Chinese Journal of Tissue Engineering Research ›› 2023, Vol. 27 ›› Issue (6): 827-834.doi: 10.12307/2023.265
Previous Articles Next Articles
Inhalation of bone marrow mesenchymal stem cells-derived exosomes alleviates inflammatory injury in chronic obstructive pulmonary disease
Wang Min1, Yin Xiushan2, Wang Yingxi1, Zhang Yan1, Zhao Long1, Xia Shuyue1
- 1Department of Respiratory Medicine, Center Hospital Affiliated to Shenyang Medical College, Shenyang 110024, Liaoning Province, China; 2Shenyang University of Chemical Technology, Shenyang 110142, Liaoning Province, China
-
Received:2022-03-04Accepted:2022-04-29Online:2023-02-28Published:2022-08-11 -
Contact:Xia Shuyue, MD, Chief physician, Department of Respiratory Medicine, Center Hospital Affiliated to Shenyang Medical College, Shenyang 110024, Liaoning Province, China Zhao Long, Master, Chief physician, Department of Respiratory Medicine, Center Hospital Affiliated to Shenyang Medical College, Shenyang 110024, Liaoning Province, China -
About author:Wang Min, Master candidate, Department of Respiratory Medicine, Center Hospital Affiliated to Shenyang Medical College, Shenyang 110024, Liaoning Province, China -
Supported by:Liaoning Province Science and Technology Project, No. LNCCC-D14-2015 (to XSY); Shenyang Science and Technology Project, No. 17-230-9-05 (to XSY); Shenyang Science and Technology Project, No. 20-205-4-073 (to ZL); Science and Technology Innovation Fund project for Graduate students of Shenyang Medical College, No. Y20210516 (to WM)
CLC Number:
Cite this article
Wang Min, Yin Xiushan, Wang Yingxi, Zhang Yan, Zhao Long, Xia Shuyue. Inhalation of bone marrow mesenchymal stem cells-derived exosomes alleviates inflammatory injury in chronic obstructive pulmonary disease[J]. Chinese Journal of Tissue Engineering Research, 2023, 27(6): 827-834.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
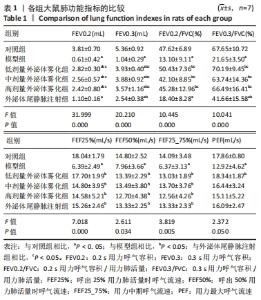
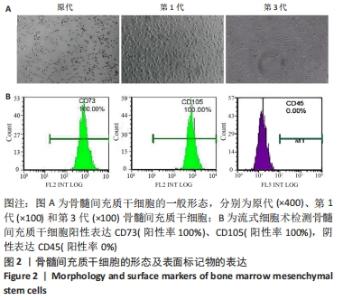
2.3 各组肺功能指标的变化 为验证大鼠慢阻肺模型是否建立成功,以及确定雾化形式的外泌体对肺功能的影响。使用小动物肺功能仪测试肺功能指标,与对照组比较,模型组FEV0.2、FEV0.3、FEV0.2/FVC、FEV0.3/FVC、FEF25%、FEF50%、FEF25_75%、PEF均显著性降低(P < 0.05),表明模型组肺功能出现严重损伤;与模型组比较,外泌体雾化低、中、高剂量组FEV0.2、FEV0.3、FEV0.2/FVC、FEV0.3/FVC、FEF25%、FEF50%、FEF25_75%均显著性升高(P < 0.05)及尾静脉注射组FEV0.3、FEV0.3/FVC、FEF25%、FEF50%、FEF25_75%均显著性升高(P < 0.05),表明外泌体治疗可以改善慢阻肺的肺功能;与外泌体尾静脉注射组相比,外泌体雾化低、中、高剂量组FEV0.2、FEV0.3、FEV0.2/FVC、FEV0.3/FVC均显著性升高(P < 0.05),表明雾化吸入外泌体对肺功能的改善程度强于尾静脉注射外泌体治疗,见表1。"

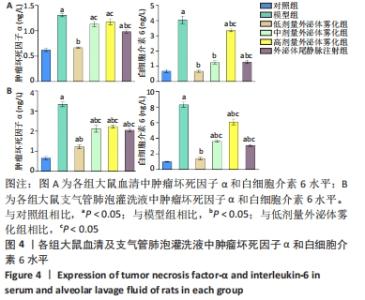
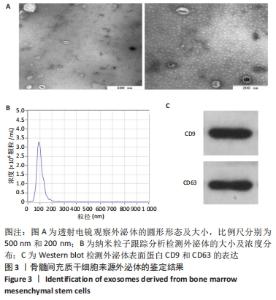
2.4 各组大鼠血清及支气管肺泡灌洗液中炎症因子水平 为明确雾化外泌体是否影响慢阻肺的炎症水平,通过ELISA实验对各组大鼠血清及肺泡灌洗液中的白细胞介素1β和肿瘤坏死因子α进行定量分析。结果显示,模型组大鼠血清和支气管肺泡灌洗液中白细胞介素1β及肿瘤坏死因子α水平明显高于对照组(P < 0.05)。与模型组相比,外泌体雾化低、中、高剂量组及外泌体尾静脉注射组大鼠血清和肺泡灌洗液中两种炎症因子水平均降低(P < 0.05)。低剂量外泌体雾化组大鼠血清和肺泡灌洗液中两种炎症因子水平显著低于外泌体雾化中、高剂量组和外泌体尾静脉注射组(P < 0.05),可见雾化形式的外泌体可减轻慢阻肺炎症反应,其中以低剂量外泌体雾化作用最为显著,见图4。"

