Chinese Journal of Tissue Engineering Research ›› 2022, Vol. 26 ›› Issue (25): 4064-4069.doi: 10.12307/2022.415
Previous Articles Next Articles
Proteoglycans in tooth development and its role in regulating stem cell homeostasis
Chen Jiawen1, 2, Sun Tianyu3, Liu Peng4, Wu Buling2, 5, Wu Jingyi6
- 1Department of Stomatology, Nanfang Hospital, Southern Medical University; 2School of Stomatology, Southern Medical University; 3Department of Periodontology, Stomatological Hospital, Southern Medical University; 4Zhujiang Hospital, Southern Medical University; 5Southern Medical University-Shenzhen Stomatology Hospital (Pingshan); 6Center of Implant Dentistry, Stomatological Hospital, Southern Medical University
-
Received:2020-09-04Accepted:2020-10-24Online:2022-09-08Published:2022-01-26 -
Contact:Wu Buling, MD, Chief physician, Professor, Doctoral supervisor, School of Stomatology, Southern Medical University, Guangzhou 510515, Guangdong Province, China; Southern Medical University-Shenzhen Stomatology Hospital (Pingshan), Shenzhen 518118, Guangdong Province, China Wu Jingyi, MD, Physician, Center of Implant Dentistry, Stomatological Hospital, Southern Medical University, Guangzhou 510280, Guangdong Province, China -
About author:Chen Jiawen, Master candidate, Department of Stomatology, Nanfang Hospital, Southern Medical University, Guangzhou 510515, Guangdong Province, China; School of Stomatology, Southern Medical University, Guangzhou 510515, Guangdong Province, China -
Supported by:the National Natural Science Foundation of China, No. 81900956 (to WJY); the Postdoctoral Special Foundation of Stomatological Hospital of Southern Medical University, No. PY2018025 (to WJY); the Scientific Research Project of Southern Medical University, No. QD2018N030 (to WJY)
CLC Number:
Cite this article
Chen Jiawen, Sun Tianyu, Liu Peng, Wu Buling, Wu Jingyi. Proteoglycans in tooth development and its role in regulating stem cell homeostasis[J]. Chinese Journal of Tissue Engineering Research, 2022, 26(25): 4064-4069.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
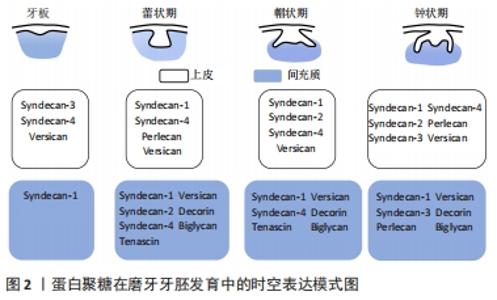
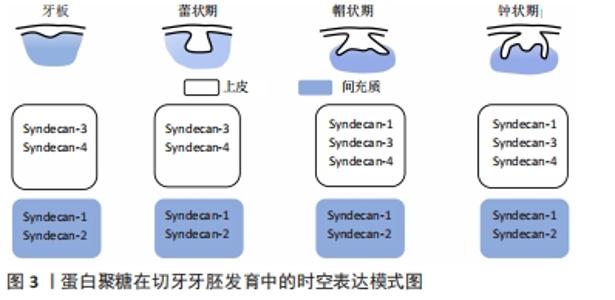
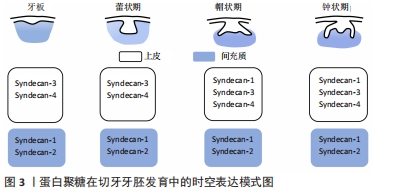
2.1 蛋白聚糖的结构及功能特点 蛋白聚糖是一类由核心蛋白和与其共价连接的一个或多个糖胺聚糖链所构成的多糖大分子[15]。蛋白聚糖分子中糖胺聚糖链的含量高,种类丰富,一个蛋白聚糖往往与一种或多种糖胺聚糖链共价连接。糖胺聚糖链是由重复二糖单位连接而成的硫化线性多糖[15],其糖链含有许多硫酸化基团,如N-SO3,6-O-SO3,2-O-SO3等。糖胺聚糖链的合成无确定的模板,具有组织和阶段特异性,通过各种合成酶的有序调控,多样的硫酸化修饰、脱乙酰基以及异构化,形成链长、聚合度、硫酸化等结构各异的糖胺聚糖链。目前根据二糖单位、单糖残基、残基连键以及硫酸基数目和位置的差异,主要将糖胺聚糖链分为7种:透明质酸、硫酸软骨素、硫酸皮肤素、硫酸乙酰肝素(heparan sulfate,HS)、肝素以及硫酸角质素。 糖胺聚糖链的结构多样性是蛋白聚糖发挥各种生物学功能的重要基础,糖胺聚糖链丰富的结构域使得蛋白聚糖能够为多种信号分子提供广泛的结合位点,协助其扩散或影响配体-受体的结合,从而调节信号通路的活性。另一方面,糖胺聚糖链能够结合并保护信号分子免受蛋白酶的降解,从而维持稳定的信号浓度[16-18]。研究表明,糖胺聚糖链能够通过上述多样的方式广泛参与调控干细胞的自我更新、分化、增殖等生物学行为[10,19]。由此可见,深入探讨蛋白聚糖对干细胞稳态的调控作用和相关机制,将有助于扩展对干细胞稳态的复杂调控网络的认识[20]。 2.2 蛋白聚糖参与牙胚发生发育 牙发生和发育是一个复杂的上皮与间充质相互作用的过程,依赖于多种信号网络的协同调控[4,21-22]。过去数十年内大量研究已针对生长因子、转录因子等细胞内信号分子建立了较为完善的牙发育信号调控网络。研究者一致认为Wnt、Hh、成纤维细胞生长因子、转化生长因子β等信号网络的稳定传导影响着牙齿的正常发育[23]。例如KLEIN等[24]构建了转化生长因子β通路过度激活的小鼠模型,观察到牙源性上皮细胞的增殖严重受阻,细胞周期停滞在G1期,小鼠的牙齿显著变小,可见稳定的信号传导是牙齿正常形态所必需的,而这些信号通路的正常激活也影响着牙齿的发育。研究发现,成纤维细胞生长因子信号通路的异常激活,会导致小鼠的无牙区以及切牙和磨牙之间的纵裂区域本应进入程序性凋亡的细胞反而大量增殖,从而导致小鼠多生牙的形成[24]。然而这些分泌性信号蛋白在传递信号时必须与细胞外信号分子(例如蛋白聚糖)相互作用从而使信号传递入胞。尽管数十年前研究者就发现蛋白聚糖参与牙齿发育的过程[11-12],近年来越来越多的研究也表明蛋白聚糖作为细胞外基质和细胞表面的重要信号分子积极参与多个组织器官的生长发育过程[25-28],但是其在牙齿发育中的作用一直未得到广泛关注。 蛋白聚糖的表达贯穿牙胚发育的全过程且具有明显的时空特异性,见图2和图3。"
