Chinese Journal of Tissue Engineering Research ›› 2019, Vol. 23 ›› Issue (34): 5516-5522.doi: 10.3969/j.issn.2095-4344.1447
Previous Articles Next Articles
Preparation of 3D printed bone tissue engineering scaffold
- Department of Orthopedics, Second Hospital of Jilin University, Changchun 130041, Jilin Province, China
-
Received:2019-06-25Online:2019-12-08Published:2019-12-08 -
Contact:Yang Xiaoyu, Chief Physician, Professor, Doctoral supervisor, Department of Orthopedics, Second Hospital of Jilin University, Changchun 130041, Jilin Province, China -
About author:Wang Kai, Master candidate, Department of Orthopedics, Second Hospital of Jilin University, Changchun 130041, Jilin Province, China -
Supported by:the National Natural Science Foundation of China, No. 81672263 (to YXY)
CLC Number:
Cite this article
Wang Kai, Zheng Shuang, Pan Su, Zhang Heng, Zhang Wei, Wang Haosheng, Yang Xiaoyu, Fu Chuan. Preparation of 3D printed bone tissue engineering scaffold[J]. Chinese Journal of Tissue Engineering Research, 2019, 23(34): 5516-5522.
share this article
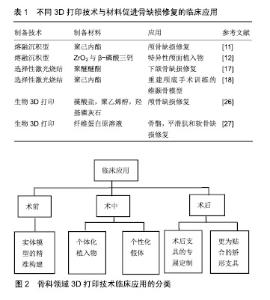
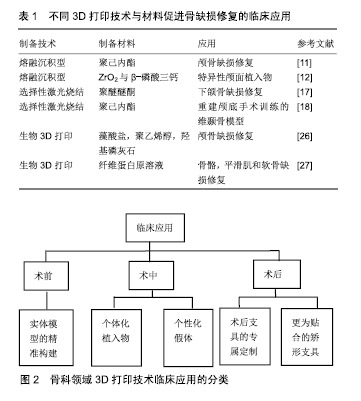
2.1 3D打印骨组织支架技术 目前应用于骨组织支架制备的3D打印技术主要有熔融沉积成型、选择性激光烧结、光固化成型、电子束熔融和近年来关注度比较大的生物3D打印等。 2.1.1 熔融沉积成型 熔融沉积成型技术主要将材料处理成熔融态后,在计算机控制下利用三维喷头逐层挤出材料,最后材料凝固成型形成三维产品。Jensen等[11]以聚已内酯为材料,应用熔融沉积技术制备了多孔3D印刷支架并应用于猪颅盖缺陷研究,发现骨缺损处表现出良好的骨连续性。Abdullah等[12]通过融合沉积技术以氧化锆及β-磷酸三钙制备生物陶瓷牙,发现该特异性颅面植入物具有良好的机械强度及生物相容性。这种技术的主要特点是工艺简单,并且成本低廉,但在打印产品精度和成型效果方面并不理想。另外,因打印过程中需对材料进行高温融溶处理,在高温状态下无法将材料与活细胞或温度敏感生物制剂结合进行打印[13-14]。 2.1.2 选择性激光烧结 选择性激光烧结所选材料多为粉末状物质,以红外激光器作为能源将平面上的粉末材料进行加热,促使材料间的分子扩散,完成颗粒材料之间的黏结,以此方式为基础在计算机控制下逐层对材料进行烧结,直至得到三维实体模型[15-16]。相对于熔融沉积成型方式,选择性激光烧结可选择的打印材料更为广泛,可利用金属粉或无机粒子等材料进行打印。Roskies等[17]利用选择性激光烧结技术制备3D梯形多孔支架并植入新西兰兔下颌骨缺损处,发现支架在颌骨重建方面具有很好的效果。Wanibuchi等[18]通过选择性激光烧结技术重建颅底手术训练的三维颞骨模型,该模型与手术患者拥有高度特异性,对于术者术前的规划有很大指导意义。选择性激光烧结与熔融沉积成型技术具有相同的缺点,即高加工温度过程中会破坏生物材料或生长因子的性能,因此不能用于结合活细胞等方面的打印[19-20]。 2.1.3 光固化成型 光固化成型是一种以数字模型为基础,通过逐层打印液态光敏树脂来构造物体的技术。光固化成型的优势包括制作周期短、制作领域广、打印精度高等。光固化成型为快速制作、批量制作、精度需求大等问题提供了新的解决途径。Zou等[21]通过对比光固化成型制备的模型尺寸与解剖学参数来确定光固化成型技术的精确度,结果证明二者之间并未存在明显差异,光固化成型3D打印模型在复杂骨科疾病诊断和治疗过程中的应用是可靠和准确的。Han等[22]针对膝关节肿瘤假体周围股骨骨折收集双侧股骨参数,以树脂为材料,应用光固化成型技术定制了股骨树脂模型并应用于病损处修复,术后X射线影像学显示位置良好,同时随访27个月,患者功能恢复良好。尽管光固化成型技术存在上述诸多优势,但是其所存在的问题同样突出:一方面,打印设备外形结构占地面积大,内部空间利用率低;另一方面,由于光敏树脂的制约导致打印材料严重受限,而且有研究表明部分打印材料对人体存在危害。 2.1.4 电子束熔融 电子束熔融技术是一种结合数控、电子设计、高能电子束等前沿技术而发展起来的当代先进技术,首先分层处理零件的三维数字模型,获得其二维截面信息,然后运用电子束熔融专用程序将有效信息导入电子束熔融设备中,以逐层增加材料的方式获得所需的三维产品。由于电子束具有功率高、扫描速度快、清洁无污染等特点,使得电子束熔融技术具有成形效率高、安全环保、适用材料范围广等优点,尤其对于难加工处理的金属材料复杂成型具有独特优势。