Chinese Journal of Tissue Engineering Research ›› 2020, Vol. 24 ›› Issue (16): 2599-2605.doi: 10.3969/j.issn.2095-4344.2016
Previous Articles Next Articles
Hot issues of research on cells and biological scaffolds in cardiac tissue engineering
Cai Zhibin, Wang Ping
- Department of Cardiac Macrovascular Surgery, Yan’an Hospital Affiliated to Kunming Medical University, Department of Cardiac Macrovascular Surgery, Yan’an Hospital of Kunming, Yunnan Provincial Institute of Cardiovascular Surgery, Yunnan Provincial Key Laboratory of Cardiovascular Diseases, Kunming 650051, Yunnan Province, China
-
Received:2019-06-18Revised:2019-06-29Accepted:2019-07-23Online:2020-06-08Published:2020-03-28 -
Contact:Wang Ping, Master, Associate professor, Master’s supervisor, Department of Cardiac Macrovascular Surgery, Yan’an Hospital Affiliated to Kunming Medical University, Department of Cardiac Macrovascular Surgery, Yan’an Hospital of Kunming, Yunnan Provincial Institute of Cardiovascular Surgery, Yunnan Provincial Key Laboratory of Cardiovascular Diseases, Kunming 650051, Yunnan Province, China -
About author:Cai Zhibin, Master candidate, Department of Cardiac Macrovascular Surgery, Yan’an Hospital Affiliated to Kunming Medical University, Department of Cardiac Macrovascular Surgery, Yan’an Hospital of Kunming, Yunnan Provincial Institute of Cardiovascular Surgery, Yunnan Provincial Key Laboratory of Cardiovascular Diseases, Kunming 650051, Yunnan Province, China -
Supported by:the Combined Project of Yunnan Department of Science and Technology and Kunming Medical University, No. 2017FE467(-096)
CLC Number:
Cite this article
Cai Zhibin, Wang Ping. Hot issues of research on cells and biological scaffolds in cardiac tissue engineering[J]. Chinese Journal of Tissue Engineering Research, 2020, 24(16): 2599-2605.
share this article
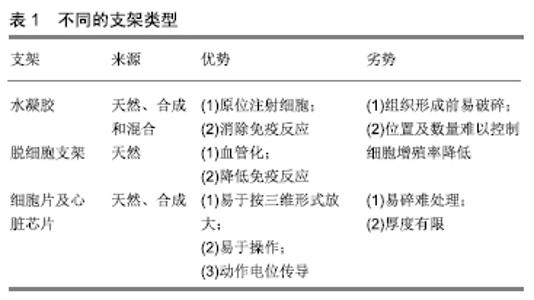
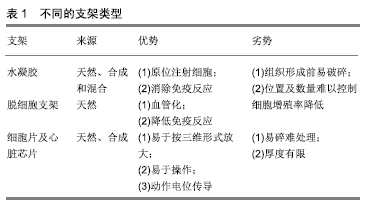
2.1 组织工程的细胞来源 目前,应用于心脏组织工程的细胞有心肌细胞、骨骼肌细胞、成纤维细胞、骨髓来源干细胞、胚胎干细胞、诱导多能干细胞、月经血干细胞,现逐一进行综述。 2.1.1 心肌细胞 研究表明,未成熟的心肌细胞如新生儿和胎儿的心肌细胞具有较强的分裂增殖能力,可经分离、纯化后应用于心脏组织工程[18]。然而,同种异体细胞的免疫排斥性、致癌风险性、数量有限和严峻的伦理合理性使其难于推广和应用。由于这些原因,需进一步探索更为合适的细胞来源以应用于心脏组织工程。 2.1.2 骨骼肌细胞 骨骼肌细胞是最早应用于心脏修复的细胞类型之一。骨骼肌细胞可通过自体肌肉组织获得,用于心脏组织工程时无免疫排斥性,同时骨骼肌细胞在体外增殖速率高[6],能耐受缺氧环境[16],无致肿瘤形成风险[19],可经多种方式移植到心脏组织[17,20],使其具有广阔的应用前景。然而,骨骼肌细胞存在发展为骨骼肌谱系细胞的倾向性,并且难与宿主心肌细胞进行电生理耦合[21]。因此,不容易融入心肌环境[22],有导致心律失常的风险[23]。 2.1.3 心脏成纤维细胞 成纤维细胞起源于间充质,呈梭形,广泛分布于人体结缔组织内,可分泌细胞外基质成分[24],且可以通过细胞外基质和细胞-细胞间的相互作用来调节心脏做功时收缩力的分配[25],此外还具有促进内皮细胞生长的作用[26]。因此,具有广泛应用于心肌细胞培养和心脏组织构建的潜力[27]。 研究发现,成纤维细胞可增强组织的血管化,成纤维细胞具有血管紧张素Ⅱ受体。当心脏组织受损时,血液中的血管紧张素可刺激受损心脏组织中的成纤维细胞和平滑肌细胞,促进其增殖以维持受损心脏组织的稳定[19,28]。在静息状态下,心脏组织的成纤维细胞具有收缩特性和类似于平滑肌细胞的特征。这些细胞被称为肌成纤维细胞,通常不存在于正常心肌组织中,而心脏组织受损后可局限性出现于受损的心脏组织中,这些肌成纤维细胞在梗死心脏的修复性纤维化中起关键作用[29],而肌成纤维细胞分泌的生长因子、细胞因子、蛋白酶等对于受损心脏组织的修复至关重要[30]。除此之外,心脏成纤维细胞可被转化为心肌细胞[31-32],以改善心脏收缩功能。然而,成纤维细胞仍然存在如数量、安全性和不可预测的细胞系形成等问题需要解决[33]。 2.1.4 骨髓来源干细胞 有研究表明,骨髓单核干细胞移植治疗可改善受损心脏功能[34]。将标记过的骨髓来源干细胞移植到受损的心脏组织中,于第9天检测发现有68%的细胞发生了向心肌细胞方向的分化,证明了骨髓来源干细胞治疗受损心脏组织的可行性及有效性[35]。骨髓来源干细胞除了可以应用于心脏组织工程外,在其他系统疾病中的应用也有相关报道。研究表明,在大鼠脊髓被球囊压迫造成脊髓损伤1 d后,通过鞘内注射100 μL CD34+和等量的CD105+骨髓干细胞悬液,治疗5周内,与对照组相比,实验组显著恢复了大鼠的运动能力并且减少了因脊髓损伤引发的其他不良伤害[36]。