Chinese Journal of Tissue Engineering Research ›› 2020, Vol. 24 ›› Issue (29): 4717-4723.doi: 10.3969/j.issn.2095-4344.2711
Previous Articles Next Articles
Organs-on-a-chip and engineered human tissues in drug development
Chen Peishan, Zhang Haiyan
Department of Cell Biology, Basic Medical College, Capital Medical University, Beijing 100069, China
-
Received:2019-11-16Revised:2019-11-20Accepted:2019-12-26Online:2020-10-18Published:2020-09-15 -
Contact:Zhang Haiyan, Professor, Doctoral supervisor, Department of Cell Biology, Basic Medical College, Capital Medical University, Beijing 100069, China -
About author:Chen Peishan, Department of Cell Biology, Basic Medical College, Capital Medical University, Beijing 100069, China -
Supported by:the National Natural Science Foundation of China, No. 81770616
CLC Number:
Cite this article
Chen Peishan, Zhang Haiyan. Organs-on-a-chip and engineered human tissues in drug development[J]. Chinese Journal of Tissue Engineering Research, 2020, 24(29): 4717-4723.
share this article
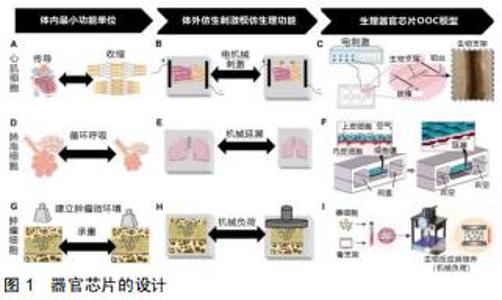
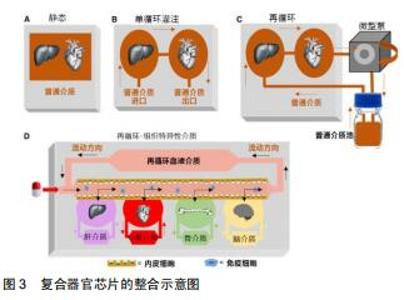
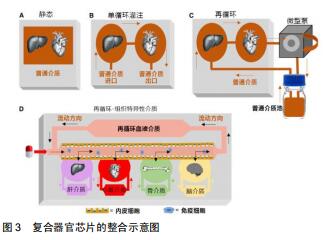
器官芯片的目的并不是建立一个完整的活体器官,而是建立一个用可控的方法来模拟某些人类生理条件下的最小功能单元。例如,将物理、化学或生物刺激施加于细胞上来模拟细胞在体内的信息传导,如电刺激下的心肌细胞;将细胞培养在特定的基膜上可以重新建立不同组织之间联系,如肺泡-毛细血管交换网和血脑屏障[1-2];以微流控芯片技术为研究平台,采用软光刻的方法制备了具有双层结构的微流控肿瘤细胞芯片[3]。对于大多数人体组织而言,器官芯片还需要考虑物理力学性能,如流体动力学、机械力学和电子学等特征。多种器官可以通过将单个的器官芯片以具有体积比和流量分布的微流体通道有机结合以实现生理功能的模拟,从而构建人体体外模型的子系统[4](见图1)。 "
|
[1] HUH D, MATTHEWS BD, MAMMOTO A, et al. Reconstituting organ-level lung functions on a chip. Science.2010;328(5986): 1662-1668.
[2] BOOTH R, KIM H. Characterization of a microfluidic in vitro model of the blood-brain barrier (muBBB). Lab Chip.2012;12(10): 1784-1792.
[3] 王帅超,杨仕平.用于肿瘤微环境研究的微流控细胞芯片构建[D]. 上海:上海师范大学,2018.
[4] WIKSWO JP, CURTIS EL, EAGLETON ZE, et al. Scaling and systems biology for integrating multiple organs-on-a-chip. Lab Chip.2013;13(18):3496-3511.
[5] PARK TH, SHULER ML. Integration of cell culture and microfabrication technology. Biotechnol Prog.2003;19(2):243-253.
[6] BHATIA SN, INGBER DE. Microfluidic organs-on-chips. Nat Biotechnol.2014;32(8):760-772.