Zhang等[23]应用电子束熔融技术制备钛金属骨小梁重建系统,用于治疗股骨头早期坏死,患者术后髋关节功能恢复良好,远期随访疗效令人满意。植入物中孔隙的存在有利于骨缺损生成,但是不同部位对孔隙要求并不一致。电子束熔融技术“复制重现”的特点使其成为制备多孔材料的不二之选。Palmquist等[24]通过电子束熔融技术制备多孔植入物并植入绵羊双侧股骨及背部,26周后取出植入物和周围组织,表现出优异的长期软组织生物相容性和高度的骨整合效果。电子束熔融技术所存在的问题包括技术难度高,造价成本高,配套软件并不完备。随着数控、电子信息、新型材料等领域的发展,电子束熔融技术也将被推向基层医院面向大众。 2.1.5 生物3D打印 以上所提到的传统3D打印技术,虽然可以精确地控制三维产物的结构与形状,但因打印过程中材料需进行高温或特定处理,因此无法实现打印支架材料与细胞、生长因子等的结合。近年来随着3D打印技术的不断革新,以可吸收材料、细胞和活性因子为基础的生物3D打印技术逐渐成为人们较为关注的一种打印方式。与传统打印方式相比,生物3D打印可以直接用于打印细胞、蛋白或生长因子等。Jakab等[25]在2008年通过3D打印技术打印出了可进行搏动的心脏组织。 de la Vega等[26]采用生物3D打印技术对人诱导的多能干细胞进行生物打印,该印刷方法保持了高水平的细胞活力(>81%)和分化能力。目前常用的生物3D打印技术主要是将细胞与材料共混后进行打印,因此如何保持细胞在材料内的活性是一个至关重要的问题,这也使能被用于生物3D打印的材料较为有限,较为常见的材料主要为具有良好生物降解性和相容的可聚合水凝胶,如藻酸盐[27]、胶原[28]、聚乙二醇等[29]。Bendtsen等[30]研究了一种新型的藻酸盐-聚乙烯醇-羟基磷灰石水凝胶制剂,并与小鼠胚胎成骨细胞前体细胞MC3T3混合后进行3D打印,MC3T3细胞在整个过程表现出高活力。这种新型骨传导性、可生物降解藻酸盐-聚乙烯醇-羟基磷灰石制剂的开发及其3D生物打印组织工程支架的能力,使其成为治疗个性化骨缺陷的有利候选者。也有研究以凝血原理为基础,将含有活细胞的凝血酶溶液喷入以纤维蛋白原溶液为基质的生物纸中,利用原位凝固形式制备含有活细胞的纤维蛋白支架。这种方式不仅可以实现活体细胞的3D打印,而且纤维蛋白本身具有促进血管生成的特性,其所制备的生物支架是骨骼、平滑肌和软骨损伤修复的理想材料[31]。然而近几年3D生物打印技术进入了瓶颈阶段,作者认为制约其发展的因素主要有2个层面:在技术层面上,存在缺乏高生物相容性的生物墨水,打印精度仍无法完全达到真实的组织结构微米级的分辨率等难题;在临床应用层面上,虽然生物3D打印在细胞乃至组织器官制造方面广泛应用,但是所打印的器官大部分还是结构上或者单一功能的实现,复杂器官的打印仍未彻底实现。 2.2 3D打印骨组织支架使用材料 骨支架材料的生物相容性、可塑性、骨诱导型和力学稳定性是影响骨组织愈合的关键。目前较常使用的3D打印材料主要为金属、生物陶瓷、高分子聚合物和多种材料构成的复合材料[32]。 2.2.1 金属材料 金属材料是临床应用较为广泛的一种3D打印材料,最为常见的为钛合金,其因具有质量轻、强度高等特性被广泛应用于临床骨缺损的治疗。由钛合金材料制备的多孔骨支架具备良好的生物相容性,在促进成骨细胞增殖和分化方面具有非常好的作用[33]。夏德林等[34]利用CAD/CAM技术针对26例颅骨缺损患者个性化制备钛合金修复假体,术后随访观察发现假体组织相容性良好,感染和假体外漏的发生率在极大程度上降低。金属材料虽然应用最为广泛,但打印过程需在高温下进行,且在长期保持细胞活性及功能方面还有待进一步研究。 2.2.2 生物陶瓷 生物陶瓷类材料是以磷酸三钙、磷酸钙、羟基磷灰石等为代表的一类骨传导能力良好的骨修复材料,这类材料主要成分与人骨无机物成分类似,具有较好的可降解性及促进新骨形成能力[35]。由双相磷酸钙(羟基磷灰石和磷酸三钙的混合物)为材料制备的骨支架,对细胞具有极好的促成骨分化能力[36]。Wang等[37]将羟基磷灰石与壳聚糖进行共混后,通过3D打印技术制备羟基磷灰石/壳聚糖复合多孔支架,并向支架内添加Ⅰ型胶原蛋白,通过动物实验发现添加Ⅰ型胶原的羟基磷灰石/壳聚糖复合多孔支架能够在极大程度上提高碱性磷酸酶的分泌,促进成骨。孙梁等[38]将聚乳酸-聚羟乙酸和磷酸三钙共混后,通过3D打印技术制备成磷酸三钙/聚乳酸-聚羟乙酸复合支架,并向支架内引入人骨形态发生蛋白,通过动物实验证明这种骨支架成功促进了15 mm骨缺损的修复。虽然生物陶瓷类材料在骨诱导能力、抗压性等方面具有优越的性能,但其与金属材料类似,同样需要在高温下进行打印,从而不能在打印过程中结合生长因子等活性分子,另外生物陶瓷类材料过高的脆性也限制着这类材料在骨组织工程的应用[39]。 2.2.3 聚合物材料 聚合物材料主要分为天然高分子聚合物和人工合成高分子聚合物2种。天然高分子聚合物主要为胶原、纤维蛋白和藻酸盐等一类生物相容性好的天然物质。这类物质因水溶性较好可与细胞或生长因子等相结合进行打印,但其所制备的材料机械强度较差且难以大量获取[40]。人工合成高分子材料主要为聚乳酸、聚已内酯等,这类材料的优点是可灵活地调节材料的机械强度和降解速度,并且可以大批量合成[41]。聚乳酸和聚乳酸-聚羟乙酸为目前已被FDA批准的医用高分子材料。以聚已内酯/聚乳酸-聚羟乙酸为打印材料,通过生物打印技术制备3D多孔支架,在细胞实验过程中支架能够很好的地维持初始结构[42]。Chou等[43]利用3D打印技术制备了一种可嵌入抗生素的聚乳酸/聚乳酸-聚羟乙酸多孔支架并用于兔股骨干重建,结果发现支架植入部位具有更好的皮质完整性、最大弯曲强度及软骨增殖效果。然而人工合成高分子也存在一定缺陷,例如这类聚合物亲水性较差,并且制备支架过程中可能需要有机溶剂的参与,有机溶剂的残留可能会对细胞活性造成一定影响[44]。 