在泌尿系统疾病中,将骨髓来源间充质干细胞移植入慢性肾病成年大鼠模型中,通过检测血浆肌酐和尿素水平、平均动脉压以及记录心电图的变化,证实了骨髓来源间充质干细胞可以减缓慢性肾病的进程,改善肾功能以及与慢性肾病相关的心脏电生理障碍和心脏功能[37]。这些研究说明骨髓单核干细胞具有较广的临床应用前景。 骨髓中含有多种干细胞,如间充质干细胞、造血干细胞、内皮干细胞和外周干细胞等[38]。间充质干细胞及造血干细胞可分化为心肌细胞,并可促进移植物血管生成和具有抗炎作用[39],而内皮干细胞则具有改善心脏功能的能力[38]。此外,骨髓干细胞还可分化为成纤维细胞,是心脏组织工程细胞来源的重要组成部分[40]。 骨髓来源干细胞可来自患者,不具有免疫原性,并且细胞的采集和培养均较容易[23,31,39],使其诱导分化后用于临床治疗。骨髓来源干细胞可根据调节信号的不同转换和改变其功能[41],因此用于治疗受损心脏组织时,其作用可能是暂时的;此外,其数量较少也限制了其应用[38]。 2.1.5 胚胎干细胞 胚胎干细胞来源于胚泡的内部细胞团,具有发育分化为多种细胞类型的潜能。研究发现,人类胚胎干细胞在含有骨形态发生蛋白4和激活素A的培养基中培养,可诱导分化为心肌细胞;通过添加外源性Wnt3a然后用Dickkopf相关蛋白1(dickkopf-related protein 1,DKK1)抑制Wnt信号传导可增强心肌细胞分化并促进心肌组织的发育形成[31]。 此外还有研究表明人类胚胎干细胞衍生的心肌细胞可改善心肌梗死后受损心脏的收缩-舒张功能,并在裸鼠心肌梗死模型中得到印证[42]。在实验中,将人类胚胎干细胞来源的心肌细胞注射到通过结扎左前降支造成永久性心脏缺血的小鼠模型受损心脏中,可改善小鼠心脏功能,抑制心肌梗死引起的心脏炎症反应[43]。将小鼠胚胎干细胞诱导成血小板衍生生长因子受体α阳性的心脏谱系定向植入梗死的小鼠心脏组织后,显示出其具有心肌细胞再生能力,是心脏再生潜在的细胞来源[44]。还有报道在行冠状动脉旁路移植手术时,同期植入人类胚胎干细胞衍生的心血管祖细胞,发现心脏收缩功能明显改善,患者症状缓解明显[45],同时临床随访结果表明人类胚胎干细胞衍生的临床级心脏祖细胞应用于心脏组织工程治疗心功能不全是安全可行的[46]。但由于不易对人类胚胎干细胞衍生的心肌细胞进行特异性标记,难以进行分离纯化[47],并且植入同种异体来源细胞需要免疫抑制治疗[48],应用于动物模型时出现的畸胎瘤及伦理合理性[39,49],使其应用依然受限。 2.1.6 诱导多能干细胞 诱导多能干细胞是一种通过对成体细胞进行重编程诱导而得到的具有多功能性的干细胞[50]。诱导多能干细胞在终末期肝衰竭替代治疗中显示出巨大的应用潜力,运用人诱导多能干细胞和大鼠肝脏脱细胞支架成功制备出了新型人工肝脏,其具有人类肝脏的功能[51]。因此可将诱导多能干细胞分化成心脏细胞,应用于心脏组织工程领域以治疗心血管疾病。 通过添加转录因子kfl-4,Myc,Oct4和Sox2对细胞进行重编程或通过添加Flk1,Isl1,Nkx2.5可诱导形成心脏祖细胞[52]。研究表明,应用改良的RNBA RNA技术可成功将诱导多能干细胞用于心脏组织工程[53]。诱导多能干细胞来源的心肌细胞,可应用于大鼠和猪的心肌梗死模型治疗。有研究报道,将人诱导多能干细胞来源的心肌细胞与促红细胞生成素、聚乙二醇水凝胶结合构建成心脏组织工程补片治疗心肌梗死大鼠,可改善心肌梗死后大鼠受损心脏功能并增加受损心脏区域的厚度和心肌细胞的数量[54]。另有研究者将心肌细胞、平滑肌细胞和人诱导多能干细胞来源的内皮细胞悬浮于纤维蛋白支架中,将其置于动态环境中培养,可产生临床级 (4 cm×2 cm×1.25 mm)的心脏补片,将其移植到心肌梗死受损心脏后,可显著减少心肌梗死猪模型梗死心肌的面积,改善心功能,且并不会增加心律失常的风险[55]。这些研究表明了诱导多能干细胞应用于心脏组织工程的可行性及有效性。 诱导多能干细胞来自患者本人,消除了免疫排斥性及伦理问题[56]。在改善心肌梗死后心脏受损区域收缩功能和心脏生物能量效率方面,人诱导多能干细胞来源的心肌细胞优于其他成体干细胞来源的心肌细胞[57],提示其广阔的临床应用前景,但致畸胎瘤性使其依然存在一定的风险。有研究报道,在脊髓损伤小鼠模型中植入诱导多能干细胞来源的神经细胞,移植后的神经细胞分化为3个神经谱系,并在47 d后促进了小鼠运动功能的恢复,同时刺激神经突触的形成;然而在为期103 d的观察内发现了肿瘤的形成[58],且肿瘤形成风险与体内未成功诱导分化的诱导多能干细胞的量成正比[59]。为了避免畸胎瘤的形成,所用于临床治疗的人诱导多能干细胞来源的心肌细胞必须具有很高的纯度。研究报道了一种具有综合和表面功能化渔网结构的微流体装置,可以高效地捕获特定的细胞。通过捕获未成功分化为心肌细胞的诱导多能干细胞,人诱导多能干细胞来源的心肌细胞纯化率超过80%,纯化率显著升高[60]。 2.1.7 月经血干细胞 月经血干细胞与其他成体干细胞相比具有许多优势,包括易获得性、样品采集方便、增殖能力强、无肿瘤形成倾向等,使其能应用于组织工程和再生医学领域。 研究者将EGFP标记的月经血来源的间充质细胞与小鼠心肌细胞共培养,利用40 μm高密度的胶原膜对2种细胞进行隔离避免直接接触,自共培养第5天起,大约一半的月经血来源间充质细胞开始自发性跳动,表现出心肌细胞特异性动作电位,将细胞片移植入裸鼠心肌梗死模型受损的心脏中,心脏功能得到显著改善,减小了裸鼠模型的心肌梗死区域[61]。月经血培养的贴壁细胞比骨髓干细胞甚至脐血干细胞来源的贴壁细胞增殖速度更快[62],而且细胞在长期扩增过程中发生核异常的风险很小[63],提示其临床应用的可行性与安全性。 还有报道,月经血干细胞与骨髓干细胞治疗小鼠急性肝衰竭的成功案例。将2种干细胞植入治疗CCL4诱导的急性肝衰竭Balb/C小鼠肝脏中,将肝脏切片用过碘酸-希夫反应染色和马松三色染色显示都有显著的肝细胞再生,血清检测发现天冬氨酸氨基转移酶、丙氨酸氨基转移酶、总胆红素、尿素和胆固醇均显著降低,同时与骨髓干细胞相比,月经血干细胞对急性肝衰竭引起的炎症反应的抑制作用更加强烈[64]。 2.2 组织工程支架的生物学特性及种类 2.2.1 组织工程支架的生物学特性 组织工程支架结构用于生物工程技术中对于细胞成分的物理支撑,因此生物支架的构成成分应该类似于心脏的细胞外基质,以减轻其植入后心肌组织对其排斥。作为细胞成分的支持结构,组织工程支架还应该具有足够的孔隙,以利于血管内皮细胞长入形成新生毛细血管,同时允许血液中的氧气和营养物质通过空隙扩散到支架外的组织结构中,为植入细胞及新生组织结构提供能量,以维持植入细胞生存及促进新生细胞增殖。此外,某些类型的组织支架,其作用在于短期内为植入细胞提供生长所需空间及物理附着作用,当植入的细胞存活或增殖后,组织工程支架需降解以排出体内,因此生物支架还需要具有一定的自身降解能力,且降解后产物对组织及机体无毒副作用。 (1)生物相容性及抗凝活性:生物支架移植入心脏后,不应具有免疫排斥性,以避免自身免疫系统对移植物的排斥及攻击。由于移植物的植入部位为心脏,其还应具有抗凝血作用,以避免血栓形成,栓塞重要脏器。 (2)生物降解性:支架应随着时间的推移缓慢降解,使得最终不会在体内留下残余物[65]。移植材料在体内可通过水解、氧化、酶和物理降解等机制分解为可吸收的无毒产物,最终通过代谢途径排出体外,在降解过程中,应保持一定的速率,以避免过快被分解以影响新生组织形成[16]。通过水解降解的生物相容性聚合物是心脏组织工程中最早应用的材料之一,这种材料的机械性能与宿主组织相匹配,能获得最佳的活细胞组织工程移植物[66]。 (3)机械性能:支架必须能够模拟天然心脏组织的机械特性,能承受移植后施加的循环压力和心脏跳动过程中的组织应力。机械特性可以改变种子细胞的排列表型,与天然聚合物相比,合成聚合物具有更高的强度和耐用性,但其大量降解后可引起局部炎症反应[67]。 2.2.2 组织工程支架类型 (1)水凝胶:水凝胶具有与心脏组织相仿的柔韧性、高含水量和理化特性,使其可用于组织工程[68]。在心脏组织工程中,水凝胶通常用于接种囊封细胞,然后将其压缩成三维组织,水凝胶可以促进细胞存活和合胞体形成,此性质与细胞外基质相似,且水凝胶的生理特性能针对不同的应用环境进行微调,使其可以更匹配应用环境[69]。水凝胶成分含有天然聚合物,如海藻酸盐、胶原蛋白或纤维蛋白,常用的海藻酸盐来源于海藻。海藻酸钠水凝胶通常用于药物输送,具有帮助细胞长效地与药物结合。海藻酸钠水凝胶可通过离子交联或通过光交联的方式捕捉细胞并释放药物与细胞结合[70],其他水凝胶可采用合成聚合物如聚乙烯醇、聚乙二醇和聚丙烯酸等进行加工合成,从而能制备最适宜的水凝胶[71]。 目前有许多先进的水凝胶应用在心脏治疗中。例如,血管生成素1衍生肽——QHREDGS,固定在壳聚糖-胶原水凝胶上,具有促进心肌梗死后心脏功能维持的能力[72]。与对照组相比,这种改良水凝胶能显著改善大鼠心肌梗死模型的心脏功能和减轻心脏重塑。此外,可注射水凝胶在心脏组织工程中的应用也受到研究者的广泛关注。可注射水凝胶利用混合液体水凝胶制备而成,可通过导管输送到人体,避免了支架输送时需要的手术操作。同时,可注射水凝胶还可用于传递细胞,传递蛋白质类物质如生长因子,并以各种组合的形式被加以制造利用[20,48,73]。注射性水凝胶的主要缺点是缺乏对最终移植部位和细胞数量的控制[18]。此外,使用核黄素-5磷酸盐对水凝胶进行光敏感化后通过紫外线介导水凝胶表面光消融,实现快速自动化地构建自定义器官芯片,表现出了坚固性和可重复性,且不改变水凝胶的机械特性。将大鼠或人心肌细胞接种到此心脏芯片中,所得到的心脏组织具有收缩功能和长期存活力,能够持续培养长达27 d,使得可以在该芯片上进行长期研究,且与传统方法相比缩短了60%的时间,为自动化和连续化制造器官芯片提供了重要技术支持[74]。 (2)脱细胞支架:脱细胞支架利用器官本身提供三维组织结构[39,75],取心脏组织,用Triton X-100和SDS等洗涤剂洗脱掉心肌细胞,使得心脏的结缔组织与细胞外基质得以保留。细胞外基质是组织构建的重要组成部分,提供组织结构支持,有助于细胞增殖和功能的维持。利用细胞洗涤剂洗脱猪的心脏瓣膜细胞,再用DNase和RNase处理去除残留的细胞,剩余下的心脏瓣膜支架仅由胶原蛋白和弹性蛋白组成,以这种支架覆盖人体心脏瓣膜细胞构建移植物,用于替换人心脏瓣膜,可降低异种移植或同种移植时发生免疫反应的可能性[41]。 (3)细胞片及心脏芯片:细胞片是一种新的无支架技术,其解决了支架内的细胞浓度比天然心脏低的问题及避免了支架降解引起炎症反应的风险。 细胞片的制作由细胞培养开始至细胞融合。细胞培养皿表面涂覆温度敏感聚合物——聚-N-异丙基丙烯酰胺,在37 ℃时具有疏水性而与细胞黏附,而当温度降低到32 ℃时会导致单层培养细胞连同细胞外基质和附着蛋白作为一个完整的细胞层分离。当二维细胞层堆积时,它们可以快速连接并形成细胞与细胞之间的连接,因此相邻心肌细胞之间的细胞外基质和细胞间连接得以保留[31]。移植后,细胞片的动作电位会改善心脏功能、血管分布和纤维化[33]。将内皮细胞夹在多层心肌细胞片之间,其血管化程度增加。同样,将间充质干细胞片植入梗死的大鼠心脏,在细胞存活数、左心室功能改善和减缓心室扩大等方面也有改善[40,52]。尽管细胞片是一种很有吸引力的选择,但其组织结构相当脆弱,临床应用时操作异常困难[76]。同时,层状心肌细胞板的厚度极限约为80 μm,当超过此厚度时,形成的血管网无法向组织提供足够的营养和氧气[77]。 心脏芯片是基于心脏组织工程原理和技术建立的生理学相关的微流体人心脏模型,以用于心脏药物的研发。研究发现,在应用聚二甲基硅氧烷压制形成的水凝胶构件中培养人诱导多能干细胞来源心肌细胞来构建纤维状心脏组织芯片,可重复生产人诱导多能干细胞来源心肌细胞纤维。此外,人诱导多能干细胞来源心肌细胞纤维可根据所施加的药物改变其收缩频率和收缩力的峰值,且收缩性质的变化与药物施加于人体时所发挥的功效是一致的,因此人诱导多能干细胞来源心肌细胞纤维可作为药物研发的有效工具,用于替代人体测试[78]。 不同支架类型及特点比较,见表1。 "
| [1] BENJAMIN EJ, VIRANI SS, CALLAWAY CW, et al. Heart Disease and Stroke Statistics-2018 Update: A Report From the American Heart Association. Circulation. 2018;137(12):e67-e492. [2] CHAUDHURI R, RAMACHANDRAN M, MOHARIL P, et al. Biomaterials and cells for cardiac tissue engineering: Current choices. Mater Sci Eng C Mater Biol Appl. 2017;79:950-957. [3] DREWS JD, MIYACHI H, SHINOKA T. Tissue-engineered vascular grafts for congenital cardiac disease: Clinical experience and current status. Trends Cardiovasc Med. 2017;27(8):521-531. [4] DOMENECH M, POLO-CORRALES L, RAMIREZ-VICK JE, et al. Tissue Engineering Strategies for Myocardial Regeneration: Acellular Versus Cellular Scaffolds. Tissue Eng Part B Rev. 2016;22(6):438-458. [5] WANG G, MCCAIN ML, YANG L, et al. Modeling the mitochondrial cardiomyopathy of Barth syndrome with induced pluripotent stem cell and heart-on-chip technologies. Nat Med. 2014;20(6):616-623. [6] KENNEDY-LYDON T, ROSENTHAL N. Cardiac regeneration: All work and no repair. Sci Transl Med. 2017;9(383): eaad9019. [7] NISHIDA K, OTSU K. Autophagy during cardiac remodeling. J Mol Cell Cardiol. 