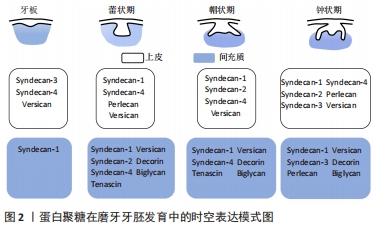

研究发现多配体聚糖(Syndecans)家族广泛表达于早期发育牙胚的不同组织结构中,从蕾状期开始syndecan-1即在间充质组织中表达,进入帽状期后则在成釉器和间充质细胞内均有广泛表达,而syndecan-2的阳性信号主要分布于蕾状期的间充质内,在帽状期其表达则主要集中在牙源性上皮的中间层,提示syndecan-1和syndecan-2可能参与了上皮和间充质之间的相互作用[13],积极调控牙胚早期发生发育。随后在牙胚内上皮细胞分化过程中,研究者观察到在牙源性上皮分化过程中syndecan-3表达水平显著上调,在内釉上皮和中间层为高表达[13],相反 syndecan-4在内釉上皮层为高表达,而在分化为成釉细胞后其表达消失[29],提示二者可能以不同的作用积极参与了牙胚内上皮细胞的分化过程。当牙胚发育进入钟状期出现釉基质的分泌和矿化时,小分子亮氨酸蛋白聚糖在前成牙本质和釉质中的表达水平显著升高[30],并参与牙本质、釉质的分泌和矿化。研究者们亦通过大量的动物模型进一步验证了小分子亮氨酸蛋白聚糖在牙本质和釉质分泌矿化中具有关键的调控作用。纤调蛋白聚糖是一种前牙本质中表达明显的小分子亮氨酸蛋白聚糖,GOLDBERG等[31]研究发现Fibromodulin-/-小鼠的釉质形成明显受阻,前牙本质的胶原纤维直径变大,牙本质的矿化程度显著降低,说明纤调蛋白聚糖能够促进牙釉质和牙本质的正常形成。核心蛋白聚糖与双糖链蛋白聚糖在牙胚发育后期亦具有广泛表达,且与牙体硬组织的分泌矿化具有密切的调控作用。核心蛋白聚糖主要在新分化的成牙本质细胞内表达,随着成牙本质细胞分化成熟,形成牙本质,其表达水平亦随着下降,并局限表达于颈环处新分化的成牙本质内[30];而双糖链蛋白聚糖的表达贯穿成牙本质细胞整个分化过程,在分化成熟的成牙本质细胞内表达最为强烈[30]。研究者对Biglycan-/-和Decorin-/-小鼠牙齿表型进行观察,发现两种小鼠牙本质的矿化程度均显著降低[32],但二者对釉质的形成则起着相反的调控作用,核心蛋白聚糖缺失导致小鼠牙釉质的形成延迟,而Biglycan-/-小鼠牙胚内成釉原蛋白的表达却显著上调,并加速了牙釉质的形成,提示二者对牙本质和釉质的形成具有相反的调控作用。由此可见,多种蛋白聚糖在牙胚发育过程中呈现了时空特异性的表达,且发挥着不同的生物学功能,调控着牙胚发育的全过程。 在牙胚发育过程中,除了主蛋白种类的变化,糖胺聚糖链及其硫酸化酶的表达也经历了显著的改变[33],GALBRAITH等[34]发现在牙形态发生时期,硫酸软骨素的含量高达91%,硫酸乙酰肝素仅占8%;但当牙胚内细胞分化开始后,硫酸乙酰肝素的含量显著上升至35%,而硫酸软骨素大幅度下降为64%。在牙胚发育进入钟状期,成釉细胞和成牙本质细胞所分泌的基质中,硫酸乙酰肝素是含量最高的糖胺聚糖链,而硫酸软骨素和硫酸角质素的含量则显著较低[30],这说明不同的糖胺聚糖链在牙胚发育的不同时期亦具有表达特异性,硫酸软骨素很可能参与调控牙齿形态发生,而硫酸乙酰肝素参与调控牙源性细胞分化和后续基质的分泌矿化。JIANG等[35]和LIU等[36]的研究亦有力证实了糖胺聚糖链在牙源性细胞分化过程中起着不可或缺的调控作用。糖胺聚糖链与核心蛋白连接的首要步骤是在糖基转移酶的催化下,半乳糖转移到核心蛋白的木糖残基上。研究者采用Xyl-MU抑制糖基转移酶的活性,导致糖胺聚糖链与核心蛋白无法连接,发现小鼠牙胚内的细胞分化过程显著改变。糖胺聚糖链缺失的小鼠牙胚,其内釉上皮的细胞折叠和中间层的形成均受抑制,成釉细胞和成牙本质细胞分化延迟,AMELX、DMP-1和DSPP等分化相关标志物的表达水平显著下降[35-36],这说明糖胺聚糖链可能正向调控牙源性干细胞向成釉细胞和成牙本质细胞的分化。但目前尚缺乏阐明蛋白聚糖和糖胺聚糖链对牙胚上皮干细胞和间充质干细胞调控作用的体外实验,因此对蛋白聚糖调控作用的分子机制的认识尚十分匮乏。 最新的研究发现蛋白聚糖上的糖胺聚糖链不仅参与牙胚的发育,还可以通过调控牙胚干细胞的稳态平衡,启动小鼠的牙齿再生。FAM20B是近年来新发现的一种非典型分泌通路上的激酶,它普遍存在于几乎所有蛋白聚糖的链接区,调控着蛋白聚糖上多种类型的糖胺聚糖链与核心蛋白之间链接区内木糖(Xylose)的磷酸化[37-38]。WU等[14,39]在上皮细胞内特异性敲除Fam20B基因,导致糖胺聚糖链无法与核心蛋白顺利连接,以此构建糖胺聚糖链缺失小鼠模型,该模型不具替牙能力的小鼠在正常切牙的近中舌侧长出一对多生的切牙,通过组织学分析发现该多生切牙牙胚的生长方式和萌出时期与继承恒牙非常类似。他们进一步利用细胞谱系示踪技术发现糖胺聚糖链缺失导致小鼠内Sox2+干细胞出现了异常激活。在正常情况下,Sox2+干细胞在牙胚发育完成后仅集中表达在唇侧颈环处,作为干细胞储备[40-41],而糖胺聚糖链缺失小鼠正常切牙胚的舌侧(即多生牙出现的区域)一直存在Sox2高表达的一群细胞,提示糖胺聚糖链的缺失很可能干预了牙板内Sox2+干细胞的归宿,激活了其成牙的潜力。 他们还在糖胺聚糖链缺失小鼠的牙胚上皮中观察到成纤维细胞生长因子通路的过表达。随后通过建立四环素诱导型转基因动物模型,特异性抑制糖胺聚糖链缺失小鼠上皮成纤维细胞生长因子受体2b,从而抑制上调的成纤维细胞生长因子通路活性,成功逆转了多生牙的表型。为了进一步探究该表型相关的分子机制,WU等[14]通过建立体外2D/3D细胞模型发现糖胺聚糖链能够抑制成纤维细胞生长因子10在细胞中的梯度扩散,从而稳定成纤维细胞生长因子10-成纤维细胞生长因子受体2b下游的传入信号,当糖胺聚糖链缺失时,失去协助的成纤维细胞生长因子10与其受体广泛结合,上调的成纤维细胞生长因子通路继而影响到Sox2+上皮干细胞的稳态平衡,最终导致小鼠多生切牙的形成。由此可见,糖胺聚糖链可能作为细胞外信号传递的重要分子,通过维持细胞信号的稳定传导,从而调控牙胚干细胞的命运。