2.2.4 复合型材料 为达到制备完美3D打印骨组织修复材料的要求,克服目前打印材料所存在的缺陷,高分子聚合物和金属、生物陶瓷等2种或多种材料相结合的复合材料,成为了3D打印骨材料的新突破口。高分子聚合物与生物陶瓷构成的骨组织支架因与天然骨基质类似,被广泛应用于3D打印骨修复材料中[45]。Matsuo等[46]以人工合成高分子聚合物聚乳酸和羟基磷灰石为原料,制备可吸收多孔托架并用于下颌骨的重建,通过对比发现:相比传统的钛合金托架,聚合物聚乳酸/羟基磷灰石托架具有更好的修复效果。一些研究也发现将磷酸三钙与高分子聚合物共混后制备的材料,能够在很大成度上促进人间充质干细胞的增殖与分化[47-48]。另外,复合材料结合具有成骨诱导作用的细胞因子、蛋白等可进一步提升3D打印骨支架在骨修复方面的作用。目前,国内并没有3D打印材料的统一制定标准,许多材料仍然依靠进口,进而造成了成本较高、使得3D打印技术难以广泛化应用于基层医院并服务于大众。因此,当前对高分子3D打印材料及其打印技术的研发应当逐步进入体系化、标准化的轨道,并完善应用标准,一方面改进原有材料,另一方面积极研发新型材料。 2.3 3D打印骨组织支架在骨缺损修复中的临床应用 目前,3D打印在骨科领域的临床应用主要有以下几大类:术前实体模型的精准构建;术中个性化内植物或假体植入;术后支具或矫形支具的设计。首先,完整的术前规划是骨科手术中至关重要的一环。传统的术前规划仅限于X射线、CT、MRI、三维重建影像技术等,而这些传统的技术在面对复杂情况时往往力不从心。3D打印实体模型可以帮助术者了解患者的异常解剖结构,从而制定最佳的手术入路及术式,提高精准性与安全性[49-50]。Debarre等[51]利用3D打印术前实体模型为手术提供了更好的解决方案,更好的实现了骨骺畸形愈合的截骨术、肩关节置换术和股骨滑车成形术。丁焕文等[52]对106例骨病患者进行术前建模以模拟手术,结果证明手术时间明显缩短,出血量减少,最大程度上简化了手术过程。 临床上传统的骨缺损治疗方式是向缺损部位植入不同材质的内植物或假体,但因为不同人群差异和损伤大小的不同,传统植入物很难与损伤部位完全匹配,3D打印技术完美地解决了这一问题,目前已经有大量3D打印产品投入到骨缺损治疗的临床应用中。Kozakiewicz等[53]根据患者损伤情况通过3D打印技术个性化设计钛合金内植物,通过手术固定眶底骨折,术后观察患者恢复情况良好。张文友等[54]成功应用3D打印技术构建韧带-骨复合支架,将支架与动物前交叉韧带进行替代实验,发现该支架在韧带-骨界面修复方面具有非常好的效果。3D打印技术制备的个体化植入物因与患者更为匹配,也在很大程度上缩短了手术和恢复时间,为患者快速康复创造了有利条件。Won 等[55]针对21例髋关节畸形患者,利用3D打印技术制备个体化人工全髋关节,手术时间明显缩短,并且假体植入的精确度有了很大程度提高。但是内植物或假体的制造过程时间过长,相关灭菌技术由于材料的制约而受限等因素导致患者有感染的风险。上述文章并未提出该弊端的存在。康复训练对于骨科术后患者的恢复尤为重要。支具的合适与否直接决定着康复的效果。传统制作支具无法满足个体差异的存在,治疗效果有限。3D打印所设计生产的支具,能够在保证疗效的前提下提高舒适度,进而增加患者的依从性。踝足矫形器、腕关节和脊柱矫形器等已被广泛应用于医疗保健及康复行业。Weiss等[56]认为针对脊柱侧弯个性化患者,使用3D打印技术制备的支具支配成功率及治疗有效率均高于传统支具,并且极大改善了穿着舒适度和患者依从性,进而提高了矫形效果。但是文章并未对支具的受力情况做定量分析。作者认为可以在支具中加入6个方向的测力传感器,进而动态记录患者恢复及矫正情况,进而同步调整支具,以期达到更好的疗效。不同3D打印技术与材料促进骨缺损修复的临床应用,见表1。3D打印技术在骨科领域临床应用的分类,见图2。 "
| [1]Liu M, Yu X, Huang FG, et al.Tissue engineering stratified scaffolds for articular cartilage and subchondral bone defects repair.Orthopedics.2013;36(11):868-873.[2]Zhu M,Zhang JH,Zhao SC,et al.Three-dimensional printing of cerium-incorporated mesoporous calcium-silicate scaffolds for bone repair.J Mater Sci.2016;51(2):836-844.[3]Placone JK,Engler AJ.Recent Advances in Extrusion-Based 3D Printing for Biomedical Applications. Adv Healthc Mater. 2018;7(8):e1701161.[4]Eltorai AE,Nguyen E,Daniels AH.Three-Dimensional Printing in Orthopedic Surgery.Orthopedics.2015;38(11): 684-687.[5]Kim JW,Lee Y,Seo J,et al.Clinical experience with three-dimensional printing techniques in orthopedic trauma.J Orthop Sci.2018;23(2):383-388.[6]Xie L,Chen C,Zhang Y,et al.Three-dimensional printing assisted ORIF versus conventional ORIF for tibial plateau fractures: A systematic review and meta-analysis.Int J Surg. 2018;57:35-44. [7]Pae HC,Kang JH,Cha JK,et al.3D-printed polycaprolactone scaffold mixed with beta-tricalcium phosphate as a bone regenerative material in rabbit calvarial defects. J Biomed Mater Res B. 2019;107(4):1254-1263.