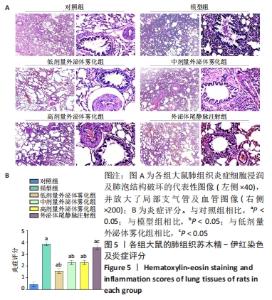
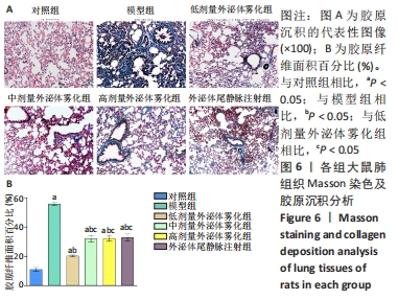
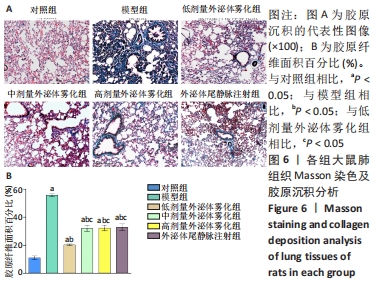
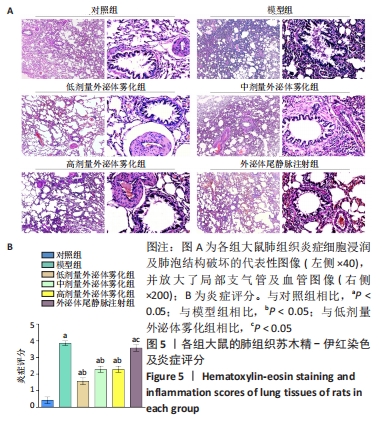
2.5 各组肺组织病理学变化 为明确雾化外泌体对慢阻肺模型大鼠肺组织病理学的影响,进行了苏木精-伊红染色和Masson染色。苏木精-伊红染色结果显示,与对照组相比,经过反复的脂多糖和香烟烟雾刺激后的慢阻肺模型组大鼠存在明显的炎症细胞浸润,且炎症细胞多聚集在血管及支气管周围,支气管管腔内可见黏液性血栓,此外,还可观察到肺泡壁变薄过度扩张,部分可见肺泡融合。然而,与模型组相比,经低、中和高剂量外泌体雾化治疗后显著降低了气道炎症和肺泡结构的破坏,其中在低剂量外泌体雾化组中,大鼠肺组织的血管及支气管周围有少量的炎症细胞浸润,未见明显的支气管黏液栓,肺泡壁轻度扩张,且有少量的肺泡融合。尾静脉注射外泌体的治疗效果并不显著,见图5A。根据各组苏木精-伊红染色结果进行了大鼠肺组织炎症评分,见图5B。与对照组相比,模型组大鼠炎症评分明显升高(P < 0.05)。与模型组相比,外泌体雾化组炎症评分显著降低(P < 0.05),其中低剂量外泌体雾化组炎症评分明显低于外泌体尾静脉注射组(P < 0.05)。"
| [1] NGUYEN JMK, ROBINSON DN, SIDHAYE VK. Why new biology must be uncovered to advance therapeutic strategies for chronic obstructive pulmonary disease. Am J Physiol Lung Cell Mol Physiol. 2021;320(1):L1-L11. [2] SUNDAR IK, RASHID K, SELLIX MT, et al. The nuclear receptor and clock gene REV-ERBα regulates cigarette smoke-induced lung inflammation. Biochem Biophys Res Commun. 2017;493(4):1390-1395. [3] SRIVASTAVA K, THAKUR D, SHARMA S, et al. Systematic review of humanistic and economic burden of symptomatic chronic obstructive pulmonary disease. Pharmacoeconomics. 2015;33(5):467-488. [4] PALLI SR, ZHOU S, SHAIKH A, et al. Effect of compliance with GOLD treatment recommendations on COPD health care resource utilization, cost, and exacerbations among patients with COPD on maintenance therapy. J Manag Care Spec Pharm. 2021;27(5):625-637. [5] OLSCHEWSKI H, BUHL R, FUNK GC, et al. Chronisch-obstruktive Lungenerkrankung 2021 – die richtige Therapie für den richtigen Patienten [Chronic obstructive pulmonary disease: the right treatment for the right patient]. Internist (Berl). 2021;62(6):679-685. [6] MAKE B, DUTRO MP, PAULOSE-RAM R, et al. Undertreatment of COPD: a retrospective analysis of US managed care and medicare patients. Int J Chron Obstruct Pulmon Dis. 2012;7:1-9. [7] CECCARIGLIA S, CARGNONI A, SILINI AR, et al. Autophagy: a potential key contributor to the therapeutic action of mesenchymal stem cells. Autophagy. 2020;16(1):28-37. [8] KIM HO, CHOI SM, KIM HS. Mesenchymal stem cell-derived secretome and microvesicles as a cell-free therapeutics for neurodegenerative disorders. Tissue Eng Regen Med. 2013;10(3):93-101. [9] MOHAMMADIPOOR A, ANTEBI B, BATCHINSKY AI, et al. Therapeutic potential of products derived from mesenchymal stem/stromal cells in pulmonary disease. Respir Res. 2018;19(1):218. [10] HARRELL CR, MILORADOVIC D, SADIKOT R, et al. Molecular and cellular mechanisms responsible for beneficial effects of mesenchymal stem cell-derived product “Exo-d-MAPPS” in attenuation of chronic airway inflammation. Anal Cell Pathol (Amst). 