但目前由于糖胺聚糖链的多样性以及研究中缺乏特异型的动物模型等各种研究条件的限制,使得关于蛋白聚糖或糖胺聚糖链在牙发生发育中的相关研究尚且匮乏,不同蛋白聚糖和糖胺聚糖链在牙发生中的具体作用仍未被完全阐明,它们对于牙胚干细胞的调控机制更是有待深入的研究。 2.3 蛋白聚糖参与维持干细胞稳态 干细胞具有自我更新、维持干性、增殖、分化的功能,维持这些功能的稳态平衡对于组织器官发挥正常功能尤为重要。近年来针对各个调控层面对干细胞活动的研究越发广泛[42-43],但蛋白聚糖对干细胞自我更新的调控作用尚无定论。 研究者们利用果蝇、线虫、斑马鱼、小鼠等多种动物模型证实蛋白聚糖广泛参与动物组织器官的早期生长发育[25-28]。大量研究认为糖胺聚糖链在干细胞稳态中的调控作用远大于蛋白聚糖中的核心蛋白。KRAUSHAAR等[44]通过建立Ext1-/-胚胎干细胞模型使胚胎干细胞中硫酸乙酰肝素糖链合成受阻,观察发现Ext1-/-胚胎干细胞在培养20 d后仍能保留典型的未分化形态;而在诱导培养条件下,与进入分化的Ext1+/+胚胎干细胞相比,Ext1-/-胚胎干细胞仍维持未分化形态和高碱性磷酸酶活性,说明硫酸乙酰肝素是胚胎干细胞退出自我更新状态进入分化程序的重要调节因素之一。在完全缺乏硫酸乙酰肝素、硫酸软骨素和硫酸皮肤素的GlcAT-I-/-胚胎干细胞中也观察到类似的结果,GlcAT-I-/-胚胎干细胞同样能长期处于自我更新状态[45],这说明糖胺聚糖链的存在可能会干扰干细胞的自我更新。 但仍有观点认为糖胺聚糖链能够有效维持干细胞自我更新的状态,并保持干细胞的多向分化潜能。在对人间充质干细胞体外培养时,外源性硫酸乙酰肝素的加入能够明显促进人间充质干细胞的增殖和自我更新,即使在长期扩增后人间充质干细胞仍维持稳定的干性[46]。外源性HS-2能够刺激G0期细胞进入细胞周期,显著促进人间充质干细胞的增殖,在培养21 d后细胞数量是对照组的8倍之多;在扩增15代后,人间充质干细胞仍维持干细胞表面标志物CD105、CD49a、STRO-1的稳定表达。研究者进一步将使用HS-2培养基长期扩增的人间充质干细胞移植至小鼠临界骨缺损模型中,发现人间充质干细胞仍保持多向分化潜能,缺损区骨组织、软骨细胞生成显著增多,较对照组表现出明显的促进骨愈合的能力。类似的研究显示,在人间充质干细胞体外培养时加入外源性HS8之后在快速扩增时期仍能保持其干性,在扩增数代后仍稳定表达其干性标志物 STRO-1[47],提示糖胺聚糖链能够有效维持干细胞自我更新的潜能。 2.4 蛋白聚糖的硫酸化修饰与干细胞稳态维持 糖胺聚糖链上多样的硫酸化修饰是蛋白聚糖发挥生物学功能的必要结构基础。糖胺聚糖链的硫化程度是间充质干细胞自我更新和分化平衡调节的重要影响因素之一。在干细胞退出自我更新进行定向分化时,糖胺聚糖链的硫酸化程度经历明显的改变。研究表明,未分化的胚胎干细胞自我更新时,所表达的糖胺聚糖链的硫酸化程度较低甚至直接缺如;相反,在胚胎干细胞进入分化状态时,所表达的糖胺聚糖链硫酸化程度明显升高[48]。糖胺聚糖链合成受阻的Ext1-/-、Extcn/cn、Ndst1/2-/-干细胞亦能够在较长的培养周期内保持稳定的自我更新能力[44-45,49]。QU等[50]认为较低的硫酸化程度可以降低糖胺聚糖链与分化相关的信号分子的结合,从而避免干细胞在诱导分化的信号通路调控下退出自我更新。透明质酸是唯一不经硫酸化修饰的糖胺聚糖链,而间充质干细胞能够合成大量的透明质酸,参与构成其所在微环境,紧密影响着间充质干细胞的自我更新。使用透明质酸预处理的培养皿进行体外培养可以延长小鼠间充质干细胞的寿命,防止细胞衰老[51],能够在人间充质干细胞传代19次后维持其干性标志(CD105、CD90)的稳定表达且保持多向分化潜能[52],这说明糖胺聚糖链的硫化程度影响着干细胞的自我更新和分化潜能。 另一方面,糖胺聚糖链上的硫酸化基团使得糖胺聚糖链能以高亲和力结合特定生长因子并调控其信号传导,从而维持间充质干细胞的干性。HS8的结构富含6-O-SO3,能以高亲和力结合并稳定成纤维细胞生长因子2,通过成纤维细胞生长因子受体1通路促进间充质干细胞的增殖。在体外长期培养中HS8亦能够上调防止细胞衰老的基因表达(CD74、CCL2、FANCD2、MDM2、SPRY2等),同时下调抑制细胞增殖的SULF2和CDKN2B表达,从而协助间充质干细胞维持干性和多向分化潜能[47]。另一方面,SULF2是一种硫酸酯酶,能够水解硫酸乙酰肝素上的硫酸化基团[53],其表达下调同样说明间充质干细胞的干性可能依赖于硫酸乙酰肝素的硫酸化基团所介导的信号传导。糖胺聚糖链的硫酸化基团通过结合成纤维细胞生长因子2及其受体形成稳定的复合体,将成纤维细胞生长因子2信号通路的活性调节在有利于间充质干细胞干性维持的水平从而促进间充质干细胞的自我更新。由此可见,糖胺聚糖链的硫酸化程度对于调控与干细胞自我更新和多向分化潜能的相关信号分子的传导而言至关重要,但其具体机制和影响作用仍有待进一步深入研究。 2.5 蛋白聚糖维持干细胞稳态的作用机制 蛋白聚糖对干细胞行为调控的作用机制仍然是目前研究的重点。近年来国内外研究认为蛋白聚糖上共价连接的糖胺聚糖链是调节干细胞稳态的重要因素。糖胺聚糖链通过不断调整其结构以选择性地结合细胞外的信号分子并协助其向细胞表面扩散。在Ext1-/-胚胎干细胞模型中,硫酸乙酰肝素无法正常合成致使干细胞一直处于自我更新状态而无法进一步分化,在这过程中研究者观察到成纤维细胞生长因子通路活性也随之受到抑制,这提示硫酸乙酰肝素可能参与调控成纤维细胞生长因子通路信号传导影响干细胞稳态。这一观点随后在成纤维细胞生长因子通路抑制细胞模型中也得到了验证,成纤维细胞生长因子通路抑制模型成功复制了Ext1-/- 胚胎干细胞持续自我更新的表型[44]。而使用硫酸乙酰肝素拮抗剂苏尔芬(Surfen)、硫酸化抑制剂NaClO3-处理胚胎干细胞[54-55],同样观察到成纤维细胞生长因子诱导的ERK1/2磷酸化受阻。不难看出糖胺聚糖链尤其是硫酸乙酰肝素与成纤维细胞生长因子通路的传导高度相关。除了内源性硫酸乙酰肝素,外源性的硫酸乙酰肝素也可以促进细胞合成成纤维细胞生长因子2,亲和力更高的游离HS8通过与内源性硫酸乙酰肝素竞争,更快地结合成纤维细胞生长因子2,使其可以扩散到其他细胞发挥作用[56]。附着于蛋白聚糖上的糖胺聚糖链通过结合配体或者与配体受体形成复合物调控下游通路的传导,最终影响干细胞活动。 目前针对蛋白聚糖调控干细胞活动的分子机制的研究广泛集中在硫酸乙酰肝素蛋白聚糖上,Sydencans家族和磷脂酰肌醇蛋白聚糖家族参与调控成纤维细胞生长因子及其受体之间的相互作用。蛋白聚糖通过硫酸乙酰肝素链上的硫酸化位点来稳定成纤维细胞生长因子1和成纤维细胞生长因子2与相应受体的结合,进一步激活ERK1/2信号通路转导,影响下游间充质干细胞的行为[17]。不同的硫酸化位点在蛋白聚糖介导的信号通路传导中发挥着特定的作用。硫酸乙酰肝素特定的硫酸化位点N-硫酸化域、艾杜糖醛酸和2-O-SO3是硫酸乙酰肝素与成纤维细胞生长因子2结合的基础,而6-O-SO3则是硫酸乙酰肝素协助成纤维细胞生长因子2与成纤维细胞生长因子受体1结合并激活下游信号通路的关键位点[57-58]。