2016;95:11-18. [8] YANCY CW, JESSUP M, BOZKURT B, et al. 2016 ACC/AHA/HFSA Focused Update on New Pharmacological Therapy for Heart Failure: An Update of the 2013 ACCF/AHA Guideline for the Management of Heart Failure: A Report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines and the Heart Failure Society of America. J Am Coll Cardiol. 2016;68(13):1476-1488. [9] ADAMS KF JR, GIBLIN EM, PEARCE N, et al. Integrating New Pharmacologic Agents into Heart Failure Care: Role of Heart Failure Practice Guidelines in Meeting This Challenge. Pharmacotherapy. 2017;37(6):645-656. [10] CECCHIN F, HALPERN DG. Cardiac Arrhythmias in Adults with Congenital Heart Disease: Pacemakers, Implantable Cardiac Defibrillators, and Cardiac Resynchronization Therapy Devices. Card Electrophysiol Clin. 2017;9(2): 319-328. [11] VALENTE TW, PITTS SR. An Appraisal of Social Network Theory and Analysis as Applied to Public Health: Challenges and Opportunities. Annu Rev Public Health. 2017;38:103-118. [12] YANAMANDALA M, ZHU W, GARRY DJ, et al. Overcoming the Roadblocks to Cardiac Cell Therapy Using Tissue Engineering. J Am Coll Cardiol. 2017;70(6):766-775. [13] WEINBERGER F, MANNHARDT I, ESCHENHAGEN T. Engineering Cardiac Muscle Tissue: A Maturating Field of Research. Circ Res. 2017;120(9):1487-1500. [14] DOGAN A, ELCIN AE, ELCIN YM. Translational Applications of Tissue Engineering in Cardiovascular Medicine. Curr Pharm Des. 2017;23(6): 903-914. [15] YACOUB MH, TAKKENBERG JJ. Will heart valve tissue engineering change the world. Nat Clin Pract Cardiovasc Med. 2005;2(2):60-61. [16] GÁLVEZ-MONTÓN C, PRAT-VIDAL C, ROURA S, et al. Innovation in cardiology (IV). Cardiac tissue engineering and the bioartificial heart. Rev Esp Cardiol (Engl Ed). 2013;66(5):391-399. [17] AVOLIO E, CAPUTO M, MADEDDU P. Stem cell therapy and tissue engineering for correction of congenital heart disease. Front Cell Dev Biol. 2015;3:39. [18] WU YK, YU J. The role of tissue engineering in cellular therapies for myocardial infarction: a review. J Mater Chem B. 2015;3(31):6401-6410. [19] CHEN QZ, HARDING SE, ALI NN, et al. Biomaterials in cardiac tissue engineering : Ten years of research survey. Materials Science and Engineering: R: Reports. 2008;59(1-6):1-37. [20] RADHAKRISHNAN J, KRISHNAN UM, SETHURAMAN S. Hydrogel based injectable scaffolds for cardiac tissue regeneration. Biotechnol Adv. 2014;32(2):449-461. [21] CURTIS MW, RUSSELL B. Cardiac tissue engineering. J Cardiovasc Nurs. 2009;24(2):87-92. [22] WANG F, GUAN J. Cellular cardiomyoplasty and cardiac tissue engineering for myocardial therapy. Adv Drug Deliv Rev. 2010;62(7-8): 784-797. [23] GEORGIADIS V, KNIGHT RA, JAYASINGHE SN, et al. Cardiac tissue engineering: renewing the arsenal for the battle against heart disease. Integr Biol (Camb). 2014;6(2):111-126. [24] SOUDERS CA, BOWERS SL, BAUDINO TA. Cardiac fibroblast: the renaissance cell. Circ Res. 2009;105(12):1164-1176. [25] COULOMBE KL, BAJPAI VK, ANDREADIS ST, et al. Heart regeneration with engineered myocardial tissue. Annu Rev Biomed Eng. 2014;16:1-28. [26] TULLOCH NL, MURRY CE. Trends in cardiovascular engineering: organizing the human heart. Trends Cardiovasc Med. 2013;23(8): 282-286. [27] HIRT MN, HANSEN A, ESCHENHAGEN T. Cardiac tissue engineering: state of the art. Circ Res. 2014;114(2):354-367. [28] SHEN H, WANG Y, ZHANG Z, et al. Mesenchymal Stem Cells for Cardiac Regenerative Therapy: Optimization of Cell Differentiation Strategy. Stem Cells Int. 2015;2015: 524756. [29] CALDERONE A, BEL-HADJ S, DRAPEAU J, et al. Scar myofibroblasts of the infarcted rat heart express natriuretic peptides. J Cell Physiol. 