Chinese Journal of Tissue Engineering Research ›› 2021, Vol. 25 ›› Issue (16): 2569-2574.doi: 10.3969/j.issn.2095-4344.3154
Previous Articles Next Articles
New strategies and problems of affinity peptide in cartilage tissue engineering
Fu Liwei1, 2, Yang Zhen1, 2, Li Hao1, 2, Gao Cangjian1, 2, Sui Xiang1, Liu Shuyun1, Guo Quanyi1
- 1Institute of Orthopedics, the First Medical Center, Chinese PLA General Hospital, Beijing Key Laboratory of Regenerative Medicine in Orthopedics, Key Laboratory of Musculoskeletal Trauma & War Injuries, PLA, Beijing 100853, China; 2Medical College of Nankai University, Tianjin 300071, China
-
Received:2020-05-30Revised:2020-06-02Accepted:2020-07-09Online:2021-06-08Published:2021-01-07 -
Contact:Guo Quanyi, Institute of Orthopedics, the First Medical Center, Chinese PLA General Hospital, Beijing Key Laboratory of Regenerative Medicine in Orthopedics, Key Laboratory of Musculoskeletal Trauma & War Injuries, PLA, Beijing 100853, China -
About author:Fu Liwei, Master candidate, Institute of Orthopedics, the First Medical Center, Chinese PLA General Hospital, Beijing Key Laboratory of Regenerative Medicine in Orthopedics, Key Laboratory of Musculoskeletal Trauma & War Injuries, PLA, Beijing 100853, China; Medical College of Nankai University, Tianjin 300071, China -
Supported by:the National Key Research and Development Plan Project, No. 2019YFA0110600 (to GQY); the National Natural Science Foundation of China, No. 81772319 (to GQY)
CLC Number:
Cite this article
Fu Liwei, Yang Zhen, Li Hao, Gao Cangjian, Sui Xiang, Liu Shuyun, Guo Quanyi . New strategies and problems of affinity peptide in cartilage tissue engineering[J]. Chinese Journal of Tissue Engineering Research, 2021, 25(16): 2569-2574.
share this article
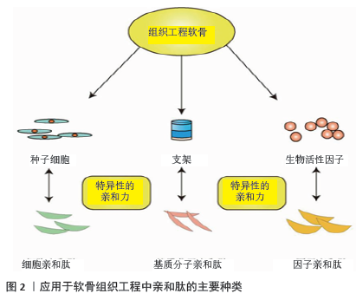
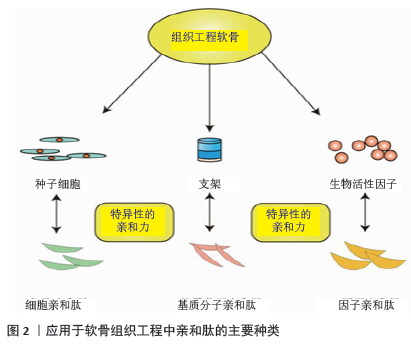
2.1 亲和肽应用于软骨组织工程的理论基础及分类 组织工程软骨是围绕种子细胞、支架及因子3个主要因素构建的,任何一个因素的性质都能够影响到最后软骨修复的质量,如修复部位干细胞募集的不足,生物活性因子浓度的缺乏等常导致修复效果不理想。而亲和肽的发现为软骨组织工程支架的构建提供了新的方向。人们在不断地筛选和鉴别中发现,许多特定氨基酸序列的多肽能够与一些细胞、因子、分子特异性地结合,具有亲和作用,根据作用对象的不同,他们可以分为细胞亲和肽、因子亲和肽及细胞外基质分子亲和肽,如图2所示,这些肽的应用会显著提高组织工程细胞和因子的作用强度,在节省成本的同时对于缺损软骨的修复效果也有很好的增强作用[15-16,18]。 "

2.2 细胞亲和肽 细胞对于生物材料的黏附性一直是评价组织工程的指标之一,这与生物材料的表面电荷和粗糙度、材料表面的生物信号等因素息息相关[19-20]。修饰支架以提高细胞的黏附性是组织工程研究的热点,比如通过修饰支架表面介导受体和细胞相互作用等[21]。对细胞具有亲和作用的多肽,作为新兴的生物材料由于其特殊的生物特性引起了人们的关注[22]。RGD肽是纤维连接蛋白中最小的基本细胞黏附肽序列,被广泛用于促进细胞在生物材料表面的黏附,它也是被最早用于软骨组织工程的多肽之一[23-24]。PARK等[25]通过将RGD肽中的羧基与壳聚糖共聚物中的残留胺基偶联来制备RGD共轭的壳聚糖水凝胶共聚物,结果表明,将RGD与壳聚糖水凝胶缀合可改善细胞活力和增殖,同时促进额外的细胞基质表达。在一项最新的研究中,AHN等[26]先利用聚富马酸丙二酯制作成3D打印生物墨水并使用微立体光刻技术创建大孔细胞支架,然后又将RGD肽固定在支架上,实验研究表明RGD肽与细胞外基质材料结合可以成功地促进软骨特异性细胞外基质的积累,即糖胺聚糖和Ⅱ型胶原在三维支架内的聚集。