糖胺聚糖链上的其他硫酸化位点同样是与生长因子结合的基础。敲除Ndst1/2的胚胎干细胞由于缺乏硫酸乙酰肝素链上的N-SO3,导致成纤维细胞生长因子4无法激活ERK1/2磷酸化,胚胎干细胞持续保持自我更新状态[54]。由此可见糖胺聚糖链正是通过其特定硫酸化位点影响着干细胞活动中成纤维细胞生长因子通路的信号传导。 糖胺聚糖链的硫酸化也被证明是调控其他通路传导所必需的,当糖胺聚糖链合成或硫酸化受阻时,Wnt信号通路的传导会受到明显的影响[59]。Wnt通路的配体能够和胞膜上的硫酸乙酰肝素蛋白聚糖结合,并受硫酸乙酰肝素硫酸酯酶重构调控的显著影响。在细胞表面硫酸乙酰肝素硫酸酯酶能够催化硫酸乙酰肝素链中的6-O-SO3水解,促进低亲和力的HS-WNT复合体形成,从而释放出大量WNT配体,游离WNT通过与卷曲蛋白和低密度脂蛋白受体相关蛋白5/6结合,使得更多的β-连锁蛋白转运到细胞核内[53,60-61],从而增强了干细胞的自我更新和维持其多能性[62]。但不同的糖胺聚糖链对信号通路的正负向调控仍存在争议。尽管研究已表明蛋白聚糖可通过介导多条信号通路的转导影响干细胞行为,但其对干细胞命运的调控机制仍有待进一步深入。"

| [1] WANG F, WU Z, FAN Z, et al. The cell re-association-based whole-tooth regeneration strategies in large animal, Sus scrofa. Cell Prolif. 2018; 51(4):e12479. [2] WU Z, WANG F, FAN Z, et al. Whole-Tooth Regeneration by Allogeneic Cell Reassociation in Pig Jawbone. Tissue Eng Part A. 2019;25(17-18): 1202-1212. [3] YANG KC, KITAMURA Y, WU CC, et al. Tooth Germ-Like Construct Transplantation for Whole-Tooth Regeneration: An In Vivo Study in the Miniature Pig. Artif Organs. 2016;40(4):E39-50. [4] YU T, KLEIN OD. Molecular and cellular mechanisms of tooth development, homeostasis and repair. Development. 2020;147(2): dev184754. [5] HU L, LIU Y, WANG S. Stem cell-based tooth and periodontal regeneration. Oral Dis. 2018;24(5):696-705. [6] Baranova J, Büchner D, Götz W, et al. Tooth Formation: Are the Hardest Tissues of Human Body Hard to Regenerate? Int J Mol Sci. 2020;21(11): 4031. [7] ABDEL MEGUID E, KE Y, Ji J, et al. Stem cells applications in bone and tooth repair and regeneration: New insights, tools, and hopes. J Cell Physiol. 2018; 233(3):1825-1835. [8] BALIC A, THESLEFF I. Tissue Interactions Regulating Tooth Development and Renewal. Curr Top Dev Biol. 2015;115:157-186. [9] CAPURRO M, IZUMIKAWA T, SUAREZ P, et al. Glypican-6 promotes the growth of developing long bones by stimulating Hedgehog signaling. J Cell Biol. 2017; 216(9):2911-2926. [10] TOWNLEY RA, BÜLOW HE. Deciphering functional glycosaminoglycan motifs in development. Curr Opin Struct Biol. 2018;50:144-154. [11] THESLEFF I, JALKANEN M, VAINIO S, et al. Cell surface proteoglycan expression correlates with epithelial-mesenchymal interaction during tooth morphogenesis. Dev Biol. 1988;129(2):565-572. [12] YAMADA K, YAMADA T, SASAKI T, et al. Light and electron microscopical immunohistochemical localization of large proteoglycans in human tooth germs at the bell stage. Histochem J. 1997;29(2):167-175. [13] WU J, LI H, HAN L, et al. The spatiotemporal expression pattern of Syndecans in murine embryonic teeth. Gene Expr Patterns. 2020;36:119109. [14] WU J, TIAN Y, HAN L, et al. FAM20B-catalyzed glycosaminoglycans control murine tooth number by restricting FGFR2b signaling. BMC Biol. 2020;18(1):87. [15] IOZZO RV, SCHAEFER L. Proteoglycan form and function: A comprehensive nomenclature of proteoglycans. Matrix Biol. 2015;42:11-55. [16] MIKAMI T, KITAGAWA H. Sulfated glycosaminoglycans: their distinct roles in stem cell biology. Glycoconj J. 2017;34(6):725-735. [17] SMOCK RG, MEIJERS R. Roles of glycosaminoglycans as regulators of ligand/receptor complexes. Open Biol. 2018;8(10):180026. [18] YU P, PEARSON CS, GELLER HM. Flexible Roles for Proteoglycan Sulfation and Receptor Signaling. Trends Neurosci. 