[8]Singh AV,Ansari MHD,Wang S,et al.The Adoption of Three-Dimensional Additive Manufacturing from Biomedical Material Design to 3D Organ Printing.Appl Sci-Basel. 2019; 9(4).[9]Bittner S,Smith B,Melchiorri A,et al.Fabrication of 3D-Printed, Bidirectional Growth Factor Gradient Scaffolds for Osteochondral Tissue Repair.Tissue Eng Pt A. 2017;23: S38-S39.[10]Sawkins M,Brown B,Bonassar L,et al.Cell, scaffold and growth factor patterning via 3D printing. J Tissue Eng Regen M. 2012; 6:374.[11]Jensen J,Rolfing JHD,Le DQS,et al.Surface-modified functionalized polycaprolactone scaffolds for bone repair: In vitro and in vivo experiments.J Biomed Mater Res A. 2014; 102(9):2993-3003.[12]Abdullah AM,Rahim TNAT,Hamad WNFW,et al.Mechanical and cytotoxicity properties of hybrid ceramics filled polyamide 12 filament feedstock for craniofacial bone reconstruction via fused deposition modelling.Dent Mater. 2018;34(11):E309-E316.[13]Ilyes K,Kovacs NK,Balogh A,et al.The applicability of pharmaceutical polymeric blends for the fused deposition modelling (FDM] 3D technique: Material considerations- printability-process modulation, with consecutive effects on in vitro release, stability and degradation.Eur J Pharm Sci. 2019; 129:110-123.[14]Linares V, Casas M,Caraballo I. Printfills: 3D printed systems combining fused deposition modeling and injection volume filling. Application to colon-specific drug delivery.Eur J Pharm Biopharm. 2019;134:138-143.[15]Chen JB,Wan N, Li JY,et al.Study on the polymer material infiltrating metallic parts by selective laser sintering of 3D printing.Rapid Prototyping J.2018;24(9):1539-1543.[16]Lahtinen E,Precker RLM,Lahtinen M,et al.Selective Laser Sintering of Metal-Organic Frameworks: Production of Highly Porous Filters by 3D Printing onto a Polymeric Matrix. Chempluschem. 2019;84(2):222-225.[17]Roskies MG,Fang DD,Abdallah MN,et al.Three-Dimensionally Printed Polyetherketoneketone Scaffolds With Mesenchymal Stem Cells for the Reconstruction of Critical-Sized Mandibular Defects. Laryngoscope. 2017;127(11):E392-E398.[18]Wanibuchi M,Noshiro S,Sugino T,et al.Training for Skull Base Surgery with a Colored Temporal Bone Model Created by Three-Dimensional Printing Technology.World Neurosurg. 2016;91:66-72.