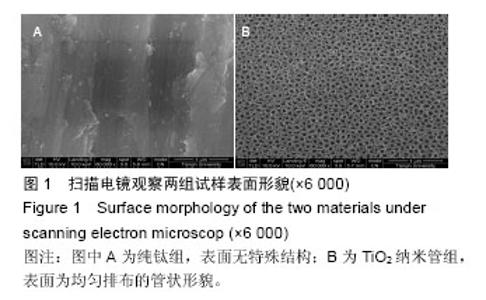
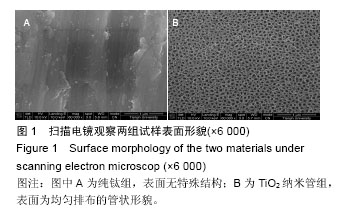
Chinese Journal of Tissue Engineering Research ›› 2016, Vol. 20 ›› Issue (43): 6416-6423.doi: 10.3969/j.issn.2095-4344.2016.43.005
Previous Articles Next Articles
Titanium dioxide nanotube composite coating: preparation and early drug-release
- 1School of Stomatology, Tianjin Medical University, Tianjin 300070, China; 2Taiping Medical University of Vietnam, Taiping 33000, Vietnam
-
Received:2016-07-23Online:2016-10-21Published:2016-10-21 -
Contact:Li Ying, M.D., Associate chief physician, School of Stomatology, Tianjin Medical University, Tianjin 300070, China -
About author:Wang Fei-fan, Studying for master’s degree, School of Stomatology, Tianjin Medical University, Tianjin 300070, China -
Supported by:the National Natural Science Foundation of China, No. 31470920, 81500886; the Natural Science Foundation of Tianjin, No. 16JCYBJC28700
CLC Number:
Cite this article
Wang Fei-fan, Song Yun-jia1, Wu Wen-meng, Lv Wu-long, Li Ying, Li Chang-yi.
share this article
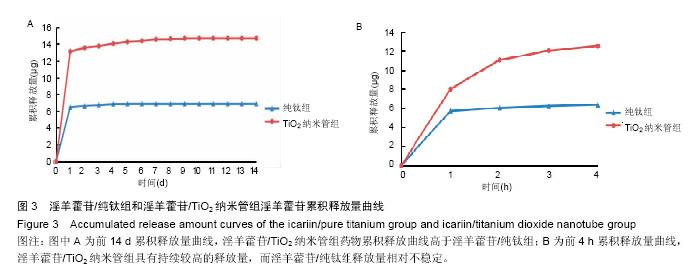
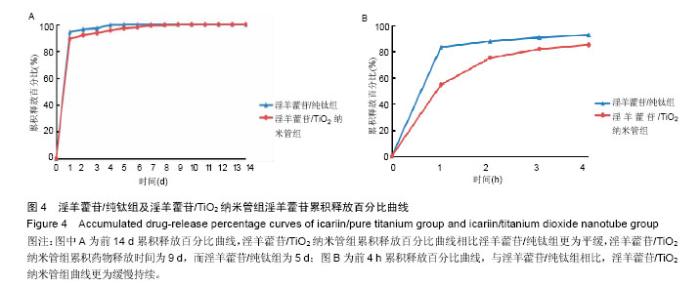
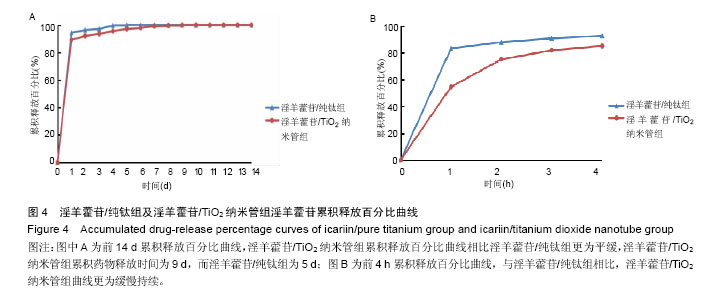
2.3 接触角检测结果分析 经过接触角检测,纯钛组为(46.3±5.4)°,TiO2纳米管组接触角为(15.8±4.2)°,组间比较差异有显著性意义(P < 0.05),TiO2纳米管组亲水性大于纯钛组。 2.4 淫羊藿苷加载量和早期释放规律检测结果分析 图3为淫羊藿苷/纯钛组和淫羊藿苷/TiO2纳米管组在前14 d及前4 h的淫羊藿苷累积释放曲线。图3A显示,淫羊藿苷/TiO2纳米管组药物累积释放曲线高于淫羊藿苷/纯钛组,经计算,前14 d淫羊藿苷/TiO2纳米管组药物累积释放量为14.68 μg,淫羊藿苷/纯钛组为6.85 μg,淫羊藿苷/TiO2纳米管组的载药量约为淫羊藿苷/纯钛组的2倍。此外,两组药物释放曲线趋势相近,可分为快速释放阶段和慢速释放阶段,前4 h为快速释放阶段,两组试样的释放量显著较高,淫羊藿苷/TiO2纳米管组在前4 h中具有持续较高的释放量,而淫羊藿苷/纯钛组的释放量相对不稳定,在1 h释放量较多,随后明显下降(图3B);在慢速释放阶段,两组均呈现线性少量释放,然而淫羊藿苷/TiO2纳米管组与淫羊藿苷/纯钛组相比具有更高更稳定的药物释放量(图3A)。 "
| [1]Portan DV,Kroustalli AA,Deligianni DD,et al.On the biocompatibility between TiO2 nanotubes layer and human osteoblasts.J Biomed Mater Res A. 2012;100 (10):2546-2553.