[7] GINTANT G, SAGER PT, STOCKBRIDGE N. Evolution of strategies to improve preclinical cardiac safety testing. Nat Rev Drug Discov.2016;15(7):457-471.
[8] GOBAA S, HOEHNEL S, ROCCIO M, et al. Artificial niche microarrays for probing single stem cell fate in high throughput. Nat Methods.2011;8(11):949-955.
[9] PASSIER R, ORLOVA V, MUMMERY C. Complex Tissue and Disease Modeling using hiPSCs. Cell Stem Cell.2016;18(3): 309-321.
[10] POLINI A, PRODANOV L, BHISE NS, et al. Organs-on-a-chip: a new tool for drug discovery. Expert Opin Drug Discov.2014; 9(4): 335-352.
[11] ZHANG B, RADISIC M. Organ-on-a-chip devices advance to market. Lab Chip.2017;17(14):2395-2420.
[12] RUSSELL WMS, BURCH RL. The Principle of Humane Experimental Technique (Wheathampstead, UK: Universities Federation of Animal Welfare). 1959.
[13] HOLMES AM, CRETON S, CHAPMAN K. Working in partnership to advance the 3Rs in toxicity testing. Toxicology.2010;267(1-3): 14-19.
[14] AHADIAN S, CIVITARESE R, BANNERMAN D, et al. Organ-On-A-Chip Platforms: A Convergence of Advanced Materials, Cells, and Microscale Technologies. Adv Healthc Mater.2018;7(14).
[15] LELIEVRE SA, KWOK T, CHITTIBOYINA S. Architecture in 3D cell culture: An essential feature for in vitro toxicology. Toxicol In Vitro.2017;45(Pt 3):287-295.
[16] ROTHBAUER M, ZIRATH H, ERTL P. Recent advances in microfluidic technologies for cell-to-cell interaction studies. Lab Chip.2018;18(2):249-270.
[17] SMITH AS, LONG CJ, BERRY BJ, et al. Microphysiological systems and low-cost microfluidic platform with analytics. Stem Cell Res Ther.2013;4 Suppl 1: S9.
[18] XU Z, LI E, GUO Z, et al. Design and Construction of a Multi-Organ Microfluidic Chip Mimicking the in vivo Microenvironment of Lung Cancer Metastasis. ACS Appl Mater Interfaces.2016;8(39): 25840-25847.
[19] ABACI H E, GLEDHILL K, GUO Z, et al. Pumpless microfluidic platform for drug testing on human skin equivalents. Lab Chip. 2015;15(3):882-888.
[20] ATAC B, WAGNER I, HORLAND R, et al. Skin and hair on-a-chip: in vitro skin models versus ex vivo tissue maintenance with dynamic perfusion. Lab Chip.2013;13(18):3555-3561.
[21] ESCH MB, UENO H, APPLEGATE DR, et al. Modular, pumpless body-on-a-chip platform for the co-culture of GI tract epithelium and 3D primary liver tissue. Lab Chip.2016;16(14):2719-2729.
[22] SHAH P, FRITZ JV, GLAAB E, et al. A microfluidics-based in vitro model of the gastrointestinal human-microbe interface. Nat. Commun. 2016; 7:11535.
[23] TAKAHASHI K, TANABE K, OHNUKI M, et al. Induction of pluripotent stem cells from adult human fibroblasts by defined factors. Cell. 2007;131(5):861-872.
[24] LIANG P, SALLAM K, WU H, et al. Patient-Specific and Genome-Edited Induced Pluripotent Stem Cell-Derived Cardiomyocytes Elucidate Single-Cell Phenotype of Brugada Syndrome. J Am Coll Cardiol.2016;68(19):2086-2096.
[25] KIM K, DOI A, WEN B, et al. Epigenetic memory in induced pluripotent stem cells. Nature.2010; 467(7313):285-290.
[26] RAJAMOHAN D, MATSA E, KALRA S, et al. Current status of drug screening and disease modelling in human pluripotent stem cells. BioEssays. 2013; 35(3):281-298.
[27] NUNES SS, MIKLAS JW, LIU J, et al. Biowire: a platform for maturation of human pluripotent stem cell-derived cardiomyocytes. Nat. Methods 2013; 10(8): 781–787.