2018;41(1):47-61. [19] ZONG C, VENOT A, LI X, et al. Heparan Sulfate Microarray Reveals That Heparan Sulfate-Protein Binding Exhibits Different Ligand Requirements. J Am Chem Soc. 2017;139(28):9534-9543. [20] BIRBRAIR A. Stem Cell Microenvironments and Beyond. Adv Exp Med Biol. 2017;1041:1-3. [21] BALIC A. Biology Explaining Tooth Repair and Regeneration: A Mini-Review. Gerontology. 2018;64(4):382-388. [22] THESLEFF I. From understanding tooth development to bioengineering of teeth. Eur J Oral Sci. 2018;126 Suppl 1:67-71. [23] LIU Z, CHEN T, BAI D, et al. Smad7 Regulates Dental Epithelial Proliferation during Tooth Development. J Dent Res. 2019;98(12):1376-1385. [24] KLEIN OD, MINOWADA G, PETERKOVA R, et al. Sprouty genes control diastema tooth development via bidirectional antagonism of epithelial-mesenchymal FGF signaling. Dev Cell. 2006;11(2):181-190. [25] NAKATO H, LI JP. Functions of Heparan Sulfate Proteoglycans in Development: Insights From Drosophila Models. Int Rev Cell Mol Biol. 2016;325:275-293. [26] DÍAZ-BALZAC CA, LÁZARO-PEÑA MI, TECLE E, et al. Complex cooperative functions of heparan sulfate proteoglycans shape nervous system development in Caenorhabditis elegans. G3 (Bethesda). 2014; 4(10):1859-1870. [27] SOARES DA COSTA D, REIS RL, PASHKULEVA I. Sulfation of Glycosaminoglycans and Its Implications in Human Health and Disorders. Annu Rev Biomed Eng. 2017;19:1-26. [28] HAYES AJ, MITCHELL RE, BASHFORD A, et al. Expression of glycosaminoglycan epitopes during zebrafish skeletogenesis. Dev Dyn. 2013;242(6):778-789. [29] YAN Z, CHEN G, YANG Y, et al. Expression and roles of syndecan-4 in dental epithelial cell differentiation. Int J Mol Med. 2014;34(5):1301-1308. [30] RANDILINI A, FUJIKAWA K, SHIBATA S. Expression, localization and synthesis of small leucine-rich proteoglycans in developing mouse molar tooth germ. Eur J Histochem. 2020;64(1):3092. [31] GOLDBERG M, SEPTIER D, OLDBERG A, et al. Fibromodulin-deficient mice display impaired collagen fibrillogenesis in predentin as well as altered dentin mineralization and enamel formation. J Histochem Cytochem. 2006;54(5):525-537. [32] GOLDBERG M, SEPTIER D, RAPOPORT O, et al. Targeted disruption of two small leucine-rich proteoglycans, biglycan and decorin, excerpts divergent effects on enamel and dentin formation. Calcif Tissue Int. 2005;77(5):297-310. [33] KERO D, BILANDZIJA TS, ARAPOVIC LL, et al. Syndecans and Enzymes Involved in Heparan Sulfate Biosynthesis and Degradation Are Differentially Expressed During Human Odontogenesis. Front Physiol. 