[2]Javed F,Romanos GE.Impact of diabetes mellitus and glycemic control on the osseointegration of dental implants: a systematic literature review.J Periodontol. 2009;80(11):1719-1730.
[3]Cochran DL,Jackson JM,Bernard JP,et al.A 5-year prospective multicenter study of early loaded titanium implants with a sandblasted and acid-etched surface. Int J Oral Maxillofac Implants.2011;26(6):1324-1332.
[4]Faeda RS,Tavares HS,Sartori R,et al.Evaluation of titanium implants with surface modification by laser beam. Biomechanical study in rabbit tibias.Braz Oral Res. 2009;23(2):137-143.
[5]吴明月,周玉琴,李全利,等.钛种植体表面磷酸化与仿生矿化的实验研究[J].中华口腔医学杂志, 2012,47(6): 354-358.
[6]Chellappa M,Anjaneyulu U,Manivasagam G,et al.Preparation and evaluation of the cytotoxic nature of TiO2 nanoparticles by direct contact method.Int J Nanomedicine.2015;10 Suppl 1:31-41.
[7]徐倩,冯青,欧俊,等.层层静电自组装构建载药种植体的研究[J].华西口腔医学杂志, 2014,32(6):537-541.
[8]Das K,Bose S,Bandyopadhyay A.TiO2 nanotubes on Ti: Influence of nanoscale morphology on bone cell-materials interaction.J Biomed Mater Res A. 2009; 90(1):225-237.
[9]Brammer KS,Oh S,Cobb CJ,et al.Improved bone-forming functionality on diameter controlled TiO2 nanotube surface.Acta Biomater. 2009;5(8): 3215-3223.
[10]Song L,Zhao J,Zhang X,et al.Icariin induces osteoblast proliferation, differentiation and mineralization through estrogen receptor-mediated ERK and JNK signal activation.Eur J Pharmacol.2013;714(1-3):15-22.
[11]Xu CQ,Liu BJ,Wu JF,et al.Icariin attenuates LPS-induced acute inflammatory responses: Involvement of PI3K/Akt and NF-kappaB signaling pathway.Eur J Pharmacol.2010;642(1-3):146-153.
[12]Zhao J,Ohba S,Komiyama Y,et al.Icariin: a potential osteoinductive compound for bone tissue engineering.Tissue Eng.2010;16(1):233-243.
[13]Luo Z,Liu M,Sun L,et al.Icariin recovers the osteogenic differentiation and bone formation of bone marrow stromal cells from a rat model of estrogen deficiency-induced osteoporosis.Mol Med Rep.2015; 12(1):382-388.
[14]Gulati K,Ramakrishnan S,Aw MS,et al.Biocompatible polymer coating of titania nanotube arrays for improved drug elution and osteoblast adhesion.Acta Biomater. 2012;8(1):449-456.
[15]Liu X,Li X,Li S,et al.An in vitro study of a titanium surface modified by simvastatin-loaded titania nanotubes-micelles.J Biomed Nanotechnol. 2014; 10(2):194-204.
[16]Zhao J,Ohba S,Shinkai M,et al.Icariin induces osteogenic differentiation in vitro in a BMP- and Runx2-dependent manner.Biochem Biophys Res Commun.2008;369(2):444-448.
