Chinese Journal of Tissue Engineering Research ›› 2023, Vol. 27 ›› Issue (7): 1126-1132.doi: 10.12307/2023.031
Previous Articles Next Articles
Piezoelectric materials for vascularized bone regeneration
Xu Yan, Li Ping, Lai Chunhua, Zhu Peijun, Yang Shuo, Xu Shulan
- Stomatology Hospital, Southern Medical University, Guangzhou 510280, Guangdong Province, China
-
Received:2021-12-29Accepted:2022-02-11Online:2023-03-08Published:2022-07-20 -
Contact:Xu Shulan, Master, Chief physician, Stomatology Hospital, Southern Medical University, Guangzhou 510280, Guangdong Province, China -
About author:Xu Yan, Master candidate, Stomatology Hospital, Southern Medical University, Guangzhou 510280, Guangdong Province, China -
Supported by:Fundamental and Applied Basic Research General Project of Guangzhou Science and Technology Bureau, No. 202102080148 (to XSL); Traditional Chinese Medicine Research Project of Guangdong Provincial Bureau of Traditional Chinese Medicine, No. 20211274 (to XSL); Cultivation Plan Project of Stomatology Hospital of Southern Medical University, No. PY2020011 (to XSL)
CLC Number:
Cite this article
Xu Yan, Li Ping, Lai Chunhua, Zhu Peijun, Yang Shuo, Xu Shulan. Piezoelectric materials for vascularized bone regeneration[J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1126-1132.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
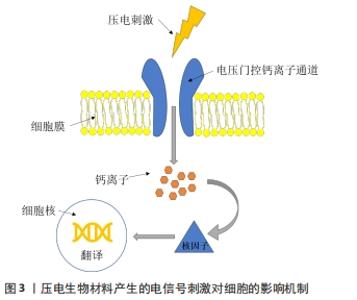
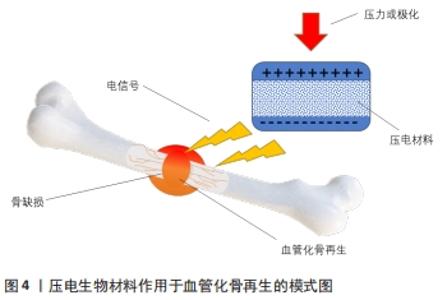
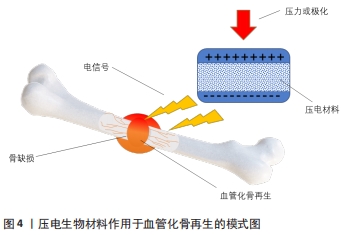
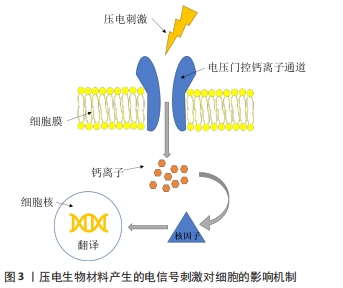
2.1 压电材料应用的生物学基础——血管、骨组织的天然压电特性 骨组织及血管组织的压电性主要源于他们的生理组成结构,总的来说可以归因于组成结构中胶原分子结晶胶束的压电效应[14]。胶原蛋白分子主要由3个多肽链组成,通过氢键连接并扭曲形成三重螺旋结构[15-16]。因此,它可以表现为晶体并通过涉及-NH和-CO基团的偶极子取向产生压电效应,在机械力的作用下,胶原蛋白纤维相互滑动在人体组织中产生压电性[14,17-18]。 骨是由细胞和细胞外基质组成的致密结缔组织,其中细胞外基质中富含大量胶原蛋白[19-20]。胶原蛋白同样也是构成血管组织的主要成分,在基底膜下,正常血管含有不同数量的弹性纤维蛋白和胶原蛋白,所有血管腔均由锚定在底层基底膜上的内皮细胞、层粘连蛋白、Ⅳ型、XV型和XⅧ型胶原以及其他生物分子构成[21]。 胶原蛋白在压力形变后的相对滑动促使产生电信号,是血管、骨组织天然压电特性的内在结构基础。这些信号通过细胞外基质传输到细胞膜中的电压门控通道,而这些通道的激活会把细胞内信号传输到细胞核,导致信号级联的激活,负责如基质产生、细胞生长和组织修复等细胞事件的发生[22],详细机制见图3。具体来讲,外力触发胶原蛋白偶极矩的重组并在表面产生负电荷,产生的负电荷可以通过打开电压门控Ca2+通道,增加细胞内Ca2+浓度,激活钙调蛋白以促进核苷酸合成和细胞增殖[23-24]。与此同时,Ca2+浓度变化也激活了钙/钙调蛋白通路,使细胞核因子去磷酸化,将其易位到细胞核中并与其他转录因子结合,进一步诱导如转化生长因子β、骨形态发生蛋白等多种生长因子的翻译[25-26]。此外,骨组织在矿化过程中会产生具有压电性晶体结构的羟基磷灰石并将其沉淀在细胞外基质中。这些羟基磷灰石矿物晶体结构具有较高的弹性模量,可以承受更多胶原纤维产生压电效应所需的变形,促进了胶原蛋白的压电性[27-28]。另外,一些体外研究也证明了烧结羟基磷灰石具有良好的压电性能[29-31]。 "

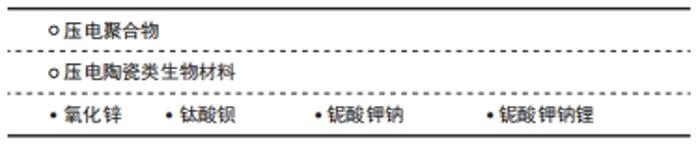
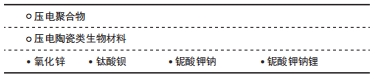