2006;207(1):165-173. [30] BROWN E, DEJANA E. Cell-to-cell contact and extracellular matrix: Editorial overview: Cell–cell and cell–matrix interactions — running, jumping, standing still. Current Opinion in Cell Biology.2003;15(5): 505-508. [31] ALREFAI MT, MURALI D, PAUL A, et al. Cardiac tissue engineering and regeneration using cell-based therapy. Stem Cells Cloning. 2015; 8:81-101. [32] DOPPLER SA, DEUTSCH MA, LANGE R, et al. Cardiac regeneration: current therapies-future concepts. J Thorac Dis. 2013;5(5):683-697. [33] DILLEY RJ, MORRISON WA. Vascularisation to improve translational potential of tissue engineering systems for cardiac repair. Int J Biochem Cell Biol. 2014;56:38-46. [34] TAYLOR DA, SAMPAIO LC, GOBIN A. Building new hearts: a review of trends in cardiac tissue engineering. Am J Transplant. 2014;14(11): 2448-2459. [35] ORLIC D, KAJSTURA J, CHIMENTI S, et al. Bone marrow cells regenerate infarcted myocardium. Nature. 2001;410(6829):701-705. [36] MUNTER JP, BEUGELS J, MUNTER S, et al. Standardized human bone marrow-derived stem cells infusion improves survival and recovery in a rat model of spinal cord injury. J Neurol Sci. 2019;402: 16-29. [37] HAMED GM, MORSY WE, HAMID MSA, et al. Effect of Bone Marrow-Derived Mesenchymal Stem Cells on Ischaemic-Reperfused Hearts in Adult Rats with Established Chronic Kidney Disease. Int J Stem Cells. 2019;12(2):304-314. [38] STRAUER BE, BREHM M, ZEUS T, et al. Regeneration of human infarcted heart muscle by intracoronary autologous bone marrow cell transplantation in chronic coronary artery disease: the IACT Study. J Am Coll Cardiol. 2005;46(9):1651-1658. [39] GHODSIZAD A, RUHPARWAR A, BORDEL V, et al. Clinical application of adult stem cells for therapy for cardiac disease. Cardiovasc Ther. 2013;31(6):323-334. [40] STRAUER BE, STEINHOFF G. 10 years of intracoronary and intramyocardial bone marrow stem cell therapy of the heart: from the methodological origin to clinical practice. J Am Coll Cardiol. 2011; 58(11):1095-1104. [41] BADER A, SCHILLING T, TEEBKEN OE, et al. Tissue engineering of heart valves--human endothelial cell seeding of detergent acellularized porcine valves. Eur J Cardiothorac Surg. 1998;14(3):279-284. [42] FERNANDES S, CHONG JJH, PAIGE SL, et al. Comparison of Human Embryonic Stem Cell-Derived Cardiomyocytes, Cardiovascular Progenitors, and Bone Marrow Mononuclear Cells for Cardiac Repair. Stem Cell Reports. 2015;5(5): 753-762. [43] YU Y, QIN N, LU XA, et al. Human embryonic stem cell-derived cardiomyocyte therapy in mouse permanent ischemia and ischemia-reperfusion models. Stem Cell Res Ther. 2019;10(1):167. [44] HONG SP, SONG S, LEE S, et al. Regenerative potential of mouse embryonic stem cell-derived PDGFRα+ cardiac lineage committed cells in infarcted myocardium. World J Stem Cells. 