[19]Eshraghi S,Das S.Micromechanical finite-element modeling and experimental characterization of the compressive mechanical properties of polycaprolactone-hydroxyapatite composite scaffolds prepared by selective laser sintering for bone tissue engineering.Acta Biomater.2012;8(8):3138-3143.[20]Wang C,Zhao QL,Wang M.Cryogenic 3D printing for producing hierarchical porous and rhBMP-2loaded Ca-P/PLLA nanocomposite scaffolds for bone tissue engineering. Biofabrication.2017;9(2):025031.[21]Zou Y,Han Q,Weng X,et al.The precision and reliability evaluation of 3-dimensional printed damaged bone and prosthesis models by stereo lithography appearance. Medicine (Baltimore].2018;97(6):e9797.[22]Han Q,Zhao X,Wang C,et al.Individualized reconstruction for severe periprosthetic fractures around the tumor prosthesis of knee under assistance of 3D printing technology: A case report. Medicine (Baltimore).2018;97(42):e12726.[23]Zhang Y,Zhang L,Sun R,et al.A new 3D printed titanium metal trabecular bone reconstruction system for early osteonecrosis of the femoral head.Medicine(Baltimore).2018;97(26):e11088.[24]Palmquist A,Snis A,Emanuelsson L,et al. Long-term biocompatibility and osseointegration of electron beam melted, free-form-fabricated solid and porous titanium alloy: Experimental studies in sheep.J Biomater Appl. 2013;27(8): 1003-1016.[25]Jakab K,Norotte C,Damon B,et al.Tissue engineering by self-assembly of cells printed into topologically defined structures.Tissue Eng Pt A.2008;14(3):413-421.[26]de la Vega L, Gomez DAR, Abelseth E,et al.3D Bioprinting Human Induced Pluripotent Stem Cell-Derived Neural Tissues Using a Novel Lab-on-a-Printer Technology. Appl Sci-Basel. 2018;8(12).https://doi.org/10.3390/app8122414[27]Axpe E,Oyen ML.Applications of Alginate-Based Bioinks in 3D Bioprinting.Int J Mol Sci. 2016;17(12).pii: E1976. Review.[28]Osidak EO,Karalkin PA,Osidak MS,et al.Viscoll collagen solution as a novel bioink for direct 3D bioprinting.J Mater Sci Mater Med.2019;30(3):31.[29]Zheng ZZ,Wu JB,Liu M,et al.3D Bioprinting of Self-Standing Silk-Based Bioink.Adv Healthc Mater. 2018;7(6):e1701026. [30]Bendtsen ST,Quinnell SP,Wei M.Development of a novel alginate-polyvinyl alcohol-hydroxyapatite hydrogel for 3D bioprinting bone tissue engineered scaffolds.J Biomed Mater Res A. 2017;105(5):1457-1468.[31]Cui X,Boland T.Human microvasculature fabrication using thermal inkjet printing technology. Biomaterials. 2009;30(31): 6221-6227.[32]Zhang L,Yang GJ,Johnson BN,et al.Three-dimensional (3D] printed scaffold and material selection for bone repair.Acta Biomater.2019;84:16-33.[33]Das S,Bourell DL,Babu SS.Metallic materials for 3D printing. Mrs Bull.2016;41(10):729-741.