不过RGD肽是广义上的亲和肽,它对于多种细胞都具有黏附和募集作用,并不具有特异性。接下来将介绍几个对特定的细胞具有亲和力的多肽序列的制备和应用。 噬菌体展示技术是指在丝状噬菌体表面表达蛋白质,其中包括抗体和肽,然后构建一个展示文库,并可从中选择和纯化出携带具有所需结合特异性序列的特定噬菌体颗粒,是一种用于分离鉴定具有特定功能分子的方法[27-29]。SHAO等[30]利用噬菌体显示生物扫描技术鉴定了一个含有7个氨基酸的多肽E7,该肽对骨髓间充质干细胞具有很高的特异性,能够有效地与骨髓间充质干细胞进行亲和相互作用。随后他们将E7共价连接到聚己内酯电纺网上并植入到大鼠膝关节软骨缺损部位,结果表明所鉴定的E7肽对间充质干细胞具有较高的特异性,并且将此肽共价连接到人工合成的聚己内酯网状物后显著提高了聚己内酯在体内的间充质干细胞募集能力。同样,MENG等[31]将设计的一种结合了E7肽修饰的脱钙骨基质颗粒和壳聚糖水凝胶复合三维支架用于软骨的再生,E7肽通过共价缀合到脱钙骨基质颗粒上,根据共聚焦激光显微图像显示脱钙骨基质-E7/壳聚糖支架显著增加了在该支架上生长的细胞数量,同时体外增殖结果表明脱钙骨基质-E7/壳聚糖支架具有出色的维持骨髓间充质干细胞募集的作用,是一种具有前景的间充质干细胞归巢装置。除此之外,LI等[32]利用E7肽修饰了一个可降解的聚酯,研究表明该生物材料在软骨组织工程中具有良好的应用价值。LU等[33]利用噬菌体展示技术将功能肽PFS鉴别为骨髓间充质干细胞亲和肽,并将其设计成功能化的自组装肽段复合水凝胶支架,用于修复兔膝关节全层软骨缺损,植入1周后在共聚焦显微镜下通过CD29和CD90双重标记监测干细胞募集,PFS复合水凝胶支架修复的软骨图像表现出相对更多的CD29+ /CD90+细胞,与微骨折或对照支架相比可增强内源性干细胞归巢并加速软骨形成过程。 除了骨髓间充质干细胞,常用于软骨组织工程的细胞还有软骨细胞和其他几种类型的间充质干细胞等[34- 35]。CHEUNG等[36]利用噬菌体展示技术鉴定了几个特异性靶向结合软骨细胞的肽序列,并且通过实验初步证明了这些肽段序列的功能性,期望可以用于软骨治疗。此后PI等[37]又通过噬菌体展示技术鉴定了一个软骨细胞亲和肽,并将聚乙烯亚胺与亲和肽进行共价结合构建了软骨靶向治疗的非病毒载体,体内实验结构表明肽修饰的聚乙烯亚胺具有明显的软骨亲和性,可作为软骨病变的特异性软骨靶向载体。此外,CHEN等[38]又利用该亲和肽制备了软骨细胞亲和肽修饰聚乙二醇化聚酰胺偶联物作为纳米材料,用于将药物定向输送到软骨。 作为间充质干细胞的一种,滑膜间充质干细胞具有很大的软骨再生方面的潜力[39]。SHAO等[40]通过噬菌体展示技术鉴定了一个与滑膜间充质干细胞具有较高特异性亲和力的具有7个氨基酸序列的肽L7,并将L7共价连接到聚己内酯电纺网和人脱钙骨支架上,荧光显微镜结果显示在人、大鼠和兔滑膜间充质干细胞中,L7的平均荧光强度分别是对照组的23.4,21.3,18.0倍,提示L7对滑膜间充质干细胞具有较高的亲和力和特异性,可与人、大鼠和兔滑膜间充质干细胞结合,无种属特异性。同时L7修饰的聚已内酯电纺丝网和人脱钙骨支架均表现出对滑膜间充质干细胞黏附性的增强,为软骨组织工程提供一个很有前景的思路和方法。 2.3 生物活性因子亲和肽 在软骨组织工程中,一些生物活性因子在组织再生过程中具有关键的作用,如生长因子可以通过促进间充质干细胞在体内的募集和分化来促进关节软骨的再生[41-43]。在软骨修复过程中,生长因子一般分为外源性生长因子和内源性生长因子[42-43]。外源性因子主要来源于工程支架的释放,这其中存在支架前期因子的突释、释放过程中活性的丧失、因子成本高等问题,而内源性的生长因子在体内往往是较难募集的,所以如何提高在组织修复过程中因子的利用率一直是研究人员关心的科学问题[44-45]。亲和肽的发现和应用为组织工程软骨中生长因子的设计和利用提供了新的思路。 转化生长因子β超家族已被证明能够促进干细胞在体外的分化和迁移,并能够在体内促进骨软骨组织的再生[46-47]。CHEN等[48]将利用噬菌体技术鉴定的转化生长因β1亲和肽引入多孔壳聚糖海绵中,将设计好的支架在转化生长因子β1溶液中浸泡 3 h,体外缓释实验结果表明海绵中释放的转化生长因子β1呈现缓慢的释放曲线,没有突发性释放。实验结果证明,含有转化生长因子β1亲和肽的壳聚糖海绵可显著促进兔骨软骨缺损模型中骨髓间充质干细胞的体外成软骨分化和体内软骨再生,这为缺乏外源性生长因子情况下的软骨修复提供了新的策略。鞠晓晶等[49]将可自主结合转化生长因子β1的多肽接枝到聚乙二醇水凝胶表面,改善聚乙二醇的生物活性,体外实验结果表明,接枝转化生长因子β1亲和肽的聚乙二醇水凝胶可自主结合转化生长因子β1,具有良好的生物相容性。WU等[50]合成了一种含有转化生长因子β1亲和肽的侧基的弹性聚氨酯,体内实验表明该支架在体内有一定的转化生长因子β1募集作用,同时可促进软骨前细胞ATDC5的存活和向软骨细胞的分化,在软骨组织工程中具有良好的应用前景。 骨形态发生蛋白2属于转化生长因子β超家族成员,具有诱导未分化间充质干细胞向成软骨细胞和成骨细胞定向分化与增殖的能力,促进成骨细胞分化成熟,参与骨和软骨生长发育及其重建过程,是骨软骨组织工程中运用相对广泛的因子之一[51-52]。CRISPIM等[53]合成了骨形态发生蛋白2亲合肽,并将它固定在聚已内酯膜上,实验结果证明了固定在膜上的亲和肽具有捕获骨形态发生蛋白的能力。同样的,LEE等[54]设计了一个可以结合内源性和外源性骨形态发生蛋白2用于骨再生的纳米纤维凝胶,其中包含了一个富含羧基的多肽结构域和一个包含有骨形态发生蛋白2结合亲和肽的结构,实验结果表明该凝胶显著减少了促进骨生长所需要的外源性骨形态发生蛋白2剂量,甚至在没有外源性骨形态发生蛋白2的情况下也促进较大程度的骨生长。 2.4 细胞外基质的亲和肽 软骨细胞外基质主要由Ⅱ型胶原(占总胶原含量的90%-95%)和糖胺聚糖 (占软骨湿质量的10%-20%)组成,在软骨细胞功能调节中起着重要的作用[55-56]。