2.2.1 压电聚合物 目前常见的压电聚合物包括聚偏氟乙烯、聚偏二氟乙烯三氟乙烯等。相比无机材料,压电聚合物具有高强度和高抗性,还能够根据使用环境及需求的不同进行灵活加工,非常适用于组织工程研究。除此之外,压电聚合物还因为具有良好的生物相容性及促成骨能力而备受关注。 聚偏氟乙烯及其共聚物是一种具有良好压电性、生物相容性、不可生物降解的高分子聚合物。RIBEIRO等[32]使用聚偏氟乙烯作为原材料,在高温高压条件下极化后获得了具有电活性的聚偏氟乙烯支架,发现极化后的聚偏氟乙烯支架可以为人脂肪干细胞的成骨分化提供必要的电刺激,模仿体内存在的机械刺激环境,从而改善干细胞成骨向分化再生,这揭示了极化聚偏氟乙烯支架良好的骨诱导性能。P?RSSINEN等[33]也同样发现聚偏氟乙烯膜可以促进人脂肪干细胞成骨分化。此外,TEIXEIRA等[34]在关于新型聚偏二氟乙烯三氟乙烯/钛酸钡复合膜对人牙槽骨源性成骨细胞作用的研究中发现,成骨细胞与所制备的压电复合材料在成骨条件下培养7,14 d后绝大多数成骨标记物的表达均显著高于对照组。AHMADI等[35]制备的新型压电聚偏氟乙烯-聚偏氟乙烯-钛酸钡0.9钙0.1-聚乙烯醇(PVDF-Ba0.9 Ca0.1TiO3/PVA)材料在没有添加成骨诱导剂的情况下能诱导骨髓间充质干细胞成骨分化。 聚偏氟乙烯及其共聚物不仅在体外实验中表现出有效的促成骨能力,在体内动物骨缺损模型中同样表现出良好的骨诱导能力。课题组前期研究将聚偏氟乙烯膜植入Wistar大鼠体内后发现,负极化的聚偏氟乙烯膜具有良好的理化性能、生物相容性与体内促成骨能力[36]。BAI等[37]制造了与人体骨骼压电系数相近的柔性钛酸钡/聚偏氟乙烯-三氟乙烯压电纳米复合膜,并将其覆盖在兔下颌骨临界尺寸骨缺损后发现,这种压电复合膜显著增强骨再生并有效促进成熟骨结构的形成。RIBEIRO等[32]在体外实验中发现极化聚偏氟乙烯薄膜可以有效促进脂肪干细胞成骨分化,然后又将这种压电薄膜植入Wistar大鼠中,4周后发现植入极化β-聚偏氟乙烯薄膜组相比对照组缺损面积愈合更多,骨重塑改建也更好[38]。REIS等[39]将聚偏氟乙烯植入绵羊胫骨内,对所获组织切片进行免疫组化染色后发现,在植入材料附近骨桥蛋白的表达增加,有效刺激了植入物界面处的骨生长。 随着研究的不断深入,人们对聚偏氟乙烯及其聚合物促进骨组织再生的作用及原理机制也有了新的认识。与未极化的聚偏氟乙烯聚合物相比,极化后聚偏氟乙烯的成骨性能在体外实验及体内实验中都显著增加。ZHOU等[40]在钛基底上制备了电活性聚偏氟乙烯-钛膜并对其进行极化处理,结果表明聚偏氟乙烯-钛膜的极化改变了其表面电荷,从而诱导细胞的黏附、增殖和成骨分化。同样,RIBEIRO等[41]也发现极化后的聚偏氟乙烯可以在体外促进成骨细胞黏附和增殖。也有学者发现,材料中压电β相含量会影响成骨分化[42],可能是由于高含量β晶相的压电聚合物在极化后产生更多的定向电偶极子,优化了材料自身的电学性能。因此,未来有望通过这两种途径,对聚偏氟乙烯及其共聚物进行改良,在骨组织的修复再生中发挥更大作用。 以下针对目前常见的压电聚合物通过压电作用促进骨再生方面的研究及发现进行讨论总结,见表1[32-33,35-39,41-51]。 "

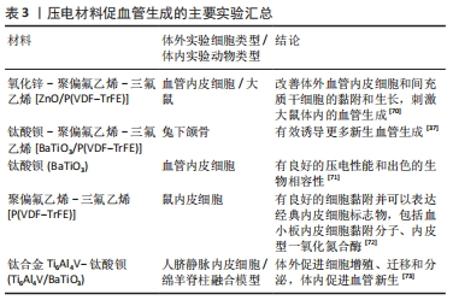
(1)氧化锌:在关于氧化锌压电陶瓷材料的应用中,有大量文献报告生物材料中氧化锌含量的增加会提高与成纤维细胞、成骨细胞、干细胞等的生物相容性。据报道,与不含氧化锌的支架相比,通过在聚己内酯-羟基磷灰石中加入1%氧化锌制备的纳米纤维支架对人成骨细胞系具有更强的骨活性[52]。LI等[53]通过静电纺丝增加氧化锌浓度和β相聚偏氟乙烯的比例,制备了掺杂氧化锌纳米粒子的聚偏氟乙烯支架,在体外实验中发现生长在压电支架上的成骨细胞密度增加了30%。AMNA等[54]将氧化锌颗粒和聚氨酯结合制成电纺纤维复合支架,可以改善小鼠成纤维细胞在复合支架上的附着和增殖。除了压电性,氧化锌压电陶瓷材料也可以通过持续释放的锌离子,调节各种细胞代谢活动[65]。 (2)钛酸钡:钛酸钡是早期应用于骨修复的无铅压电陶瓷材料,压电性能高效而稳定。以往研究工作也证明了钛酸钡具有良好的生物相容性,并发现它能够增强间充质干细胞的成骨分化[66-67]。此外,也有研究发现钛酸钡纳米颗粒的存在会显著增加成骨过程中羟基磷灰石沉积物的形成[68]。鉴于这一发现,目前钛酸钡多与羟基磷灰石材料复合使用,将羟基磷灰石的生物活性、铁电性与钛酸钡的压电性高效结合,增加材料的机械性能,同时又能够有效促进成骨。POLLEY等[55]结合3D打印工艺制造的多孔钛酸钡和羟基磷灰石复合支架表现出良好的细胞相容性、促细胞黏附性能以及更强的骨再生潜力。TANG等[57]使用注塑成型工艺制备了含不同比例羟基磷灰石的钛酸钡复合材料,并通过极化获得压电性能,在其设计的模拟人体骨骼动态环境的压电材料体外生物培养装置中,通过磷灰石沉积和成骨细胞培养实验研究了压电相含量对生物特性的影响。在静态空载条件下,钛酸钡含量的变化对羟基磷灰石/钛酸钡复合材料的生物相容性和骨诱导活性影响不大,但所有复合压电陶瓷的性能均低于纯羟基磷灰石支架;当羟基磷灰石/钛酸钡表面上施加周期性载荷后,其生物相容性和骨诱导活性显著提高且均高于纯羟基磷灰石支架;这一发现证实了将二者结合使用在动态条件下触发材料压电作用后确实会促进更好地成骨。随着研究的深入,人们发现孔隙率也会对羟基磷灰石/钛酸钡材料性能产生影响,LIU等[56]报道具有不同孔隙率的多孔羟基磷灰石/钛酸钡支架在体外促进了MG63成骨样细胞增殖、分化和黏附,其中当孔隙率为50%时整体表现最佳。EHTERAMI等[58]在传统钛酸钡支架中添加明胶/纳米羟基磷灰石涂层,保持原有多孔结构的同时提高了抗压强度和弹性模量,细胞培养实验表明,所研制的支架具有良好的生物相容性,细胞能够黏附、增殖和迁移到支架的孔隙中。 (3)铌酸钾钠、铌酸钾钠锂:铌酸钾钠和铌酸钾钠锂是具有强压电系数的无铅压电陶瓷,其压电系数远远大于骨组织,因此具有很高的应用价值,同时体外实验也证明了其良好的生物相容性,在铌酸钾钠材料上培养小鼠成骨细胞表现出良好的活力[62]。YU等[63]以铌酸钾钠为基础,模拟天然骨骼表面的微尺度压电区,在体外实验中能诱导小鼠骨髓间充质干细胞的成骨分化,在兔体内可以促进新骨生成。而最近关于这类压电陶瓷的研究发现,极化同样会影响材料的性能,YAO等[64]发现与未极化的铌酸钾钠相比,极化的铌酸钾钠能更好地引导骨髓间充质干细胞的迁移和增殖。与铌酸钾钠相比,铌酸钾钠锂的强度和压电系数提高,然而锂离子的释放导致铌酸钾钠锂的细胞毒性略高于铌酸钾钠,限制了它的发展及使用[69]。 2.3 压电材料在血管修复及内皮再生中的应用 血管组织的压电特性主要来源于细胞外基质中存在的胶原蛋白和弹性蛋白。有关电生理的研究发现血管内皮细胞也可以直接响应电信号刺激,因此使用压电材料,借助材料的电学特性促进血管再生大有可为,见表3[37,70-73]。 "
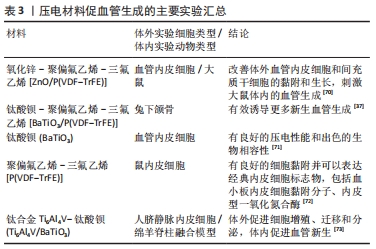