2018;9:732. [34] GALBRAITH DB, CUTLER LS, KOLLAR EJ. The correlation of temporal regulation of glycosaminoglycan synthesis with morphogenetic events in mouse tooth development. Arch Oral Biol. 1992;37(8):623-628. [35] JIANG B, XU F, LI L, et al. The inhibition of glycosaminoglycan incorporation influences the cell proliferation and cytodifferentiation in cultured embryonic mouse molars. J Mol Histol. 2019;50(1):11-19. [36] LIU L, CHEN W, LI L, et al. Inhibition of chondroitin sulfate glycosaminoglycans incorporation affected odontoblast differentiation in cultured embryonic mouse molars. J Mol Histol. 2017;48(5-6):337-345. [37] KOIKE T, IZUMIKAWA T, TAMURA J, et al. FAM20B is a kinase that phosphorylates xylose in the glycosaminoglycan-protein linkage region. Biochem J. 2009; 421(2):157-162. [38] ZHANG H, ZHU Q, CUI J, et al. Structure and evolution of the Fam20 kinases. Nat Commun. 2018;9(1):1218. [39] TIAN Y, MA P, LIU C, et al. Inactivation of Fam20B in the dental epithelium of mice leads to supernumerary incisors. Eur J Oral Sci. 2015;123(6):396-402. [40] JUURI E, JUSSILA M, SEIDEL K, et al. Sox2 marks epithelial competence to generate teeth in mammals and reptiles. Development. 2013;140(7): 1424-1432. [41] JUURI E, SAITO K, AHTIAINEN L, et al. Sox2+ stem cells contribute to all epithelial lineages of the tooth via Sfrp5+ progenitors. Dev Cell. 2012; 23(2):317-328. [42] SAEI AREZOUMAND K, ALIZADEH E, PILEHVAR-SOLTANAHMADI Y, et al. An overview on different strategies for the stemness maintenance of MSCs. Artif Cells Nanomed Biotechnol. 2017;45(7):1255-1271. [43] JIN S, COLLIN J, ZHU L, et al. A Novel Role for miR-1305 in Regulation of Pluripotency-Differentiation Balance, Cell Cycle, and Apoptosis in Human Pluripotent Stem Cells. Stem Cells. 2016;34(9):2306-2317. [44] KRAUSHAAR DC, YAMAGUCHI Y, WANG L. Heparan sulfate is required for embryonic stem cells to exit from self-renewal. J Biol Chem. 2015; 290(38):23023. [45] IZUMIKAWA T, SATO B, KITAGAWA H. Chondroitin sulfate is indispensable for pluripotency and differentiation of mouse embryonic stem cells. Sci Rep. 2014;4:3701. [46] HELLEDIE T, DOMBROWSKI C, RAI B, et al. Heparan sulfate enhances the self-renewal and therapeutic potential of mesenchymal stem cells from human adult bone marrow. Stem Cells Dev. 2012;21(11):1897-1910. [47] LING L, REN X, CAO X, et al. Enhancing the Efficacy of Stem Cell Therapy with Glycosaminoglycans. Stem Cell Reports. 2020;14(1):105-121. [48] KRAUSHAAR DC, DALTON S, WANG L. Heparan sulfate: a key regulator of embryonic stem cell fate. Biol Chem. 2013;394(6):741-751. [49] KRAUSHAAR DC, RAI S, CONDAC E, et al. Heparan sulfate facilitates FGF and BMP signaling to drive mesoderm differentiation of mouse embryonic stem cells. J Biol Chem. 2012;287(27):22691-22700. [50] QU C, RILLA K, TAMMI R, et al. Extensive CD44-dependent hyaluronan coats on human bone marrow-derived mesenchymal stem cells produced by hyaluronan synthases HAS1, HAS2 and HAS3. Int J Biochem Cell Biol. 2014;48:45-54. [51] CHEN PY, HUANG LL, HSIEH HJ. Hyaluronan preserves the proliferation and differentiation potentials of long-term cultured murine adipose-derived stromal cells. Biochem Biophys Res Commun. 