNA-AK131850可促进内皮祖细胞的增殖、分化、迁移及血管发生[89]。 通常骨缺损处往往具有不同的生理病理环境,如炎症反应、骨质疏松等。炎症微环境的存在可减弱干细胞的成骨分化能力,从而抑制成骨细胞介导的骨形成,导致净骨丢失,因此人们越来越关注炎症微环境中的成骨 分化。研究发现,lncRNA MIR31HG通过促进IκBα磷酸化和NF-κB通路活化抑制成骨分化[90],因此靶向这种MIR31HG -NF-κB调节环可能有助于提高炎症微环境下人脂肪间充质干细胞的成骨能力。另外,与牙周炎相关的lncRNA-POIR与miR-182相互作用,促进牙周炎患者牙周间充质干细胞的成骨分化[91]。 非编码RNA也可以参与调节骨质疏松症环境下的骨形成。miR-874通过靶向抑制SUFU和激活Hedgehog信号通路促进骨质疏松大鼠成骨细胞的增殖和分化[92]。此外,一些非编码RNA则抑制骨质疏松病症下的骨形成,例如miR-106b[93]、lncRNA AK016739[94]、lncRNA MEG3等[95],其抑制剂可能是治疗骨质疏松症和骨缺损的潜在药物。 表1,2分别详细列举了文内涉及的非编码RNA的功能与应用研究。"

| [1] PAMELA H.Strategic Directions in Osteoinduction and Biomimetics. Tissue Eng Part A. 2017;23(23-24):1295-1296. [2] AMINI AR, LAURENCIN CT, NUKAVARAPU SP. Bone Tissue Engineering: Recent Advances and Challenges. Crit Rev Biomed Eng. 2012;40(5):363-408. [3] HO-SHUI-LING A, BOLANDER J, RUSTOM LE, et al.Bone regeneration strategies: Engineered scaffolds,bioactive molecules and stem cells current stage and future perspectives. Biomaterials. 2018;180:143-162. [4] LIENEMANN PS, LUTOLF MP, EHRBAR M.Biomimetic hydrogels for controlled biomolecule delivery to augment bone regeneration.Adv Drug Deliv Rev.2012;64(12):1078-1089. [5] ZHANG Y, MA W, ZHAN Y, et al.Nucleic acids and analogs for bone regeneration. Bone Res. 2018;6:331-339 [6] NGUYEN M K, JEON O, DANG P N,et al.RNA interfering molecule delivery from in situ forming biodegradable hydrogels for enhancement of bone formation in rat calvarial bone defects.Acta Biomater. 2018;75:105-114. [7] BEAVERS KR, NELSON CE, DUVALL CL.MiRNA inhibition in tissue engineering and regenerative medicine.Adv Drug Deliv Rev. 2015; 88:123-137. [8] HUYNH NP, ANDERSON BA, GUILAK F, et al.Emerging roles for long noncoding RNAs in skeletal biology and disease.Connect Tissue Res. 2017;58(1):116-141. [9] CHEN G, DENG C, LI YP.TGF-β and BMP Signaling in Osteoblast Differentiation and Bone Formation.Int J Biol Sci.2012;8(2):272-288. [10] LIU W, KONERMANN A, GUO T, et al.Canonical Wnt signaling differently modulates osteogenic differentiation of mesenchymal stem cells derived from bone marrow and from periodontal ligament under inflammatory conditions.Biochim Biophys Acta. 2014;1840(3):1125-1134. [11] ZHANG Y, GAO Y, CAI L, et al.MicroRNA-221 is involved in the regulation of osteoporosis through regulates RUNX2 protein expression and osteoblast differentiation.Am J Transl Res. 2017; 9(1):126-135. [12] KUREEL J, JOHN AA, DIXIT M, et al.MicroRNA-467g inhibits new bone regeneration by targeting Ihh/Runx-2 signaling.Int J Biochem Cell B. 2017;85:35-43. [13] ZHANG W, WU Y, SHIOZAKI Y, et al.miRNA-133a-5p Inhibits the Expression of Osteoblast Differentiation-Associated Markers by Targeting the 3' UTR of RUNX2. DNA Cell Biol. 2018;37(3):199-209. [14] FANG S, DENG Y, GU P, et al.MicroRNAs regulate bone development and regeneration.Int J Mol Sci.2015;16(4):8227-8253. [15] TANG X, LIN J, WANG G, et al.MicroRNA-433-3p promotes osteoblast differentiation through targeting DKK1 expression.PLoS One. 2017; 12(6):e0179860. [16] ZHANG X, ZHU Y, ZHANG C, et al.miR-542-3p prevents ovariectomy -induced osteoporosis in rats via targeting SFRP1.J Cell Physiol.2018; 233(9):6798-6806. [17] ARFAT Y, BASRA M A R, SHAHZAD M, et al.miR-208a-3p Suppresses Osteoblast Differentiation and Inhibits Bone Formation by Targeting ACVR1.Mol Ther-nucl Acids.2018;11:323-336. [18] PAIS H, NICOLAS F E, SOOND S M, et al.Analyzing mRNA expression identifies Smad3 as a microRNA-140 target regulated only at protein level.RNA.2010;16(3):489-494. [19] CAO R.Role of Histone H3 Lysine 27 Methylation in Polycomb-Group Silencing.Science. 