2019;11(1):44-54. [45] MENASCHÉ P, VANNEAUX V, HAGÈGE A, et al. Human embryonic stem cell-derived cardiac progenitors for severe heart failure treatment: first clinical case report. Eur Heart J. 2015;36(30):2011-2017. [46] MENASCHÉ P, VANNEAUX V, HAGÈGE A, et al. Transplantation of Human Embryonic Stem Cell-Derived Cardiovascular Progenitors for Severe Ischemic Left Ventricular Dysfunction. J Am Coll Cardiol. 2018; 71(4):429-438. [47] MARTINS AM, VUNJAK-NOVAKOVIC G, REIS RL. The current status of iPS cells in cardiac research and their potential for tissue engineering and regenerative medicine. Stem Cell Rev Rep. 2014;10(2):177-190. [48] FUJITA J, FUKUDA K. Future prospects for regenerated heart using induced pluripotent stem cells. J Pharmacol Sci. 2014;125(1):1-5. [49] RUSSO V, YOUNG S, HAMILTON A, et al. Mesenchymal stem cell delivery strategies to promote cardiac regeneration following ischemic injury. Biomaterials. 2014;35(13):3956-3974. [50] CHACHQUES JC, PRADAS MM, BAYES-GENIS A, et al. Creating the bioartificial myocardium for cardiac repair: challenges and clinical targets. Expert Rev Cardiovasc Ther. 2013;11(12):1701-1711. [51] MINAMI T, ISHII T, YASUCHIKA K, et al. Novel hybrid three-dimensional artificial liver using human induced pluripotent stem cells and a rat decellularized liver scaffold. Regen Ther. 2019;10:127-133. [52] EMMERT MY, HITCHCOCK RW, HOERSTRUP SP. Cell therapy, 3D culture systems and tissue engineering for cardiac regeneration. Adv Drug Deliv Rev. 2014;69-70:254-269. [53] DOPPLER SA, DEUTSCH MA, LANGE R, et al. Cardiac regeneration: current therapies-future concepts. J Thorac Dis. 2013;5(5):683-697. [54] CHOW A, STUCKEY DJ, KIDHER E, et al. Human Induced Pluripotent Stem Cell-Derived Cardiomyocyte Encapsulating Bioactive Hydrogels Improve Rat Heart Function Post Myocardial Infarction. Stem Cell Reports. 2017;9(5): 1415-1422. [55] GAO L, GREGORICH ZR, ZHU W, et al. Large Cardiac Muscle Patches Engineered From Human Induced-Pluripotent Stem Cell-Derived Cardiac Cells Improve Recovery From Myocardial Infarction in Swine. Circulation. 2018;137(16):1712-1730. [56] ZHAO Y, FERIC NT, THAVANDIRAN N, et al. The role of tissue engineering and biomaterials in cardiac regenerative medicine. Can J Cardiol. 2014;30(11):1307-1322. [57] ISHIDA M, MIYAGAWA S, SAITO A, et al. Transplantation of Human-induced Pluripotent Stem Cell-derived Cardiomyocytes Is Superior to Somatic Stem Cell Therapy for Restoring Cardiac Function and Oxygen Consumption in a Porcine Model of Myocardial Infarction. Transplantation. 2019;103(2):291-298. [58] NORI S, OKADA Y, NISHIMURA S, et al. Long-term safety issues of iPSC-based cell therapy in a spinal cord injury model: oncogenic transformation with epithelial-mesenchymal transition. Stem Cell Reports. 2015;4(3):360-373. [59] HONG SG, WINKLER T, WU C, et al. Path to the clinic: assessment of iPSC-based cell therapies in vivo in a nonhuman primate model. Cell Rep. 2014;7(4):1298-1309. [60] LI X, YU L, LI J, et al. On chip purification of hiPSC-derived cardiomyocytes using a fishnet-like microstructure. Biofabrication. 2016;8(3):035017. [61] HIDA N, NISHIYAMA N, MIYOSHI S, et al. Novel cardiac precursor-like cells from human menstrual blood-derived mesenchymal cells. Stem Cells. 