2007;360(1):1-6. [52] WONG TY, CHANG CH, YU CH, et al. Hyaluronan keeps mesenchymal stem cells quiescent and maintains the differentiation potential over time. Aging Cell. 2017;16(3):451-460. [53] AI X, DO AT, LOZYNSKA O, et al. QSulf1 remodels the 6-O sulfation states of cell surface heparan sulfate proteoglycans to promote Wnt signaling. J Cell Biol. 2003;162(2):341-351. [54] LANNER F, LEE KL, SOHL M, et al. Heparan sulfation-dependent fibroblast growth factor signaling maintains embryonic stem cells primed for differentiation in a heterogeneous state. Stem Cells. 2010; 28(2):191-200. [55] HUANG ML, MICHALAK AL, FISHER CJ, et al. Small Molecule Antagonist of Cell Surface Glycosaminoglycans Restricts Mouse Embryonic Stem Cells in a Pluripotent State. Stem Cells. 2018;36(1):45-54. [56] TITMARSH DM, TAN CL, GLASS NR, et al. Microfluidic Screening Reveals Heparan Sulfate Enhances Human Mesenchymal Stem Cell Growth by Modulating Fibroblast Growth Factor-2 Transport. Stem Cells Transl Med. 2017;6(4):1178-1190. [57] YAMADA T, KEREVER A, YOSHIMURA Y, et al. Heparan sulfate alterations in extracellular matrix structures and fibroblast growth factor-2 signaling impairment in the aged neurogenic niche. J Neurochem. 2017;142(4):534-544. [58] QIU H, SHI S, YUE J, et al. A mutant-cell library for systematic analysis of heparan sulfate structure-function relationships. Nat Methods. 2018;15(11):889-899. [59] WANG X, CORNELIS FMF, LORIES RJ, et al. Exostosin-1 enhances canonical Wnt signaling activity during chondrogenic differentiation. Osteoarthritis Cartilage. 2019;27(11):1702-1710. [60] MORIMOTO-TOMITA M, UCHIMURA K, WERB Z, et al. Cloning and characterization of two extracellular heparin-degrading endosulfatases in mice and humans. J Biol Chem. 2002;277(51):49175-49185. [61] DHOOT GK, GUSTAFSSON MK, AI X, et al. Regulation of Wnt signaling and embryo patterning by an extracellular sulfatase. Science. 2001; 293(5535):1663-1666. [62] SASAKI N, OKISHIO K, UI-TEI K, et al. Heparan sulfate regulates self-renewal and pluripotency of embryonic stem cells. J Biol Chem. 2008; 283(6):3594-3606. [63] CHEN YH, NARIMATSU Y, CLAUSEN TM, et al. The GAGOme: a cell-based library of displayed glycosaminoglycans. Nat Methods. 2018; 15(11):881-888. [64] ZHANG X, PAGADALA V, JESTER HM, et al. Chemoenzymatic synthesis of heparan sulfate and heparin oligosaccharides and NMR analysis: paving the way to a diverse library for glycobiologists. Chem Sci. 2017; 8(12):7932-7940. |
| [1] | Zhu Chan, Han Xuke, Yao Chengjiao, Zhou Qian, Zhang Qiang, Chen Qiu. Human salivary components and osteoporosis/osteopenia [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(9): 1439-1444. |
| [2] | Jin Tao, Liu Lin, Zhu Xiaoyan, Shi Yucong, Niu Jianxiong, Zhang Tongtong, Wu Shujin, Yang Qingshan. Osteoarthritis and mitochondrial abnormalities [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(9): 1452-1458. |