2020;2020:3153891. [11] WILLIS GR, FERNANDEZ-GONZALEZ A, ANASTAS J, et al. Mesenchymal stromal cell exosomes ameliorate experimental bronchopulmonary dysplasia and restore lung function through macrophage immunomodulation. Am J Respir Crit Care Med. 2018;197(1):104-116. [12] MONSEL A, ZHU YG, GENNAI S, et al. Therapeutic effects of human mesenchymal stem cell-derived microvesicles in severe pneumonia in mice. Am J Respir Crit Care Med. 2015;192(3):324-336. [13] DU YM, ZHUANSUN YX, CHEN R, et al. Mesenchymal stem cell exosomes promote immunosuppression of regulatory T cells in asthma. Exp Cell Res. 2018;363(1): 114-120. [14] WAN X, CHEN S, FANG Y, et al. Mesenchymal stem cell-derived extracellular vesicles suppress the fibroblast proliferation by downregulating FZD6 expression in fibroblasts via micrRNA-29b-3p in idiopathic pulmonary fibrosis. J Cell Physiol. 2020;235(11):8613-8625. [15] LEE C, MITSIALIS SA, ASLAM M, et al. Exosomes mediate the cytoprotective action of mesenchymal stromal cells on hypoxia-induced pulmonary hypertension. Circulation. 2012;126(22):2601-2011. [16] O’FARRELL HE, YANG IA. Extracellular vesicles in chronic obstructive pulmonary disease (COPD). J Thorac Dis. 2019;11(Suppl 17):S2141-S2154. [17] BORGHARDT JM, KLOFT C, SHARMA A. Inhaled therapy in respiratory disease: the complex interplay of pulmonary kinetic processes. Can Respir J. 2018;2018: 2732017. [18] GRÄSSEL S, STÖCKL S, JENEI-LANZL Z. Isolation, culture, and osteogenic/chondrogenic differentiation of bone marrow-derived mesenchymal stem cells. Methods Mol Biol. 2012;879:203-267. [19] SUN D, OUYANG Y, GU Y, et al. Cigarette smoke-induced chronic obstructive pulmonary disease is attenuated by CCL20-blocker: a rat model. Croat Med J. 2016;57(4):363-370. [20] HERR C, HAN G, LI D, et al. Combined exposure to bacteria and cigarette smoke rebles characteristic phenotypes of human COPD in a murine disease model. Exp Toxicol Pathol. 2015;67(3):261-269. [21] GUAN XJ, SONG L, HAN FF, et al. Mesenchymal stem cells protect cigarette smoke-damaged lung and pulmonary function partly via VEGF-VEGF receptors. J Cell Biochem. 2013;114(2):323-335. [22] GU W, SONG L, LI XM, et al. Mesenchymal stem cells alleviate airway inflammation and emphya in COPD through down-regulation of cyclooxygenase-2 via p38 and ERK MAPK pathways. Sci Rep. 2015;5:8733. [23] CHURG A, WANG RD, TAI H, et al. Macrophage metalloelastase mediates acute cigarette smoke-induced inflammation via tumor necrosis factor-alpha release. Am J Respir Crit Care Med. 2003;167(8):1083-1089. [24] KIM YS, KIM JY, HUH JW, et al. The therapeutic effects of optimal dose of mesenchymal stem cells in a murine model of an elastase induced-emphya. Tuberc Respir Dis (Seoul). 2015;78(3):239-245. [25] ZHEN G, LIU H, GU N, et al. Mesenchymal stem cells transplantation protects against rat pulmonary emphya. Front Biosci. 2008;13:3415-3422. [26] KIM SY, LEE JH, KIM HJ, et al. Mesenchymal stem cell-conditioned media recovers lung fibroblasts from cigarette smoke-induced damage. Am J Physiol Lung Cell Mol Physiol. 