[17]Zhang X,Guo Y,Li DX,et al.The effect of loading icariin on biocompatibility and bioactivity of porous β-TCP ceramic.J Mater Sci Mater Med.2011;22(2):371-379.
[18]Geetha M,Singh AK,Asokamani R,et al.Ti based biomaterials, the ultimate choice for orthopaedic implants–A review.Prog Mater Sci.2009;54:397-425.
[19]Beppu K,Kido H,Watazu A,et al.Peri-implant bone density in senile osteoporosis-changes from implant placement to osseointegration.Clin Implant Dent Relat Res.2013;15(2):217-226.
[20]Alghamdi HS,Jansen JA.Bone regeneration associated with nontherapeutic and therapeutic surface coatings for dental implants in osteoporosis. Tissue Eng Part B Rev. 2013;19(3):233-253.
[21]Macak JM,Tsuehiya H,Schmuki P.High-aspect-ratio TiO2 nanotubes by anodization of titanium. Angew Chem Int Ed Engl.2009;44(14):2100-2102.
[22]Oh S,Brammer KS,Li YS,et al.Stem cell fate dictated solely by altered nanotube dimension.Proc Natl Acad Sci USA.2009;106(7):2130-2135.
[23]Das K,Bose S,Bandyopadhyay A,et al.TiO2 nanotubes on Ti:Influence of nanoscale morphology on bone cell-materials interaction.J Biomed Mater Res A. 2009; 90(1):225-237.
[24]Brammer KS,Oh S,Cobb CJ,et al.Improved bone-forming functionality on diameter controlled TiO2 nanotube surface.Acta Biomater.2009;5(8):3215-3223.
[25]于卫强,蒋欣泉,张益琳,等.TiO2纳米管的制备及其对成骨细胞增殖和碱性磷酸酶活性的影响[J].中华口腔医学杂志,2009,44(12):751-755.
[26]Kim JH,Cho KP,Chung YS,et al.The effect of nanotubular titanium surfaces on osteoblast differentiation.J Nanosci Nanotechnol. 2010;10(5): 3581-3585.
[27]Peng Z,Ni J,Zheng K,et al.Dual effects and mechanism of TiO2 nanotube arrays in reducing bacterial colonization and enhancing C3H10T1/2 cell adhesion. Int J Nanomedicine.2013;8:3093-3105.
[28]Yu WQ,Xu L,Zheng K,et al.The effect of Ti anodized nano-foveolae structure on preosteoblast growth and osteogenic gene expression.J Nanosci Nanotechnol. 2014;14(6):4387-4393.
[29]Ma M,Kazemzadeh-Narbat M,Hui Y,et al.Local delivery of antimicrobial peptides using self-organized TiO2 nanotube arrays for peri-implant infections.J Biomed Mater Res A.2012;100(2):278-285.
[30]Zhao L,Wang H,Huo K,et al.Antibacterial nano-structured titania coating incorporated with silver nanoparticles.Biomaterials.2011;32(24):5706-5716.
[31]Ma Y,Zhang Z,Liu Y,et al.Nanotubes Functionalized with BMP2 Knuckle Peptide Improve the Osseointegration of Titanium Implants in Rabbits.J Biomed Nanotechnol. 2015;11(2):236-244.
[32]Li B,Hao J,Min Y,et al.Biological properties of nanostructured Ti incorporated with Ca, P and Ag by electrochemical method.Mater Sci Eng C Mater Biol Appl. 2015;51:80-86.
[33]Moon SH,Lee SJ,Park IS,et al.Bioactivity of Ti-6Al-4V alloy implants treated with ibandronate after the formation of the nanotube TiO2 layer.J Biomed Mater Res B Appl Biomater.2012;100(8):2053-2059.
[34]杨丽,张荣华,朱晓峰,等.淫羊藿苷对大鼠间充质干细胞骨向分化过程中转化生长因子β1、骨形态发生蛋白2 表达的影响[J].中国组织工程研究与临床康复, 2010,14(19): 3518-3522.
[35]Fu SP,Yang L,Hong H,et al.Icariin Promoted Osteogenic Differentiation of SD Rat Bone Marrow Mesenchymal Stem Cells: an Experimental Study.Zhongguo Zhong Xi Yi Jie He Za Zhi. 2015; 35(7):839-846.