对于各种各样的软骨组织工程支架而言,细胞外基质分子的加入对软骨细胞自然生存环境模拟很有帮助[57-58]。KATSUMATA等[59-60]通过筛选发现,胎盘生长因子2的特定肽段结构区域对多种软骨细胞外基质蛋白具有异常高的亲和力,并且使用交联剂将该肽段与肿瘤坏死因子α共价缀合注射到小鼠关节炎模型中,发现肽功能化的肿瘤坏死因子α能够增长保留时间,显示出非常强大的治疗潜力。 NGUYEN 等[61]根据软骨细胞外基质的生物学结构去构建能够产生不同胶原和糖胺聚糖含量的功能化水凝胶,其中将基质金属蛋白酶敏感肽结合到支架设计中,发现在聚乙二醇水凝胶中加入硫酸软骨素和基质金属蛋白酶敏感肽可以诱导高水平的Ⅱ型胶原蛋白和低水平的蛋白多糖表达,导致与关节软骨表面区相似的生物学结构。在之后的研究中,他们构建了一种基于聚乙二醇的3层水凝胶,分别为掺入硫酸软骨素和基质金属蛋白酶敏感肽的表层、掺入硫酸软骨素的中间层、掺入透明质酸的底层,实验结果表明该支架成功模拟了天然软骨空间变化的生物学特性[62]。 CHOW等[63]通过噬菌体展示技术鉴定了2个糖胺聚糖的特异性结合序列,一个是透明质酸结合肽,另一个是硫酸软骨素结合肽,然后将这两个特定序列与聚己内酯结合用于电纺,通过依次电纺含有质量浓度0-3 g/L的肽-聚已内酯结合物溶液形成了单一的肽浓度梯度,从而达到模拟从关节面到骨糖胺聚糖浓度递增梯度的目的。该实验是一种创建具有生物分子仿生梯度的支架策略,运用透明质酸和硫酸软骨素结合肽对电纺纤维表面进行功能化,这一策略提供了一个平台来创建临床相关的工程化组织结构,这些结构可以实现天然生物组织的生物学功能。TSANAKTSIDOU等[64]用硫酸软骨素结合肽对可注射甲基丙烯酸透明质酸水凝胶对进行功能化,实验结果证明人工合成的含有人间充质干细胞的水凝胶可用于局部注射修复软骨损伤。 HESSE等[65]采用不同降解率的水凝胶联合RGD、CKLER和CWYRGRL三种多肽培养人间充质干细胞和猪软骨细胞,其中RGD为细胞黏附肽段;CKLER是一种核心蛋白聚糖衍生的肽序列,它与Ⅱ型胶原强烈结合;CWYRGRL是人工Ⅱ型胶原结合位点。实验结果表明,肽功能化的水凝胶能够指导细胞有丝分裂、细胞形态和软骨细胞外基质在整个构建过程中的分布,但其中胶原沉积的效果并不是很显著,需要进一步的探索。JIANG等[66]开发了一种软骨靶向短肽序列WYRGRL修饰的聚乙二醇/聚乳酸-羟基乙酸共聚物制成的纳米颗粒,并且证明了该纳米颗粒在体外与软骨组织有较强的结合力,是一种有前景的药物递送系统。 应用于软骨组织工程的亲和肽总结见表1。 "

| [1] MOLLON B, KANDEL R, CHAHAL J, et al. The clinical status of cartilage tissue regeneration in humans. Osteoarthritis Cartilage. 2013;21(12): 1824-1833. [2] BEDI A, FEELEY BT, WILLIAMS RJ 3RD. Management of articular cartilage defects of the knee. J Bone Joint Surg Am. 2010;92(4):994-1009. [3] 章亚东.软骨损伤修复技术的既往与未来[J].中国骨与关节杂志, 2018,7(8):561-563. [4] MARTEL-PELLETIER J, BARR AJ, CICUTTINI FM, et al. Osteoarthritis. Nat Rev Dis Primers. 2016;2:160-172. [5] 张志成,孙天胜.软骨损伤临床治疗进展[J].中华外科杂志,2006, 44(12):862-864. [6] KWON H, BROWN WE, LEE CA, et al. Surgical and tissue engineering strategies for articular cartilage and meniscus repair. Nat Rev Rheumatol. 2019;15(9):550-570. [7] VASILIADIS HS, WASIAK J. Autologous chondrocyte implantation for full thickness articular cartilage defects of the knee. Cochrane Database Syst Rev. 2010;(10):CD003323. [8] BHATTACHARJEE M, COBURN J, CENTOLA M, et al. Tissue engineering strategies to study cartilage development, degeneration and regeneration. Adv Drug Deliv Rev. 2015;84:107-122. [9] MAKRIS EA, GOMOLL AH, MALIZOS KN, et al. Repair and tissue engineering techniques for articular cartilage. Nat Rev Rheumatol. 