由于聚偏氟乙烯及其共聚物良好的压电性能和生物相容性,其在血管修复及内皮再生中同样被广泛应用。HITSCHERICH等[72]发现小鼠内皮细胞在聚偏二氟乙烯三氟乙烯支架上有良好的细胞黏附并可以表达血小板内皮细胞黏附分子、内皮型一氧化氮合酶等经典内皮细胞标志物。AUGUSTINE等[70]在聚偏二氟乙烯三氟乙烯支架中添加氧化锌纳米颗粒后,在体外促进了血管内皮细胞和间充质干细胞的黏附和生长,在体内也刺激大鼠的血管生成。BAI等[37]制造了与人体骨骼压电系数相近的柔性钛酸钡/聚偏二氟乙烯-三氟乙烯压电纳米复合膜,植入兔骨缺损处诱导产生了更多的新生血管。 无铅压电陶瓷良好的生物相容性、机械性能使得它在血管修复及内皮再生中同样表现出色。研究显示掺杂钛酸钡压电陶瓷纳米颗粒的压电复合血管移植物有良好的压电性能和出色的生物相容性[71]。LIU等[73]开发了一种由多孔钛合金Ti6Al4V和钛酸钡组成的生物活性复合支架,这种复合支架不仅具有合适的压电性能还能够重建电磁微环境,人脐静脉内皮细胞在支架材料上增殖和迁移以及分泌因子的增加反映了支架支持血管化的能力,使用绵羊脊柱融合模型在体内同样证明了这一点。此外,氧化锌压电材料在促进血管生成中也有不俗的表现,有研究推断可能是因为材料中主要成分氧化锌可以增加促血管再生的活性氧生成,从而促进了血管新生[65]。 总而言之,体外细胞实验和体内动物研究的结果均表明,压电材料在血管新生方面同样可以发挥作用,有利于血管生成,具有巨大的临床转化潜力,是一种很有前途的复合生物材料。 "

| [1] KARALASHVILI L, KAKABADZE A, UHRYN M, et al. Bone grafts for reconstruction of bone defects. Georgian Med News. 2018;(282):44-49. [2] SCHROEDER JE, MOSHEIFF R. Tissue engineering approaches for bone repair: Concepts and evidence. Injury. 2011;42(6):609-613. [3] LIU Y, LIM J, TEOH SH. Review: development of clinically relevant scaffolds for vascularised bone tissue engineering. Biotechnol Adv. 2013;31(5):688-705. [4] DIOMEDE F, MARCONI GD, FONTICOLI L, et al. Functional relationship between osteogenesis and angiogenesis in tissue regeneration. Int J Mol Sci. 2020;21(9):3242. [5] SARAN U, GEMINI PIPERNI S, CHATTERJEE S. Role of angiogenesis in bone repair. Arch Biochem Biophys. 2014;561:109-117. [6] SABERIANPOUR S, HEIDARZADEH M, GERANMAYEH MH, et al. Tissue engineering strategies for the induction of angiogenesis using biomaterials. J Biol Eng. 2018;12(1):36. [7] ZHANG ZY, TEOH SH, CHONG MS, et al. Neo-vascularization and bone formation mediated by fetal mesenchymal stem cell tissue-engineered bone grafts in critical-size femoral defects. Biomaterials. 2010;31(4):608-620. [8] CHAZONO M, TANAKA T, KITASATO S, et al. Electron microscopic study on bone formation and bioresorption after implantation of β-tricalcium phosphate in rabbit models. J Orthop Sci. 2008;13(6):550-555. [9] LEPPIK L, OLIVEIRA KMC, BHAVSAR MB, et al. Electrical stimulation in bone tissue engineering treatments. Eur J Trauma Emerg Surg. 2020; 46(2):231-244. [10] SPADARO JA. Mechanical and electrical interactions in bone remodeling. Bioelectromagnetics. 1997;18(3):193-202. [11] 熊莹,许燕,周建平,等.组织工程研究中的电活性生物材料[J].中国组织工程研究,2019,23(34):5523-5530. [12] KHARE D, BASU B, DUBEY AK. Electrical stimulation and piezoelectric biomaterials for bone tissue engineering applications. Biomaterials. 2020;258:120280. [13] TANDON B, BLAKER JJ, CARTMELL SH. Piezoelectric materials as stimulatory biomedical materials and scaffolds for bone repair. Acta Biomater. 2018;73:1-20. [14] KAO FC, CHIU PY, TSAI TT, et al. The application of nanogenerators and piezoelectricity in osteogenesis. Sci Technol Adv Mater. 2019;20(1): 1103-1317. [15] VEIS A. Collagen fibrillogenesis. Connect Tissue Res. 1982;10(1):11-24. [16] RAMACHANDRAN GN, KARTHA G. Structure of Collagen. Nature. 