[36]Ma HP,Ming LG,Ge BF,et al.Icariin is More Potent Than Genistein in Promoting Osteoblast Differentiation and Mineralization In vitro.J Cell Biochem. 2011;112(3): 916-923.
[37]Ma XN,Zhou J,Ge BF,et al.Icariin induces osteoblast differentiation and mineralization without dexamethasone in vitro.Planta Med. 2013;79(16):1501-1508.
[38]Çal??kan N,Bayram C,Erdal E,et al.Titania nanotubes with adjustable dimensions for drug reservoir sites and enhanced cell adhesion.Mater Sci Eng C Mater Biol Appl. 2014;35:100-105.
[39]Ding X,Zhou L,Wang J,et al.The effects of hierarchical micro/nanosurfaces decorated with TiO2 nanotubes on the bioactivity of titanium implants in vitro and in vivo.Int J Nanomedicine.2015;10:6955-6973.
[40]Jia H,Kerr LL.Sustained ibuprofen release using composite poly(lactic-co-glycolic acid)/titanium dioxide nanotubes from Ti implant surface.J Pharm Sci. 2013; 102(7):2341-2348.
[41]Balasundaram G,Yao C,Webster TJ.TiO2 nanotubes functionalized with regions of bone morphogenetic protein-2 increases osteoblast adhesion.J Biomed Mater Res A.2008; 84(2):447-453.
[42]Popat KC,Eltgroth M,Latempa TJ,et al.Decreased Staphylococcus epidermis adhesion and increased osteoblast functionality on antibiotic-loaded titania nanotubes. Biomaterials.2007;28(32):4880-4888.
[43]Yang W,Yu XC,Chen XY,et al.Pharmacokinetics and tissue distribution profile of icariin propylene glycol-liposome intraperitonea.J Pharm Pharmacol. 2012;64(2):190-198.
[44]Lee YH,Bhattarai G,Park IS,et al.Bone regeneration around N-acetyl cysteine-loaded nanotube titanium dental implant in rat mandible.Biomaterials. 2013; 34(38):10199-21008.
[45]Gulati K,Aw MS,Losic D.Drug-eluting Ti wires with titania nanotube arrays for bone fixation and reduced bone infection.Nanoscale Res Lett.2011;6:571.
[46]Eaninwene G,Yao C,Webster TJ.Enhanced osteoblast adhesion to drug-coated anodized nanotubular titanium surfaces.Int J Nanomedicine. 2008;3(2): 257-264.
[47]Aw MS,Khalid KA,Gulati K,et al.Characterization of drug-release kinetics in trabecular bone from titania nanotube implants.Int J Nanomedicine. 2012;7: 4883-4892.
[48]吴涛,徐俊昌,南开辉,等.淫羊藿苷促进羊骨髓间充质干细胞的增殖和成骨分化[J].中国组织工程研究与临床康复, 2009,13(19):3725-3729.
[49]Wu Y,Xia L,Zhou Y,et al.Icariin induces osteogenic differentiation of bone mesenchymal stem cells in a MAPK-dependent manner.Cell Prolif. 2015;48(3): 375-384.
[50]Zhang J,Li AM,Liu BX,et al.Effect of icarisid II on diabetic rats with erectile dysfunction and its potential mechanism via assessment of AGEs, autophagy, mTOR and the NO-cGMP pathway. Asian J Androl. 2013;15(1):143-148.
[51]Nuschke A,Rodrigues M,Stolz DB,et al.Human mesenchymal stem cells/multipotent stromal cells consume accumulated autophagosomes early in differentiation.Stem Cell Res Ther.2014;5(6):140.
[52]Pantovic A,Krstic A,Janjetovic K,et al.Coordinated time-dependent modulation of AMPK/Akt/mTOR signaling and autophagy controls osteogenic differentiation of human mesenchymal stem cells. Bone.2013;52:7.
[53]Oh S,Daraio C,Chen LH,et al.Significantly accelerated osteoblast cell growth on aligned TiO2 nanotuhes. J Biomed Mater Res A.2006;78(1):97-103.
[54]Minagar S,Wang J,Berndt CC,et al.Cell response of anodized nanotubes on titanium and titanium alloys - A review.J Biomed Mater Res A.2013;101(9):2726-2739.
[55]Shekaran A,García AJ.Extracellular matrix-mimetic adhesive biomaterials for bone repair.J Biomed Mater Res A.2011;96(1):261-272.
[56]曹馨,于卫强,张富强.RGD肽修饰TiO2纳米管对成骨细胞黏附的影响[J].口腔医学研究, 2012,28(6):522-525,530.