[34]夏德林,付光新,马征,等.CAD/CAM预制个体化钛合金修复体颅骨缺损[J].中国组织工程研究与临床康复, 2010,14(39): 7254-7258.[35]Ma HS,Feng C,Chang J,et al.3D-printed bioceramic scaffolds: From bone tissue engineering to tumor therapy.Acta Biomater. 2018;79:37-59.[36]Shim KS,Kim SE,Yun YP,et al.Surface immobilization of biphasic calcium phosphate nanoparticles on 3D printed poly(caprolactone) scaffolds enhances osteogenesis and bone tissue regeneration. J Ind Eng Chem.2017;55:101-109.[37]Wang H,Wu G,Zhang J,et al.Osteogenic effect of controlled released rhBMP-2 in 3D printed porous hydroxyapatite scaffold.Colloid Surface B.2016;141:491-498.[38]孙梁,熊卓.快速成型聚乳酸-聚羟乙酸/磷酸三钙支架修复兔桡骨缺损[J].中国组织工程研究与临床康复, 2011,15(12):2091-2094.[39]Inzana JA,Olvera D,Fuller SM,et al.3D printing of composite calcium phosphate and collagen scaffolds for bone regeneration. Biomaterials.2014;35(13):4026-4034.[40]于强,田京.构建骨组织工程支架中应用的3D打印技术[J].中国组织工程研究,2015,19(30):4870-4875.[41]Rocha CR,Perez ART,Roberson DA,et al.Novel ABS-based binary and ternary polymer blends for material extrusion 3D printing. J Mater Res.2014;29(17):1859-1866.[42]Kim BS,Jang J,Chae S,et al.Three-dimensional bioprinting of cell-laden constructs with polycaprolactone protective layers for using various thermoplastic polymers.Biofabrication. 2016; 8(3):035013.[43]Chou YC,Lee D,Chang TM,et al.Combination of a biodegradable three-dimensional (3D) - printed cage for mechanical support and nanofibrous membranes for sustainable release of antimicrobial agents for treating the femoral metaphyseal comminuted fracture.J Mech Behav Biomed.2017;72:209-218.[44]Lao LH,Wang YJ,Zhu Y,et al.Poly (lactide-co-glycolide)/ hydroxyapatite nanofibrous scaffolds fabricated by electrospinning for bone tissue engineering. J Mater Sci-Mater M.2011;22(8):1873-1884.[45]Xia ZY,Hou JY,Li K.Composite materials and bone tissue engineering in sports injury.Adv Mater Res Switz. 2012;583: 91-94.[46]Matsuo A,Chiba H,Takahashi H,et al.Clinical application of a custom-made bioresorbable raw particulate hydroxyapatite/ poly-l-lactide mesh tray for mandibular reconstruction. Odontology. 2010;98(1):85-88.[47]Rai B,Lin JL,Lim ZXH,et al.Differences between in vitro viability and differentiation and in vivo bone-forming efficacy of human mesenchymal stem cells cultured on PCL-TCP scaffolds. Biomaterials. 2010;31(31):7960-7970.[48]Kim J,McBride S,Tellis B,et al.Rapid-prototyped PLGA/beta-TCP/hydroxyapatite nanocomposite scaffolds in a rabbit femoral defect model.Biofabrication.2012;4(2):025003.