[28] VILLASANTE A, MARTURANO-KRUIK A, VUNJAK-NOVAKOVIC G. Bioengineered human tumor within a bone niche. Biomaterials. 2014;35(22):5785-5794.
[29] VILLASANTE A, MARTURANO-KRUIK A, ROBINSON ST, et al. Tissue-Engineered Model of Human Osteolytic Bone Tumor. Tissue Eng Part C Methods.2017;23(2):98-107.
[30] ZHANG YS, ALEMAN J, SHIN SR, et al. Multisensor-integrated organs-on-chips platform for automated and continual in situ monitoring of organoid behaviors. Proc Natl Acad Sci U S A.2017; 114(12): E2293-E2302.
[31] SHIRURE VS, GEORGE SC. Design considerations to minimize the impact of drug absorption in polymer-based organ-on-a-chip platforms. Lab Chip.2017;17(4):681-690.
[32] VAN MEER BJ, DE VRIES H, FIRTH K, et al. Small molecule absorption by PDMS in the context of drug response bioassays. Biochem Biophys Res Commun.2017;482(2):323-328.
[33] TSAMANDOURAS N, CHEN W, EDINGTON CD, et al. Integrated Gut and Liver Microphysiological Systems for Quantitative In Vitro Pharmacokinetic Studies. AAPS J.2017;19(5):1499-1512.
[34] ONAKPOYA IJ, HENEGHAN CJ, ARONSON JK. Post-marketing withdrawal of 462 medicinal products because of adverse drug reactions: a systematic review of the world literature. BMC Med. 2016,14:10.
[35] OLSSON S, EDWARDS IR. Tachycardia during cisapride treatment. BMJ.1992;305(6856):748-749.
[36] GIACOMELLI E, BELLIN M, SALA L, et al. Three-dimensional cardiac microtissues composed of cardiomyocytes and endothelial cells co-differentiated from human pluripotent stem cells. Development.2017;144(6):1008-1017.
[37] BURRIDGE PW, HOLMSTROM A, and WU JC. Chemically defined culture and cardiomyocyte differentiation of human pluripotent stem cells. Curr Protoc Hum Genet. 2015;87: 1-15.
[38] MANNHARDT I, BRECKWOLDT K, LETUFFE-BRENIERE D, et al. Human Engineered Heart Tissue: Analysis of Contractile Force. Stem Cell Reports.2016;7(1):29-42.
[39] KOSTADINOVA R, BOESS F, APPLEGATE D, et al. A long-term three dimensional liver co-culture system for improved prediction of clinically relevant drug-induced hepatotoxicity. Toxicol Appl Pharmacol.2013;268(1):1-16.
[40] SCHEPERS A, LI C, CHHABRA A, et al. Engineering a perfusable 3D human liver platform from iPS cells. Lab Chip.2016;16(14): 2644-2653.
[41] DOMANSKY K, INMAN W, SERDY J, et al. Perfused multiwell plate for 3D liver tissue engineering. Lab Chip.2010;10(1):51-58.
[42] BHISE NS, MANOHARAN V, MASSA S, et al. A liver-on-a-chip platform with bioprinted hepatic spheroids. Biofabrication.2016; 8(1):14101.
[43] KHETANI SR, BHATIA SN. Microscale culture of human liver cells for drug development. Nat Biotechnol.2008;26(1):120-126.
[44] CHO CH, PARK J, TILLES AW, et al. Layered patterning of hepatocytes in co-culture systems using microfabricated stencils. Biotechniques.2010;48(1):47-52.
[45] RAMAIAHGARI SC, DEN BRAVER MW, HERPERS B, et al. A 3D in vitro model of differentiated HepG2 cell spheroids with improved liver-like properties for repeated dose high-throughput toxicity studies. Arch Toxicol.2014;88(5):1083-1095.
[46] GORAL VN, HSIEH YC, PETZOLD ON, et al. Perfusion-based microfluidic device for three-dimensional dynamic primary human hepatocyte cell culture in the absence of biological or synthetic matrices or coagulants. Lab Chip.2010;10(24):3380-3386.