2015;11(1):21-34. [10] NUKAVARAPU SP, DORCEMUS DL. Osteochondral tissue engineering: current strategies and challenges. Biotechnol Adv. 2013;31(5):706-721. [11] LONGLEY R, FERREIRA AM, GENTILE P. Recent Approaches to the Manufacturing of Biomimetic Multi-Phasic Scaffolds for Osteochondral Regeneration. Int J Mol Sci. 2018;19(6):1755-1772. [12] GADJANSKI I, VUNJAK-NOVAKOVIC G. Challenges in engineering osteochondral tissue grafts with hierarchical structures. Expert Opin Biol Ther. 2015;15(11):1583-1599. [13] LEE AC, HARRIS JL, KHANNA KK, et al. A Comprehensive Review on Current Advances in Peptide Drug Development and Design. Int J Mol Sci. 2019;20(10):2383-2404. [14] CLAASSEN H, SCHICHT M, PAULSEN F. Impact of sex hormones, insulin, growth factors and peptides on cartilage health and disease. Prog Histochem Cytochem. 2011;45(4):239-293. [15] HOLMES TC. Novel peptide-based biomaterial scaffolds for tissue engineering. Trends Biotechnol. 2002;20(1):16-21. [16] SHAH RN, SHAH NA, DEL ROSARIO LIM MM, et al. Supramolecular design of self-assembling nanofibers for cartilage regeneration. Proc Natl Acad Sci USA. 2010;107(8):3293-3298. [17] BONZANI IC, GEORGE JH, STEVENS MM. Novel materials for bone and cartilage regeneration. Curr Opin Chem Biol. 2006;10(6):568-575. [18] HASTAR N, ARSLAN E, GULER MO, et al. Peptide-Based Materials for Cartilage Tissue Regeneration. Adv Exp Med Biol. 2017;1030:155-166. [19] STEWARD AJ, LIU Y, WAGNER DR. Engineering cell attachments to scaffolds in cartilage tissue engineering. JOM Journal of the Minerals, Metals and Materials Society. 2011;63(4):74-82. [20] 余劭婷,秦金桥,关国平,等.丝素纤维表面改性提高细胞粘附性能[J].生物医学工程学进展,2016,37(3):144-149. [21] RAHMANY MB, VAN DYKE M. Biomimetic approaches to modulate cellular adhesion in biomaterials: A review. Acta Biomater. 2013;9(3): 5431-5437. [22] LIU Q, JIA Z, DUAN L, et al. Functional peptides for cartilage repair and regeneration. Am J Transl Res. 2018;10(2):501-510. [23] HERSEL U, DAHMEN C, KESSLER H. RGD modified polymers: biomaterials for stimulated cell adhesion and beyond. Biomaterials. 2003;24(24):4385-4415. [24] ALIPOUR M, BANESHI M, HOSSEINKHANI S, et al. Recent progress in biomedical applications of RGD-based ligand: From precise cancer theranostics to biomaterial engineering: A systematic review. J Biomed Mater Res A. 2020;108(4):839-850. [25] PARK KD, PARK KM, JOUNG YK. RGD-Conjugated Chitosan-Pluronic Hydrogels as a Cell Supported Scaffold for Articular Cartilage Regeneration. Macromol Res. 2008;16(6):517-523. [26] AHN CB, KIM Y, PARK SJ, et al. Development of arginine-glycine-aspartate-immobilized 3D printed poly(propylene fumarate) scaffolds for cartilage tissue engineering. J Biomater Sci Polym Ed. 2018; 29(7-9):917-931. [27] HOOGENBOOM HR, CHAMES P. Natural and designer binding sites made by phage display technology. Immunol Today. 2000;21(8): 371-378. [28] AZZAZY HM, HIGHSMITH WE JR. Phage display technology: clinical applications and recent innovations. Clin Biochem. 