2012;302(9):L891-908. [27] LI J, LI D, LIU X, et al. Human umbilical cord mesenchymal stem cells reduce systemic inflammation and attenuate LPS-induced acute lung injury in rats. J Inflamm (Lond). 2012;9(1):33. [28] FREDENBURGH LE, PERRELLA MA, MITSIALIS SA. The role of heme oxygenase-1 in pulmonary disease. Am J Respir Cell Mol Biol. 2007;36(2):158-165. [29] WEISS DJ, CASABURI R, FLANNERY R, et al. A placebo-controlled, randomized trial of mesenchymal stem cells in COPD. Chest. 2013;143(6):1590-1598. [30] STOLK J, BROEKMAN W, MAUAD T, et al. A phase I study for intravenous autologous mesenchymal stromal cell administration to patients with severe emphya. QJM. 2016;109(5):331-336. [31] LE THI BICH P, NGUYEN THI H, DANG NGO CHAU H, et al. Allogeneic umbilical cord-derived mesenchymal stem cell transplantation for treating chronic obstructive pulmonary disease: a pilot clinical study. Stem Cell Res Ther. 2020;11(1):60. [32] AVERYANOV A, KOROLEVA I, KONOPLYANNIKOV M, et al. First-in-human high-cumulative-dose stem cell therapy in idiopathic pulmonary fibrosis with rapid lung function decline. Stem Cells Transl Med. 2020;9(1):6-16. [33] VOLAREVIC V, MARKOVIC BS, GAZDIC M, et al. Ethical and safety issues of stem cell-based therapy. Int J Med Sci. 2018;15(1):36-45. [34] GLASSBERG MK, MINKIEWICZ J, TOONKEL RL, et al. Allogeneic Human Mesenchymal Stem Cells in Patients With Idiopathic Pulmonary Fibrosis via Intravenous Delivery (AETHER): A Phase I Safety Clinical Trial. Chest. 2017;151(5): 971-981. [35] DUIJVESTEIN M, VOS AC, ROELOFS H, et al. Autologous bone marrow-derived mesenchymal stromal cell treatment for refractory luminal Crohn’s disease: results of a phase I study. Gut. 2010;59(12):1662-1669. [36] DHERE T, COPLAND I, GARCIA M, et al. The safety of autologous and metabolically fit bone marrow mesenchymal stromal cells in medically refractory Crohn’s disease - a phase 1 trial with three doses. Aliment Pharmacol Ther. 2016;44(5):471-481. [37] GAZDIC M, VOLAREVIC V, ARSENIJEVIC N, et al. Mesenchymal stem cells: a friend or foe in immune-mediated diseases. Stem Cell Rev Rep. 2015;11(2):280-287. [38] HARRELL CR, FELLABAUM C, JOVICIC N, et al. Molecular mechanisms responsible for therapeutic potential of mesenchymal stem cell-derived secretome. Cells. 2019;8(5):467. [39] KARDIA E, HALIM NSSA, YAHAYA BH. Aerosol-based cell therapy for treatment of lung diseases. Methods Mol Biol. 2016;1516:243-255. [40] KIM WY, HONG SB. Sepsis and Acute Respiratory Distress Syndrome: Recent Update. Tuberc Respir Dis (Seoul). 2016;79(2):53-57. [41] ZHANG E, GENG X, SHAN S, et al. Exosomes derived from bone marrow mesenchymal stem cells reverse epithelial-mesenchymal transition potentially via attenuating Wnt/β-catenin signaling to alleviate silica-induced pulmonary fibrosis. Toxicol Mech Methods. 