2002;298(5595):1039-1043. [20] ZHU XX, YAN YW, CHEN D, et al.Long non-coding RNA HoxA-AS3 interacts with EZH2 to regulate lineage commitment of mesenchymal stem cells.Oncotarget.2016;7(39):63561-63570. [21] ZHU L, XU PC. Downregulated LncRNA-ANCR promotes osteoblast differentiation by targeting EZH2 and regulating Runx2 expression. Biochem Bioph Res Co.2013;432(4):612-617. [22] YI J, LIU D, XIAO J.LncRNA MALAT1 sponges miR-30 to promote osteoblast differentiation of adipose-derived mesenchymal stem cells by promotion of Runx2 expression.Cell Tissue Res. 2019;376(1):113-121. [23] YU C, LI L, XIE F, et al.LncRNA TUG1 sponges miR-204-5p to promote osteoblast differentiation through upregulating Runx2 in aortic valve calcification. Cardiovasc Res. 2018;114(1):168-179. [24] YU L, QU H, YU Y, et al.LncRNA-PCAT1 targeting miR-145-5p promotes TLR4-associated osteogenic differentiation of adipose-derived stem cells.J Cell Mol Med.2018;22(12):6134-6147. [25] WU R, RUAN J, SUN Y, et al.Long non-coding RNA HIF1A-AS2 facilitates adipose-derived stem cells (ASCs) osteogenic differentiation through miR-665/IL6 axis via PI3K/Akt signaling pathway. Stem Cell Res Ther.2018;9(1):348. [26] ZHANG J, TAO Z, WANG Y. Long noncoding RNA DANCR regulates the proliferation and osteogenic differentiation of human bone-derived marrow mesenchymal stem cells via the p38 MAPK pathway. Int J Mol Med.2018;41(1):213-219. [27] ZHUANG W,GE X,YANG S,et al.Upregulation of lncRNA MEG3 Promotes Osteogenic Differentiation of Mesenchymal Stem Cells From Multiple Myeloma Patients By Targeting BMP4 Transcription. Stem Cells. 2015;33(6):1985-1997. [28] CURTIN CM, CASTANO IM, O'BRIEN FJ.Scaffold-Based microRNA Therapies in Regenerative Medicine and Cancer.Adv Healthc Mater. 2018;7(1):1700695. [29] HEINRICH E M, DIMMELER S. MicroRNAs and stem cells: control of pluripotency,reprogramming,and lineage commitment.Circ Res. 2012; 110(7):1014-1022. [30] LIU Z,CHANG H,HOU Y,et al.Lentivirusmediated microRNA26a overexpression in bone mesenchymal stem cells facilitates bone regeneration in bone defects of calvaria in mice.Mol Med Rep. 2018; 18(6):5317-5326. [31] HU B, LI Y, WANG M, et al.Functional reconstruction of critical-sized load-bearing bone defects using a Sclerostin-targeting miR-210-3p- based construct to enhance osteogenic activity.Acta Biomaterialia. 2018;76:275-282. [32] LI K C, CHANG Y H, YEH CL, et al.Healing of osteoporotic bone defects by baculovirus-engineered bone marrow-derived MSCs expressing MicroRNA sponges. Biomaterials. 2016;74:155-166. [33] YAN J,CHANG B,HU X,et al.Titanium implant functionalized with antimiR-138 delivered cell sheet for enhanced peri-implant bone formation and vascularization.Mater Sci Eng C Mater. 