| [3] | Zhang Lichuang, Xu Hao, Ma Yinghui, Xiong Mengting, Han Haihui, Bao Jiamin, Zhai Weitao, Liang Qianqian. Mechanism and prospects of regulating lymphatic reflux function in the treatment of rheumatoid arthritis [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(9): 1459-1466. |
| [4] | Wang Baojuan, Zheng Shuguang, Zhang Qi, Li Tianyang. Miao medicine fumigation can delay extracellular matrix destruction in a rabbit model of knee osteoarthritis [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(8): 1180-1186. |
| [5] | Wang Jing, Xiong Shan, Cao Jin, Feng Linwei, Wang Xin. Role and mechanism of interleukin-3 in bone metabolism [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(8): 1260-1265. |
| [6] | Zhu Chan, Han Xuke, Yao Chengjiao, Zhang Qiang, Liu Jing, Shao Ming. Acupuncture for Parkinson’s disease: an insight into the action mechanism in animal experiments [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(8): 1272-1277. |
| [7] | Gao Yujin, Peng Shuanglin, Ma Zhichao, Lu Shi, Cao Huayue, Wang Lang, Xiao Jingang. Osteogenic ability of adipose stem cells in diabetic osteoporosis mice [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 999-1004. |
| [8] | Guo Jia, Ding Qionghua, Liu Ze, Lü Siyi, Zhou Quancheng, Gao Yuhua, Bai Chunyu. Biological characteristics and immunoregulation of exosomes derived from mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1093-1101. |
| [9] | Wu Weiyue, Guo Xiaodong, Bao Chongyun. Application of engineered exosomes in bone repair and regeneration [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1102-1106. |
| [10] | Zhou Hongqin, Wu Dandan, Yang Kun, Liu Qi. Exosomes that deliver specific miRNAs can regulate osteogenesis and promote angiogenesis [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1107-1112. |
| [11] | Zhang Jinglin, Leng Min, Zhu Boheng, Wang Hong. Mechanism and application of stem cell-derived exosomes in promoting diabetic wound healing [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1113-1118. |
| [12] | Huang Chenwei, Fei Yankang, Zhu Mengmei, Li Penghao, Yu Bing. Important role of glutathione in stemness and regulation of stem cells [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1119-1124. |
| [13] | Hui Xiaoshan, Bai Jing, Zhou Siyuan, Wang Jie, Zhang Jinsheng, He Qingyong, Meng Peipei. Theoretical mechanism of traditional Chinese medicine theory on stem cell induced differentiation [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1125-1129. |
| [14] | An Weizheng, He Xiao, Ren Shuai, Liu Jianyu. Potential of muscle-derived stem cells in peripheral nerve regeneration [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1130-1136. |
| [15] | Fan Yiming, Liu Fangyu, Zhang Hongyu, Li Shuai, Wang Yansong. Serial questions about endogenous neural stem cell response in the ependymal zone after spinal cord injury [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1137-1142. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||