2021;31(9):655-666. [42] RIDZUAN N, ZAKARIA N, WIDERA D, et al. Human umbilical cord mesenchymal stem cell-derived extracellular vesicles ameliorate airway inflammation in a rat model of chronic obstructive pulmonary disease (COPD). Stem Cell Res Ther. 2021;12(1):54. [43] KIM SY, BURGESS JK, WANG Y, et al. Atomized Human Amniotic Mesenchymal Stromal Cells for Direct Delivery to the Airway for Treatment of Lung Injury. J Aerosol Med Pulm Drug Deliv. 2016;29(6):514-524. [44] HALIM NSS, CH’NG ES, KARDIA E, et al. Aerosolised Mesenchymal Stem Cells Expressing Angiopoietin-1 Enhances Airway Repair. Stem Cell Rev Rep. 2019; 15(1):112-125. [45] DINH PC, PAUDEL D, BROCHU H, et al. Inhalation of lung spheroid cell secretome and exosomes promotes lung repair in pulmonary fibrosis. Nat Commun. 2020; 11(1):1064. [46] BARI E, FERRAROTTI I, TORRE ML, et al. Mesenchymal stem/stromal cell secretome for lung regeneration: The long way through “pharmaceuticalization” for the best formulation. J Control Release. 2019;309:11-24. [47] A Pilot Clinical Study on Inhalation of Mesenchymal Stem Cells Exosomes Treating Severe Novel Coronavirus Pneumonia. https://clinicaltrials.gov/ct2/show/NCT04276987[2022-03-04] [48] DOLOVICH MB, AHRENS RC, HESS DR, et al. Device selection and outcomes of aerosol therapy: Evidence-based guidelines: American College of Chest Physicians/American College of Asthma, Allergy, and Immunology. Chest. 2005;127(1):335-371. [49] OTERO-ORTEGA L, GÓMEZ DE FRUTOS MC, LASO-GARCÍA F, et al. Exosomes promote restoration after an experimental animal model of intracerebral hemorrhage. J Cereb Blood Flow Metab. 2018;38(5):767-779. [50] CHANDEL A, GOYAL AK, GHOSH G, et al. Recent advances in aerosolised drug delivery. Biomed Pharmacother. 2019;112:108601. [51] MCCARTHY SD, HORGAN E, ALI A, et al. Nebulized mesenchymal stem cell derived conditioned medium retains antibacterial properties against clinical pathogen isolates. J Aerosol Med Pulm Drug Deliv. 2020;33(3):140-152. [52] SHI MM, YANG QY, MONSEL A, et al. Preclinical efficacy and clinical safety of clinical-grade nebulized allogenic adipose mesenchymal stromal cells-derived extracellular vesicles. J Extracell Vesicles. 2021;10(10):e12134. |
| [1] | Fang Xingyan, Tian Zhenli, Zhao Zheyi, Wen Ping, Xie Tingting. Effects of sodium arsenite on human umbilical vein endothelial cell injury and sphingosine kinases 1/sphingosine 1-phosphate signaling axis [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(在线): 1-7. |
| [2] | Pan Zhongjie, Qin Zhihong, Zheng Tiejun, Ding Xiaofei, Liao Shijie. Targeting of non-coding RNAs in the pathogenesis of the osteonecrosis of the femoral head [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(9): 1441-1447. |
| [3] | Dang Yi, Du Chengyan, Yao Honglin, Yuan Nenghua, Cao Jin, Xiong Shan, Zhang Dingmei, Wang Xin. Hormonal osteonecrosis and oxidative stress [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(9): 1469-1476. |