1955; 176(4482):593-595. [17] MINARY-JOLANDAN M, YU MF. Nanoscale characterization of isolated individual type I collagen fibrils: polarization and piezoelectricity. Nanotechnology. 2009;20(8):085706. [18] FUKADA E, YASUDA I. Piezoelectric effects in collagen. Jpn J Appl Phys. 1964;3(2):117. [19] MILAZZO M, DAVID A, JUNG GS, et al. Molecular origin of viscoelasticity in mineralized collagen fibrils. Biomater Sci. 2021;9(9):3390-3400. [20] MILAZZO M, JUNG GS, DANTI S, et al. Mechanics of mineralized collagen fibrils upon transient loads. ACS Nano. 2020;14(7):8307-8316. [21] XU J, SHI GP. Vascular wall extracellular matrix proteins and vascular diseases. Biochim Biophys Acta. 2014;1842(11):2106-2119. [22] MIARA B, ROHAN E, ZIDI M, et al. Piezomaterials for bone regeneration design—homogenization approach. J Mech Phys Solids. 2005;53(11): 2529-2556. [23] BRIGHTON CT, WANG W, SELDES R, et al. Signal transduction in electrically stimulated bone cells. J Bone Joint Surg Am. 2001;83(10): 1514-1523. [24] AHN AC, GRODZINSKY AJ. Relevance of collagen piezoelectricity to “Wolff’s Law”: a critical review. Med Eng Phys. 2009;31(7):733-741. [25] ZHUANG H, WANG W, SELDES RM, et al. Electrical stimulation induces the level of TGF-β1 mRNA in osteoblastic cells by a mechanism involving calcium/calmodulin pathway. Biochem Biophys Res Commun. 1997;237(2):225-229. [26] XU J, WANG W, CLARK CC, et al. Signal transduction in electrically stimulated articular chondrocytes involves translocation of extracellular calcium through voltage-gated channels. Osteoarthr Cartil. 2009;17(3):397-405. [27] NORIS-SUÁREZ K, LIRA-OLIVARES J, FERREIRA AM, et al. In vitro deposition of hydroxyapatite on cortical bone collagen stimulated by deformation-induced piezoelectricity. Biomacromolecules. 2007;8(3):941-948. [28] ABOU NEEL EA, ALJABO A, STRANGE A, et al. Demineralization-remineralization dynamics in teeth and bone. INT J NANOMED. 2016; 11:4743-4763. [29] CALDERíN L, STOTT MJ, RUBIO A. Electronic and crystallographic structure of apatites. Phys Rev B Condens Matter. 2003;67(13):134106. [30] FLORENCIO-SILVA R, SASSO GR, SASSO-CERRI E, et al. Biology of Bone Tissue: Structure, Function, and Factors That Influence Bone Cells. Biomed Res Int. 2015;2015:421746. [31] LANG SB, TOFAIL SA, KHOLKIN AL, et al. Ferroelectric polarization in nanocrystalline hydroxyapatite thin films on silicon. Sci Rep. 2013;3:2215. [32] RIBEIRO C, PäRSSINEN J, SENCADAS V, et al. Dynamic piezoelectric stimulation enhances osteogenic differentiation of human adipose stem cells. J Biomed Mater Res A. 2015;103(6):2172-2175. [33] PäRSSINEN J, HAMMARéN H, RAHIKAINEN R, et al. Enhancement of adhesion and promotion of osteogenic differentiation of human adipose stem cells by poled electroactive poly(vinylidene fluoride). J Biomed Mater Res A. 2015;103(3):919-928. [34] TEIXEIRA LN, CRIPPA GE, GIMENES R, et al. Response of human alveolar bone-derived cells to a novel poly(vinylidene fluoride-trifluoroethylene)/barium titanate membrane. J Mater Sci Mater Med. 2011;22(1):151-158. [35] AHMADI N, KHARAZIHA M, LABBAF S. Core-shell fibrous membranes of PVDF-Ba0.9Ca0.1TiO3/PVA with osteogenic and piezoelectric properties for bone regeneration. Biomed Mater. 