[47] LEE PJ, HUNG PJ, LEE LP. An artificial liver sinusoid with a microfluidic endothelial-like barrier for primary hepatocyte culture. Biotechnol Bioeng.2007;97(5):1340-1346.
[48] RENNERT K, STEINBORN S, GROGER M, et al. A microfluidically perfused three dimensional human liver model. Biomaterials.2015;71:119-131.
[49] GODOY P, HEWITT NJ, ALBRECHT U, et al. Recent advances in 2D and 3D in vitro systems using primary hepatocytes, alternative hepatocyte sources and non-parenchymal liver cells and their use in investigating mechanisms of hepatotoxicity, cell signaling and ADME. Arch Toxicol.2013;87(8):1315-1530. [50] LUBBERSTEDT M, MULLER-VIEIRA U, MAYER M, et al. HepaRG human hepatic cell line utility as a surrogate for primary human hepatocytes in drug metabolism assessment in vitro. J Pharmacol Toxicol Methods.2011;63(1):59-68.
[51] WARE BR, BERGER DR, KHETANI SR. Prediction of Drug-Induced Liver Injury in Micropatterned Co-cultures Containing iPSC-Derived Human Hepatocytes. Toxicol Sci.2015; 145(2):252-262.
[52] LI Y, YANG Y, XIONG A, et al. Comparative Gene Expression Analysis of Lymphocytes Treated with Exosomes Derived from Ovarian Cancer and Ovarian Cysts. Front Immunol.2017;8:607.
[53] YIN P, LV H, LI Y, et al. Exosome-Mediated Genetic Information Transfer, a Missing Piece of Osteoblast-Osteoclast Communication Puzzle. Front Endocrinol (Lausanne).2017;8:336.
[54] LI AP, UZGARE A, LAFORGE YS. Definition of metabolism- dependent xenobiotic toxicity with co-cultures of human hepatocytes and mouse 3T3 fibroblasts in the novel integrated discrete multiple organ co-culture (IdMOC) experimental system: results with model toxicants aflatoxin B1, cyclophosphamide and tamoxifen. Chem Biol Interact.2012;199(1):1-8.
[55] LI AP, BODE C, SAKAI Y. A novel in vitro system, the integrated discrete multiple organ cell culture (IdMOC) system, for the evaluation of human drug toxicity: comparative cytotoxicity of tamoxifen towards normal human cells from five major organs and MCF-7 adenocarcinoma breast cancer cells. Chem Biol Interact. 2004;150(1):129-136.
[56] LAU YY, CHEN YH, LIU TT, et al. Evaluation of a novel in vitro Caco-2 hepatocyte hybrid system for predicting in vivo oral bioavailability. Drug Metab Dispos.2004;32(9):937-942.
[57] PHAN D, WANG X, CRAVER BM, et al. A vascularized and perfused organ-on-a-chip platform for large-scale drug screening applications. Lab Chip.2017;17(3):511-520.
[58] OLEAGA C, BERNABINI C, SMITH AS, et al. Multi-Organ toxicity demonstration in a functional human in vitro system composed of four organs. Sci Rep.2016; 6:20030.
[59] MA L, BARKER J, ZHOU C, et al. Towards personalized medicine with a three-dimensional micro-scale perfusion-based two-chamber tissue model system. Biomaterials.2012; 33(17): 4353-4361.
[60] MILLER PG, SHULER ML. Design and demonstration of a pumpless 14 compartment microphysiological system. Biotechnol Bioeng.2016;113(10):2213-2227.
[61] UPADHYAY RK. Drug delivery systems, CNS protection, and the blood brain barrier. Biomed Res Int. 2014; 2014: 869269.
[62] KIMURA H, IKEDA T, NAKAYAMA H, et al. An on-chip small intestine-liver model for pharmacokinetic studies. J Lab Autom. 2015;20(3):265-273.
[63] HABERT R, MUCZYNSKI V, GRISIN T, et al. Concerns about the widespread use of rodent models for human risk assessments of endocrine disruptors. Reproduction.2014;147(4): R119-R129.
[64] CHO I, BLASER MJ. The human microbiome: at the interface of health and disease. Nat Rev Genet.2012;13(4):260-270.