[49]Corona PS,Vicente M,Tetsworth K,et al. Preliminary results using patient-specific 3d printed models to improve preoperative planning for correction of post-traumatic tibial deformities with circular frames.Injury.2018;49:S51-S59.[50]Dekker TJ,Steele JR,Federer AE,et al.Use of Patient-Specific 3D-Printed Titanium Implants for Complex Foot and Ankle Limb Salvage, Deformity Correction, and Arthrodesis Procedures. Foot Ankle Int. 2018;39(8):916-921.[51]Debarre E,Hivart P,Baranski D,et al.Speedy skeletal prototype production to help diagnosis in orthopaedic and trauma surgery.Methodology and examples of clinical applications. Orthop Traumatol Surg Res.2012;98(5):597-602.[52]丁焕文,沈健坚,涂强,等.计算机辅助技术在骨关节疾病中的应用[J].中国组织工程研究与临床康复,2011,15(17):3113-3118.[53]Kozakiewicz M,Elgalal M,Loba P,et al.Clinical application of 3D pre-bent titanium implants for orbital floor fractures.J Cranio Maxill Surg.2009;37(4):229-234.[54]张文友,贺健康,李翔,等.基于3-D打印技术的韧带-骨复合支架制造与体内植入研究[J].中国修复重建外科杂志, 2014,28(3): 314-317.[55]Won SH,Lee YK,Ha YC,et al.Improving pre-operative planning for complex total hip replacement with a Rapid Prototype model enabling surgical simulation.Bone Joint J. 2013;95b(11): 1458-1463.[56]Weiss HR, Tournavitis N, Nan X,et al.Workflow of CAD / CAM Scoliosis Brace Adjustment in Preparation Using 3D Printing. Open Med Inform J.2017;11:44-51. |
| [1] | Chen Jinping, Li Kui, Chen Qian, Guo Haoran, Zhang Yingbo, Wei Peng. Meta-analysis of the efficacy and safety of tranexamic acid in open spinal surgery [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1458-1464. |
| [2] | Hua Haotian, Zhao Wenyu, Zhang Lei, Bai Wenbo, Wang Xinwei. Meta-analysis of clinical efficacy and safety of antibiotic artificial bone in the treatment of chronic osteomyelitis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(6): 970-976. |
| [3] | Zhan Fangbiao, Cheng Jun, Zou Xinsen, Long Jie, Xie Lizhong, Deng Qianrong. Intraoperative intravenous application of tranexamic acid reduces perioperative bleeding in multilevel posterior spinal surgery: a meta-analysis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(6): 977-984. |
| [4] | Zhang Bin, Sun Lihua, Zhang Junhua, Liu Yusan, Cui Caiyun. A modified flap immediate implant is beneficial to soft tissue reconstruction in maxillary aesthetic area [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 707-712. |
| [5] | Li Chenjie, Lü Linwei, Song Yang, Liu Jingna, Zhang Chunqiu. Measurement and statistical analysis of trabecular morphological parameters of titanium alloy peri-prosthesis under preload [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 516-520. |