2019;15(1):015007. [36] 何逸恒,程鸣威,朱培君,等.电活性生物膜促进大鼠的体内成骨[J].中国组织工程研究,2022,26(28):4446-4451. [37] BAI Y, DAI X, YIN Y, et al. Biomimetic piezoelectric nanocomposite membranes synergistically enhance osteogenesis of deproteinized bovine bone grafts. Int J Nanomedicine. 2019;14:3015-3026. [38] RIBEIRO C, CORREIA DM, RODRIGUES I, et al. In vivo demonstration of the suitability of piezoelectric stimuli for bone reparation. Mater Lett. 2017;209:118-121. [39] REIS J, FRIAS C, CANTO E CASTRO C, et al. A new piezoelectric actuator induces bone formation in vivo: a preliminary study. J Biomed Biotechnol. 2012;2012:613403. [40] ZHOU Z, LI W, HE T, et al. Polarization of an electroactive functional film on titanium for inducing osteogenic differentiation. Sci Rep. 2016; 6:35512. [41] RIBEIRO C, MOREIRA S, CORREIA V, et al. Enhanced proliferation of pre-osteoblastic cells by dynamic piezoelectric stimulation. RSC Advances. 2012;2(30):11504-11509. [42] ZHANG C, LIU W, CAO C, et al. Modulating Surface Potential by Controlling the β Phase Content in Poly(vinylidene fluoridetrifluoroethylene) Membranes Enhances Bone Regeneration. Adv Healthc Mater. 2018;7(11):e1701466. [43] KITSARA M, BLANQUER A, MURILLO G, et al. Permanently hydrophilic, piezoelectric PVDF nanofibrous scaffolds promoting unaided electromechanical stimulation on osteoblasts. Nanoscale. 2019;11(18): 8906-8917. [44] SOBREIRO-ALMEIDA R, TAMAÑO-MACHIAVELLO MN, CARVALHO EO, et al. Human Mesenchymal Stem Cells Growth and Osteogenic Differentiation on Piezoelectric Poly(vinylidene fluoride) Microsphere Substrates. Int J Mol Sci. 2017;18(11):2391. [45] DAMARAJU SM, SHEN Y, ELELE E, et al. Three-dimensional piezoelectric fibrous scaffolds selectively promote mesenchymal stem cell differentiation. Biomaterials. 2017;149:51-62. [46] ZHANG X, ZHANG C, LIN Y, et al. Nanocomposite membranes enhance bone regeneration through restoring physiological electric microenvironment. ACS Nano. 2016;10(8):7279-7286. [47] 张晨光.通过控制P(VDF-Trfe)膜β相含量调控其表面电势促进骨再生[D]. 武汉:华中科技大学,2018. [48] JEONG HG, HAN YS, JUNG KH, et al. Poly(vinylidene fluoride) Composite Nanofibers Containing Polyhedral Oligomeric Silsesquioxane⁻Epigallocatechin Gallate Conjugate for Bone Tissue Regeneration. Nanomaterials (Basel). 2019;9(2):184. [49] AZADIAN E, ARJMAND B, ARDESHIRYLAJIMI A, et al. Polyvinyl alcohol modified polyvinylidene fluoride-graphene oxide scaffold promotes osteogenic differentiation potential of human induced pluripotent stem cells. J Cell Biochem. 