2008;26(7):1695-1704. [62] RAHIMI M, MOHSENI-KOUCHESFEHANI H, Zarnani AH, et al. Evaluation of menstrual blood stem cells seeded in biocompatible Bombyx mori silk fibroin scaffold for cardiac tissue engineering. J Biomater Appl. 2014;29(2):199-208. [63] ALLICKSON JG, SANCHEZ A, YEFIMENKO N, et al. Recent Studies Assessing the Proliferative Capability of a Novel Adult Stem Cell Identified in Menstrual Blood. Open Stem Cell J. 2011;3(2011):4-10. [64] FATHI-KAZEROONI M, TAVOOSIDANA G, TAGHIZADEH-JAHED M, et al. Comparative restoration of acute liver failure by menstrual blood stem cells compared with bone marrow stem cells in mice model. Cytotherapy. 2017;19(12):1474-1490. [65] RADISIC M, PARK H, GERECHT S, et al. Biomimetic approach to cardiac tissue engineering. Philos Trans R Soc Lond B Biol Sci. 2007; 362(1484):1357-1368. [66] ZAMMARETTI P, JACONI M. Cardiac tissue engineering: regeneration of the wounded heart. Curr Opin Biotechnol. 2004;15(5):430-434. [67] LEOR J, AMSALEM Y, COHEN S. Cells, scaffolds, and molecules for myocardial tissue engineering. Pharmacol Ther. 2005;105(2):151-163. [68] DHINGRA S, WEISEL RD, LI RK. Synthesis of aliphatic polyester hydrogel for cardiac tissue engineering. Methods Mol Biol. 2014;1181: 51-59. [69] DUAN Y, LIU Z, O'NEILL J, et al. Hybrid gel composed of native heart matrix and collagen induces cardiac differentiation of human embryonic stem cells without supplemental growth factors. J Cardiovasc Transl Res. 2011;4(5):605-615. [70] TANDON V, ZHANG B, RADISIC M, et al. Generation of tissue constructs for cardiovascular regenerative medicine: from cell procurement to scaffold design. Biotechnol Adv. 2013;31(5):722-735. [71] HASAN A, KHATTAB A, ISLAM MA, et al. Injectable Hydrogels for Cardiac Tissue Repair after Myocardial Infarction. Adv Sci (Weinh). 2015;2(11):1500122. [72] REIS LA, CHIU LL, WU J, et al. Hydrogels with integrin-binding angiopoietin-1-derived peptide, QHREDGS, for treatment of acute myocardial infarction. Circ Heart Fail. 2015;8(2):333-341. [73] PAVO N, CHARWAT S, NYOLCZAS N, et al. Cell therapy for human ischemic heart diseases: critical review and summary of the clinical experiences. J Mol Cell Cardiol. 2014;75:12-24. [74] NAWROTH JC, SCUDDER LL, HALVORSON RT, et al. Automated fabrication of photopatterned gelatin hydrogels for organ-on-chips applications. Biofabrication. 2018;10(2): 025004. [75] ZIMMERMANN WH, DIDIÉ M, DÖKER S, et al. Heart muscle engineering: an update on cardiac muscle replacement therapy. Cardiovasc Res. 2006;71(3):419-429. [76] FAROUZ Y, CHEN Y, TERZIC A, et al. Concise review: growing hearts in the right place: on the design of biomimetic materials for cardiac stem cell differentiation. Stem Cells. 2015;33(4):1021-1035. [77] MATSUURA K, SHIMIZU T, OKANO T. Toward the development of bioengineered human three-dimensional vascularized cardiac tissue using cell sheet technology. Int Heart J. 2014;55(1):1-7. [78] MORIMOTO Y, MORI S, SAKAI F, et al. Human induced pluripotent stem cell-derived fiber-shaped cardiac tissue on a chip. Lab Chip. 2016;16(12):2295-2301. |
| [1] | Xu Dongzi, Zhang Ting, Ouyang Zhaolian. The global competitive situation of cardiac tissue engineering based on patent analysis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 807-812. |