2018;89:52-64. [34] XIE Q, WANG Z, ZHOU H, et al.The role of miR-135-modified adipose-derived mesenchymal stem cells in bone regeneration. Biomaterials.2016;75:279-294. [35] WANG Z, ZHANG D, HU Z, et al.MicroRNA-26a-modified adipose- derived stem cells incorporated with a porous hydroxyapatite scaffold improve the repair of bone defects.Mol Med Rep. 2015;12(3):3345-3350. [36] DENG Y, ZHOU H, ZOU D, et al.The role of miR-31-modified adipose tissue-derived stem cells in repairing rat critical-sized calvarial defects. Biomaterials.2013;34(28):6717-6728. [37] CASTANO I M, CURTIN C M, SHAW G, et al.A novel collagen-nanohydroxyapatite microRNA-activated scaffold for tissue engineering applications capable of efficient delivery of both miR-mimics and antagomiRs to human mesenchymal stem cells.J Control Release.2015;200:42-51. [38] MENCIA CASTANO I, CURTIN CM, DUFFY GP, et al.Harnessing an Inhibitory Role of miR-16 in Osteogenesis by Human Mesenchymal Stem Cells for Advanced Scaffold-Based Bone Tissue Engineering. Tissue Eng Part A.2019;25(1-2):24-33. [39] JIA S, YANG X, SONG W, et al. Incorporation of osteogenic and angiogenic small interfering RNAs into chitosan sponge for bone tissue engineering.Int J Nanomed.2014;9:5307-5316. [40] MOORTHI A, VIMALRAJ S, AVANI C,et al.Expression of microRNA- 30c and its target genes in human osteoblastic cells by nano-bioglass ceramic-treatment.Int J Biol Macromol.2013;56:181-185. [41] JAMES E N, DELANY A M, NAIR L S. Post-transcriptional regulation in osteoblasts using localized delivery of miR-29a inhibitor from nanofibers to enhance extracellular matrix deposition. Acta Biomaterialia.2014;10(8):3571-3580. [42] PINESE C, LIN J, MILBRETA U, et al.Sustained delivery of siRNA/mesoporous silica nanoparticle complexes from nanofiber scaffolds for long-term gene silencing.Acta Biomaterialia. 2018;76: 164-177. [43] ZHANG X, LI Y, CHEN Y E, et al.Cell-free 3D scaffold with two-stage delivery of miRNA-26a to regenerate critical-sized bone defects.Nat Commun.2016;7:10376. [44] IZADPANAHI M, SEYEDJAFARI E, AREFIAN E, et al.Nanotopographical cues of electrospun PLLA efficiently modulate non-coding RNA network to osteogenic differentiation of mesenchymal stem cells during BMP signaling pathway. Mat Sci Eng C Mater. 2018;93:686-703. [45] WU G, FENG C, QUAN J, et al.In situ controlled release of stromal cell-derived factor-1alpha and antimiR-138 for on-demand cranial bone regeneration.Carbohydr Polym.2018;182:215-224. [46] NGUYEN MK, MCMILLAN A, HUYNH CT, et al. Photocrosslinkable, biodegradable hydrogels with controlled cell adhesivity for prolonged siRNA delivery to hMSCs to enhance their osteogenic differentiation.J Mater Chem B.2017;5(3):485-495. [47] HUYNH CT, NGUYEN MK, NARIS M, et al.Light-triggered RNA release and induction of hMSC osteogenesis via photodegradable, dual-crosslinked hydrogels.Nanomedicine. 