2020;121(5-6):3185-3196. [50] AUGUSTINE A, AUGUSTINE R, HASAN A, et al. Development of titanium dioxide nanowire incorporated poly(vinylidene fluoride-trifluoroethylene) scaffolds for bone tissue engineering applications. J Mater Sci Mater Med. 2019;30(8):96. [51] GENCHI GG, SINIBALDI E, CESERACCIU L, et al. Ultrasound-activated piezoelectric P(VDF-TrFE)/boron nitride nanotube composite films promote differentiation of human SaOS-2 osteoblast-like cells. Nanomedicine. 2018;14(7):2421-2432. [52] FELICE B, SáNCHEZ MA, SOCCI MC, et al. Controlled degradability of PCL-ZnO nanofibrous scaffolds for bone tissue engineering and their antibacterial activity. Mater Sci Eng C. 2018;93:724-738. [53] LI Y, SUN L, WEBSTER TJ. The Investigation of ZnO/Poly(vinylidene fluoride) nanocomposites with improved mechanical, piezoelectric, and antimicrobial properties for orthopedic applications. J Biomed Nanotechnol. 2018;14(3):536-545. [54] AMNA T, HASSAN MS, SHEIKH FA, et al. Zinc oxide-doped poly(urethane) spider web nanofibrous scaffold via one-step electrospinning: a novel matrix for tissue engineering. Appl Microbiol Biotechnol. 2013;97(4):1725-1734. [55] POLLEY C, DISTLER T, DETSCH R, et al. 3D Printing of Piezoelectric Barium Titanate-Hydroxyapatite Scaffolds with Interconnected Porosity for Bone Tissue Engineering. Materials (Basel). 2020;13(7):1773. [56] LIU B, CHEN L, SHAO C, et al. Improved osteoblasts growth on osteomimetic hydroxyapatite/BaTiO3 composites with aligned lamellar porous structure. Mater Sci Eng C Mater Biol Appl. 2016;61:8-14. [57] TANG Y, WU C, WU Z, et al. Fabrication and in vitro biological properties of piezoelectric bioceramics for bone regeneration. Sci Rep. 2017;7: 43360. [58] EHTERAMI A, KAZEMI M, NAZARI B, et al. Fabrication and characterization of highly porous barium titanate based scaffold coated by Gel/HA nanocomposite with high piezoelectric coefficient for bone tissue engineering applications. J Mech Behav Biomed Mater. 2018;79:195-202. [59] POON KK, WURM MC, EVANS DM, et al. Biocompatibility of (Ba,Ca)(Zr,Ti)O3 piezoelectric ceramics for bone replacement materials. J Biomed Mater Res B Appl Biomater. 2020;108(4):1295-1303. [60] LIU W, YANG D, WEI X, et al. Fabrication of piezoelectric porous BaTiO3 scaffold to repair large segmental bone defect in sheep. J Biomater Appl. 2020;35(4-5):544-552. [61] SHOKROLLAHI H, SALIMI F, DOOSTMOHAMMADI A. The fabrication and characterization of barium titanate/akermanite nano-bio-ceramic with a suitable piezoelectric coefficient for bone defect recovery. J Mech Behav Biomed Mater. 2017;74:365-370. [62] CHEN W, YU Z, PANG J, et al. Fabrication of Biocompatible Potassium Sodium Niobate Piezoelectric Ceramic as an Electroactive Implant. Materials (Basel). 