| [6] | Wu Zijian, Hu Zhaoduan, Xie Youqiong, Wang Feng, Li Jia, Li Bocun, Cai Guowei, Peng Rui. Three-dimensional printing technology and bone tissue engineering research: literature metrology and visual analysis of research hotspots [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 564-569. |
| [7] | Li Xiaozhuang, Duan Hao, Wang Weizhou, Tang Zhihong, Wang Yanghao, He Fei. Application of bone tissue engineering materials in the treatment of bone defect diseases in vivo [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 626-631. |
| [8] | Li Jun, Zuo Xinhui, Liu Xiaoyuan, Zhang Kai, Han Xiangzhen, He Huiyu, . Effect of over expression of miR-378a on osteogenic and vascular differentiation of bone marrow mesenchymal stem cell sheet [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(31): 4939-4944. |
| [9] | He Jie, Chang Qi. Biological reconstruction of large bone defects after resection of malignant tumor of extremities [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(3): 420-425. |
| [10] | Xing Hao, Zhang Yonghong, Wang Dong. Advantages and disadvantages of repairing large-segment bone defect [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(3): 426-430. |
| [11] | Kang Bo, Wang Jindong. Development status and strategy of outpatient total knee arthroplasty [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(27): 4408-4414. |
| [12] | Liu Jinyu, Ding Yiwei, Lu Zhengcao, Gao Tianjun, Cui Hongpeng, Li Wen, Du Wei, Ding Yu. Finite element biomechanical study of full endoscopic fenestration decompression for cervical spondylotic myelopathy [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(24): 3850-3854. |
| [13] | Chen Siqi, Xian Debin, Xu Rongsheng, Qin Zhongjie, Zhang Lei, Xia Delin. Effects of bone marrow mesenchymal stem cells and human umbilical vein endothelial cells combined with hydroxyapatite-tricalcium phosphate scaffolds on early angiogenesis in skull defect repair in rats [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3458-3465. |
| [14] | Zhou Anqi, Tang Yufei, Wu Bingfeng, Xiang Lin. Designing of periosteum tissue engineering: combination of generality and individuality [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3551-3557. |
| [15] | Chen Song, He Yuanli, Xie Wenjia, Zhong Linna, Wang Jian. Advantages of calcium phosphate nanoparticles for drug delivery in bone tissue engineering research and application [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3565-3570. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||