2002;35(6): 425-445. [29] 侯利丹,孟祥军.噬菌体展示技术与肿瘤靶向治疗[J].肿瘤研究与临床,2017,29(3):214-216. [30] SHAO Z, ZHANG X, PI Y, et al. Polycaprolactone electrospun mesh conjugated with an MSC affinity peptide for MSC homing in vivo. Biomaterials. 2012;33(12):3375-3387. [31] MENG Q, MAN Z, DAI L, et al. A composite scaffold of MSC affinity peptide-modified demineralized bone matrix particles and chitosan hydrogel for cartilage regeneration. Sci Rep. 2015;5:17802. [32] LI Q, XING D, MA L, et al. Synthesis of E7 peptide-modified biodegradable polyester with the improving affinity to mesenchymal stem cells. Mater Sci Eng C Mater Biol Appl. 2017;73:562-568. [33] LU J, SHEN X, SUN X, et al. Increased recruitment of endogenous stem cells and chondrogenic differentiation by a composite scaffold containing bone marrow homing peptide for cartilage regeneration. Theranostics. 2018;8(18):5039-5058. [34] SONG L, BAKSH D, TUAN RS. Mesenchymal stem cell-based cartilage tissue engineering: cells, scaffold and biology. Cytotherapy. 2004; 6(6):596-601. [35] MARSANO A, MILLWARD-SADLER SJ, SALTER DM, et al. Differential cartilaginous tissue formation by human synovial membrane, fat pad, meniscus cells and articular chondrocytes. Osteoarthritis Cartilage 2007;15(1):48-58. [36] CHEUNG CS, LUI JC, BARON J. Identification of chondrocyte-binding peptides by phage display. J Orthop Res. 2013;31(7):1053-1058. [37] PI Y, ZHANG X, SHI J, et al. Targeted delivery of non-viral vectors to cartilage in vivo using a chondrocyte-homing peptide identified by phage display. Biomaterials. 2011;32(26):6324-6332. [38] CHEN D, DING B, YAN X, et al. Chondrocyte affinity peptide modified PAMAM conjugate as a nanoplatform for targeting and retention in cartilage. Nanomedicine (Lond). 2018;13(7):749-767. [39] SHIRASAWA S, SEKIYA I, SAKAGUCHI Y, et al. In vitro chondrogenesis of human synovium-derived mesenchymal stem cells: optimal condition and comparison with bone marrow-derived cells. J Cell Biochem. 2006; 97(1):84-97. [40] SHAO Z, ZHANG X, PI Y, et al. Surface modification on polycaprolactone electrospun mesh and human decalcified bone scaffold with synovium-derived mesenchymal stem cells-affinity peptide for tissue engineering. J Biomed Mater Res A. 2015;103(1):318-329. [41] BLUNK T, SIEMINSKI AL, GOOCH KJ, et al. Differential effects of growth factors on tissue-engineered cartilage. Tissue Eng. 2002;8(1):73-84. [42] FORTIER LA, BARKER JU, STRAUSS EJ, et al. The role of growth factors in cartilage repair. Clin Orthop Relat Res. 2011;469(10):2706-2715. [43] VINATIER C, GUICHEUX J. Cartilage tissue engineering: From biomaterials and stem cells to osteoarthritis treatments. Ann Phys Rehabil Med. 2016;59(3):139-144. [44] WHITAKER MJ, QUIRK RA, HOWDLE SM, et al. Growth factor release from tissue engineering scaffolds. J Pharm Pharmacol. 