2017;10(4):345. [63] YU P, NING C, ZHANG Y, et al. Bone-inspired spatially specific piezoelectricity induces bone regeneration. Theranostics. 2017;7(13): 3387-3397. [64] YAO T, CHEN J, WANG Z, et al. The antibacterial effect of potassium-sodium niobate ceramics based on controlling piezoelectric properties. Colloids Surf B Biointerfaces. 2019;175:463-468. [65] LAURENTI M, CAUDA V. ZnO Nanostructures for Tissue Engineering Applications. Nanomaterials (Basel). 2017;7(11):374. [66] ROCCA A, MARINO A, ROCCA V, et al. Barium titanate nanoparticles and hypergravity stimulation improve differentiation of mesenchymal stem cells into osteoblasts. INT J NANOMED. 2015;10:433-445. [67] LI Y, DAI X, BAI Y, et al. Electroactive BaTiO3 nanoparticle-functionalized fibrous scaffolds enhance osteogenic differentiation of mesenchymal stem cells. Int J Nanomed. 2017;12:4007-4018. [68] CIOFANI G, RICOTTI L, CANALE C, et al. Effects of barium titanate nanoparticles on proliferation and differentiation of rat mesenchymal stem cells. Colloid Surface B. 2013;102:312-320. [69] YU SW, KUO ST, TUAN WH, et al. Cytotoxicity and degradation behavior of potassium sodium niobate piezoelectric ceramics. Ceram Int. 2012; 38(4):2845-2850. [70] AUGUSTINE R, DAN P, SOSNIK A, et al. Electrospun poly(vinylidene fluoride-trifluoroethylene)/zinc oxide nanocomposite tissue engineering scaffolds with enhanced cell adhesion and blood vessel formation. Nano Res. 2017;10(10):3358-3376. [71] CAFARELLI A, LOSI P, SALGARELLA AR, et al. Small-caliber vascular grafts based on a piezoelectric nanocomposite elastomer: Mechanical properties and biocompatibility. J Mech Behav Biomed Mater. 2019; 97:138-148. [72] HITSCHERICH P, WU S, GORDAN R, et al. The effect of PVDF-TrFE scaffolds on stem cell derived cardiovascular cells. Biotechnol Bioeng. 2016;113(7):1577-1585. [73] LIU W, LI X, JIAO Y, et al. Biological Effects of a Three-Dimensionally Printed Ti6Al4V Scaffold Coated with Piezoelectric BaTiO3 Nanoparticles on Bone Formation. ACS Appl Mater Interfaces. 2020;12(46):51885-51903. |
| [1] | Sun Kexin, Zeng Jinshi, Li Jia, Jiang Haiyue, Liu Xia. Mechanical stimulation enhances matrix formation of three-dimensional bioprinted cartilage constructs [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(在线): 1-7. |
| [2] | Wen Xinghua, Ding Huanwen, Cheng Kai, Yan Xiaonan, Peng Yuanhao, Wang Yuning, Liu Kang, Zhang Huiwu. Three-dimensional finite element model analysis of intramedullary nailing fixation design for large femoral defects in Beagle dogs [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(9): 1371-1376. |
| [3] | Xu Xingxing, Wen Chaoju, Meng Maohua, Wang Qinying, Chen Jingqiao, Dong Qiang. Carbon nanomaterials in oral implant [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1062-1070. |