[57]Richard M Thomas AE.The biology of fracture healing. Injury.2011;42(6):551-555.
[58]Leighton Y,Carvajal JC,Valenzuela F,et al.Synthesis of nanostructured porous silica coatings on titanium and their cell adhesive and osteogenic differentiation properties.J Biomed Mater Res A.2014;102(1):37-48.
[59]Hsieh TP,Sheu SY,Sun JS,et al.Icariin isolated from Epimedium pubescens regulates osteoblasts anabolism through BMP-2, Smad4, and Cbfa1 expression.Phytomedicine. 2010;17(6):414-423. |
| [1] | Zhang Tongtong, Wang Zhonghua, Wen Jie, Song Yuxin, Liu Lin. Application of three-dimensional printing model in surgical resection and reconstruction of cervical tumor [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1335-1339. |
| [2] | Zeng Yanhua, Hao Yanlei. In vitro culture and purification of Schwann cells: a systematic review [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1135-1141. |
| [3] | Xu Dongzi, Zhang Ting, Ouyang Zhaolian. The global competitive situation of cardiac tissue engineering based on patent analysis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 807-812. |
| [4] | Li Xingping, Xiao Dongqin, Zhao Qiao, Chen Shuo, Bai Yiguang, Liu Kang, Feng Gang, Duan Ke. Preparation and properties of copper-loaded antibacterial functional film on titanium surface [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 553-557. |
| [5] | Wu Zijian, Hu Zhaoduan, Xie Youqiong, Wang Feng, Li Jia, Li Bocun, Cai Guowei, Peng Rui. Three-dimensional printing technology and bone tissue engineering research: literature metrology and visual analysis of research hotspots [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 564-569. |
| [6] | Shi Xiaoxiu, Mao Shilong, Liu Yang, Ma Xingshuang, Luo Yanfeng. Comparison of tantalum and titanium (alloy) as orthopedic materials: physical and chemical indexes, antibacterial and osteogenic ability [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 593-599. |
| [7] | Chang Wenliao, Zhao Jie, Sun Xiaoliang, Wang Kun, Wu Guofeng, Zhou Jian, Li Shuxiang, Sun Han. Material selection, theoretical design and biomimetic function of artificial periosteum [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 600-606. |
| [8] | Liu Fei, Cui Yutao, Liu He. Advantages and problems of local antibiotic delivery system in the treatment of osteomyelitis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 614-620. |
| [9] | Li Xiaozhuang, Duan Hao, Wang Weizhou, Tang Zhihong, Wang Yanghao, He Fei. Application of bone tissue engineering materials in the treatment of bone defect diseases in vivo [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 626-631. |
| [10] | Zhang Zhenkun, Li Zhe, Li Ya, Wang Yingying, Wang Yaping, Zhou Xinkui, Ma Shanshan, Guan Fangxia. Application of alginate based hydrogels/dressings in wound healing: sustained, dynamic and sequential release [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 638-643. |
| [11] | Chen Jiana, Qiu Yanling, Nie Minhai, Liu Xuqian. Tissue engineering scaffolds in repairing oral and maxillofacial soft tissue defects [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 644-650. |
| [12] | Xing Hao, Zhang Yonghong, Wang Dong. Advantages and disadvantages of repairing large-segment bone defect [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(3): 426-430. |
| [13] | Chen Siqi, Xian Debin, Xu Rongsheng, Qin Zhongjie, Zhang Lei, Xia Delin. Effects of bone marrow mesenchymal stem cells and human umbilical vein endothelial cells combined with hydroxyapatite-tricalcium phosphate scaffolds on early angiogenesis in skull defect repair in rats [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3458-3465. |
| [14] | Wang Hao, Chen Mingxue, Li Junkang, Luo Xujiang, Peng Liqing, Li Huo, Huang Bo, Tian Guangzhao, Liu Shuyun, Sui Xiang, Huang Jingxiang, Guo Quanyi, Lu Xiaobo. Decellularized porcine skin matrix for tissue-engineered meniscus scaffold [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3473-3478. |
| [15] | Mo Jianling, He Shaoru, Feng Bowen, Jian Minqiao, Zhang Xiaohui, Liu Caisheng, Liang Yijing, Liu Yumei, Chen Liang, Zhou Haiyu, Liu Yanhui. Forming prevascularized cell sheets and the expression of angiogenesis-related factors [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3479-3486. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||