2016;11(12):1535-1550. [48] HUYNH C T, ZHENG Z, NGUYEN M K, et al.Cytocompatible Catalyst-Free Photodegradable Hydrogels for Light-Mediated RNA Release To Induce hMSC Osteogenesis.Acs Biomater Sci Eng. 2017;3(9):2011-2023. [49] FRITH JE,KUSUMA GD,CARTHEW J,et al.Mechanically-sensitive miRNAs bias human mesenchymal stem cell fate via mTOR signalling. Nat Commun.2018;9(1):257. [50] WANG Z, WANG K, LU X, et al.BMP-2 encapsulated polysaccharide nanoparticle modified biphasic calcium phosphate scaffolds for bone tissue regeneration.J Biomed Mater Res A. 2015;103(4):1520-1532. [51] ZHANG Y, YANG S, ZHOU W, et al.Addition of a Synthetically Fabricated Osteoinductive Biphasic Calcium Phosphate Bone Graft to BMP2 Improves New Bone Formation.J Biomed Mater Res. 2016; 18(6):1238-1247. [52] SUKUL M, NGUYEN TB, MIN YK, et al.Effect of Local Sustainable Release of BMP2-VEGF from Nano-Cellulose Loaded in Sponge Biphasic Calcium Phosphate on Bone Regeneration. Tissue Eng Part A. 2015;21(11-12):1822-1836. [53] YAN H, LIU X, ZHU M, et al.Hybrid use of combined and sequential delivery of growth factors and ultrasound stimulation in porous multilayer composite scaffolds to promote both vascularization and bone formation in bone tissue engineering.J Biomed Mater Res A. 2016;104(1):195-208. [54] SALIFU A A, LEKAKOU C, LABEED F. Multilayer cellular stacks of gelatin-hydroxyapatite fiber scaffolds for bone tissue engineering.J Biomed Mater Res A.2017;105(3):779-789. [55] GEBREMARIAM B, SHIMA T, HAMID A J, et al. Dexamethasone Loaded Multi-Layer Poly-L-Lactic Acid/Pluronic P123 Composite Electrospun Nanofiber Scaffolds for Bone Tissue Engineering and Drug Delivery. Pharm Dev Technol. 2018:1-30. [56] DING S, LI L, LIU X, et al.A nano-micro alternating multilayer scaffold loading with rBMSCs and BMP-2 for bone tissue engineering. Colloid Surface B.2015;133:286-295. [57] SONG W, YANG C, LE D QS, et al.Calcium-MicroRNA Complex- Functionalized Nanotubular Implant Surface for Highly Efficient Transfection and Enhanced Osteogenesis of Mesenchymal Stem Cells. Acs Appl Mater Inter. 2018;10(9):7756-7764. [58] HUANG Y, ZHENG Y, XU Y, et al.Titanium Surfaces Functionalized with siMIR31HG Promote Osteogenic Differentiation of Bone Marrow Mesenchymal Stem Cells.Acs Biomater-Sci Eng. 2018;4(8):2986-2993. [59] BAO X, ZHU L, HUANG X, et al.3D biomimetic artificial bone scaffolds with dual-cytokines spatiotemporal delivery for large weight-bearing bone defect repair.Sci Rep. 2017;7(1):7814-7814. [60] YOUNG PARK J, SHIM JH, CHOI SA, et al.3D printing technology to control BMP-2 and VEGF delivery spatially and temporally to promote large-volume bone regeneration.J Mater Chem B. 2015;3:5379-5646. [61] PAOLINI A, LEONI L,GIANNICCHI I, et al. MicroRNAs delivery into human cells grown on 3D-printed PLA scaffolds coated with a novel fluorescent PAMAM dendrimer for biomedical applications. Sci Rep. 2018;8(1):13888. [62] LUO Y, CAO X, CHEN J, et al. MicroRNA-224 suppresses osteoblast differentiation by inhibiting SMAD4. J Cell Physiol. 2018;233(10): 6929-6937. [63] ZHANG S, LIU Y, ZHENG Z, et al.MicroRNA-223 Suppresses Osteoblast Differentiation by Inhibiting DHRS3. Cell Physiol Biochem. 2018;47(2):667-679. [64] YU D, ZHAO X, CHENG JZ, et al.Downregulated microRNA-488 enhances odontoblast differentiation of human dental pulp stem cells via activation of the p38 MAPK signaling pathway.J Cell Physiol. 2019;234(2):1442-1451. [65] XUE N, QI L, ZHANG G, et al.miRNA-125b Regulates Osteogenic Differentiation of Periodontal Ligament Cells Through NKIRAS2/NF-kappa B Pathway. Cell Physiol Biochem. 