| [4] | Yang Yitian, Wang Lu, Yao Wei, Zhao Bin. Application of the interaction between biological scaffolds and macrophages in bone regeneration [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1071-1079. |
| [5] | Li Cheng, Zheng Guoshuang, Kuai Xiandong, Yu Weiting. Alginate scaffold in articular cartilage repair [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1080-1088. |
| [6] | Lu Di, Zhang Cheng, Duan Rongquan, Liu Zongxiang. Osteoinductive properties of calcium phosphate ceramic bone repair materials [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1103-1109. |
| [7] | Shi Yehong, Wang Cheng, Chen Shijiu. Early thrombosis and prevention of small-diameter blood vessel prosthesis [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1110-1116. |
| [8] | Tang Haotian, Liao Rongdong, Tian Jing. Application and design of piezoelectric materials for bone defect repair [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1117-1125. |
| [9] | Zong Mingrui, Liu Haiyan, Li Bing, Wu Xiuping. Application of carboxymethyl chitosan in tissue engineering of stomatology [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(3): 447-452. |
| [10] | Dai Xianglin, Zhang Wenfeng, Yao Xijun, Shang Jiaqi, Huang Qiujin, Ren Yifan, Deng Jiupeng. Barium titanate/polylactic acid piezoelectric composite film affects adhesion, proliferation, and osteogenic differentiation of MC3T3-E1 cells [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(3): 367-373. |
| [11] | Liu Huan, Li Han, Ma Yunhao, Zhong Weijian, Ma Guowu. Osteogenic capacity of partially demineralized dentin particles in the maxillary sinus lift [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(3): 354-359. |
| [12] | Ning Ziwen, Wang Xu, Shi Zhengliang, Qin Yihua, Wang Guoliang, Jia Di, Wang Yang, Li Yanlin. Meniscal injury repair methods for non-blood supply area [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(3): 420-426. |
| [13] | Xie Pingjin, Luo Zhen, Lu Qigui, Guo Yanxing, Chen Qunqun, Li Feilong. Effect of ligustrazine and overexpression of miR-20b-5p on synovial, cartilage and subchondral bone angiogenesis in rats with early-stage knee osteoarthritis: a histological observation [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(2): 237-245. |
| [14] | Liu Gang, Deng Bowen, Jiang Shengyuan, Xu Lin, Fan Xiao, Tao Jingwei, Zhang Houjun, He Feng, Zhao Yi, Mu Xiaohong. Tetramethylpyrazine improves hemorheological indexes in rats with complete spinal cord transection: a dynamic observation [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(2): 282-286. |
| [15] | Chen Feng, Ren Guowu, Zhang Xiaoyun, Chen Yueping, Shi Rusheng. Receptor activator of nuclear factor-kappa B ligand signal transduction mechanism and osteoclast activation [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(2): 293-299. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||