| [4] | Tang Liang, Li Xiheng, Niu Ruijuan, Li Xinyue, Zou Xinying, Mao Tianjiao, Li Jiang. Naringin regulates the function of RAW264.7 macrophages to affect the osteogenic differentiation of MC-3T3-E1 cells [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(8): 1205-1210. |
| [5] | Liang Jiaqi, Liu Hengxu, Yang Jinxin, Yang Yi, Deng Xuhui, Tan Mingjian, Luo Jiong. Health benefit relationship between exercise and intestinal bacteria [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(8): 1292-1299. |
| [6] | Gao Yu, Han Jiahui, Ge Xin. Immunoinflammatory microenvironment after spinal cord ischemia-reperfusion injury [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(8): 1300-1305. |
| [7] | Yuan Bo, Xie Lide, Fu Xiumei. Schwann cell-derived exosomes promote the repair and regeneration of injured peripheral nerves [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(6): 935-940. |
| [8] | Xu Qijing, Yang Yichun, Lei Wei, Yang Ying, Yu Jiang, Xia Tingting, Zhang Meng, Zhang Tao, Zhang Qian. Advances and problems in cell-free treatment of diabetic skin chronic wounds [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(6): 962-969. |
| [9] | Chen Guanting, Zhang Linqi, Li Qingru. Meta-analysis of the value of exosomal miRNA for the diagnosis of chronic kidney disease [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(6): 970-976. |
| [10] | Liu Wentao, Feng Xingchao, Yang Yi, Bai Shengbin. Effect of M2 macrophage-derived exosomes on osteogenic differentiation of bone marrow mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(6): 840-845. |
| [11] | Long Yanming, Xie Mengsheng, Huang Jiajie, Xue Wenli, Rong Hui, Li Xiaojie. Casein kinase 2-interaction protein-1 regulates the osteogenic ability of bone marrow mesenchymal stem cells in osteoporosis rats [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(6): 878-882. |
| [12] | Li Qicheng, Deng Jin, Fu Xiaoyang, Han Na. Effects of bone marrow mesenchymal stem cells-derived exosomes on hypoxia-treated myoblasts [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(6): 853-859. |
| [13] | Zhang Houjun, Deng Bowen, Jiang Shengyuan, Zhao Yi, Ren Jingpei, Xu Lin, Mu Xiaohong. Proteomic analysis of cerebrospinal fluid exosomes derived from cerebral palsy children [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(6): 903-908. |
| [14] | Gao Ting, Ma Xiaohong, Li Xiaorong. Extraction and identification of exosomes from three different sources of ovarian granulosa cells [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(6): 860-865. |
| [15] | Li Zhichao, Tan Guoqing, Su Hui, Xu Zhanwang, Xue Haipeng. Regulatory role of non-coding RNAs as potential therapeutic targets in spinal cord injury [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(5): 758-764. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||