2001;53(11): 1427-1437. [45] SOHIER J, MORONI L, VAN BLITTERSWIJK C, et al. Critical factors in the design of growth factor releasing scaffolds for cartilage tissue engineering. Expert Opin Drug Deliv. 2008;5(5):543-566. [46] RAMOSHEBI LN, MATSABA TN, TEARE J, et al. Tissue engineering: 转化生长因子-beta superfamily members and delivery systems in bone regeneration. Expert Rev Mol Med. 2002;4(20):1-11. [47] MADRY H, REY-RICO A, VENKATESAN JK, et al. Transforming growth factor Beta-releasing scaffolds for cartilage tissue engineering. Tissue Eng Part B Rev. 2014;20(2):106-125. [48] CHEN J, LI Y, WANG B, et al. TGF-β1 affinity peptides incorporated within a chitosan sponge scaffold can significantly enhance cartilage regeneration. J Mater Chem B. 2018;6(4):675-687. [49] 鞠晓晶,潘国庆,刘星志,等.可自主结合转化生长因子β1聚乙二醇水凝胶的制备及其生物相容性[J]. 中国组织工程研究,2018, 22(14):2209-2214. [50] WU G, XIAO M, XIAO J, et al. Elastic polyurethane bearing pendant TGF-β1 affinity peptide for potential tissue engineering applications. Mater Sci Eng C Mater Biol Appl. 2018;83:67-77. [51] CARREIRA ACO, ZAMBUZZI WF, ROSSI MC, et al. Bone Morphogenetic Proteins: Promising Molecules for Bone Healing, Bioengineering, and Regenerative Medicine. Vitam Horm. 2015;99:293-322. [52] DRAGOO JL, CHOI JY, LIEBERMAN JR, et al. Bone induction by BMP-2 transduced stem cells derived from human fat. J Orthop Res. 2003; 21(4):622-629. [53] CRISPIM JF, FU SC, LEE YW, et al. Bioactive Tape With BMP-2 Binding Peptides Captures Endogenous Growth Factors and Accelerates Healing After Anterior Cruciate Ligament Reconstruction. Am J Sports Med. 2018;46(12):2905-2914. [54] LEE SS, HSU EL, MENDOZA M, et al. Gel scaffolds of BMP-2-binding peptide amphiphile nanofibers for spinal arthrodesis. Adv Healthc Mater. 2015;4(1):131-141. [55] VAN DER KRAAN PM, BUMA P, AND VAN KUPPEVELT T, et al. Interaction of chondrocytes, extracellular matrix and growth factors: relevance for articular cartilage tissue engineering. Osteoarthritis Cartilage. 2002; 10(8):631-637. [56] GENTILI C, CANCEDDA R. Cartilage and bone extracellular matrix. Curr Pharm Des. 2009;15(12):1334-1348. [57] CHOI B, KIM S, LIN B, et al. Cartilaginous extracellular matrix-modified chitosan hydrogels for cartilage tissue engineering. ACS Appl Mater Interfaces. 