2018;48(4):1771-1781. [66] ZHANG G-P, ZHANG J, ZHU C-H, et al.MicroRNA-98 regulates osteogenic differentiation of human bone mesenchymal stromal cells by targeting BMP2.J Cell Mol Med.2017;21(2):254-264. [67] LIU X, ZHU W, WANG L, et al.miR-145-5p suppresses osteogenic differentiation of adipose-derived stem cells by targeting semaphorin 3A.In Vitro Cell Dev-An.2019;55(3):189-202. [68] SEENPRACHAWONG K, TAWORNSAWUTRUK T, NANTASENAMAT C, et al.miR-130a and miR-27b Enhance Osteogenesis in Human Bone Marrow Mesenchymal Stem Cells via Specific Down-Regulation of Peroxisome Proliferator-Activated Receptor gamma.Front Genet. 2018;9:543. [69] FAN L, FAN J, LIU Y, et al.miR-450b Promotes Osteogenic Differentiation In Vitro and Enhances Bone Formation In Vivo by Targeting BMP3.Stem Cells Dev.2018;27(9):600-611. [70] ZHANG L, TANG Y, ZHU X, et al.Overexpression of MiR-335-5p Promotes Bone Formation and Regeneration in Mice.J Bone Miner Res. 2017;32(12):2466-2475. [71] FENG L, SHI L, LU Y-F, et al.Linc-ROR Promotes Osteogenic Differentiation of Mesenchymal Stem Cells by Functioning as a Competing Endogenous RNA for miR-138 and miR-145. Mol Ther-Nucl Acids. 2018;11:345-353. [72] JIA B, WANG Z, SUN X, et al.Long noncoding RNA LINC00707 sponges miR-370-3p to promote osteogenesis of human bone marrow-derived mesenchymal stem cells through upregulating WNT2B. Stem Cell Res Ther.2019;10(1):67. [73] TAO S, LIN Q, HUIJUN C, et al.Cyclic tension promotes osteogenic differentiation in human periodontal ligament stem cells.Int J Clin Exp Patho.2014;7(11):7872-7880. [74] CHANG M, LIN H, FU H, et al.MicroRNA-195-5p Regulates Osteogenic Differentiation of Periodontal Ligament Cells Under Mechanical Loading.J Cell Physiol.2017;232(12):3762-3774. [75] DAI Z, JIN Y, ZHENG J, et al.MiR-217 promotes cell proliferation and osteogenic differentiation of BMSCs by targeting DKK1 in steroid-associated osteonecrosis. Biomed Pharmacoth. 2019;109: 1112-1119. [76] XIAO W, WEN S, CHEN H, et al.MicroRNA-133b negatively regulates the proliferation and osteogenic differentiation of bone marrow mesenchymal stem cells.Int J Clin Exp Patho. 2016;9(12): 12609-12614. [77] TU XM, GU YL, REN GQ.miR-125a-3p targetedly regulates GIT1 expression to inhibit osteoblastic proliferation and differentiation.Exp Ther Med.2016;12(6):4099-4106. [78] LV C, YANG S, CHEN X, et al.MicroRNA-21 promotes bone mesenchymal stem cells migration in vitro by activating PI3K/Akt/MMPs pathway.J Clin Neurosci.2017;46:156-162. [79] LI X, JI J, WEI W, et al.MiR-25 promotes proliferation,differentiation and migration of osteoblasts by up-regulating Rac1 expression. Biomed Pharmacoth.2018;99:622-628. [80] ZHENG X, DAI J, ZHANG H, et al.MicroRNA-221 promotes cell proliferation,migration,and differentiation by regulation of ZFPM2 in osteoblasts.Bra J Med Biol Res.2018;51(12)-51(12):e7574. [81] JIA HL, ZHOU DS.Downregulation of microRNA-367 promotes osteoblasts growth and proliferation of mice during fracture by activating the PANX3-mediated Wnt/beta-catenin pathway. J Cell Biochem. 