[65] DIMASI JA, HANSEN RW, GRABOWSKI HG. The price of innovation: new estimates of drug development costs. J Health Econ.2003;22(2):151-185.
[66] JEKUNEN A. Decision-making in product portfolios of pharmaceutical research and development--managing streams of innovation in highly regulated markets. Drug Des Devel Ther. 2014;8:2009-2016.
[67] NG KS, THAM LS, TAN HH, et al. Cisapride and torsades de pointes in a pacemaker patient. Pacing Clin Electrophysiol.2000; 23(1):130-132.
[68] INGBER DE. Reverse Engineering Human Pathophysiology with Organs-on-Chips. Cell.2016;164(6):1105-1109. |
| [1] | Pu Rui, Chen Ziyang, Yuan Lingyan. Characteristics and effects of exosomes from different cell sources in cardioprotection [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(在线): 1-. |
| [2] | Lin Qingfan, Xie Yixin, Chen Wanqing, Ye Zhenzhong, Chen Youfang. Human placenta-derived mesenchymal stem cell conditioned medium can upregulate BeWo cell viability and zonula occludens expression under hypoxia [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(在线): 4970-4975. |
| [3] | Zhang Tongtong, Wang Zhonghua, Wen Jie, Song Yuxin, Liu Lin. Application of three-dimensional printing model in surgical resection and reconstruction of cervical tumor [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1335-1339. |
| [4] | Zhang Xiumei, Zhai Yunkai, Zhao Jie, Zhao Meng. Research hotspots of organoid models in recent 10 years: a search in domestic and foreign databases [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1249-1255. |
| [5] | Wang Zhengdong, Huang Na, Chen Jingxian, Zheng Zuobing, Hu Xinyu, Li Mei, Su Xiao, Su Xuesen, Yan Nan. Inhibitory effects of sodium butyrate on microglial activation and expression of inflammatory factors induced by fluorosis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1075-1080. |
| [6] | Wang Xianyao, Guan Yalin, Liu Zhongshan. Strategies for improving the therapeutic efficacy of mesenchymal stem cells in the treatment of nonhealing wounds [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1081-1087. |
| [7] | Liao Chengcheng, An Jiaxing, Tan Zhangxue, Wang Qian, Liu Jianguo. Therapeutic target and application prospects of oral squamous cell carcinoma stem cells [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1096-1103. |
| [8] | Xie Wenjia, Xia Tianjiao, Zhou Qingyun, Liu Yujia, Gu Xiaoping. Role of microglia-mediated neuronal injury in neurodegenerative diseases [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1109-1115. |
| [9] | Li Shanshan, Guo Xiaoxiao, You Ran, Yang Xiufen, Zhao Lu, Chen Xi, Wang Yanling. Photoreceptor cell replacement therapy for retinal degeneration diseases [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1116-1121. |
| [10] | Jiao Hui, Zhang Yining, Song Yuqing, Lin Yu, Wang Xiuli. Advances in research and application of breast cancer organoids [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1122-1128. |
| [11] | Wang Shiqi, Zhang Jinsheng. Effects of Chinese medicine on proliferation, differentiation and aging of bone marrow mesenchymal stem cells regulating ischemia-hypoxia microenvironment [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1129-1134. |
| [12] | Zeng Yanhua, Hao Yanlei. In vitro culture and purification of Schwann cells: a systematic review [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1135-1141. |
| [13] | Kong Desheng, He Jingjing, Feng Baofeng, Guo Ruiyun, Asiamah Ernest Amponsah, Lü Fei, Zhang Shuhan, Zhang Xiaolin, Ma Jun, Cui Huixian. Efficacy of mesenchymal stem cells in the spinal cord injury of large animal models: a meta-analysis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1142-1148. |
| [14] | Hou Jingying, Yu Menglei, Guo Tianzhu, Long Huibao, Wu Hao. Hypoxia preconditioning promotes bone marrow mesenchymal stem cells survival and vascularization through the activation of HIF-1α/MALAT1/VEGFA pathway [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 985-990. |
| [15] | Shi Yangyang, Qin Yingfei, Wu Fuling, He Xiao, Zhang Xuejing. Pretreatment of placental mesenchymal stem cells to prevent bronchiolitis in mice [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 991-995. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||