| [2] | Xia Guoming, Xu Qiang, Liu Xuqiang, Yu Xiaolong, Dai Min. Application and effect of induced pluripotent stem cells in bone surgery tissue engineering [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(19): 3077-3082. |
| [3] | Ni Jinghua, Luo Jia, Jiang Sen, Li Heng, Zhu Jianzhong. Application and prospect of induced pluripotent stem cells in tumor diseases [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(13): 2127-2132. |
| [4] | Chen Peishan, Zhang Haiyan. Organs-on-a-chip and engineered human tissues in drug development [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(29): 4717-4723. |
| [5] | Liu Zu, Han Shen, Li Yaxiong, Li Kunlin, Zhang Yayong, Jiang Lihong. Applications, roles and problems of exosomes derived from different stem cells in the treatment of cardiovascular diseases [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(19): 3063-3070. |
| [6] | Fan Wenjuan, Chen Xudong, Deng Jinbo. Cerebral organoids culture and application in central nervous system diseases [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(11): 1756-1761. |
| [7] | Liu Tingting, Han Changxu, Wang Guoqiang. New insight into cell transplantation for repairing intervertebral discs [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(1): 154-158. |
| [8] | Cheng Gang, Huang Denggao, Liang Ying. Hepatocyte-like cells derived from induced pluripotent stem cells inhibit liver fibrosis in rats [J]. Chinese Journal of Tissue Engineering Research, 2019, 23(9): 1384-1389. |
| [9] | Liu Shenggang, Yang Hongzhong, He Baimei. Autologous induced pluripotent stem cell transplantation for acute lung injury in rats [J]. Chinese Journal of Tissue Engineering Research, 2019, 23(9): 1403-1409. |
| [10] | Zeng Jianyin, Han Shen, Li Yaxiong, Liu Bin, Zhang Yayong, Jiang Lihong. Application of induced pluripotent stem cells in cardiovascular diseases: efficiency and safety [J]. Chinese Journal of Tissue Engineering Research, 2019, 23(9): 1448-1454. |
| [11] | Pan Lijuan, Wang Rongli. Therapeutic effect of autologous source induced pluripotent stem cell transplantation on chronic hepatitis B-induced liver injury mice [J]. Chinese Journal of Tissue Engineering Research, 2019, 23(5): 773-778. |
| [12] | Ma Linjie, Xue Wentao, Tan Jupeng. Induced pluripotent stem cell transplantation for systemic lupus erythematosus in a mouse model [J]. Chinese Journal of Tissue Engineering Research, 2019, 23(33): 5286-5292. |
| [13] | Gong Jiao, Liu Ming. Human induced pluripotent stem cell transplantation for hypoxic-ischemic encephalopathy in neonatal mice [J]. Chinese Journal of Tissue Engineering Research, 2019, 23(33): 5322-5327. |
| [14] | Shen Mengjie, Yang Kun, Liu Qi. Application potential of different origin stem cells in oral bone regeneration [J]. Chinese Journal of Tissue Engineering Research, 2019, 23(25): 4068-4074. |
| [15] | Ke Min-xia, Ji Meng, Wang Hao, Hong Dan-ping, Wu Yue-hong, Qi Nian-min. Stem cell models for commercialization [J]. Chinese Journal of Tissue Engineering Research, 2018, 22(5): 766-773. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||