2014;6(22):20110-20121. [58] LEVETT PA, MELCHELS FP, SCHROBBACK K, et al. A biomimetic extracellular matrix for cartilage tissue engineering centered on photocurable gelatin, hyaluronic acid and chondroitin sulfate. Acta Biomater. 2014;10(1):214-223. [59] KATSUMATA K, ISHIHARA J, FUKUNAGA K, et al. Conferring extracellular matrix affinity enhances local therapeutic efficacy of anti-TNF-α antibody in a murine model of rheumatoid arthritis. Arthritis Res Ther. 2019;21(1):298. [60] MARTINO MM, BRIQUEZ PS, GÜÇ E, et al. Growth factors engineered for super-affinity to the extracellular matrix enhance tissue healing. Science. 2014;343(6173):885-888. [61] NGUYEN LH, KUDVA AK, GUCKERT NL, et al. Unique biomaterial compositions direct bone marrow stem cells into specific chondrocytic phenotypes corresponding to the various zones of articular cartilage. Biomaterials. 2011;32(5):1327-1338. [62] NGUYEN LH, KUDVA AK, SAXENA NS, et al. Engineering articular cartilage with spatially-varying matrix composition and mechanical properties from a single stem cell population using a multi-layered hydrogel. Biomaterials. 2011;32(29):6946-6952. [63] CHOW LW, ARMGARTH A, ST-PIERRE JP, et al. Peptide-directed spatial organization of biomolecules in dynamic gradient scaffolds. Adv Healthc Mater. 2014;3(9):1381-1386. [64] TSANAKTSIDOU E,KAMMON O,KIPARISSIDES C. On the synthesis and characterization of biofunctional hyaluronic acid based injectable hydrogels for the repair of cartilage lesions. Eur Polym J. 2019;114: 45-76. [65] HESSE E, FREUDENBERG U, NIEMIETZ T, et al. Peptide-functionalized starPEG/heparin hydrogels direct mitogenicity, cell morphology and cartilage matrix distribution in vitro and in vivo. J Tissue Eng Regen Med. 2018;12(1):229-239. [66] JIANG T, KAN HM, RAJPURA K, et al. Development of Targeted Nanoscale Drug Delivery System for Osteoarthritic Cartilage Tissue. J Nanosci Nanotechnol. 2018;18(4):2310-2317. |
| [1] | Lin Qingfan, Xie Yixin, Chen Wanqing, Ye Zhenzhong, Chen Youfang. Human placenta-derived mesenchymal stem cell conditioned medium can upregulate BeWo cell viability and zonula occludens expression under hypoxia [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(在线): 4970-4975. |
| [2] | Pu Rui, Chen Ziyang, Yuan Lingyan. Characteristics and effects of exosomes from different cell sources in cardioprotection [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(在线): 1-. |
| [3] | Zhang Chao, Lü Xin. Heterotopic ossification after acetabular fracture fixation: risk factors, prevention and treatment progress [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1434-1439. |