2019;120(5):8247-8258. [82] HE Y, HUANG C, LIN X, et al.MicroRNA-29 family,a crucial therapeutic target for fibrosis diseases.Biochimie.2013;95(7):1355-1359. [83] LI QS, MENG FY, ZHAO YH, et al.Inhibition of microRNA-214-5p promotes cell survival and extracellular matrix formation by targeting collagen type IV alpha 1 in osteoblastic MC3T3-E1 cells.Bone Joint Res. 2017;6(8):464-471. [84] WANG Z, XIE Q, YU Z, et al. A regulatory loop containing miR-26a,GSK3β and C/EBPα regulates the osteogenesis of human adipose-derived mesenchymal stem cells.Sci Rep.2015;5:15280. [85] BASAK I, WARA A K M, JAVID M, et al.MicroRNA-26a Regulates Pathological and Physiological Angiogenesis by Targeting BMP/SMAD1 Signaling.Circ Res.2013;113(11):1231-U127. [86] ZHANG B, LI Y, YU Y, et al.MicroRNA-378 Promotes Osteogenesis- Angiogenesis Coupling in BMMSCs for Potential Bone Regeneration. Anal Cell Pathol.2018;2018: 8402390. [87] FAN X, TENG Y, YE Z, et al.The effect of gap junction-mediated transfer of miR-200b on osteogenesis and angiogenesis in a co-culture of MSCs and HUVECs.J Cell Sci. 2018;131(13):jcs216135. [88] LIU L,LIU Y,FENG C,et al.Lithium-containing biomaterials stimulate bone marrow stromal cell-derived exosomal miR-130a secretion to promote angiogenesis.Biomaterials. 2019;192:523-536. [89] QUAN H, LIANG M, LI N, et al.LncRNA-AK131850 Sponges MiR-93-5p in Newborn and Mature Osteoclasts to Enhance the Secretion of Vascular Endothelial Growth Factor a Promoting Vasculogenesis of Endothelial Progenitor Cells.Cell Physiol Biochem. 2018;46(1): 401-417. [90] JIN C, JIA L, HUANG Y, et al.Inhibition of lncRNA MIR31HG Promotes Osteogenic Differentiation of Human Adipose-Derived Stem Cells. Stem Cells.2016;34(11):2707-2720. [91] WANG L, WU F, SONG Y, et al. Long noncoding RNA related to periodontitis interacts with miR-182 to upregulate osteogenic differentiation in periodontal mesenchymal stem cells of periodontitis patients. Cell Death Dis. 2016;7(8):e2327. [92] LIN JC, LIU ZG, YU B, et al.MicroRNA-874 targeting SUFU involves in osteoblast proliferation and differentiation in osteoporosis rats through the Hedgehog signaling pathway. Biochem Bioph Res Co. 2018;506(1): 194-203. [93] LIU K, JING Y, ZHANG W, et al. Silencing miR-106b accelerates osteogenesis of mesenchymal stem cells and rescues against glucocorticoid-induced osteoporosis by targeting BMP2. Bone. 2017;97:130-138. [94] YIN C, TIAN Y, YU Y, et al.A novel long noncoding RNA AK016739 inhibits osteoblast differentiation and bone formation.J Cell physiol. 2019;234(7):11524-11536. [95] WANG Q, LI Y, ZHANG Y, et al.LncRNA MEG3 inhibited osteogenic differentiation of bone marrow mesenchymal stem cells from postmenopausal osteoporosis by targeting miR-133a-3p.Biomed Pharmacoth.2017;89:1178-1186. |
| [1] | Pu Rui, Chen Ziyang, Yuan Lingyan. Characteristics and effects of exosomes from different cell sources in cardioprotection [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(在线): 1-. |
| [2] | Lin Qingfan, Xie Yixin, Chen Wanqing, Ye Zhenzhong, Chen Youfang. Human placenta-derived mesenchymal stem cell conditioned medium can upregulate BeWo cell viability and zonula occludens expression under hypoxia [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(在线): 4970-4975. |