| [4] | Zhou Jihui, Li Xinzhi, Zhou You, Huang Wei, Chen Wenyao. Multiple problems in the selection of implants for patellar fracture [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1440-1445. |
| [5] | Wang Debin, Bi Zhenggang. Related problems in anatomy mechanics, injury characteristics, fixed repair and three-dimensional technology application for olecranon fracture-dislocations [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1446-1451. |
| [6] | Ji Zhixiang, Lan Changgong. Polymorphism of urate transporter in gout and its correlation with gout treatment [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1290-1298. |
| [7] | Yuan Mei, Zhang Xinxin, Guo Yisha, Bi Xia. Diagnostic potential of circulating microRNA in vascular cognitive impairment [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1299-1304. |
| [8] | Wu Xun, Meng Juanhong, Zhang Jianyun, Wang Liang. Concentrated growth factors in the repair of a full-thickness condylar cartilage defect in a rabbit [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1166-1171. |
| [9] | Li Jiacheng, Liang Xuezhen, Liu Jinbao, Xu Bo, Li Gang. Differential mRNA expression profile and competitive endogenous RNA regulatory network in osteoarthritis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1212-1217. |
| [10] | Geng Qiudong, Ge Haiya, Wang Heming, Li Nan. Role and mechanism of Guilu Erxianjiao in treatment of osteoarthritis based on network pharmacology [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1229-1236. |
| [11] | Wan Ran, Shi Xu, Liu Jingsong, Wang Yansong. Research progress in the treatment of spinal cord injury with mesenchymal stem cell secretome [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1088-1095. |
| [12] | Liao Chengcheng, An Jiaxing, Tan Zhangxue, Wang Qian, Liu Jianguo. Therapeutic target and application prospects of oral squamous cell carcinoma stem cells [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1096-1103. |
| [13] | Zhao Min, Feng Liuxiang, Chen Yao, Gu Xia, Wang Pingyi, Li Yimei, Li Wenhua. Exosomes as a disease marker under hypoxic conditions [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1104-1108. |
| [14] | Xie Wenjia, Xia Tianjiao, Zhou Qingyun, Liu Yujia, Gu Xiaoping. Role of microglia-mediated neuronal injury in neurodegenerative diseases [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1109-1115. |
| [15] | Li Shanshan, Guo Xiaoxiao, You Ran, Yang Xiufen, Zhao Lu, Chen Xi, Wang Yanling. Photoreceptor cell replacement therapy for retinal degeneration diseases [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1116-1121. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||