| [3] | Zhang Xiumei, Zhai Yunkai, Zhao Jie, Zhao Meng. Research hotspots of organoid models in recent 10 years: a search in domestic and foreign databases [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1249-1255. |
| [4] | Wang Zhengdong, Huang Na, Chen Jingxian, Zheng Zuobing, Hu Xinyu, Li Mei, Su Xiao, Su Xuesen, Yan Nan. Inhibitory effects of sodium butyrate on microglial activation and expression of inflammatory factors induced by fluorosis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1075-1080. |
| [5] | Wang Xianyao, Guan Yalin, Liu Zhongshan. Strategies for improving the therapeutic efficacy of mesenchymal stem cells in the treatment of nonhealing wounds [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1081-1087. |
| [6] | Liao Chengcheng, An Jiaxing, Tan Zhangxue, Wang Qian, Liu Jianguo. Therapeutic target and application prospects of oral squamous cell carcinoma stem cells [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1096-1103. |
| [7] | Xie Wenjia, Xia Tianjiao, Zhou Qingyun, Liu Yujia, Gu Xiaoping. Role of microglia-mediated neuronal injury in neurodegenerative diseases [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1109-1115. |
| [8] | Li Shanshan, Guo Xiaoxiao, You Ran, Yang Xiufen, Zhao Lu, Chen Xi, Wang Yanling. Photoreceptor cell replacement therapy for retinal degeneration diseases [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1116-1121. |
| [9] | Jiao Hui, Zhang Yining, Song Yuqing, Lin Yu, Wang Xiuli. Advances in research and application of breast cancer organoids [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1122-1128. |
| [10] | Wang Shiqi, Zhang Jinsheng. Effects of Chinese medicine on proliferation, differentiation and aging of bone marrow mesenchymal stem cells regulating ischemia-hypoxia microenvironment [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1129-1134. |
| [11] | Zeng Yanhua, Hao Yanlei. In vitro culture and purification of Schwann cells: a systematic review [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1135-1141. |
| [12] | Kong Desheng, He Jingjing, Feng Baofeng, Guo Ruiyun, Asiamah Ernest Amponsah, Lü Fei, Zhang Shuhan, Zhang Xiaolin, Ma Jun, Cui Huixian. Efficacy of mesenchymal stem cells in the spinal cord injury of large animal models: a meta-analysis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1142-1148. |
| [13] | Hou Jingying, Yu Menglei, Guo Tianzhu, Long Huibao, Wu Hao. Hypoxia preconditioning promotes bone marrow mesenchymal stem cells survival and vascularization through the activation of HIF-1α/MALAT1/VEGFA pathway [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 985-990. |
| [14] | Shi Yangyang, Qin Yingfei, Wu Fuling, He Xiao, Zhang Xuejing. Pretreatment of placental mesenchymal stem cells to prevent bronchiolitis in mice [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 991-995. |
| [15] | Liang Xueqi, Guo Lijiao, Chen Hejie, Wu Jie, Sun Yaqi, Xing Zhikun, Zou Hailiang, Chen Xueling, Wu Xiangwei. Alveolar echinococcosis protoscolices inhibits the differentiation of bone marrow mesenchymal stem cells into fibroblasts [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 996-1001. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||